Abstract
Background:
The recommended delivery mode for bronchodilators in bronchodilator responsiveness (BDR) testing remains controversial.
Objective:
To compare the efficacy of salbutamol administration using a nebulizer
Design:
A retrospective study.
Methods:
This study examined the data of patients with chronic obstructive pulmonary disease who completed BDR testing between 1 December 2021 and 30 June 2022, at Xiangya Hospital, Central South University. After administering 400 μg of salbutamol through an MDI with spacer or 2.5 mg using a nebulizer, the changes in forced expiratory volume in one second (FEV1) and forced vital capacity (FVC) were analyzed in patients with moderate-to-very severe spirometric abnormalities [pre-bronchodilator FEV1 percentage predicted values (FEV1%pred) ⩽59%]. Significant responsiveness was assessed as >12% and >200 mL improvement in FEV1 and/or FVC or >10% increase in FEV1%pred or FVC percentage predicted values (FVC%pred) from pre- to post-bronchodilator administration.
Results:
Of the enrolled 894 patients, 83.2% were male (median age, 63 years). After propensity score matching, 240 pairs of patients were selected. The increment in FEV1 and increased FEV1 relative to the predicted value (ΔFEV1%pred) were significantly higher in patients <65 years and those with severe spirometric abnormalities in the nebulization group than patients in the MDI group (all
Conclusion:
Salbutamol delivered using a nebulizer may be preferable to an MDI with spacer in certain circumstances. Nebulization has the potential to increase responsiveness to salbutamol in BDR testing.
Plain language summary
Bronchodilator responsiveness testing is commonly undertaken as an important part of spirometry testing to determine the degree of volume and airflow improvement after bronchodilator administration. BDR testing results may affect patients’ diagnosis and treatment. This study compared the effects of two delivery models (a metered dose inhaler (MDI) with spacer and nebulization) on responsiveness to bronchodilators and the results of bronchodilator responsiveness testing among patients with chronic obstructive pulmonary disease. We found that the increment in forced expiratory volume in one second were significantly higher in patients aged <65 years and in those with severe spirometric abnormalities in the nebulization group than in those in the MDI group. The study provides evidence that salbutamol delivered by a nebulizer is preferable to an MDI with spacer in patients <65 years and in those with severe spirometric abnormalities and could increase positive responsiveness to bronchodilators. The study will assist in clinical decision-making by selecting the appropriate dosing regimen for different patients.
Keywords
Introduction
Bronchodilator responsiveness (BDR) testing determines the degree of volume and airflow improvement after bronchodilator administration, which is commonly undertaken as an essential part of spirometry testing. 1 BDR testing results may affect patients’ diagnosis and treatment. Administration of four separate doses of 100 μg of short-acting β2-agonist salbutamol using a metered-dose inhaler (MDI) with spacer is recommended by the American Thoracic Society (ATS)–European Respiratory Society (ERS) joint guidelines since 2005 to minimize the variance between different laboratories. 2 The guidelines in 2005 and 2019 outlined another procedure for BDR testing with salbutamol placed in a nebulizer driven by a compressor.1,2 However, there is no consensus on how to choose the modality of administering bronchodilators for different participants in the BDR testing protocol.
For adults using an MDI with spacer, the deposition is expected to be 10–20% of a 100-μg actuation, which is much higher than that without a spacer.3,4 A pressurized MDI and spacer is considered to be at least as good as a nebulizer and is recommended by the guidelines as the preferred delivery method for β2-agonists in children and adults with mild-to-moderate asthma.
5
However, the poor coordination of patients when pressing and breathing and the inability of patients to hold their breath inevitably reduces the deposition of bronchodilators. As previously reported, the choice of delivery modality in BDR testing mainly depends on the severity of baseline pulmonary defects and coordination concerns.
6
Notably, several studies have evaluated the acute bronchodilator effects of β2-agonists delivered by an MDI with spacer compared with the effects of delivery by a nebulizer in patients with chronic obstructive pulmonary disease (COPD) or asthma in emergency department settings;2,7–13 however, studies focused on the effects of the two delivery techniques on BDR testing are rare. This study assessed the effects of salbutamol administered by an MDI with spacer
Materials and methods
Study design and participants
This study retrospectively analyzed patients’ data who completed BDR testing between 1 December 2021 and 30 June 2022, at Xiangya Hospital, Central South University (China), a 3500-bed tertiary-care center. The eligibility criteria for this study were as follows: (1) aged⩾18 years; (2) spirometry-defined COPD with relevant symptoms, and a post-bronchodilator ratio forced expiratory volume in one second and forced vital capacity (FEV1/FVC) < 0.70 14 ; (3) moderate-to-very severe spirometric abnormalities, based on pre-bronchodilator FEV1 percentage predicted values (FEV1%pred) ⩽ 59% on initial spirometry testing.2,15 Patients with upper airway obstruction or a history of pneumonectomy were excluded. 15 Patients with a diagnosis of respiratory tract infection were also excluded. Because of the variances in baseline characteristics among the patients using different modes of salbutamol administration, propensity score (PS) matching was applied to reduce potential bias. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement.
Spirometry
All spirometers (Jaeger Masterscreen, Germany) met the instrument standardization criteria of the ATS/ERS. 2 Fully trained and certified spirometry technicians carried out the testing procedures. The spirometry technique met the standardized ATS/ERS quality criteria.2,15 Using the ATS/ERS criteria for acceptable and repeatable spirometry, the participants performed ⩾3 and ⩽8 forced expiratory maneuvers until the largest and second-largest FEV1 and FVC measurements were within 150 mL of each other. 2 The predicted values of FEV1 and FVC were determined from the statement of the European Committee of Steel and Coal 16 and adjusted for the Chinese population according to the recommendations of Zheng and Zhong.17,18
Before the pulmonary function test, patients were requested to adhere to medication wash-out requirements, including withholding of short- and long-acting β2-agonists (for ⩾8 and ⩾12 h, respectively), short- and long-acting anticholinergic agents (for ⩾6 and ⩾24 h, respectively), short- and long-acting theophylline (for ⩾24 and ⩾48 h, respectively), and anti-leukotrienes (for ⩾48 h). The patients were not permitted to smoke, exercise, or consume tea/coffee for 6 h before spirometry. 18 At the beginning of the test, we will document whether the patient has used a bronchodilator before the test, as well as the name and time of medication used. This information is provided as a reference for the doctor in interpreting the results.
Spirometry was performed before and 15 min after administration of 400 μg of salbutamol (Ventolin, GlaxoSmithKline, Cairo, Egypt) through an MDI with spacer or administration of 2.5 mg of salbutamol solution (Ventolin) in saline (2.5 mL) through a nebulizer driven by a PulmoAide compressor (Omron, Dalian, China), 2 which are routinely performed in our laboratory. Administration of 2.5 mg of salbutamol using a nebulizer is nominally equivalent to 400 μg by an MDI and spacer with a relative potency of 6:1. 8 Salbutamol was administered in the recommended four doses of 100 μg through an MDI with spacer (a valved holding chamber, 750 mL in volume, Volumatic™, GlaxoSmithKline, UK). A single MDI actuation of salbutamol should be slowly inhaled, followed by a breath hold (5s) before exhalation. Patients with severe baseline lung function impairment and poor coordination during spirometry tests were scheduled for nebulization. Nebulization generated a consistent mist of salbutamol diluted in saline for 15 min driven by room air. Air flow to the nebulizer is the range of 6–10 L/min. 1 The mist was inhaled through a mouthpiece during tidal breathing.
Sample size
After PS matching, 240 pairs of patients were selected. In this study, ΔFEV1%pred or ΔFVC%pred after and before bronchodilator administration were two of the major primary outcomes. The null hypothesis stated that ΔFEV1%pred or ΔFVC%pred were identical between the two groups, while the alternative hypothesis was the opposite. We used the PASS 15.0.5 software (NCSS, Kaysville, UT, USA) ‘Means – Paired means – Test (inequality)’ to calculate the sample size. Based on a previous study, 11 we assumed that the mean ± standard deviation (SD) change in ΔFEV1%pred was 18 ± 10 mL for the Nebulizer group and 16 ± 10 mL for the inhaler group; the mean ± SD change in ΔFVC%pred was 17 ± 10 mL for the Nebulizer group and 14 ± 10 mL for the inhaler group, respectively. Then the mean of paired differences (alternative assumption) was 2 mL, and the SD of paired differences was 10 mL. The statistical significance level was set as 0.05, and the statistical power was set as 0.80. For the primary outcome of ΔFEV1%pred, a minimum number of 199 patients per group was required. Likewise, at least 90 patients per group were needed if ΔFVC%pred was the primary outcome. Our current study sample fulfilled the aforementioned assumptions.
Outcomes
The predicted and test values of FEV1 and FVC before and after bronchodilator administration were recorded. The primary outcome was the effect of administering salbutamol using a nebulizer or an MDI with spacer on the following variables: absolute change, percentage change from baseline, and change relative to the predicted value. Changes in FEV1 or FVC after and before bronchodilator administration were expressed as (I) absolute change over baseline (ΔFEV1 or ΔFVC); (II) FEV1 or FVC percentage change over baseline (ΔFEV1% or ΔFVC%); and (III) change relative to the predicted value for FEV1 or FVC (ΔFEV1%pred or ΔFVC%pred). The secondary outcome was to explore whether the two delivery methods could affect the positive rate of BDR testing based on the two different criteria of the ATS/ERS.15,19 An absolute increase of >200 mL and a relative increase of >12% in FEV1 and/or FVC from baseline were classified as a significant BDR response. 15 According to the 2021 updated guidelines, ΔFEV1%pred or ΔFVC%pred > 10% was considered a significant BDR response. 19
The following equations were used to calculate the ΔFEV1%pred and ΔFVC%pred 19 :

Covariates and definitions
The severity of pulmonary function abnormalities, including obstructive, restrictive, and mixed, was based on pre-bronchodilator FEV1%pred. FEV1%pred >70, 60–69, 50–59, 35–49, and <35% represented mild, moderate, moderately severe, severe, and very severe spirometric abnormalities, respectively. 15 Data on demographic characteristics, including sex, age, height, and smoking status, were collected. As the degree of coordination may be related to age, we stratified the patients into aged (⩾65 years) and younger groups (<65 years). We categorized sex by female and male, smoking status by non-smoker and ex-smoker or current-smoker. We also collected clinical data of the patients, namely respiratory symptoms, disease course, and primary diagnosis. There is no missing data in this study.
Statistical analysis
We performed PS matching to reduce potential bias. A logistic regression model was run with 1:1 matching using a caliper width of 0.0007 with the following covariates: age, sex, smoking status, and baseline FEV1%pred.19,20–22 Categorical variables were compared using the Chi-squared test, and continuous variables were compared using
Results
Of the 972 eligible patients in the database, 894 patients (median age, 63 years; 83.2% male) were included in the study. Of these, 578 patients received bronchodilators through an MDI with spacer, while 316 patients received them

Flowchart of patient enrollment.
Characteristics of the patients at baseline.
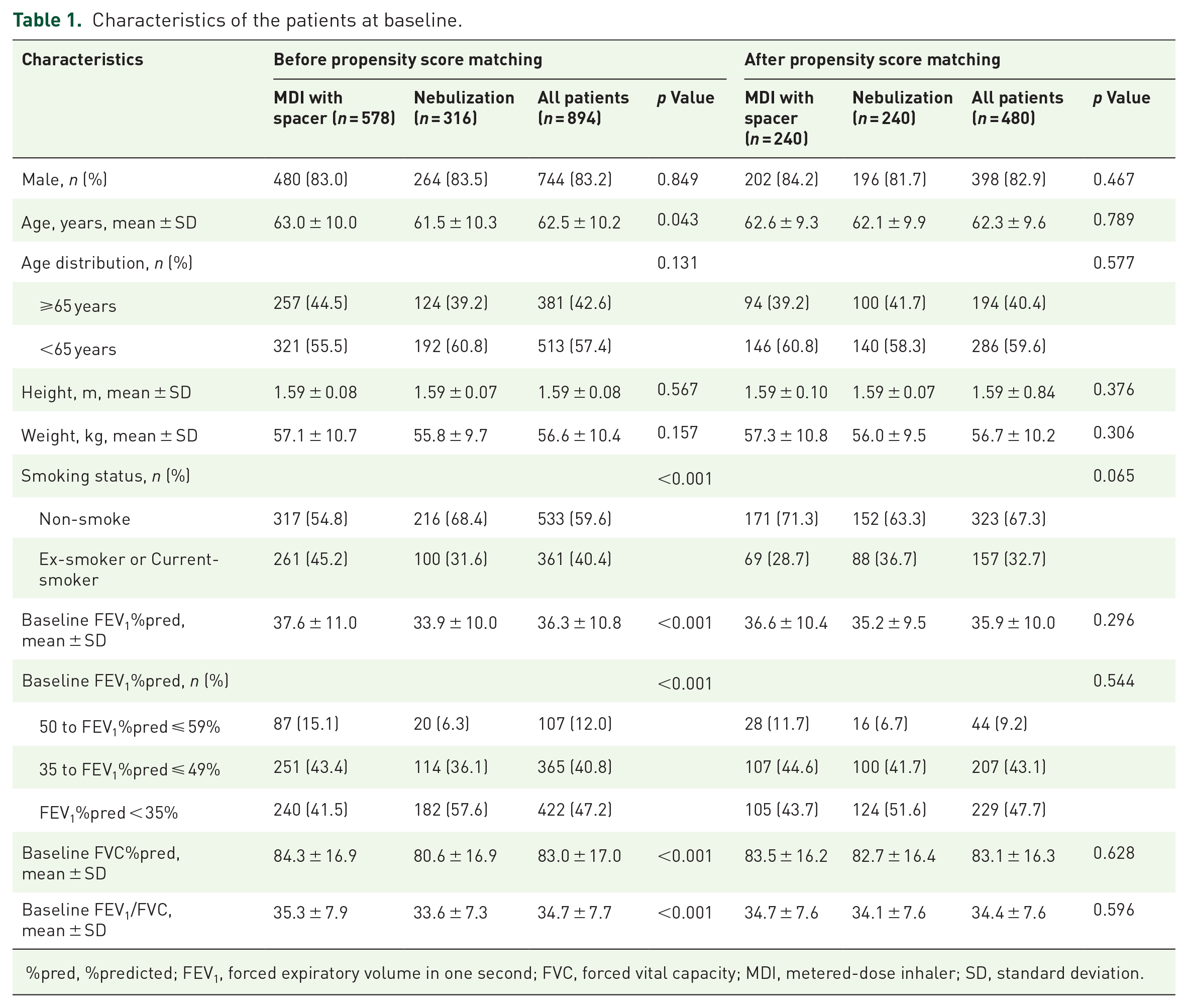
%pred, %predicted; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; MDI, metered-dose inhaler; SD, standard deviation.
In the matched cohorts (Table 2), ΔFEV1 and ΔFVC, ΔFEV1% and ΔFVC%, and ΔFVC%pred were similar between the two delivery techniques. Only ΔFEV1%pred was significantly higher in the nebulization group than in the MDI group. For conciseness, only results with statistical significance in the subgroup variables are shown in Table 2. In the younger group (<65 years), the increment in the FEV1 from pre- to post-bronchodilator administration in the nebulization group was considerably higher than that in the MDI group (0.11
Summary of outcome measures in all patients among the matched cohorts.

An increase in FEV1 and/or FVC > 12% and >200 mL from pre-bronchodilator baseline.
An increase >10% in FEV1 or FVC from pre- bronchodilator baseline relative to the predicted value.
%pred, %predicted; BDR, bronchodilator responsiveness; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; MDI, metered-dose inhaler.
We also analyzed variables associated with changes in FEV1 and FVC (Tables 3 and 4). After controlling for confounders, patients who used nebulization had a 30 mL greater increase in ΔFEV1 and a 1.09% greater increase in ΔFEV1%pred from baseline compared to MDI with spacer (Table 3). There was no significant linear association between the delivery modes and changes in FVC (Table 4).
Multivariate linear regression of the changes in FEV1 from pre- to post-bronchodilator administration in the matched cohorts.
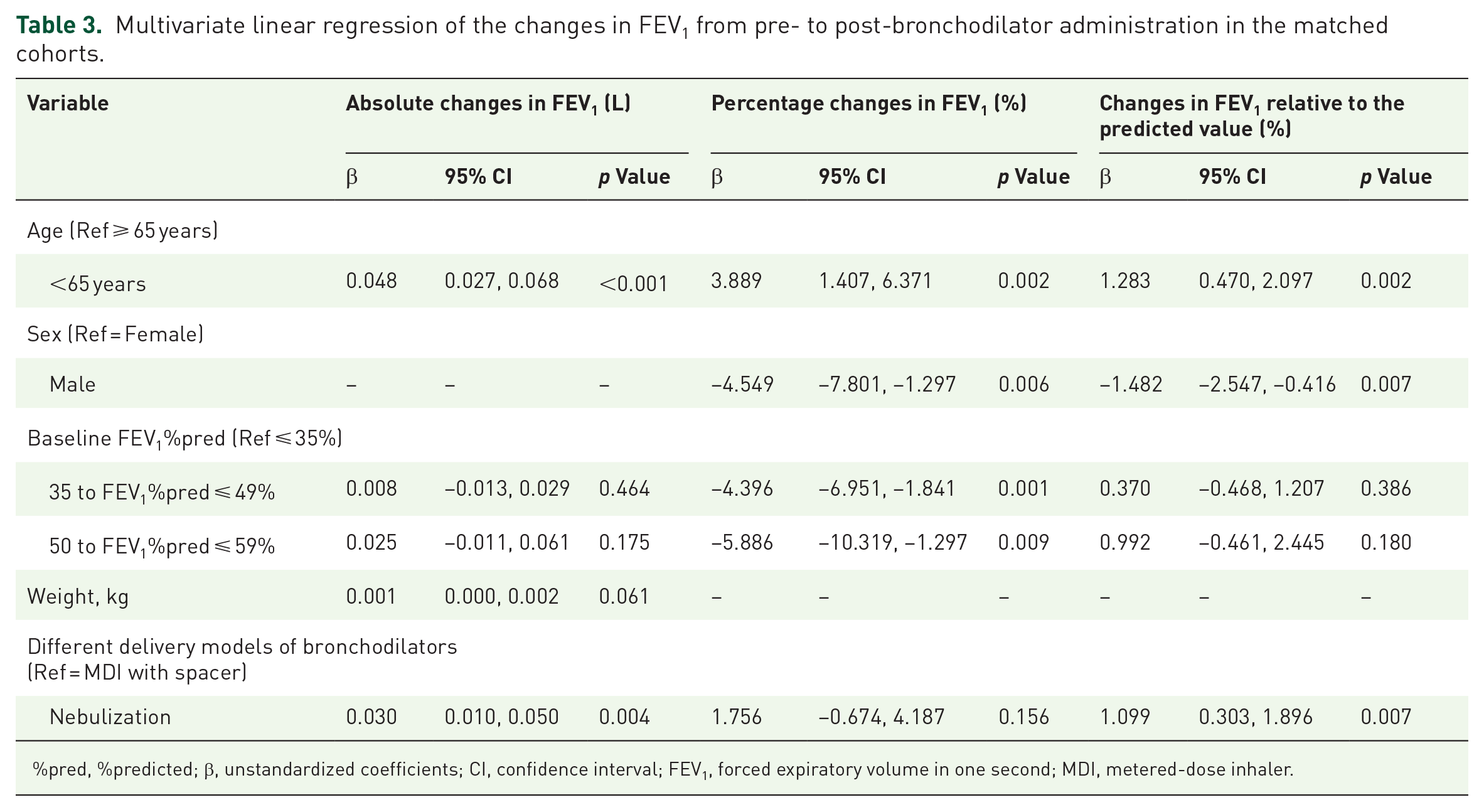
%pred, %predicted; β, unstandardized coefficients; CI, confidence interval; FEV1, forced expiratory volume in one second; MDI, metered-dose inhaler.
Multivariate linear regression of the changes in FVC from pre- to post-bronchodilator administration in the matched cohorts.

%pred, %predicted; β, unstandardized coefficients; CI, confidence interval; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; MDI, metered-dose inhaler.
We further evaluated the significant bronchodilator responsiveness using the two criteria and performed subgroup analysis to find differences in subgroups (Supplemental Table S1). Overall, 37.7% of the patients in our study met one of these criteria. Of the 480 patients, a significant BDR response was diagnosed in 171 (35.6%) according to the absolute and percent improvements in FEV1 or FVC, and 152 patients (31.7%) met the FEV1%pred and FVC%pred criteria. There was a large overlap of individuals diagnosed with significant BDR by the two diagnostic methods in our study (all kappa values ⩾0.75); however, the variance between the two criteria was significant (
As Table 5 shows, the positive rate of BDR in patients aged <65 years was 1.65 times higher than that in the patients aged ⩾65 years based on absolute and percentage change in FEV1 or FVC (OR = 1.65, 95% CI:1.10–2.46). In patients with severe lung function impairment, the significance BDR rate was 0.52 times that in patients with very severe lung function impairment (OR = 0.52, 95% CI: 0.35–0.79). The significant BDR rate by a nebulizer was 1.67 times higher than that by an MDI with spacer (OR = 1.67, 95% CI: 1.13–2.47) in multivariate logistic regression. There was no significant association between available variables and significant BDR rate according to the ΔFEV1%pred or ΔFVC%pred criteria.
Logistic regression for significant bronchodilator responsiveness in the matched cohorts.

An increase in post-bronchodilator FEV1 and/or FVC > 12% and >200 mL from pre-bronchodilator baseline.
An increase >10% in FEV1 or FVC from pre-bronchodilator baseline relative to the predicted value.
%pred, %predicted; CI, credibility interval; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; OR, odds ratios; MDI, metered-dose inhaler.
Discussion
BDR testing was used to assess acute respiratory response to bronchodilators. The results of BDR testing are vital to the diagnosis of respiratory conditions. The ATS/ERS guidelines provide the recommended type and dose of bronchodilators for BDR testing.1,2 Meanwhile, the guidelines also proposed another dosing option.1,2 However, the impact of delivery techniques on BDR testing is yet to be identified. To our knowledge, this is the first study to compare the effects of two delivery models (an MDI with spacer and nebulization) on the results of BDR testing. To determine practical strategies for real-world situations, we stratified the patients with COPD into moderate-to-very severe spirometric abnormality groups according to the baseline FEV1%pred before bronchodilator administration rather than the post-bronchodilator FEV1%pred as per the GOLD guideline.14,15 We found that the absolute increment in FEV1 from the baseline and the increase relative to FEV1%pred were significantly higher in patients <65 years and in those with severe spirometric abnormalities in the nebulization group than in those in the MDI group. Compared with MDI with spacer, patients who used nebulization had a 30 mL greater increase in ΔFEV1 and a 1.09% greater increase in ΔFEV1%pred from baseline. Nebulization improved the significant BDR rate when using absolute and percentage changes in FEV1 or FVC from baseline as the criterion.
After matching the baseline lung function (FEV1%pred) and demographic characteristics of all enrolled patients, our results demonstrated that the overall improvements in FEV1 values were similar between the two groups, in line with the results of a few previous studies.7,8,10 Notably, some previous studies showed that an MDI with spacer was as effective as a nebulizer in improving the FEV1 in patients with acute airflow obstruction conditions, such as asthma and COPD.7,8,10,11,13,23 Consequently, most investigations of the treatment response to various bronchodilators focused on the pediatric population in an emergency situation; however, the current study evaluated the effect of administering salbutamol using different delivery methods on responsiveness to salbutamol in the BDR testing. Furthermore, previous studies did not stratify associated factors for subgroup analysis, such as age, sex, and baseline FEV1 levels. Previous studies also did not evaluate the effect of delivery modes using different variables, such as ΔFEV1% and ΔFEV1%pred, and may have missed some critical findings. In our study, the increase in FEV1 values after stratification analysis was significantly higher in patients aged <65 years and in those with severe spirometric abnormalities in the nebulization group than those in the MDI group, and the difference in the increment in ΔFEV1%pred was statistically significant between the two groups, which has not been previously reported.
A variety of methodologies have been used in previous studies to assess acute bronchodilator responses with the two delivery modes, including different types and doses of bronchodilators.2,7–13 As reported, the efficacy of an MDI and spacer is influenced by the characteristics of the spacer including volume, valve and antistatic features, and the coordination of breath hold after inhalation.3,24,25 Different methodologies and characteristics of the spacer may be resulting in divergence between studies. To date, nebulized β2-agonists are recommended for patients with severe asthma.5,26 In contrast with previous studies; we focused on the discrepancy between different delivery approaches in the BDR testing procedure. Guidelines for selecting an appropriate delivery device for bronchodilators in BDR testing are yet to be specified. Six other laboratory studies reported that the choice between an MDI and spacer and jet nebulizer mainly depended on criteria such as the patient’s age, current mode of therapy, FEV1/FVC ratio at baseline, and clinical condition at the testing time. 6 In our study, patients scheduled for nebulization had more severe pulmonary defects (e.g. lower FEV1%pred and FEV1/FVC, and lower FVC%pred) than those who used an MDI with spacer. Therefore, the choice of delivery mode primarily depended on baseline spirometry values. How to find suitable patients for the different models need further investigation. We showed the demonstrable advantages of a nebulizer over an MDI with spacer; the former led to a significant increase of ΔFEV1%pred. The changes were evident in patients <65 years and those with severe initial spirometric abnormalities. Therefore, in the BDR testing protocol, salbutamol inhalation using a nebulizer may be more suitable for patients aged <65 years and those with baseline FEV1%pred between 35% and 49%.
Does the elevation of FEV1 caused by nebulization affect the results of the BDR testing? Compared with MDI with spacer, nebulization induced a more significant increase in ΔFEV1 and ΔFEV1%pred. When a nebulizer delivered bronchodilators, they had higher odds (1.67 times) of significant BDR rate based on absolute and percentage changes criteria than an MDI with spacer. However, there were no differences between the two delivery models when bronchodilator responsiveness was classified according to the ΔFEV1%pred or ΔFVC%pred >10%. The above results demonstrated that nebulization led to a more marked increase in FEV1 values than an MDI with spacer, which upregulated the positive rate of BDR testing based on absolute and percentage changes criterion. However, the elevation of ΔFEV1%pred by nebulization was inadequate to significantly improve the positive rate using the criterion based on percentage predicted values.
There was a large overlap of individuals diagnosed with significant BDR by the two diagnostic methods;19,27 however, compared with the criterion based on the changes relative to the predicted value, the criterion based on absolute and percentage changes from baseline FEV1 or FVC values detected a large percentage of patients with significant responsiveness to bronchodilators, in line with the results of earlier studies.18,20,22,27 The primary reason for this is that the diagnostic category according to absolute and percentage change over baseline in FEV1 or FVC is dependent on the initial values and exaggerates the bronchodilator response in patients with low baseline values in some extent. In our analysis, the responsiveness to a bronchodilator increased in patients with very severe respiratory impairment based on the absolute and relative changes from baseline compared to the changes relative to the predicted values. Low baseline FEV1 values are associated with large increases in ΔFEV1%, which is more dependent on the initial FEV1 value than ΔFEV1 and ΔFEV1%pred.21,22,27 Therefore, individuals with low initial values tend to satisfy the criterion of a 12% increase from the baseline. In addition, compared with the absolute and percentage thresholds, the percentage predicted threshold prevented misinterpretation due to the magnitude of the baseline lung function in assessing bronchodilator responsiveness, as recommended in the guidelines. 19 Although we collected clinical symptoms of the patients and excluded patients with a diagnosis of respiratory tract infection, it was difficult to distinguish whether the patients with COPD were at their stable or exacerbation stage based on symptoms because of the retrospective study design. Therefore, the proportion of patients with significant BDR response in this study was higher than in previous studies focusing on stable COPD patients in China, 18 which demonstrated that 21.4% of patients were diagnosed using the absolute and percent improvements criteria in FEV1 or FVC, and 13.5% of patients were diagnosed as positive using the FEV1%pred and FVC%pred criteria.
Our study has several limitations. First, it is a retrospective study. Although we performed PS matching analysis to minimize bias, half of the sample was eliminated to adjust for confounders. The results of this study should be validated by a randomized crossover design study. In addition, we only included patients with moderate-to-very severe lung function impairment based on FEV1%pred. 15 Patients with obstructive lung function impairments, such as COPD, have lower aerosol delivery than healthy subjects. 28 Patients with mild spirometric defects may not need nebulized bronchodilators for maximal bronchodilation. Besides, due to the retrospective design of this study, the included patients with COPD were not differentiated between stable and exacerbation status. This lack of distinction may result in an overestimation of the bronchodilator response rate. However, from another perspective, not distinguishing the stages of COPD patients may make the findings of this study more applicable to real-world situations. Furthermore, we did not include patients with other airway limitations, such as asthma, because different underlying airway diseases influence the response to bronchodilators in the BDR testing. Therefore, our study findings should be verified for other obstructive airway diseases.
Conclusion
In conclusion, salbutamol delivered using a nebulizer may be preferable to salbutamol delivered by an MDI with spacer in some circumstances and could increase responsiveness to bronchodilators. Our results can provide valuable insights for clinical decision-making when determining the appropriate dosing regimen (400 μg by an MDI with spacer or 2.5 mg
Supplemental Material
sj-docx-1-tar-10.1177_17534666231214134 – Supplemental material for Nebulization versus metered-dose inhaler and spacer in bronchodilator responsiveness testing: a retrospective study
Supplemental material, sj-docx-1-tar-10.1177_17534666231214134 for Nebulization versus metered-dose inhaler and spacer in bronchodilator responsiveness testing: a retrospective study by Rongli Lu, Ying Li, Chengping Hu, Pinhua Pan, Qiaohong Zhao and Ruoxi He in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666231214134 – Supplemental material for Nebulization versus metered-dose inhaler and spacer in bronchodilator responsiveness testing: a retrospective study
Supplemental material, sj-docx-2-tar-10.1177_17534666231214134 for Nebulization versus metered-dose inhaler and spacer in bronchodilator responsiveness testing: a retrospective study by Rongli Lu, Ying Li, Chengping Hu, Pinhua Pan, Qiaohong Zhao and Ruoxi He in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
