Abstract
Background:
In the phase III, 52-week ETHOS study in patients with moderate to very severe chronic obstructive pulmonary disease (COPD), triple therapy with budesonide/glycopyrrolate/formoterol fumarate (BGF), at two inhaled corticosteroid dose levels, resulted in significantly lower moderate/severe exacerbation rates
Methods:
ETHOS (NCT02465567) was a randomized, double-blind, multi-center, parallel-group study in patients with moderate to very severe COPD who had experienced ⩾1 moderate/severe exacerbation in the previous year. Patients received BGF 320/18/9.6 µg, BGF 160/18/9.6 μg, GFF 18/9.6 µg, or BFF 320/9.6 µg twice daily
Results:
The PFT modified intent-to-treat population included 3088 patients (mean age 64.4 years; mean reversibility post-albuterol 16.7%; mean post-albuterol FEV1% predicted 42.8). BGF 320/18/9.6 µg and 160/18/9.6 µg significantly improved morning pre-dose trough FEV1 at week 24
Conclusions:
BGF 320/18/9.6 µg and 160/18/9.6 µg significantly improved trough FEV1
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is characterized by airflow limitation and is ranked as the third leading cause of mortality worldwide.1–3 In 2017, COPD had a global prevalence of approximately 300 million cases, 4 was associated with approximately 3.2 million deaths, 5 and was ranked as the seventh leading cause of disability worldwide. 5
The use of inhaled corticosteroid/long-acting muscarinic antagonist/long-acting β2-agonist (ICS/LAMA/LABA) triple therapy is recommended for patients with COPD who experience persistent exacerbations, defined as an acute worsening of respiratory symptoms resulting in the need for additional therapy, or symptoms despite the use of dual LAMA/LABA, or ICS/LABA inhaled therapies. 3 In such patients, triple therapies have been shown to improve lung function and symptoms and reduce the frequency of COPD exacerbations relative to dual combination therapies. 3
In the ETHOS study (NCT02465567), the efficacy and safety of the ICS/LAMA/LABA triple fixed-dose combination therapy budesonide/glycopyrrolate/formoterol fumarate (BGF), at two ICS dose levels, delivered twice daily
A previous study of BGF, KRONOS, assessed symptomatic patients with moderate to very severe COPD without a requirement for a history of exacerbations (NCT02497001); primary endpoints were change from baseline in morning pre-dose trough forced expiratory volume in one second (FEV1)
Here, we report data from the ETHOS pulmonary function test (PFT) sub-study, which assessed the effect of BGF relative to GFF and BFF on lung function, including the effect on the rate of decline, in a subset of patients in the ETHOS study, throughout the 52-week treatment period. In addition, we performed subgroup analyses based on FEV1 severity and blood eosinophil count at baseline.
Methods
Study design
Details of the primary ETHOS study design have been published.10,11 Briefly, ETHOS was a 52-week, randomized, double-blind, parallel-group trial conducted across 26 countries. Patients received twice daily dosing with BGF 320/18/9.6
Eligible patients for the ETHOS study were 40–80 years of age, with symptomatic COPD (COPD assessment test score ⩾10 at screening despite receiving ⩾2 inhaled maintenance therapies), had a post-bronchodilator FEV1 25–65% of predicted normal, and had a smoking history of ⩾10 pack-years. If post-bronchodilator FEV1 was <50% of predicted normal, patients required a history of ⩾1 moderate or severe COPD exacerbation in the previous year, and if post-bronchodilator FEV1 was ⩾50% of predicted normal, a history of ⩾2 moderate or ⩾1 severe COPD exacerbations was required. Patients with a current diagnosis of asthma, respiratory disease other than COPD, or other significant uncontrolled diseases (including cardiac disease and cancer) were excluded.10,11
A subset of study sites was designated for participation in the PFT sub-study, which was conducted concurrently with the full study. For inclusion in the PFT sub-study, the average of the 60- and 30-minute pre-dose FEV1 assessments was required to be <65% predicted normal value at visit 4. In addition, patients were excluded from the PFT sub-study if they failed to meet American Thoracic Society/European Respiratory Society spirometry criteria for acceptability and repeatability. 12 In order to ensure that baseline FEV1 values were stable and reflective of their true COPD severity during the screening period but prior to randomization, patients who did not meet FEV1 baseline stability criteria were also excluded; this was defined as the average of the 60- and 30-minute pre-dose FEV1 assessments at the randomization visit being within ±20% or 200 mL of the mean pre-dose FEV1 obtained at the two previous visits.
Lung function endpoints and assessments
Primary endpoints of the PFT sub-study included change from baseline in morning pre-dose trough FEV1 at week 24 and over 24 weeks for BGF
During the PFT sub-study, spirometry assessments were obtained at day 1 and weeks 4, 12, 24, 36, and 52. At these visits, spirometry assessments were conducted at 60 minutes and 30 minutes pre-dose and 5 (day 1 only), 15, and 30 minutes, and 1, 2, and 4 hours post-dose.
The FEV1 AUC0–4 was calculated using the trapezoidal rule, after first having subtracted the baseline FEV1 value, and the AUC was transformed into a weighted average by dividing by the time in hours from dosing to the last measurement included (typically 4 hours).
Statistical analyses
The PFT sub-study population was a subset of the patients in the modified intent-to-treat (mITT) population of the ETHOS study. The overall mITT population included all patients who were randomly assigned and treated and had post-randomization data obtained before discontinuation of treatment. The change from baseline in morning pre-dose trough FEV1 and differences between treatment groups in FEV1 AUC0–4 were analyzed using a repeated measures linear mixed model. The model included baseline FEV1, log baseline blood eosinophil count, and percentage reversibility to bronchodilator as continuous covariates, and visit, treatment, treatment by visit interaction, and ICS use at baseline as categorical covariates. Other endpoints were analyzed using a similar repeated measures linear mixed model such as morning pre-dose trough FEV1. The rates of decline in morning pre-dose trough FEV1 and FEV1 AUC0–4 were analyzed with a linear mixed model with random patient slopes of FEV1
The primary endpoints were part of a type I error control procedure used for reporting the full ETHOS study. Other endpoints were not multiplicity controlled and were reported in terms of unadjusted
In order to examine further the potential impact of lung function severity and eosinophil counts on lung function decline over one year, we performed an exploratory analysis of lung function decline in a pooled cohort of ICS-containing therapies (BGF 320/18/9.6 µg, BGF 160/18/9.6 μg, and BFF 320/9.6 µg)
Results
Study population
A total of 3088 patients were included in the 4-hour PFT sub-study (36.3% of the total ETHOS mITT population; mean age 64.4 years; mean reversibility post-albuterol 16.7%; mean post-albuterol FEV1% predicted, 42.8%; Table 1). Patient demographics were generally similar to those of the overall ETHOS mITT population. 10
Demographics and baseline characteristics (PFT sub-study mITT population).

BGF 320/18/9.6 µg,
BGF 160/18/9.6 µg,
Number of pack-years smoked = (number of cigarettes per day/20) × number of years smoked.
BGF 320/18/9.6 µg,
BFF, budesonide/formoterol fumarate; BGF, budesonide/glycopyrrolate/formoterol fumarate; CAT, COPD Assessment Test; COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 second; GFF, glycopyrrolate/formoterol fumarate; ICS, inhaled corticosteroid; mITT, modified intent-to-treat; PFT, pulmonary function test; SD, standard deviation.
Morning pre-dose trough FEV1
BGF 320/18/9.6 μg treatment resulted in significant improvements in least squares (LS) mean change from baseline morning pre-dose trough FEV1 at week 24 compared with GFF (35 mL;
Change from baseline a in morning pre-dose trough FEV1 and FEV1 AUC0–4 (efficacy estimand; PFT sub-study mITT population b ).
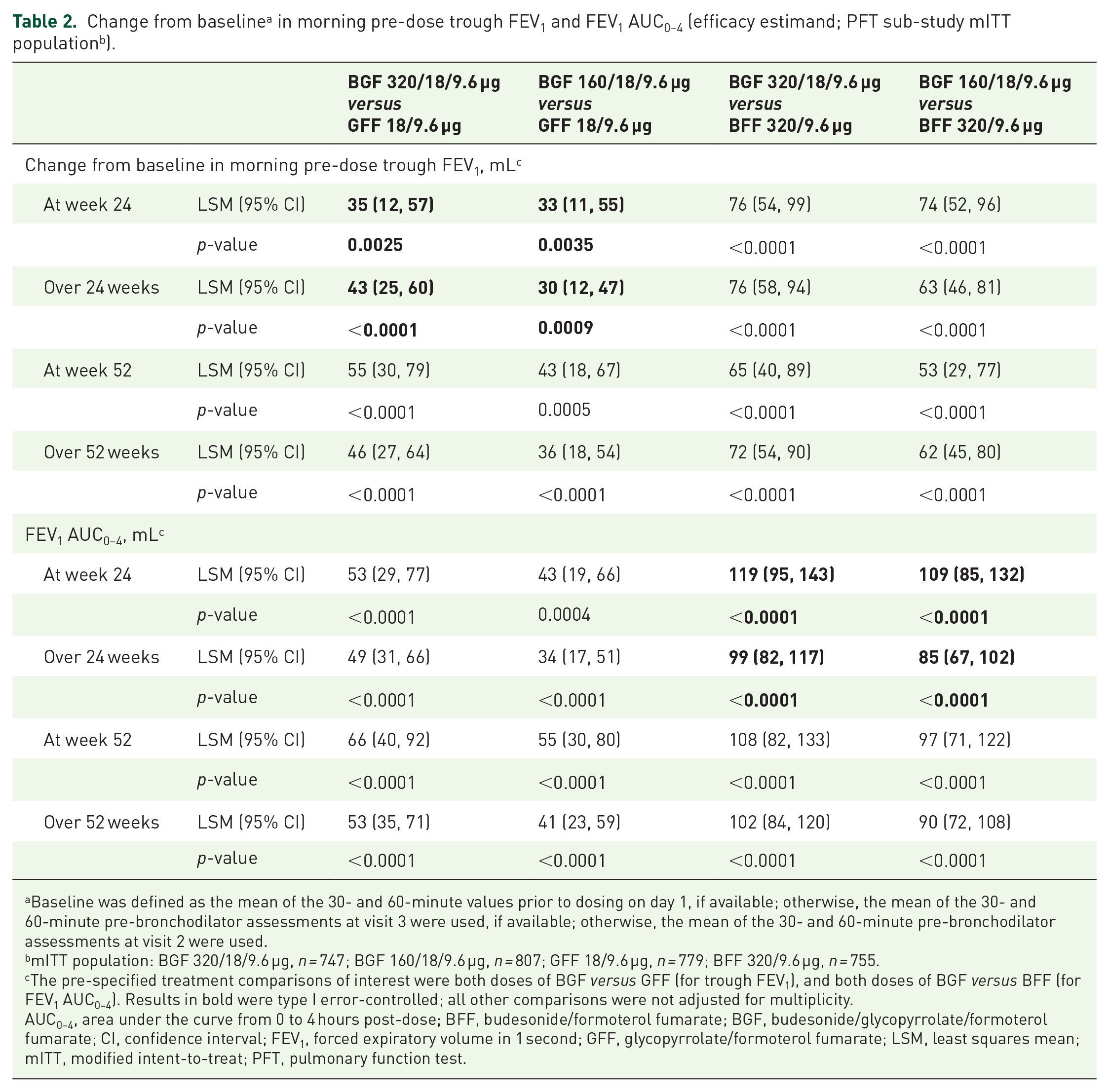
Baseline was defined as the mean of the 30- and 60-minute values prior to dosing on day 1, if available; otherwise, the mean of the 30- and 60-minute pre-bronchodilator assessments at visit 3 were used, if available; otherwise, the mean of the 30- and 60-minute pre-bronchodilator assessments at visit 2 were used.
mITT population: BGF 320/18/9.6 μg,
The pre-specified treatment comparisons of interest were both doses of BGF
AUC0–4, area under the curve from 0 to 4 hours post-dose; BFF, budesonide/formoterol fumarate; BGF, budesonide/glycopyrrolate/formoterol fumarate; CI, confidence interval; FEV1, forced expiratory volume in 1 second; GFF, glycopyrrolate/formoterol fumarate; LSM, least squares mean; mITT, modified intent-to-treat; PFT, pulmonary function test.
Significant improvements in morning pre-dose trough FEV1 were maintained at week 52 for BGF 320/18/9.6 µg and BGF 160/18/9.6 µg

Change from baseline in morning pre-dose trough FEV1 over study duration (efficacy estimand; PFT mITT sub-study population).
FEV1 AUC0–4
BGF 320/18/9.6 μg treatment resulted in significant improvements in LS mean FEV1 AUC0–4 at week 24 compared with BFF (119 mL;

FEV1 AUC0–4 over study duration (efficacy estimand; PFT mITT sub-study population).
Significant improvements in FEV1 AUC0–4 were maintained at week 52 for BGF 320/18/9.6 μg and BGF 160/18/9.6 μg
Analyses by FEV1% predicted at baseline (<50% versus ⩾50%)
The effects of BGF
Change from baseline in morning pre-dose trough FEV1 and FEV1 AUC0–4 at week 24 by post-bronchodilator FEV1% predicted (efficacy estimand; PFT sub-study mITT population).

AUC0–4, area under the curve from 0 to 4 hours post-dose; BFF, budesonide/formoterol fumarate; BGF, budesonide/glycopyrrolate/formoterol fumarate; CI, confidence interval; FEV1, forced expiratory volume in 1 second; GFF, glycopyrrolate/formoterol fumarate; LSM, least squares mean; mITT, modified intent-to-treat; PFT, pulmonary function test.
Analyses by baseline blood eosinophil count (<150 cells/mm3 versus ⩾150 cells/mm3)
Similarly, the effects of BGF
Change from baseline in morning pre-dose trough FEV1 and FEV1 AUC0–4 at week 24 by baseline blood eosinophil count (efficacy estimand; PFT sub-study mITT population).
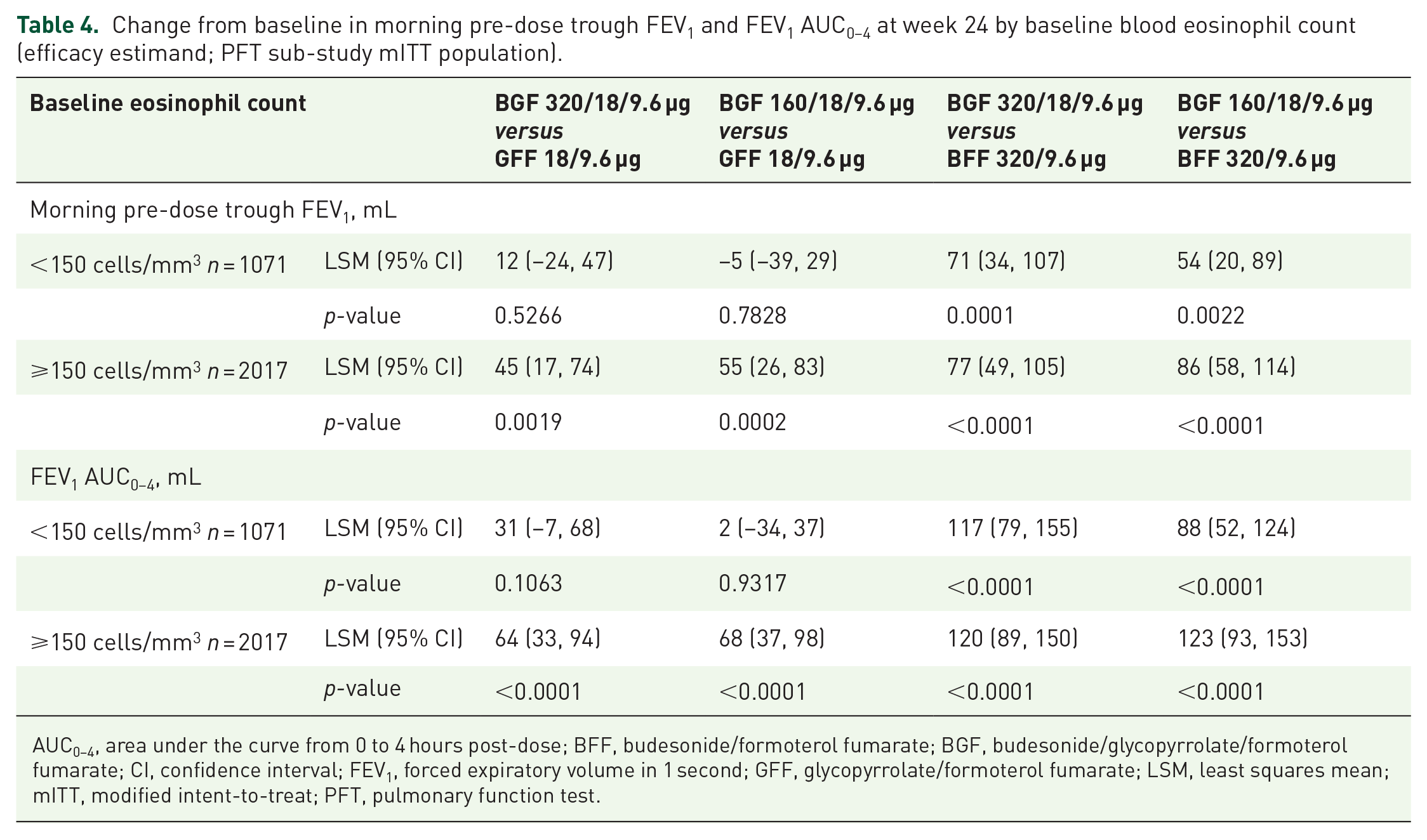
AUC0–4, area under the curve from 0 to 4 hours post-dose; BFF, budesonide/formoterol fumarate; BGF, budesonide/glycopyrrolate/formoterol fumarate; CI, confidence interval; FEV1, forced expiratory volume in 1 second; GFF, glycopyrrolate/formoterol fumarate; LSM, least squares mean; mITT, modified intent-to-treat; PFT, pulmonary function test.
The change from baseline in morning pre-dose trough FEV1 and FEV1 AUC0–4 at week 24 for both doses of BGF
Onset of action
Improvements in FEV1 were achieved rapidly with post-dose changes from baseline in FEV1 being >100 mL for all four treatment groups at the 5-minute post-dose measurement (Figure 3).
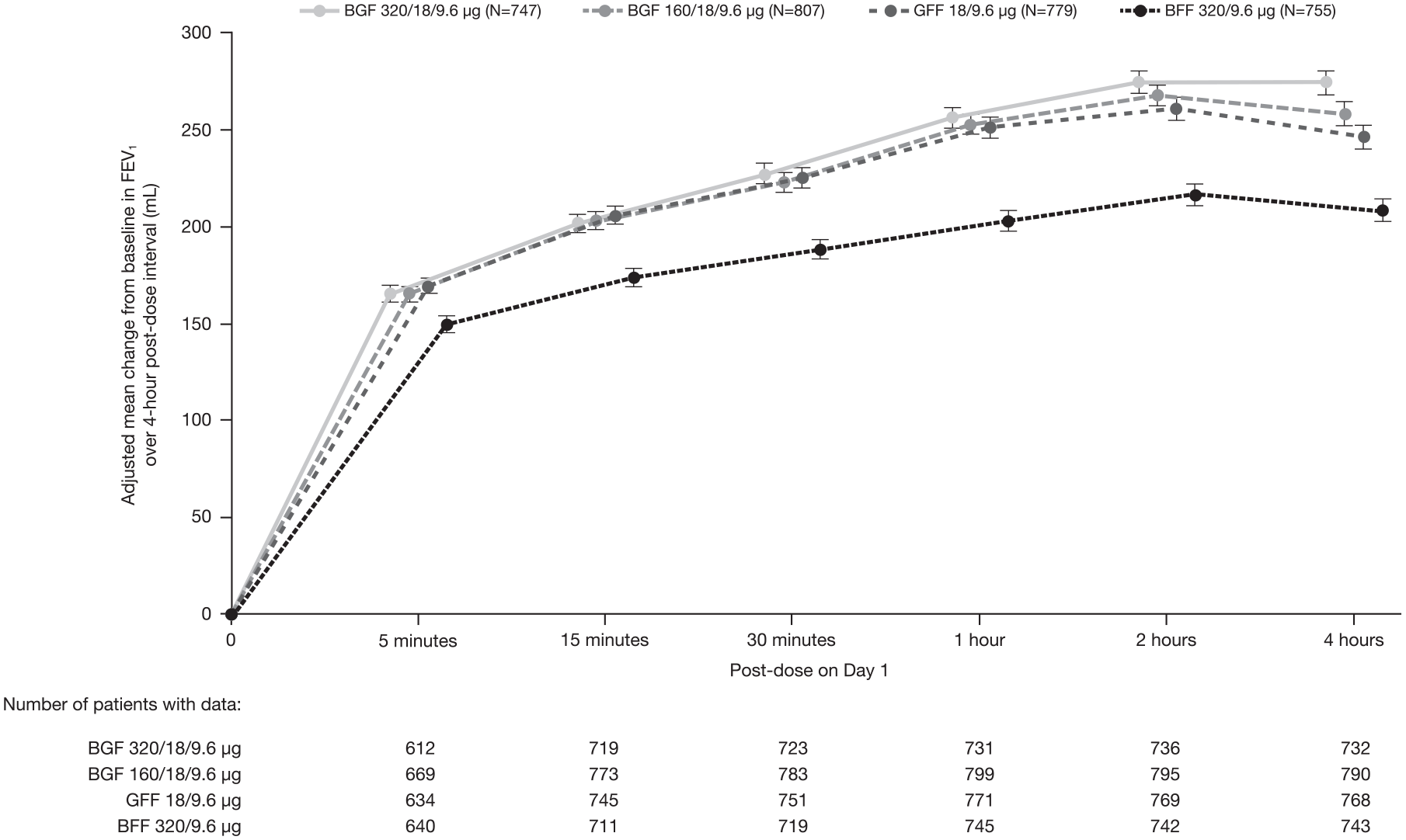
Change from baseline in FEV1 over 4-hour post-dose interval at day 1 (efficacy estimand; PFT mITT sub-study population).
Analyses of ICS-containing therapies versus GFF on lung function decline
A trend for a lower rate of decline in morning pre-dose trough FEV1 over 52 weeks was observed in the BGF treatment groups relative to GFF (Supplemental Table 2). However, no consistent effects were observed for a lower rate of decline in FEV1 AUC0–4 for BGF relative to BFF (Supplemental Table 2).
Patients treated with ICS-containing therapies were pooled to assess whether benefits
Adjusted rate of decline in morning pre-dose trough FEV1 and FEV1 AUC0–4 over 52 weeks by ICS-containing therapy (efficacy estimand; PFT sub-study mITT population).
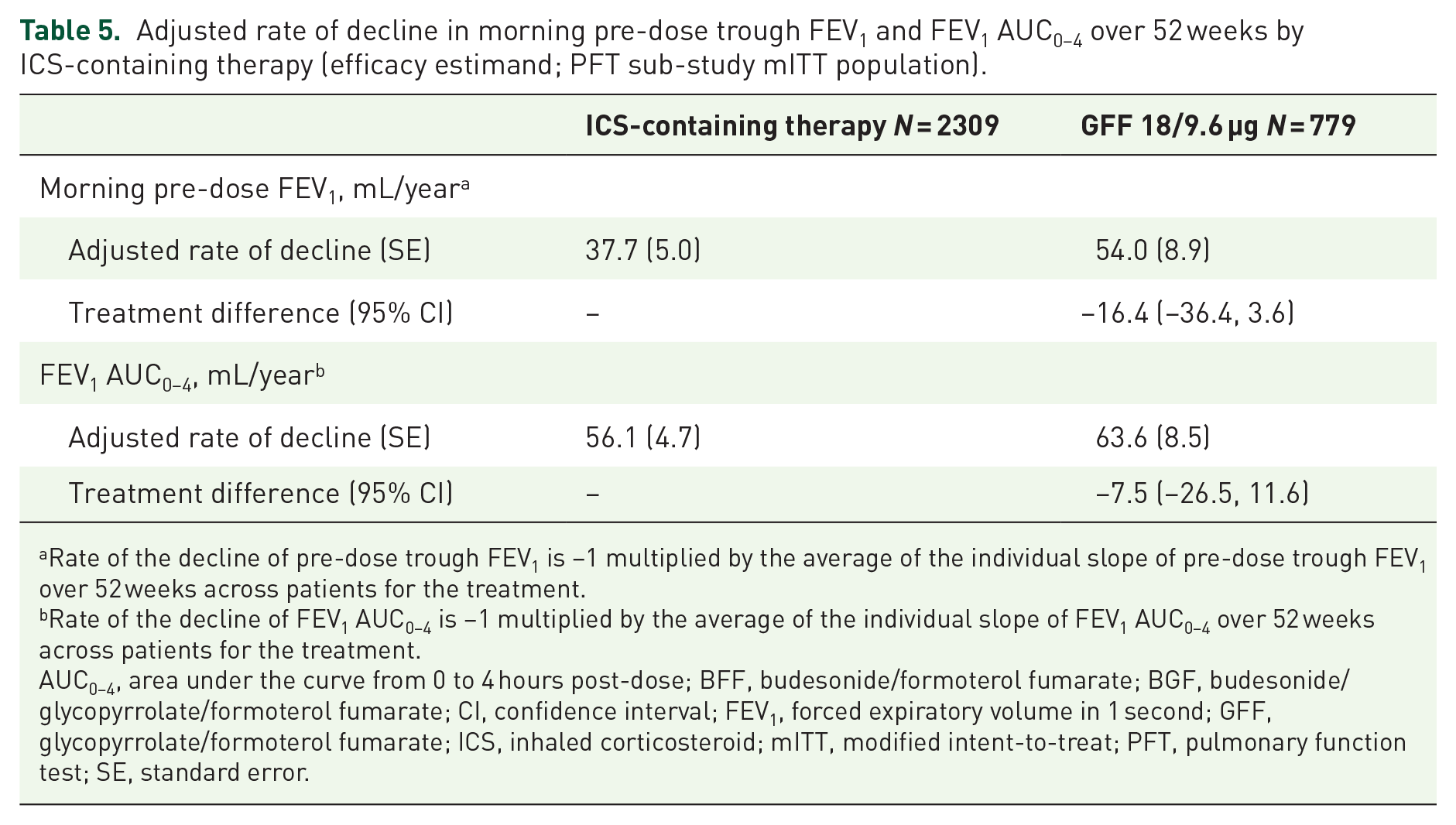
Rate of the decline of pre-dose trough FEV1 is –1 multiplied by the average of the individual slope of pre-dose trough FEV1 over 52 weeks across patients for the treatment.
Rate of the decline of FEV1 AUC0–4 is –1 multiplied by the average of the individual slope of FEV1 AUC0–4 over 52 weeks across patients for the treatment.
AUC0–4, area under the curve from 0 to 4 hours post-dose; BFF, budesonide/formoterol fumarate; BGF, budesonide/glycopyrrolate/formoterol fumarate; CI, confidence interval; FEV1, forced expiratory volume in 1 second; GFF, glycopyrrolate/formoterol fumarate; ICS, inhaled corticosteroid; mITT, modified intent-to-treat; PFT, pulmonary function test; SE, standard error.

Adjusted rate of decline in pre-dose trough FEV1 (a) and FEV1 AUC0–4 (b) over 52 weeks.
Discussion
The findings of this 4-hour PFT sub-study of ETHOS demonstrated the benefit of BGF
The benefits of BGF 320/18/9.6 μg on lung function at week 24 observed in ETHOS are consistent with data from the KRONOS study, in which BGF demonstrated significant improvements in lung function relative to ICS/LABA dual therapy.
9
As in ETHOS, a statistically significant improvement in FEV1 AUC0–4 at week 24 was also observed for BGF 320/18/9.6 μg compared with BFF in KRONOS. However, unlike ETHOS, the improvement in change from baseline in morning pre-dose trough FEV1 at week 24 compared with GFF in KRONOS was numerical, but did not achieve statistical significance.
9
Nonetheless, it is important to note that analyses at week 24 with the attributable estimand in KRONOS, which factored in missing data, did demonstrate a significant difference between BGF 320/18/9.6 μg and GFF for trough FEV1 (24 mL; unadjusted
As reported previously, lung function improvements with an ICS are known to be associated with blood eosinophil counts.15–20 In this regard, the percentage of patients with baseline blood eosinophil levels <150 cells/mm3 was 48.2% and 40.0% in the overall populations for KRONOS and ETHOS, respectively. The percentage of patients with blood eosinophil levels <150 cells/mm3 was considerably higher in KRONOS compared with ETHOS, which may explain the lower magnitude of benefit observed in pre-dose trough FEV1 for BGF relative to GFF at week 24 in KRONOS. Nonetheless, the improvements observed in morning pre-dose trough FEV1 at week 24 in ETHOS for BGF relative to GFF (35 mL) were consistent with values observed for the ICS component for this endpoint in other triple fixed-dose combinations at week 24 or week 26 (20 mL to 81 mL).15,18,21
Although the current study was not designed to evaluate lung function decline, a trend for a lower rate of decline in morning pre-dose trough FEV1 over 52 weeks was observed for both doses of BGF relative to GFF. No consistent effects were observed for rate of decline in FEV1 AUC0–4 over 52 weeks for BGF relative to BFF. However, exploratory analyses pooling ICS-containing therapies were also conducted to evaluate the effects of blood eosinophils and lung function severity on lung function decline, as these factors are known to modulate the lung function benefits of ICS in COPD. Results using the pooled data suggested that greater reductions in the annual rate of lung function decline relative to the LAMA/LABA occurred in patients with moderate airflow obstruction and those with a baseline blood eosinophil count ⩾100 cells/mm3. Although speculative, these findings suggest that there may be value in initiating ICS therapy to prevent lung function decline in COPD patients with eosinophil counts ⩾100 cells/mm3 and less severe lung function impairment, rather than waiting until a marked loss of lung function has already occurred. Clearly, prospective studies aimed at reducing the rate of lung function decline earlier in the COPD disease process are needed.
In conclusion, both BGF 320/18/9.6 μg and BGF 160/18/9.6 μg provided significant improvements in lung function at week 24
Supplemental Material
sj-pdf-1-tar-10.1177_17534666211034329 – Supplemental material for Improvements in lung function with budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler versus dual therapies in patients with COPD: a sub-study of the ETHOS trial
Supplemental material, sj-pdf-1-tar-10.1177_17534666211034329 for Improvements in lung function with budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler versus dual therapies in patients with COPD: a sub-study of the ETHOS trial by Klaus F. Rabe, Fernando J. Martinez, Dave Singh, Roopa Trivedi, Martin Jenkins, Patrick Darken, Magnus Aurivillius and Paul Dorinsky in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-2-tar-10.1177_17534666211034329 – Supplemental material for Improvements in lung function with budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler versus dual therapies in patients with COPD: a sub-study of the ETHOS trial
Supplemental material, sj-pdf-2-tar-10.1177_17534666211034329 for Improvements in lung function with budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler versus dual therapies in patients with COPD: a sub-study of the ETHOS trial by Klaus F. Rabe, Fernando J. Martinez, Dave Singh, Roopa Trivedi, Martin Jenkins, Patrick Darken, Magnus Aurivillius and Paul Dorinsky in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-3-tar-10.1177_17534666211034329 – Supplemental material for Improvements in lung function with budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler versus dual therapies in patients with COPD: a sub-study of the ETHOS trial
Supplemental material, sj-pdf-3-tar-10.1177_17534666211034329 for Improvements in lung function with budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler versus dual therapies in patients with COPD: a sub-study of the ETHOS trial by Klaus F. Rabe, Fernando J. Martinez, Dave Singh, Roopa Trivedi, Martin Jenkins, Patrick Darken, Magnus Aurivillius and Paul Dorinsky in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-4-tar-10.1177_17534666211034329 – Supplemental material for Improvements in lung function with budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler versus dual therapies in patients with COPD: a sub-study of the ETHOS trial
Supplemental material, sj-pdf-4-tar-10.1177_17534666211034329 for Improvements in lung function with budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler versus dual therapies in patients with COPD: a sub-study of the ETHOS trial by Klaus F. Rabe, Fernando J. Martinez, Dave Singh, Roopa Trivedi, Martin Jenkins, Patrick Darken, Magnus Aurivillius and Paul Dorinsky in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-5-tar-10.1177_17534666211034329 – Supplemental material for Improvements in lung function with budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler versus dual therapies in patients with COPD: a sub-study of the ETHOS trial
Supplemental material, sj-pdf-5-tar-10.1177_17534666211034329 for Improvements in lung function with budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler versus dual therapies in patients with COPD: a sub-study of the ETHOS trial by Klaus F. Rabe, Fernando J. Martinez, Dave Singh, Roopa Trivedi, Martin Jenkins, Patrick Darken, Magnus Aurivillius and Paul Dorinsky in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-6-tar-10.1177_17534666211034329 – Supplemental material for Improvements in lung function with budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler versus dual therapies in patients with COPD: a sub-study of the ETHOS trial
Supplemental material, sj-pdf-6-tar-10.1177_17534666211034329 for Improvements in lung function with budesonide/glycopyrrolate/formoterol fumarate metered dose inhaler versus dual therapies in patients with COPD: a sub-study of the ETHOS trial by Klaus F. Rabe, Fernando J. Martinez, Dave Singh, Roopa Trivedi, Martin Jenkins, Patrick Darken, Magnus Aurivillius and Paul Dorinsky in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The authors would like to thank all the patients, their families, and the investigators, research nurses, and operations staff involved in ETHOS. The authors also thank Julie McLaren, Earl St Rose, Shaila Ballal, and Colin Reisner, who were employees of AstraZeneca when this study was conducted, and held stock options in the company, for their valuable contribution to the study. Medical writing support, under the direction of the authors, was provided by Jake Casson, CMC Connect, McCann Health Medical Communications, and was funded by AstraZeneca in accordance with Good Publication Practice (GPP3) guidelines. 22
Dave Singh is supported by the National Institute for Health Research (NIHR) Manchester Biomedical Research Centre (BRC)
Author contributions
The authors meet the criteria for authorship as recommended by the International Committee of Medical Journal Editors, take responsibility for the integrity of the work as a whole, contributed to the writing and reviewing of the manuscript, and have given final approval for the version to be published. All authors had full access to the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Klaus F Rabe, Fernando J Martinez, Dave Singh, Roopa Trivedi, and Magnus Aurivillius: Acquisition of data; data interpretation.
Martin Jenkins, Patrick Darken, and Paul Dorinsky: Conception/design; data analysis/interpretation.
Conflict of interest statement
Klaus F Rabe reports personal fees from AstraZeneca, Berlin-Chemie, Boehringer Ingelheim, Chiesi Pharmaceuticals, InterMune, Novartis, Sanofi, and Teva; and grants from the Ministry of Education and Science, Germany, outside the submitted work.
Fernando J Martinez reports grants from AstraZeneca during the conduct of the study; personal fees and non-financial support from the American College of Chest Physicians, AstraZeneca, Boehringer Ingelheim, Chiesi Pharmaceuticals, Concert, Continuing Education, Genentech, GlaxoSmithKline, Inova Fairfax Health System, Miller Communications, the National Association for Continuing Education, Novartis, PeerView Communications, Prime Communications, the Puerto Rican Respiratory Society, Roche, Sunovion, and Theravance; non-financial support from ProterixBio; and personal fees from the American Thoracic Society, Columbia University, Haymarket Communications, Integritas, inThought Research, MD Magazine, Methodist Hospital Brooklyn, New York University, Unity, Up-To-Date, WebMD/MedScape, and Western Connecticut Health Network; and grants from the National Institutes of Health, outside the submitted work.
Dave Singh reports personal fees from Apellis, Cipla, Genentech, Peptinnovate, and Skyepharma; and grants and personal fees from AstraZeneca, Boehringer Ingelheim, Chiesi Pharmaceuticals, GlaxoSmithKline, Glenmark, Menarini, Merck, Mundipharma, Novartis, Pfizer, Pulmatrix, Teva, Theravance, and Verona, outside the submitted work.
Roopa Trivedi, Martin Jenkins, Patrick Darken, Magnus Aurivillius, and Paul Dorinsky are employees of AstraZeneca and hold stock and/or stock options in the company.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by AstraZeneca. The funder of the study had a role in study design, data collection, data analysis, data interpretation, and writing of the report. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and the accuracy of the data analysis.
Data availability
Supplemental material
The reviews of this paper are available via the supplemental material section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
