Abstract
Background:
Gut microbiota assumes an essential role in the development and progression of pulmonary arterial hypertension (PAH). Trimethylamine N-oxide (TMAO), a gut microbiota-dependent metabolite, is correlated with the prognosis of patients with PAH. However, the correlation between changes in TMAO (ΔTMAO) and the prognosis of PAH remains elusive.
Objectives:
To investigate the association between ΔTMAO and prognosis of PAH, and explore whether dynamic assessment of TMAO level was superior to measurement at a single time point in predicting prognosis.
Design:
Single-center cohort study.
Methods:
Consecutive patients diagnosed with PAH and had at least two TMAO measurements taken from May 2019 to June 2020 were eligible. The outcome events of this study were defined as adverse clinical events.
Results:
A total of 117 patients with PAH who had two TMAO measurements and follow-up were included in this study. Patients with ΔTMAO ⩾1.082 μmol/L had over four times increased risk of adverse clinical events than their counterparts after adjusting for confounders [hazard ratio (HR) 4.050, 95% confidence interval (CI): 1.468–11.174; p = 0.007]. Patients with constant high TMAO levels at both time points had the highest risk of adverse clinical events compared with patients with constant low TMAO levels (HR 3.717, 95% CI: 1.627–8.492; p = 0.002). ΔTMAO was also associated with changes in parameters reflecting PAH severity (p < 0.05).
Conclusion:
Changes in TMAO were independently correlated with prognosis in patients with PAH, irrespective of baseline level of TMAO. ΔTMAO also correlated with alteration in disease severity. Repeated assessment of TMAO level contributes to better identification of patients with increased risk of adverse clinical events.
Introduction
Accumulating evidence revealed that gut microbiota plays an essential role in several diseases. As one of the common metabolites of gut microbiota, trimethylamine N-oxide (TMAO) raises great concerns.
1
Animal food-derived choline and
Diet discrepancy in individuals results in the heterogeneity of circulating plasma TMAO levels, and the surrounding milieu also plays a substantial role in gut microbiome shaping, which indirectly influences the synthesis of TMAO.9,10 Therefore, exploring the impact of changes in plasma TMAO levels (ΔTMAO) could dynamically reflect the influence of microbial metabolite alteration on disease condition and prognosis, providing insights into management strategies. 11 Previous studies have found that elevation of TMAO over several years was correlated with increased risk of coronary heart diseases and dynamic observation of TMAO level enhanced the discernment of individuals at higher risk of experiencing coronary heart diseases than single time point assessment. 12 The association between ΔTMAO and prognosis of pulmonary arterial hypertension (PAH), however, remains elusive.
In this study, we aim to investigate the association between ΔTMAO and prognosis of PAH, and explore whether dynamic assessment of TMAO level was superior to measurement at a single time point in predicting prognosis.
Methods
Study design and population
This cohort study was conducted in Fuwai Hospital. The current study was conducted in line with the Declaration of Helsinki, and the Ethics Committee of Fuwai Hospital approved the protocol of study. Written informed consent was signed by study participants. Consecutive patients diagnosed with idiopathic PAH and PAH associated with congenital heart disease in accordance with guideline 13 between May 2019 and June 2020 were eligible. Inclusion criteria were as follows: (1) patients who had at least two TMAO measurements taken and (2) patients who were able to be followed up regularly. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 14
Demographics, comorbidities, World Health Organization functional class (WHO-FC), 6-min walk distance (6MWD), and parameters derived from echocardiography, laboratory tests, and right heart catheterization (RHC) were obtained from an electronic medical record system.
RHC
RHC was performed as previously reported. 15 Hemodynamic variables were measured including right atrial and ventricular pressure, pulmonary arterial pressure, and pulmonary artery wedge pressure. Pulmonary vascular resistance was determined using the formula: (mean pulmonary arterial pressure − pulmonary artery wedge pressure) divided by cardiac output.
Examination of TMAO
Supernatants of fasting blood samples were obtained for TMAO examination by liquid chromatography with mass spectrometry. The specific procedure was demonstrated in our previous work. 16 Briefly, 20 μL was added to a 1.5-mL tube and mixed with 80 μL of a 10 μM internal standard consisting of d9-metabolites in methanol. The proteins in the samples were vortex-precipitated for 1 min, centrifuged at 4°C for 10 min at 20,000 g, and the supernatant was recovered. A standard curve was constructed using 20 μL of standards with different concentrations (0–100 μM) in parallel to obtain the exact concentration of analytes. The standard curve was acceptable when the coefficient of determination (R 2 ) reached 0.99. The supernatant (70 μL) was injected into a silica column at a flow rate of 0.5 mL/min using an LC-20AD Shimadzu pump system interfaced with an API 5500Q-TRAP mass spectrometer. Discontinuous gradients were generated to resolve analytes by mixing solvent A (0.1% propanoic acid in water) with solvent B (0.1% acetic acid in methanol) at different ratios starting from 2% B linearly to 95% B over 5 min, then held for 1 min, and then back to 2% B. Electrospray ionization was employed to monitor the precursor and characterize the production of transitions using multiple reactions monitoring precursors in the positive-ion mode for TMAO at m/z 76→58 and d9-TMAO at m/z 85→66. Within every set of 20 samples, three quality control specimens characterized by varying concentrations of TMAO were assessed.
Baseline TMAO was measured at hospitalization and the second measurement was performed after an average of 6 months in this study.
Follow-up
Patients were regularly followed up through outpatient clinical visits, rehospitalization assessment, or telephone calls every 6 months after discharge until the occurrence of outcome or censored date (19 November 2022). The median times of follow-up in this study was 4.3,6 Patients with at least one follow-up were eventually included in the study. The time interval between the first measurement of TMAO and follow-up was 34 (24, 38) months, and the time interval between the second measurement of TMAO and follow-up was 25 (19, 33) months.
Outcome
The primary outcome events of this study were defined as adverse clinical events, which included death, rehospitalization due to heart failure, or deterioration of PH involving the presence of deteriorating symptoms, an elevated WHO-FC relative to the baseline assessment, or a minimum decline of 15% in the 6MWD from the baseline measurement.8,17 Time to adverse clinical events was calculated from the time of the second TMAO examination to adverse clinical events or censored date. Outcomes were adjudicated by two independent senior clinicians (CX and ZL). We also used death as the secondary endpoint.
Statistical analysis
ΔTMAO was calculated as plasma TMAO level at the second examination time − plasma TMAO level at the first examination time. Continuous data were presented as either the mean ± standard deviation or as the median (25th, 75th percentiles) contingent upon the normal distribution of the data, and categorical data were presented as numbers (percentage). For continuous variables, an independent sample t-test or nonparametric Mann–Whitney U test was used to identify the difference between groups as appropriate. For categorical parameters, the chi-square test or Fisher exact test was used as appropriate. A receiver operator characteristic curve (ROC) analysis was applied to determine the optimal cutoff of ΔTMAO in predicting adverse clinical events. Associations between ΔTMAO and other parameters were examined by Spearman’s coefficient. Univariable Cox analysis was performed to explore parameters correlated with adverse clinical events. Then parameters demonstrating statistical or clinical significance were incorporated into the multivariable Cox regression model. Kaplan–Meier method with the log-rank test was employed to compare time to adverse clinical events between groups. Sensitivity analysis was conducted by excluding patients with chronic kidney disease given that chronic kidney disease had a negative impact on the prognosis of patients and TMAO was secreted from the kidney. A significance threshold was established at a two-sided p value <0.05. Statistical analysis was executed using IBM SPSS Statistics version 25.0 (IBM SPSS Corp., Armonk, NY, USA) and R (version 4.3.2; R Foundation for Statistical Computing, Vienna, Austria).
Results
Study population
From May 2019 to June 2020, a total of 117 patients including 45 idiopathic PAH and 72 PAH associated with congenital heart disease were encompassed within this study. Patients’ plasma was obtained both in the first and second hospitalization for TMAO measurement. After a median of 25 (19, 33) months of follow-up, 39 patients experienced adverse clinical events, including 29 cases of rehospitalization due to heart failure or deterioration of PH, and 10 deaths. The optimal cutoff of ΔTMAO identified by ROC was 1.082 μmol/L, and accordingly patients were classified into two groups, namely ΔTMAO <1.082 μmol/L and ΔTMAO ⩾1.082 μmol/L.
Comparison of patients with ΔTMAO <1.082 μmol/L and ΔTMAO ⩾1.082 μmol/L
At baseline, patients with ΔTMAO ⩾1.082 μmol/L had comparable demographics, hemodynamics, echocardiographic parameters, and biochemistry examination results, while higher proportion in WHO-FC III or IV (56.4% versus 29.5%, p = 0.005) and higher prevalence of chronic kidney diseases (15.8% versus 1.0%, p = 0.001) than patients with ΔTMAO < 1.082 μmol/L (Table 1). The levels of baseline TMAO were also similar in both groups.
Characteristics at the first TMAO examination between ΔTMAO <1.082 μmol/L and ΔTMAO ⩾1.082 μmol/L groups.

Data are presented as mean ± standard deviation, median (25th, 75th percentiles), or number (percentage). Significant p values (p < 0.05) are bolded.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; CI, cardiac index; CKD, chronic kidney disease; EF, ejection fraction; Medicine, pulmonary hypertension specific therapy; mPAP, mean pulmonary arterial pressure; mRAP, mean right atrial pressure; 6MWD, 6-min walk distance; NT-proBNP, N-terminal pro-brain natriuretic peptide; PAWP, pulmonary artery wedge pressure; PVR, pulmonary vascular resistance; RVED/LVED, right ventricular end-diastolic diameter/left ventricular end-diastolic diameter; SvO2, mixed venous oxygen saturation; TAPSE, tricuspid annular plane systolic excursion; TMAO, trimethylamine N-oxide; TRV, tricuspid regurgitation velocity; WHO-FC, World Health Organization function class.
At the second time of measurement, despite comparable demographics and echocardiographic parameters, patients with ΔTMAO ⩾1.082 μmol/L had a significantly higher prevalence of chronic kidney disease, lower level of albumin (40.95 ± 6.54 versus 43.69 ± 4.03 g/L, p = 0.017), higher uric acid (469.02 ± 108.82 versus 402.32 ± 121.88 μmol/L, p = 0.028), and higher level of TMAO [3.35 (2.41, 4.46) versus 1.23 (0.75, 1.78) μmol/L, p < 0.001] than counterparts (Table 2).
Characteristic at second examination between ΔTMAO <1.082 μmol/L and ΔTMAO ⩾1.082 μmol/L groups.

Data are presented as mean ± standard deviation, median (25th, 75th percentiles), or number (percentage). Significant p values (p < 0.05) are bolded.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; CKD, chronic kidney disease; EF, ejection fraction; Medicine, pulmonary hypertension specific therapy; 6MWD, 6-min walk distance; NT-proBNP, N-terminal pro-brain natriuretic peptide; RVED/LVED, right ventricular end-diastolic diameter/left ventricular end-diastolic diameter; TAPSE, tricuspid annular plane systolic excursion; TMAO, trimethylamine N-oxide; TRV, tricuspid regurgitation velocity; WHO-FC, World Health Organization function class.
Associations between ΔTMAO and changes of well-validated parameters reflecting PAH severity
Supplemental Table S1 and Figure 1 illustrated associations between ΔTMAO and changes in well-validated parameters reflecting PAH severity. ΔTMAO correlated positively with changes in right ventricular end-diastolic diameter/left ventricular end-diastolic diameter (r = 0.38, p < 0.001) and N-terminal pro-brain natriuretic peptide (ΔNT-proBNP) (r = 0.34, p < 0.001), and negatively with changes in tricuspid annular plane systolic excursion (r = −0.21, p = 0.022).
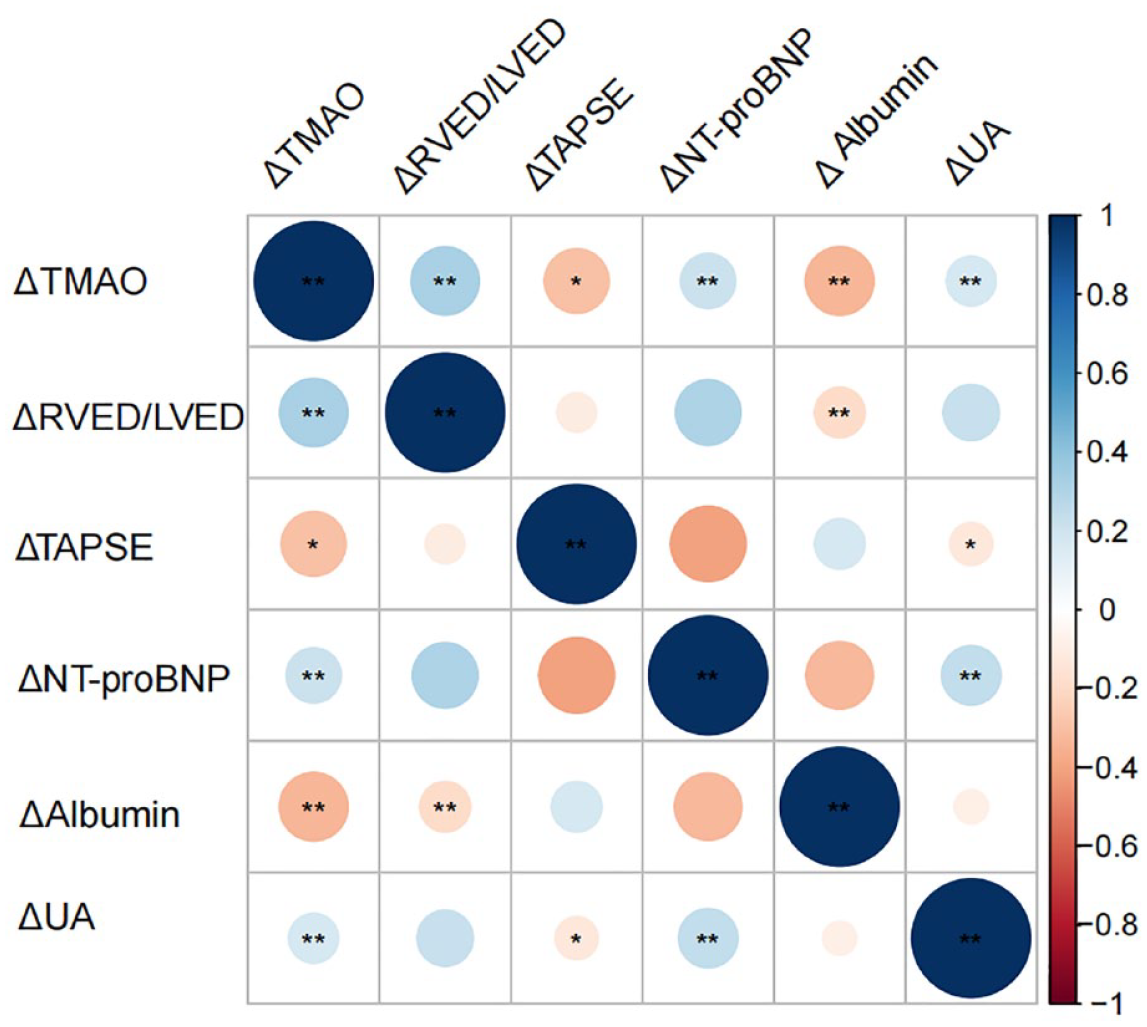
Association between ΔTMAO and changes of well-validated parameters reflecting PAH severity ΔTMAO correlated positively with Δ right ventricular end-diastolic diameter/left ventricular end-diastolic diameter, ΔNT-proBNP, and Δ uric acid. On the contrary, ΔTMAO correlated negatively with ΔTAPSE and Δ albumin. Δ was defined as the level of variables at the second examination time – the level of variables at the first examination time.
High ΔTMAO levels were associated with poor prognosis PAH patients
Univariable Cox regression analysis revealed parameters associated with adverse clinical events (Supplemental Table S2). Higher levels of TMAO at baseline and the second time point as well as ΔTMAO were correlated with increased risk of adverse clinical events. Multivariable Cox analysis indicated that patients with ΔTMAO ⩾ 1.082 μmol/L had about four times increased risk of adverse events than counterparts after adjusting for baseline TMAO level, demographics, comorbidities, severity of PAH, and biochemical parameters [Model 1: hazard ratio (HR) = 3.955, 95% confidence interval (CI): 1.486–10.527; p = 0.006]. Further adjustment for laboratory parameters at the second time point increased the association between ΔTMAO and prognosis in patients with PAH (Model 2: HR = 4.050, 95% CI: 1.468–11.174; p = 0.007). The two Cox models included baseline TMAO level and ΔTMAO concurrently, and both parameters remained statistically significant after adjusting for confounders, indicating that baseline TMAO and its alteration were independently correlated with the prognosis of patients with PAH (Table 3). A combination of baseline TMAO and ΔTMAO could significantly increase the predictive ability of baseline TMAO (combination versus baseline TMAO concordance index: 0.659 versus 0.579, p = 0.019) (Supplemental Table S3).
Multivariable Cox in identifying patients with clinical worsening.
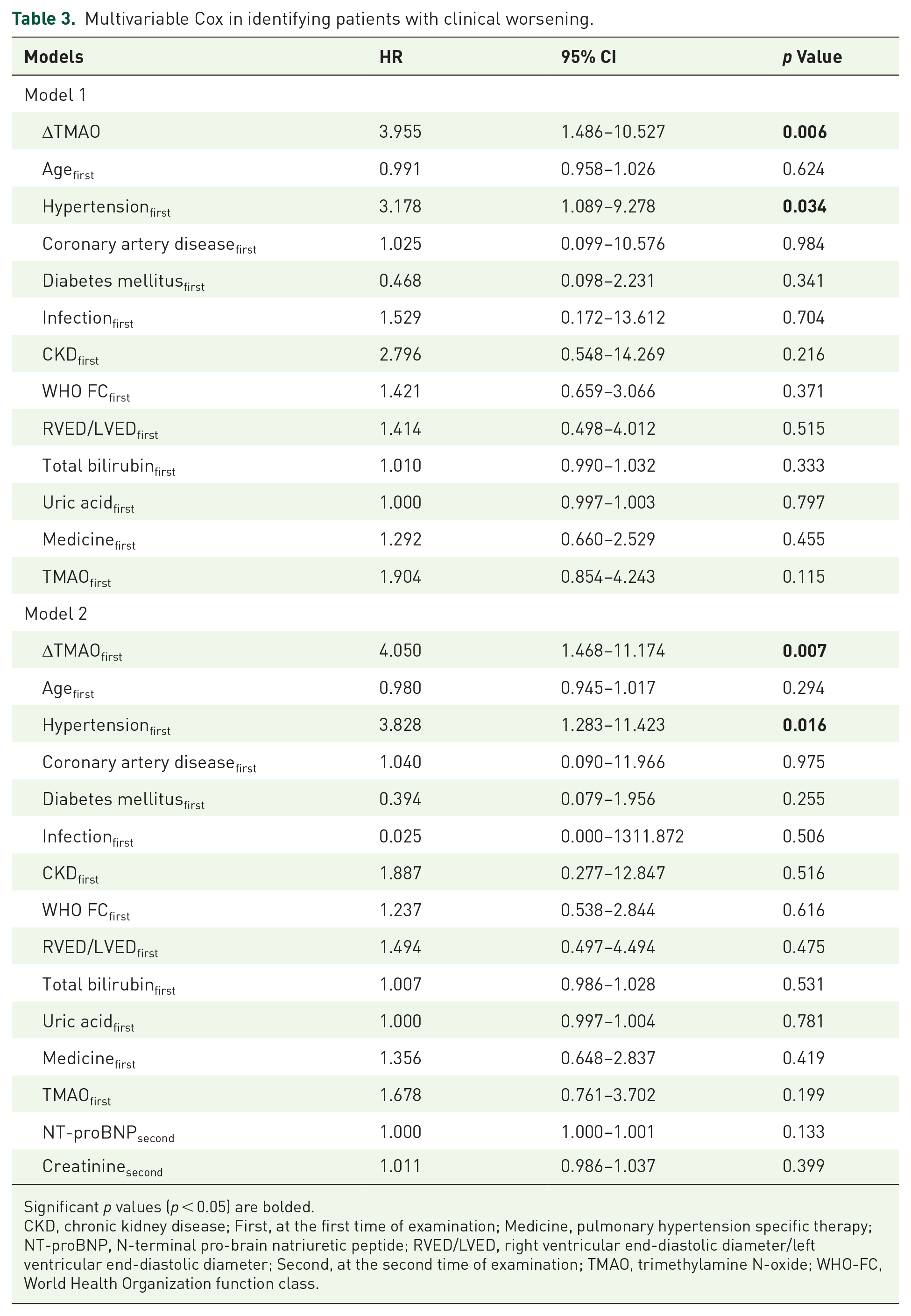
Significant p values (p < 0.05) are bolded.
CKD, chronic kidney disease; First, at the first time of examination; Medicine, pulmonary hypertension specific therapy; NT-proBNP, N-terminal pro-brain natriuretic peptide; RVED/LVED, right ventricular end-diastolic diameter/left ventricular end-diastolic diameter; Second, at the second time of examination; TMAO, trimethylamine N-oxide; WHO-FC, World Health Organization function class.
Kaplan–Meier analysis (Figure 2) indicated that patients with ΔTMAO ⩾1.082 μmol/L had significantly poorer prognosis and shorter time to adverse clinical events than counterparts.

Kaplan–Meier analysis among ΔTMAO <1.082 μmol/L and ΔTMAO ⩾1.082 μmol/L groups. Patients with ΔTMAO ⩾1.082 μmol/L had significantly poorer prognosis and shorter time to experience adverse clinical events than counterparts (log-rank p = 0.002). ΔTMAO was calculated as plasma TMAO level at the second examination time − plasma TMAO level at the first examination time.
We further explored whether constant high levels of TMAO were correlated with a significantly higher risk of adverse clinical events according to the TMAO levels at two times of measurement. We classified patients into four groups according to the cutoffs identified by ROC (TMAOfirst 2.495 μmol/L, TMAOsecond 1.655 μmol/L). Patients with constant high TMAO levels at both time points had the highest risk of adverse clinical events among the four groups (constant high TMAO levels/constant low TMAO levels: HR 3.717, 95% CI: 1.627–8.492; p = 0.002) and patients with low baseline TMAO level but a high level of TMAO at the second time point had the second highest risk of experiencing adverse clinical events (Figure 3).

Association between TMAO levels at two times and patients’ prognosis. Constant high levels of TMAO were correlated with a significantly higher risk of adverse clinical events according to the TMAO levels at two times of measurement. Patients with low baseline TMAO levels but high levels of TMAO at the second time point had the second highest risk of experiencing adverse clinical events.
Using death as a secondary endpoint, ΔTMAO ⩾ 1.082 μmol/L was also correlated with an elevated risk of death in patients with PAH (HR 3.980, 95% CI: 1.088–13.910, p = 0.037). Besides, after excluding patients with chronic kidney disease, we found that ΔTMAO ⩾ 1.082 μmol/L was still correlated with adverse clinical events in patients with PAH (HR = 2.792, 95% CI: 1.291–6.040, p = 0.009).
Discussion
In the current study, we demonstrated that elevation of TMAO level was independently correlated with poor prognosis in patients with PAH, irrespective of baseline level of TMAO. The combination of ΔTMAO and baseline level of TMAO was superior to the single baseline TMAO in predicting adverse clinical events, which highlights the importance of repeated assessment of TMAO level in monitoring the prognosis of patients with PAH. Furthermore, patients with a constant high level of TMAO had a higher risk of experiencing adverse clinical events than patients who experienced a reduction in TMAO or remained constant low level of TMAO.
A higher level of TMAO was observed in patients with PAH compared with normal controls, 18 and our previous study found that a high level of baseline TMAO was correlated with both the severity of the condition and prognostic outcomes of PAH.7,8 In this study, we observed the level of TMAO longitudinally and found that repeated measurement of TMAO could provide better discrimination of prognosis. Briefly, after controlling for the confounders that might influence the level of TMAO, including cardiovascular and metabolic comorbidities, chronic kidney diseases, baseline TMAO level, and PH severity, we found that ΔTMAO was independently correlated with the prognosis of patients with PAH. ΔTMAO also positively associated with changes in well-validated parameters that reflect PAH severity, indicating that ΔTMAO can also reflect the progression of PAH.
Several factors contribute to changes in TMAO, including altered bacterial communities, kidney function, and dietary patterns. Dysbiosis of the gut microbiota has been observed in both patients with PAH and animal models. 18 Advanced PAH can result in right heart failure and gastrointestinal congestion, which reduces bowel perfusion and contributes to gut bacterial translocation as well as intestinal bacterial imbalance. Disordered gut microbiota impairs the intestinal barrier and normal metabolic processes. Previous studies have found increased expression of enzymes for TMAO production in the feces of PH animal model. 18 Increased number of bacterial communities, which are associated with the metabolism of TMAO, was also observed in patients with PAH. Besides altered gut microbiota, a complication of chronic kidney diseases in PAH further exaggerates the accumulation of TMAO. Renal excretion is the major pathway for the clearance of TMAO. Patients with advanced kidney impairment fail to clear TMAO adequately, resulting in chronic accumulation of TMAO. Consistently, a higher prevalence of chronic kidney diseases was observed in patients with increased ΔTMAO in this study. Notably, chronic kidney disease has been adjusted in the multivariable Cox analysis to reveal the true impact of TMAO level changes on the prognosis of PAH patients. Our results showed that patients with ΔTMAO ⩾1.082 μmol/L still had a higher risk of clinical adverse events after controlling for the effects of chronic kidney disease. In addition, we also performed sensitivity analysis by excluding patients with chronic kidney disease, and the result was consistent with the multivariable Cox result.
The elevated levels of TMAO might further aggravate PH, leading to a vicious cycle. The underlying mechanism is attributable to increased pulmonary perivascular inflammation and proliferation of pulmonary artery smooth muscle cells mediated by TMAO. TMAO can induce macrophage-producing proinflammatory cytokines, contributing to pulmonary vascular remodeling. Previous studies have found that elevated ΔTMAO can contribute to coronary heart diseases, diabetes, heart failure, and atrial fibrillation by impairing cholesterol transport, triggering atherosclerosis, and impairing glucose homeostasis and insulin sensitivity.19–22 However, Videja et al. 23 pointed out the potential therapeutic function of TMAO for its alleviation effects in the monocrotaline-induced PH model. It raised curiosity about the specific effects of TMAO in PH, which promotes further investigation to reveal the real role of the metabolite in disease pathogenesis.
PAH is an important comorbidity of systemic sclerosis. Most recently, the concentrations of TMAO in patients with systemic sclerosis and the potential role of TMAO in this immune-mediated connective tissue disease have been explored. 24 The study concluded that the levels of TMAO were increased in patients with systemic sclerosis, particularly in patients with advanced organ involvement. 24 The axis of TMAO-protein R-like endoplasmic reticulum kinase has been highlighted in the pathogenesis of systemic sclerosis, 25 suggesting the decrease in TMAO levels might represent a novel therapeutic approach in disease management.
This study had a few limitations. The study is based on a single center and the sample size is relatively small, which warrants multicenter studies with larger sample sizes. In addition, the data on diet are not available in this study, precluding evaluating the impact of dietary patterns on the level of TMAO. Gut microbiota are involved in the production of TMAO and we have not included the microbiota assessment in PAH patients which may omit the relevant inner information between gut microbiota and the disease. Regardless, we first demonstrated the association between the dynamic change of TMAO levels and patients with PH, which highlighted the importance of dynamic monitoring TMAO in the clinic practice. In the course of this investigation, a formal sample size calculation was not conducted, and the study encompassed all eligible patients meeting the predefined inclusion criteria, potentially resulting in underpowered analyses and constraining the generalizability of the findings.
Conclusion
Changes in TMAO demonstrated independent associations with the prognostic outcomes among patients diagnosed with PAH, independent of baseline level of TMAO. Changes in TMAO also correlated with alteration in disease severity. Repeated assessment of TMAO level contributes to better identification of patients with elevated risk of adverse clinical events.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231224692 – Supplemental material for The significance of dynamic monitoring plasma TMAO level in pulmonary arterial hypertension – a cohort study
Supplemental material, sj-docx-1-tar-10.1177_17534666231224692 for The significance of dynamic monitoring plasma TMAO level in pulmonary arterial hypertension – a cohort study by Yicheng Yang, Xin Li, Peizhi Wang, Songren Shu, Bingyang Liu, Yanru Liang, Beilan Yang, Zhihui Zhao, Qin Luo, Zhihong Liu, Lemin Zheng, Qixian Zeng and Changming Xiong in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The authors thank Chuangshi Wang (National Clinical Research Center for Cardiovascular Diseases, Fuwai Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China) for her statistical guidance and also thank the support of English Language Editing Service from MASCU (Medical Association with Science.Creativity.Unity), Inc., Shenzhen, China.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
