Abstract
Nanoparticles have attracted extensive attention due to their high degree of cell targeting, biocompatibility, controllable biological activity, and outstanding pharmacokinetics. Changing the size, morphology, and surface chemical groups of nanoparticles can increase the biological distribution of agents to achieve precise tissue targeting and optimize therapeutic effects. Examples of their use include nanoparticles designed for increasing antigen-specific immune responses, developing vaccines, and treating inflammatory diseases. Nanoparticles show the potential to become a new generation of therapeutic agents for regulating inflammation. Recently, many nanomaterials with targeted properties have been developed to treat acute lung injury/acute respiratory distress syndrome (ALI/ARDS). In this review, we provide a brief explanation of the pathological mechanism underlying ALI/ARDS and a systematic overview of the latest technology and research progress in nanomedicine treatments of ALI, including improved nanocarriers, nanozymes, and nanovaccines for the targeted treatment of lung injury. Ultimately, these nanomedicines will be used for the clinical treatment of ALI/ARDS.
Introduction
Acute respiratory distress syndrome (ARDS) is the most advanced form of acute lung injury (ALI), and it is caused by harmful extrapulmonary factors and pathogenic factors. 1 The mortality rate of patients with ALI/ARDS has decreased over the past decade, but it remains as high as 30–40%; moreover, ALI/ARDS is an important cause of death in critically ill patients. The pathological features of ALI/ARDS are diffuse alveolar damage, which involves the destruction of type I alveolar epithelial cells and pulmonary vascular endothelial cells; pulmonary interstitial fibrosis; and type II epithelial cell proliferation, which results in pulmonary edema and inflammatory cell infiltration in lung tissues. 2 Some patients eventually develop ARDS, which causes irreversible acute respiratory failure and multiple organ dysfunction. As medical interventions have improved, novel drugs and therapies for ALI/ARDS have gradually been developed; however, the case fatality rate of ALI/ARDS has not significantly decreased, and treatment of ALI/ARDS remains difficult. Therefore, searching for safer and more effective therapies has been the focus of recent research. The pathogenesis of ALI/ARDS is complex, and to date, it has not been fully elucidated. Currently, it is thought that ALI/ARDS represents an extensive and excessive inflammatory response in lung tissues after severe infection, trauma, shock, massive blood transfusion, infusion, etc.3,4 Therefore, the inflammatory response plays an important role in the occurrence and development of ALI/ARDS. The pathophysiological mechanism underlying ALI/ARDS consists mainly of inflammatory cell migration, fiber proliferation, and apoptosis. Inflammatory cells and inflammatory mediators constitute the ‘cell network’ and ‘cytokine network’ of the inflammatory response and regulate the immune response in ALI/ARDS. These networks regulate the inflammatory response through different signal transduction pathways. 5 Therefore, overactivation or imbalance of the inflammatory response is a primary cause of ALI/ARDS.
Nanoparticles are very small particles with diameters ranging from 1 to 100 nm. Because of their unique physical and chemical properties, nanoparticles are widely used in the fields of engineering and medicine as well as other scientific fields. Nanoparticles have attracted substantial attention due to their strong ability to target cells, excellent biocompatibility, controllable biological activity, and outstanding pharmacokinetics.6,7 Changing the size, morphology, and surface chemical groups of nanoparticles can increase the biological distribution of these agents to precisely target tissues and optimize therapeutic effects. Nanoparticles are thus powerful tools for the treatment of human immune-related diseases. Nanoparticles have been designed for applications including increasing antigen-specific immune responses, developing vaccines, and treating inflammatory diseases. Moreover, studies have shown that nanoparticles can target phagocytes. 8 Therefore, nanoparticles have the potential to become a new generation of therapeutic agents for regulating inflammation. Recently, many nanomaterials with targeted properties have been developed to treat ALI/ARDS. The emergence of nanomedicine has led to new strategies for overcoming limitations in the field of traditional drug therapy, but challenges remain, limiting the likelihood of the clinical translation of these agents. In particular, toxicity and safety are the major challenges facing the clinical translation of nanomedicines.
In this review, we provide a brief explanation of the pathological mechanism underlying ALI/ARDS, and a systematic overview of the latest technology and research progress in the treatment of ALI with nanomedicine, including improved nanocarriers, nanozymes, and nanovaccines for the targeted treatment of lung injury. Ultimately, these nanomedicines will be used for the clinical treatment of ALI/ARDS.
Pathological mechanism underlying ALI/ARDS
ALI is a common clinical syndrome in critically ill patients, and its severe form is called ARDS. 1 The pathophysiological features of this condition are alveolar epithelial cell and pulmonary microvascular endothelial cell injury and increased permeability of the alveolar membrane, resulting in diffuse pulmonary interstitial and alveolar edema. 2 Moreover, decreased levels of active alveolar surface substances and dystonia of the alveolar surface ultimately lead to decreased lung compliance and severe ventilation/blood flow imbalance. The clinical manifestations of this condition are progressive hypoxemia and respiratory distress, and the radiological manifestations of ALI are heterogeneous exudative lesions, which can develop into severe disease and lead to ARDS.
Many factors cause ALI/ARDS, including factors that act either directly or indirectly; among these factors, the most common factor that acts directly is infection, such as bacterial or viral infection. In addition, stomach content inhalation and lung contusions cause ALI/ARDS. The most common factor that acts indirectly is sepsis, which is a consequence of severe trauma, massive transfusion or infusion, acute severe pancreatitis, etc. The pathogenesis of ALI is complex, and the pathological process mainly includes the following three stages. 9 During the acute exudation stage, pulmonary vascular endothelial cells and type I alveolar epithelial cells are damaged; a large number of inflammatory cells, such as neutrophils, disseminate and infiltrate lung tissues; and the excessive release of inflammatory factors in the lungs leads to an imbalance in the proinflammatory/anti-inflammatory system and the oxidation/antioxidant system, triggering an inflammatory ‘waterfall-like’ cascade reaction and ultimately exacerbating the initial lung injury. With increased microvascular permeability, alveolar edema occurs and causes disordered lung function. During lung tissue remodeling, chronic inflammation develops, and the inflammatory cascade plays a key role in disease-related cell apoptosis, proliferation, and migration.10,11 Extracellular matrix deposition occurs in alveoli, type II alveolar epithelial cells undergo significant proliferation, and squamous metaplasia may also develop during this stage. In the fibrosis stage, a continuous inflammatory response and hyperplasia lead to the degeneration of dense collagen in the alveoli and hyalinotic changes in the alveolar wall. The deposition of cellulose in alveoli leads to pulmonary tissue fibrosis and irreversible damage to respiratory function. Due to excessive inflammatory responses and oxidative stress, the vascular endothelium, and alveolar epithelial cells are damaged, which disrupts their barrier function and leads to respiratory failure in patients with impaired gas exchange capacity.
One of the critical mechanisms underlying the development of ARDS involves an imbalance in the inflammatory response, which exacerbates alveolar epithelial and endothelial cell damage and leads to increased alveolar-capillary permeability, then fibrosis, and ultimately ARDS. 12 During ALI/ARDS, the inflammatory response can be initiated by exogenous and endogenous stimuli. Exogenous stimuli, such as bacterial antigens, initiate inflammatory responses by activating Toll-like receptors (TLRs) and intracellular kinases, leading to cell membrane rupture and oxidative stress. 13 Endogenous stimuli primarily include damage-associated molecular pattern molecules that are released by dead cells or local inflammatory cells, and these molecules activate and recruit immune cells by binding to different receptors, such as TLRs or interleukin-1 receptor (IL-1R), thereby activating proinflammatory pathways.
The main inflammatory cells that are involved in ARDS are neutrophils and lung macrophages. Neutrophils, which are important inflammatory cells, release a large number of inflammatory factors and produce reactive oxygen species (ROS), which directly damage lung tissue. 14 During the initial (acute) phase of inflammation, neutrophils respond to the migration of inflammatory cells to the site of inflammation, activating these inflammatory cells, initiating inflammatory cascades, and contributing to uncontrolled inflammation in the lung. Alveolar endothelial cells are damaged by proinflammatory factors that are released by neutrophils as well as by ROS, and this damage results in pathological symptoms. Alveolar endothelial cells synthesize and release vasoactive substances and inflammatory mediators, which participate in vascular permeability, coagulation, and systemic inflammatory responses and thus play important roles in ALI/ARDS. The development of ALI/ARDS leads to the release of a variety of proinflammatory factors, including cytokines and lipid products, and the release of these factors is accompanied by the production of procoagulant factors and the activation of neutrophils, platelets, and monocytes, all of which disrupt the endothelial barrier and lead to deregulated tissue perfusion and oxygenation. In addition, alveolar macrophages can produce proinflammatory factors. Alveolar macrophages account for approximately 90% of all macrophages in the lung, they are activated at the beginning of inflammatory responses in the lungs, and they are sources of inflammatory mediators and cytokines. 15 During the initial inflammatory phase, alveolar macrophages are activated and release neutrophil-activating factors, such as tumor necrosis factor α (TNF-α), interleukin-1β (IL-1β), and IL-8. These early inflammatory factors activate neutrophils, which, in turn, activate more macrophages in the alveoli; thus, a series of inflammatory responses is initiated.
However, studies have shown that neutrophils play both proinflammatory and anti-inflammatory roles in ALI/ARDS. The main mechanism may be related to the production of chemokines by macrophages. The balance between the concentration of chemokines produced by local macrophages and the concentration of chemokines produced at distant inflammatory sites affects the direction of neutrophil migration in the lung. In response to this concentration gradient, neutrophils cross the endothelium and are recruited to infected or inflammatory sites where they release many proteolytic enzymes and ROS as well as stimulate the release of the anti-inflammatory factor IL-10 by macrophages.
Despite the severity of ARDS, there is no effective treatment. Inhaled nitric oxide, glucocorticoids, and β-agonists have been used to treat ARDS. However, these agents either only temporarily alleviate symptoms without reducing mortality rates or cause adverse effects on other organs. Therefore, effective targeted drugs with low toxicity need to be developed for the treatment of ALI/ARDS.
Application of nanoparticles in the treatment of ALI
Nanoparticles have attracted substantial attention due to their ability to effectively target therapeutic agents to particular sites, excellent biocompatibility, controllable biological activity, and outstanding pharmacokinetics.
16
Changing the size, morphology, and surface chemical groups of nanoparticles can allow better biological distribution of agents
The emergence of nanomedicine will lead to new approaches for addressing the current limitations in the field of traditional drug therapy, but challenges to the clinical translation of nanomedicines remain. Toxicity and safety are the most important problems that need to be resolved for the clinical translation of nanomedicine. Additional novel nanoparticles for use in the targeted treatment of lung injury are urgently needed; examples of such nanoparticles include improved nanocarriers, nanozymes, and nanovaccines, which will become novel nanomedicines for the clinical treatment of ALI.
Novel nanocarriers for the treatment of ALI
Liposome-based nanoparticles
Liposomes are artificial membrane-bound vesicles. In water, the hydrophilic heads of phospholipid molecules interface with water molecules, and the hydrophobic tails of liposomes extend into the air. After agitation, spherical liposomes with a double lipid membrane form. Liposomes are among the most well-studied and relatively mature nanocarriers due to their advantages of high selectivity, low toxicity, lack of immunogenicity, and easy degradation
Li

Lipid nanoparticles based on the formation of protein corona for treatment of acute lung injury. (a) n-DOCPs targeted activated neutrophils in inflammatory mice. (b) Neutrophils-mediated delivery of drug-loaded n-DOCPs mitigated acute lung injury and pneumococcal pneumonia.
The therapy application of nanomedicine in ARDS.

ACM, apoptotic cell membrane; ALI, acute lung injury; DMON, dendritic mesoporous silicon nanoparticles; GNPs, gold nanoparticles; MOFs, metal-organic frameworks; MSN, mesoporous silica nanoparticles; PBA, phenylboronic acid.
Novel polymeric nanoparticles
The use of polymer nanoparticles as carriers for pulmonary drug delivery has attracted substantial attention. Polymer nanomaterials are designed from natural or synthetic polymers, most of which are biodegradable and biocompatible. Commonly used polymer materials include natural products (chitosan, alginate, and cyclodextrin) and synthetic products [polyethylene glycol (PEG), polylactic acid-
Dendrimers are macromolecules that exhibit regular branching. Recently, novel dendrimer–drug conjugates and dendrimer gene carriers have been developed. The outer layer of dendrimers can be polymerized to establish higher-order dendrimer structures, and the outer layer of dendrimers can be engrafted with a large number of functional groups to enhance their functionality. Based on their clear structural advantages, dendrimers can be used as nanocarriers to deliver different types of drugs, and they exhibit a combination of various characteristics that render them useful as a therapeutic strategy for efficient and precise treatment; thus, dendrimers play an important role in drug delivery.
Poly(amidoamine) (PAMAM) dendrimers
PAMAM dendrimers are polymers with special structures that have increasingly attracted attention. PAMAM dendrimers are perfectly formed monodisperse polymers with an organized and highly branched three-dimensional structure, and these structures are easy to modify to change their solubility because they not only form an internal cavity but also have a large number of functional groups on their surface. Therefore, PAMAM dendrimers have the potential to be used as drug delivery systems in biomedicine and other fields. In many studies, PAMAM dendrimers have been shown to mediate effective cell uptake and siRNA transfection

Poly(amidoamine) dendrimer mediated intracellular delivery to alleviate acute lung injury. (a) The synthesis of G5.NH2-PBA and formation of Den/FN complexes. (b) Treatment of LPS-induced ALI mouse models with G5. NH2-PBA effectively relieved the symptoms of ALI.
Phosphorus dendrimers
Phosphorus dendrimers are commonly synthesized by introducing phosphate-containing units (such as hachlorocyclotrimonitrile and thiophosphate) into PAMAM dendrimers. Phosphorus dendrimers are widely used as nanocarriers to treat several diseases due to the highly branched, symmetrical structure and highly uniform molecular weight distribution of traditional dendrimers as well as the good biocompatibility that is conferred by phosphorus. Studies have shown that phosphorus dendrimers can be used as effective nanocarriers of oligodeoxynucleotides or small interfering RNAs (siRNAs).46,47 As the molar ratio between the positively charged groups of the dendrimer and the phosphate in the siRNA skeleton increases, the surface potential of the cationic phosphorus dendrimer/siRNA complex changes from negative to positive, and the hydrodynamic diameter gradually increases. More importantly, the siRNA molecules in the complex do not change, indicating that cationic phosphorus dendrimers can effectively prevent RNA digestion and can be used as effective nanocarriers for RNA-based disease therapy.
46
Bohr
Novel inorganic nanoparticles
Inorganic nanoparticles (NPs) are a class of nanoparticles that can be synthesized by physical or chemical methods; these NPs have a variety of morphologies and a size range of 1–100 nm. They are usually composed of inert metals and biocompatible metals. NPs include metal nanomaterials, such as gold, silver, gadolinium, hafnium oxide, iron oxide, and quantum dots, and inorganic nonmetallic nanomaterials, such as carbon and black phosphorus. The surface of NPs is easily modified, and it can be combined with drug molecules in different ways, such as through electrostatic interactions, hydrophobic interactions, and covalent bonds between enzyme-sensitive groups, to achieve responsive cargo release; thus, these NPs are ideal choices for drug delivery. In addition to the ease of surface modification, NPs are simpler to prepare than other nanomaterials, and they have higher drug-loading capacity and better biocompatibility for drug delivery; these NPs exert greater therapeutic effects. For example, gold nanoparticles (GNPs) are becoming the preferred delivery system for anti-inflammatory drugs because of their easy synthesis, controllable size, and relative safety, and their surface can be easily modified with peptides, allowing them to exhibit novel biological activities. Wang
Mesoporous silica nanoparticles (MSNs), which are novel inorganic materials, possess excellent characteristics, such as a high specific surface area, large pore volume, controllable particle size, and good biocompatibility. Compared to other carriers, MSNs have a higher loading efficiency, and thus, they are widely used in the field of biomedicine. Importantly, due to the ability of MSNs to preferentially accumulate in lung tissues and sites of inflammation, they may have advantages over other nanoparticles in the treatment of ALI.49,50 For instance, García-Fernández

The application of mesoporous silica nanoparticles (MSN) to treat acute lung injury. (a) Design of the TNFR-MSNs nanodevices for acute lung injury therapy. (b) Evaluation of the therapeutic effect of TNFR-Dex-MSNs in LPS-ALI mice.
Metal-organic frameworks
Metal-organic frameworks (MOFs) are coordination polymers that are composed of metal ions and organic ligands. Due to the diversity of metal ions and the different modes of organic ligand coordination, synthesized MOFs exhibit high porosity, structural diversity, and controllable functionality. Modifying MOFs allows the generation of novel functional materials. Compared to other porous nanomaterials, MOFs have strong adaptability to nanoparticle pore sizes. Nanomedicines that include MOFs as carriers can be adapted to the size of drug particles, increasing drug loading. In addition, MOFs have high permeability and retention properties, enabling passive targeting in the treatment of disease.
51
Furthermore, recent studies have revealed the potential efficacy of MOFs in the treatment of ALI.
52
For instance, Wang

The application of MOFs to treat acute lung injury. (a) The scheme for ZPM@PDE-siZEB1/2 preparation and intratracheal therapeutic mechanism of ZPM@PDE-siZEB1/2 in ALI model mice. (b) Histopathological changes of lung tissue in ALI mice after treatment with ZPM@PDE-siZEB1/2.
Nanozymes for the treatment of ALI
Nanozymes are novel nanomaterials with enzyme-like activities. Nanozymes, which are recently developed artificial enzymes, have contributed to the field of simulated enzymes. Nanozymes catalyze substrates under mild or extreme conditions and convert them into products through enzymatic kinetics, such as the property represented by the Michaelis–Menten equation. Compared with traditional enzymes, nanozymes have advantages such as high catalytic activity, functionality under mild reaction conditions, good stability, and inexpensive and large-scale production. Nanozymes are classified as oxidoreductases, hydrolases, synthases, and isomerases according to their catalytic activity. Many nanomaterials have been reported to exhibit unique oxidoreductase-mimicking activities. For example, Fe3O4, Au, and carbon nanotubes have very high peroxidase (POD)-like catalytic activity.56–58 CeO2 exhibits oxidase (OXD)-like activity.
59
In addition, Pt, Pd, MnO2, etc., show unique catalase (CAT)- and superoxide dismutase (SOD)-like activities, enabling them to effectively scavenge harmful ROS and play important roles in cell protection, especially
Inflammation is a normal immune response and is normally beneficial for the body. However, when inflammation in the body is dysregulated, acute inflammation can become chronic and lethal. The inflammatory response is closely related to the excessive production of ROS; therefore, antioxidants are extremely important in the treatment of inflammation.
63
In recent years, many nanozymes, such as SOD- and CAT-like nanozymes, have been shown to exhibit activity similar to endogenous antioxidants. Specifically, nanozymes can mimic endogenous antioxidant systems and participate in cell protection. Nanozymes with CAT-, SOD-, glutathione peroxidase (GPx)-like activities and other activities can decompose ROS into molecules that are characterized by low reactivity, such as H2O or O2, to relieve cellular oxidative stress; therefore, they can be used as antioxidant therapeutic agents. For example, Huang
Nanozymes benefit anti-inflammatory therapy due to their potent anti-inflammatory properties, ROS-scavenging capabilities, and ability to function as combination drug carriers that can be loaded with certain antioxidant active ingredients, thereby enhancing overall anti-inflammatory effects. For instance, nanozymes that are constructed using FeCl3 and poly(vinylpyrrolidone) (PVP) can efficiently encapsulate curcumin, functioning as a combined antioxidant nanomedicine. The Fe nanoparticles in the nanozymes can scavenge ROS, while curcumin is a natural antioxidant that suppresses inflammation. Fe-Cur NP treatment significantly reduced the levels of TNF-α, IL-1β, and IL-6 in the serum and bronchoalveolar lavage fluid (BALF) of mice with ALI, reduced the release of intracellular Ca2+ and inhibited NLRP3 inflammasome and NF-κB signaling pathway activation. In addition, Fe-Cur NPs were intratracheally and intravenously injected into mice with ALI, and the accumulation of these nanozymes in lung tissues was higher than that of free curcumin, confirming the therapeutic efficacy of nanozymes in ALI. Moreover, Fe-Cur NPs decreased the numbers of macrophages (CD11bloF4/80hi) and CD3+CD45+ T cells in the lungs of mice with ALI, contributing to decreasing the inflammatory cytokine storm that was induced by ALI. More importantly, these nanoparticles were noncytotoxic
The modifiable properties of nanozymes allow them to potentially be applied for targeted therapy. For example, the preparation of AOzyme@ACM, which uses an apoptotic cell membrane (ACM) to encapsulate antioxidant nanozymes (AOzyme), enhances macrophage recognition of ‘eat me’ signals by simulating apoptotic bodies, thereby effectively enhancing the clearance of apoptotic cells. In addition, the AOzyme nanozyme portion comprises HMnO2 and Cu NPs. Cu NPs can scavenge a wide range of ROS, including H2O2, O2•−, ·OH, and other free radicals. MnO2 effectively eliminates H2O2. Therefore, the HMnO2-Cu composite in AOzyme@ACM exhibits antioxidase-mimicking activity and efficiently scavenges ROS. Therefore, AOzyme@ACM treatment significantly inhibited inflammation in mice with LPS-induced ALI, promoted macrophage polarization toward the M2 phenotype, and suppressed the activation of the NF-κB signaling pathway, thereby reducing inflammatory cell infiltration and alleviating edema in mice with ALI (Figure 5 and Table 1).
37
Wu
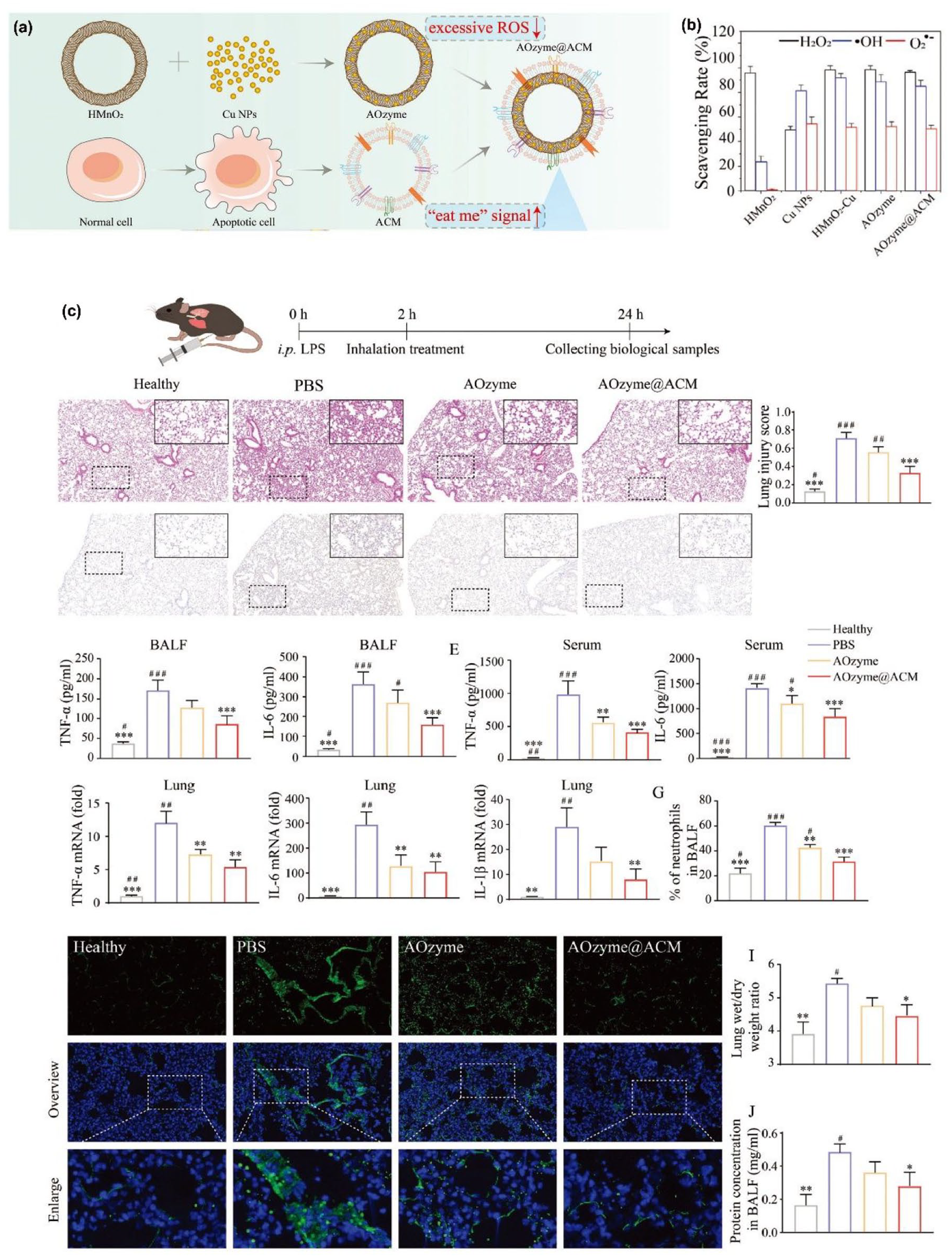
Nanozymes for the treatment of acute lung injury. (a) The synthesis of apoptotic cell membrane-coated antioxidant nanozymes (AOzyme@ACM). (b) ROS scavenging capabilities of AOzyme@ACM. (c) Pro-efferocytic AOzyme@ACM alleviated sepsis-related acute lung injury.
Carbon nanodots (C-dots), which are fluorescent nanomaterials, have attracted considerable attention due to their unique properties.
65
There are abundant oxygen-containing functional groups on the surface of C-dots, such as hydroxyl, carboxyl, and amino groups, which endow C-dots with effective antioxidant enzymatic activities.
66
In addition, C-dots exhibit catalytic activity due to their small size and abundant active sites. C-dots show great potential for application in the treatment of diseases, especially ROS-related diseases.
67
Therefore, C-dots have been extensively studied in the treatment of inflammatory diseases, such as ALI. For example, Wang
Nanovaccines for the treatment of lung injury
Nanovaccines are a new generation of vaccines based on nanotechnology, and vector design and surface ligand modification allow nanovaccines to elicit long-term and effective immune responses. Nanovaccines have advantages over conventional vaccines in terms of lymph node accumulation, antigen assembly, and antigen presentation. Nanotechnology can promote the processing of nanovaccines as targets for the immune response. Once nanovaccines are internalized by antigen-presenting cells (APCs), nanoparticles induce the formation of inflammasome complexes, the secretion of inflammatory cytokines, and the recruitment of immune cells. Nanovaccines increase antigen stability and target antigen-presenting cells, promote the maturation of these cells, and activate specific immune responses. In addition, by adjusting the shape, particle size, and charge of nanovaccines or by applying surface modification, scientists can enhance the tissue targeting capacity of these vaccines, increase the efficiency of their uptake by cells, and reduce their cytotoxic effects. Therefore, nanovaccines show great promise in the treatment of ALI caused by pneumonia.
Notably, Wu

Nanovaccines for the treatment of acute lung injury. (a) A schematic diagram of the antibacterial immune regulation of nanovaccine encapsulated by bacterial OMVs. The nanovaccine induced bacteria-specific immunity against the source bacteria and produced specific antibodies and toxic T cells, which were used to treat acute pneumonia and prevent infection. (b) The treatment effect of pneumonia by injecting serum containing the antibody and pure serum
Limitations and prospects
One advantage of nanomedicines over traditional drugs is their ability to be used in combination therapy. Nanocarriers can efficiently deliver anti-inflammatory or antioxidant drugs, such as dexamethasone, resveratrol, and curcumin. In addition, polymer- or lipid-based nanocarriers and peptides can be used to deliver various nucleic acids, including cDNA, plasmid DNA, siRNA, and miRNA. Multiple drugs can be incorporated into nanocarriers to achieve codelivery, enhance the efficiency of drug delivery, reduce off-target side effects, and exert synergistic therapeutic effects. Furthermore, targeted delivery through nanocarriers can improve the pharmacokinetic properties of therapeutic agents. Many nanocarriers have characteristics that are desirable for drug delivery, especially characteristics that improve the solubility, stability, or activity of hydrophobic agents, nucleic acids, enzymes, and peptides.28,47 Some traditional drugs, such as the Src tyrosine kinase inhibitor (PP2) and sivelestat, have disadvantages such as insufficient solubility, poor stability, and short
The emergence of nanomedicine provides new directions for overcoming the limitations of traditional drug treatment, but challenges remain in enhancing the clinical translation of these agents. Although nanoparticles have certain advantages in terms of particle size, easy synthesis methods, surface modification capacity, and biocompatibility,
Some nanomedicines are in clinical use, but many are still in clinical trials, and these clinical trials are mostly focused on the use of nanomedicines for
Conclusion
The clinical treatment of ALI prioritizes supportive care and mechanical ventilation. Although some preclinical studies demonstrated the benefits of drug interventions, to date, these measures have not been proven to be entirely effective, and no pharmacological therapy has been established as a fully effective treatment for ALI. In the past few decades, a variety of nanocarriers, such as liposomes, microemulsions, nanoemulsions, nanogels, polymeric micelles, and polymeric nanoparticles, have been widely used for the efficient delivery of drugs. Changing the size, morphology, and surface chemical groups of nanoparticles can improve the biological distribution of these agents in the body as well as tissue targeting, thereby optimizing therapeutic effects. Therefore, nanoparticles have gradually become a powerful tool for the treatment of human immune-related diseases. In addition to optimized nanocarriers, nanozymes and nanovaccines have been used as effective nanomaterials for the treatment of inflammatory diseases in recent years. Recent advances in nanotechnology may provide new perspectives for the treatment of ALI with drugs.
