Abstract
Background:
The relationships between spirometric assessment of lung function and symptoms (including exacerbations) in patients with asthma and/or chronic obstructive pulmonary disease (COPD) in a real-life setting are uncertain.
Objectives:
To assess the relationships between baseline post-bronchodilator (post-BD) spirometry measures of lung function and symptoms and exacerbations in patients with a physician-assigned diagnosis of asthma and/or COPD.
Design:
The NOVEL observational longiTudinal studY (NOVELTY) is a global, prospective, 3-year observational study.
Methods:
Logistic regression analysis was used to evaluate relationships. Spirometry measures were assessed as percent predicted (%pred). Symptoms were assessed at baseline, and exacerbations were assessed at baseline and Year 1.
Results:
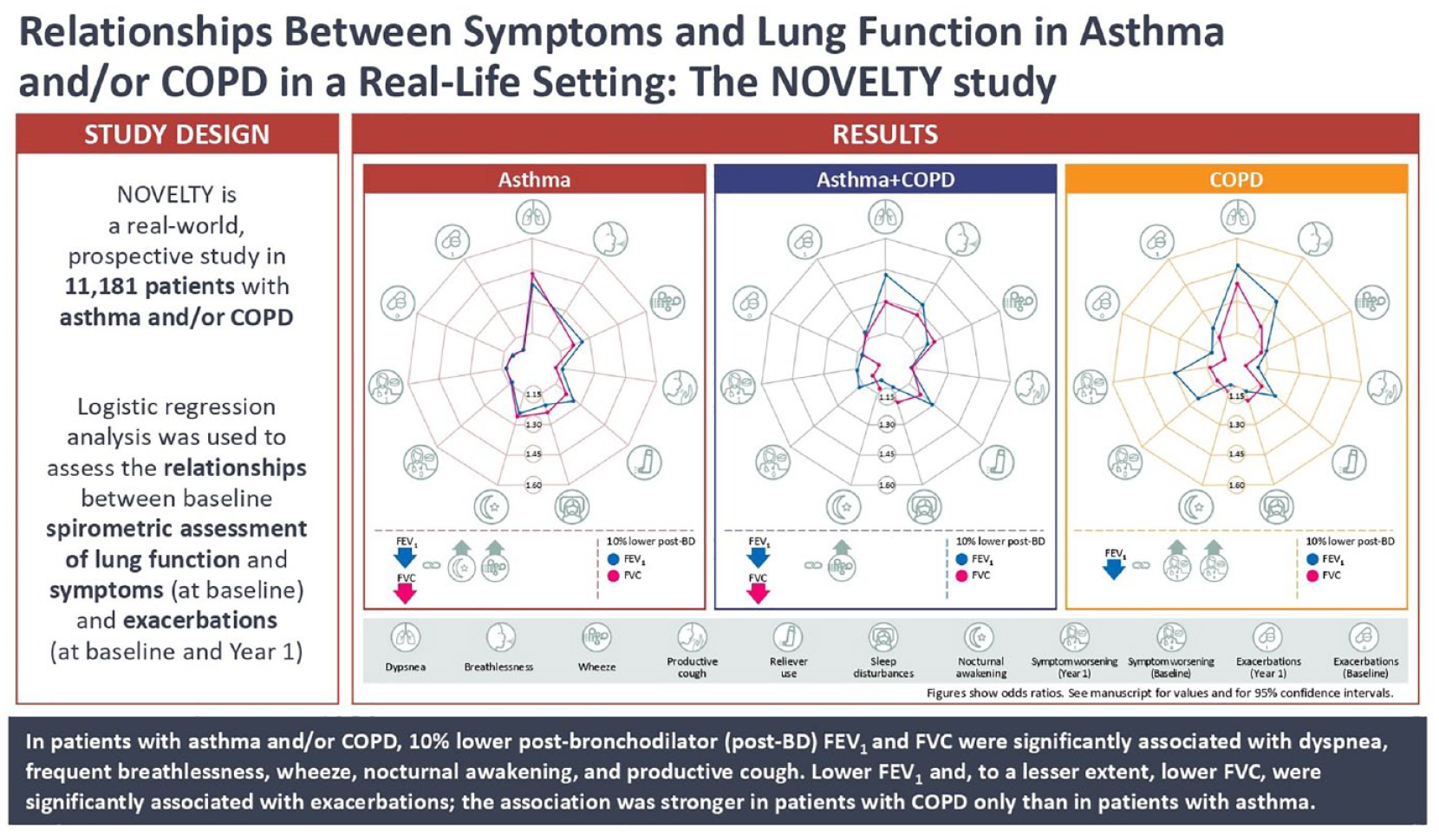
A total of 11,181 patients in NOVELTY had spirometry data (asthma, n = 5903; COPD, n = 3881; asthma + COPD, n = 1397). A 10% lower post-BD %pred forced expiratory volume in 1 s (FEV1) and forced vital capacity (FVC) – adjusted for age and sex – were significantly associated with dyspnea (modified Medical Research Council ⩾ grade 2), frequent breathlessness [St George’s Respiratory Questionnaire (SGRQ)], frequent wheeze attacks (SGRQ), nocturnal awakening (Respiratory Symptoms Questionnaire; ⩾1 night/week), and frequent productive cough (SGRQ). Lower post-BD %pred FEV1 and, to a lesser extent, lower post-BD %pred FVC were significantly associated with ⩾1 physician-reported exacerbation at baseline or Year 1. This association was stronger in patients with COPD than in those with asthma.
Conclusion:
In a real-life setting, reduced lung function is consistently associated with symptoms in patients with asthma, COPD, or asthma + COPD. The relationship with exacerbations is stronger in COPD only than in asthma.
Trail registration:
clinicaltrials.gov identifier: NCT02760329 (www.clinicaltrials.gov).
Plain Language Summary
- forced expiratory volume in 1 second (FEV1) – the amount of air that can be blown out of the lungs in 1 second - forced vital capacity (FVC) – the amount of air that can be forcibly breathed out from the lungs after taking the deepest breath possible
Graphical abstract
Introduction
Patients with asthma, chronic obstructive pulmonary disease (COPD), or asthma + COPD typically present with respiratory symptoms, such as breathlessness and productive coughing.1,2 It is generally believed that the symptoms experienced by the patient relate directly to the degree of lung function impairment that the patient suffers. However, although this is corroborated by population-level data,3,4 at the individual level, there is a large degree of variability between airflow limitation and the presence of symptoms.4,5
Clinical trials in patients with asthma and/or COPD, upon which guidelines for treatment and management are based, require patients to satisfy strict criteria for enrollment; as such, the vast majority of patients who present with obstructive lung diseases in clinical practice are excluded.6,7 There are limited real-world data to demonstrate whether there is a relationship between symptoms and lung function impairment in patients who present with asthma and/or COPD in clinical practice. NOVEL observational longiTudinal studY (NOVELTY; clinicaltrials.gov identifier: NCT02760329) is a large (n = 11,215 patients), global, prospective, observational study that aims to identify phenotypes and endotypes of obstructive lung disease that go beyond diagnostic labels and that will help to drive a shift toward patient-tailored care based on the presence of treatable traits. To enable this, NOVELTY enrolled patients with a physician-assigned diagnosis of asthma and/or COPD from primary or secondary care settings. 8 Thus, the recruitment of patients receiving routine clinical care in NOVELTY provides a unique opportunity to directly compare the relationship between respiratory symptoms and outcomes with lung function in a large and broad population of patients with obstructive lung disease in a single cohort.
Methods
The NOVELTY study design, population, and methodology have been described previously8,9 and are summarized below. This report follows the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) guidelines for reporting observational studies. 10
Study design and patients
Patients were enrolled by primary care physicians, allergists, or pulmonologists between September 2015 and March 2017, across 371 sites in community and hospital outpatient settings in 19 countries.8,9 Patients were recruited by physician-assigned diagnosis and assessment of disease severity (mild, moderate, or severe) in roughly equal numbers without prespecified diagnostic criteria or guidance. 9 Briefly, all patients included in NOVELTY were aged ⩾12 years and had not participated in any respiratory interventional trial within the 12 months prior to enrollment; all patients were likely to complete 3 years of follow-up. 8 Patients with missing smoking status were excluded from the current analysis. Data from patients enrolled in China were excluded from the present analysis due to a change in regulations on data transfer in May 2019. The current analysis only included data for patients with a physician-assigned diagnosis of asthma and/or COPD for whom technically acceptable spirometry measurements were available at baseline. No additional inclusion or exclusion criteria were specified for this analysis. Data were stratified by physician-assigned diagnosis and assessment of disease severity (mild, moderate, or severe).
The NOVELTY study was approved by the relevant institutional review board or independent ethics committee and the local health authority as applicable (Supplemental Table S1). All patients provided written informed consent.
Assessments
As detailed previously,8,9 at recruitment, demographics, smoking exposure and status, clinical characteristics, comorbidities, medication use, rate of physician-reported moderate to severe exacerbations, and patient-reported symptoms and worsenings were recorded using the St George’s Respiratory Questionnaire (SGRQ), 11 modified Medical Research Council (mMRC) Dyspnoea Scale, 12 and Respiratory Symptom Questionnaire (RSQ). 13 A patient-reported worsening was defined as an episode of breathing worsening beyond that usually experienced in a typical day. Frequent breathlessness, frequent wheeze, and frequent productive cough were defined using the SGRQ as shortness of breath, attacks of wheezing, or bringing up phlegm, respectively, on several or most days a week over the preceding 3 months. 11 For this analysis, physician-reported exacerbations in the previous 12 months and patient-reported worsenings in the previous 3 months were recorded at baseline and at Year 1. Pre- and post-bronchodilator (post-BD) percent predicted (%pred) forced expiratory volume in 1 s (FEV1) and forced vital capacity (FVC), and post-BD %pred inspiratory capacity (IC) were determined by trained personnel following international recommendations. 8 Reference values were those of the Global Lung Function Initiative. 14
Statistical analysis
Results are presented for the overall analysis set and stratified by physician-assigned diagnosis (asthma, COPD, asthma + COPD) and severity (mild, moderate, severe). The distributions of spirometric variables by physician-assigned diagnosis and severity are represented by density histograms. Potential associations between spirometric variables and each symptom (all binary) were evaluated using logistic regression models adjusted for age and sex (within each diagnostic group and overall) where the symptom (e.g. frequent breathlessness: yes/no) was the outcome and spirometric variable (e.g. FEV1) was the independent variable. The changes in spirometric measurements of lung function were standardized to 10% increments relevant to the distribution of the change observed for a particular outcome. Associations were reported as odds ratio (OR) with 95% confidence interval per 10% lower post-BD %pred spirometric measure. All data analyses were performed in RStudio (version 3.6.1 or higher).
A sensitivity analysis was performed to assess the same associations in patients with a high likelihood of asthma (physician-assigned diagnosis of asthma only, asthma symptom onset at age <40 years, <5 pack-years smoking exposure) versus physician-assigned asthma, and a high-likelihood of COPD (physician-assigned diagnosis of COPD only, COPD symptom onset at age >40 years, ⩾10 pack-year smoking exposure, FEV1/FVC < 0.7) versus physician-assigned COPD.
A second sensitivity analysis was performed to assess the same associations in patients recruited from primary care versus those recruited from non-primary care settings.
Results
Patient characteristics
This analysis included 11,181 patients with spirometric data at baseline enrolled in NOVELTY across 18 countries: 5903 (53%) with asthma, 3881 (35%) with COPD, and 1397 (13%) with asthma + COPD. The main demographics and clinical characteristics are presented in Table 1. The asthma group included a greater proportion of females and younger patients than the COPD and asthma + COPD groups (Table 1). Region of enrollment and baseline comorbidities are shown in Supplemental Tables S2 and S3.
Baseline demographics and clinical characteristics of patients with spirometric assessment in NOVELTY by physician-assigned diagnosis and severity of asthma and/or COPD. Data are presented as means (± SD) or n (%).

Thirty-seven patients had missing data for smoking status. Patients with missing data were excluded from the denominator when calculating percentages.
CAAT, chronic airways assessment test; COPD, chronic obstructive pulmonary disease; mMRC, modified Medical Research Council Dyspnoea Scale; RSQ, Respiratory Symptom Questionnaire; SD, standard deviation; SGRQ, St George’s Respiratory Questionnaire.
Symptom burden
The symptom burden by disease label and physician-assessed severity is presented in Table 1. The proportions of patients with at least one physician-reported exacerbation in the previous 12 months showed little variation by physician-assigned diagnosis either at baseline or Year 1 (Table 1). The proportions of patients with at least one patient-reported worsening in the previous 3 months were lowest in the COPD group at baseline and Year 1 (Table 1).
Spirometric characteristics
Post-BD (%pred) FEV1, FVC, and FEV1 z-scores were highest in the asthma group and lowest in the COPD group, with good correlation between lower FEV1, FVC, and FEV1 z-scores and disease severity (Table 2; Supplemental Figure S1). The relationships between %pred FEV1/FVC and %pred FEV1 for most patients indicated airflow limitation compatible with a physician-assigned diagnosis of asthma and/or an assigned diagnosis of COPD, according to established diagnostic criteria 2 (Figure 1).
Spirometric characteristics at baseline by physician-assigned diagnosis and severity of asthma and/or COPD. Data are presented as means (±SD).
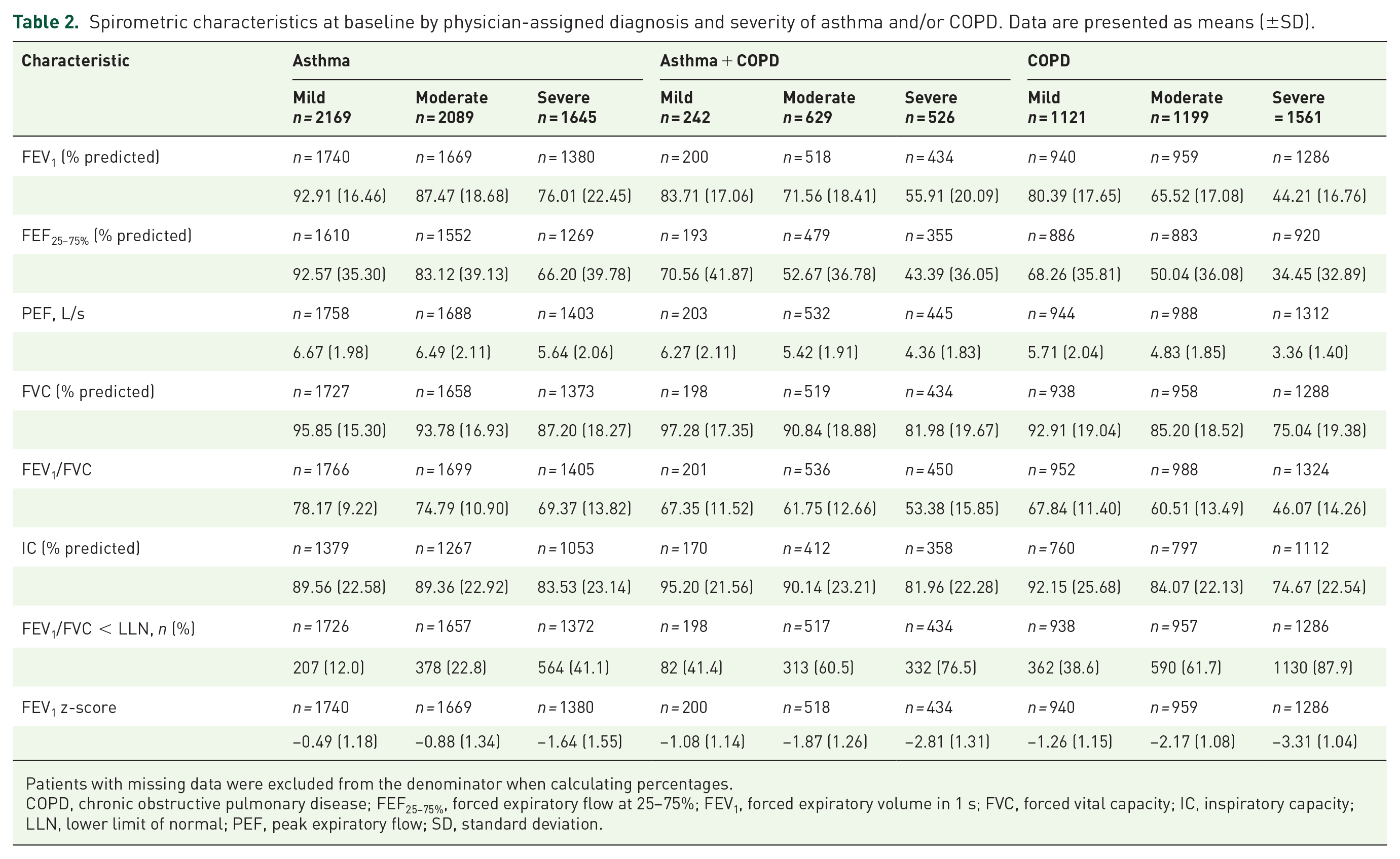
Patients with missing data were excluded from the denominator when calculating percentages.
COPD, chronic obstructive pulmonary disease; FEF25–75%, forced expiratory flow at 25–75%; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; IC, inspiratory capacity; LLN, lower limit of normal; PEF, peak expiratory flow; SD, standard deviation.

Relationship between post-bronchodilator FEV1/FVC ratio and FEV1 % predicted in patients with physician-assigned diagnosis of asthma and/or COPD and spirometric assessment in NOVELTY.
Relationship between lung function and symptoms
Overall study population
Logistic regression analysis adjusted for age and sex showed that for the overall study population, most OR were >1, meaning that for each 10% lower FEV1 or FVC at baseline, the odds of experiencing symptoms were higher (Figure 2). Specifically, lower FEV1 and FVC were each significantly associated with more frequent dyspnea (mMRC dyspnea score ⩾2), breathlessness (SGRQ), wheeze attacks (SGRQ), and nocturnal awakening (RSQ; ⩾1 night/week) with weaker but still significant associations between lower FEV1 and FVC, and frequent productive cough (SGRQ) (Figure 2). The association between lower FEV1 and ⩾1 physician-reported exacerbation at baseline or Year 1 was stronger than for lower FVC (Figure 2).

Associations between FEV1 and FVC, symptoms, and exacerbations in patients with physician-assigned diagnosis of asthma and/or COPD and spirometric assessment in NOVELTY (analysis adjusted for age and sex).
By disease label
Lower FEV1 and FVC were each significantly associated with mMRC score ⩾2 and frequent wheeze attacks across all groups (Figure 2). By contrast, lower FEV1 and FVC were significantly and more strongly associated with nocturnal awakening in patients with asthma than in patients with asthma + COPD or COPD (Figure 2). The association between lower FEV1 and ⩾1 physician-reported exacerbation at baseline and Year 1 was stronger for patients with COPD than those with asthma (Figure 2). In comparison with the data for physician-reported exacerbation, the association between lower FEV1 or FVC and patient-reported worsening in the asthma and COPD groups was significant but weaker at baseline and Year 1 (Figure 2). These observations (adjusted for age and sex) remained essentially unchanged in the unadjusted analysis (Supplemental Figure S2) and in the analysis adjusted for age, sex, and smoking status (Supplemental Figure S3). There was a similar association between FEV1 %pred and symptoms/exacerbations in the physician-assigned and the high likelihood asthma and COPD groups (Supplemental Figure S4). Additionally, there were similar associations between FEV1 %pred and symptoms/exacerbations in patients recruited from primary care settings and non-primary care settings (Supplemental Figure S5).
Discussion
The main results show that: (1) more severe airflow limitation was significantly associated with increased prevalence of dyspnea, wheeze attacks, nocturnal awakening, and frequent productive cough; (2) the relationships were similar between lower FEV1 and FVC across physician-assigned diagnoses of asthma, asthma + COPD, and COPD; and (3) lower FEV1 was more strongly associated with physician-reported exacerbation in patients with COPD than those with asthma.
Previous studies
Asthma and COPD are heterogenous diseases with some overlapping symptoms.1,7 COPD is characterized by inflammatory and structural changes in the lungs that result in airflow limitation. 1 Patients with COPD present differently in clinical practice because there are many genetic, biological, clinical, and environmental factors that interplay in its pathogenesis. 15 This results in a heterogeneous population in which there are several non-linear components of disease that are not present in every patient with COPD, nor in a single patient at every timepoint assessed. 15 In this context, spirometric measures do not fully capture the complexity of the disease. 5 This is demonstrated by findings that patients within each category of disease severity, classified according to the Global Initiative for Obstructive Lung Disease (GOLD) spirometric criteria, 1 have varying levels of symptoms, exacerbation frequency, exercise tolerance, and comorbidities. 5 Therefore, a move toward treating patients with obstructive lung disease according to the presence of treatable traits that go beyond diagnostic labels has been suggested. 16
Given that using %pred cut-offs for spirometry metrics is subject to bias, a shift toward defining severity based on z-scores has been recommended; however, recommendations are not consistent across international guidelines, and so z-scores are not in routine use in clinical practice.1,7,14,17,18 To present findings that can be easily interpreted by clinicians, and for consistency with previously published NOVELTY publications, we have used %pred values in this analysis.
Clinical management across diagnoses in obstructive lung diseases aims to improve disease control by decreasing exacerbations and presenting symptoms, thereby increasing patient quality of life.1,2 In our analysis, symptoms of dyspnea, productive cough, wheeze, and breathlessness were reported across physician-assigned diagnoses of asthma and/or COPD. The diagnosis of both asthma and COPD requires spirometric assessment of airflow limitation. However, the inter-relationship between the presenting symptoms, spirometric assessment, and physician-assigned diagnosis across obstructive lung diseases is currently unclear. Our findings of a significant association between lower FEV1 and more frequent wheeze attacks, and exacerbations across asthma and COPD are consistent with a previous review and regression analysis of randomized controlled trials in COPD, indicating significant associations between a higher FEV1 and SGRQ total score and lower risk of exacerbations. 19 For patients with asthma, while the association between lower FEV1 and patient-reported worsenings was weaker than that between lower FEV1 and physician-reported exacerbations, the correlation was significant both at baseline and Year 1; Asthma Control Questionnaire (ACQ)-5 and ACQ-6 scores were found to be only weakly correlated to %pred FEV1 in a previous longitudinal study of patients with persistent asthma. 4
Considerable variation is reported in the literature as to the proportion of patients with either asthma or COPD who have a concurrent asthma + COPD diagnosis. 20 However, one systematic review and meta-analysis reported that the pooled prevalence of asthma + COPD was 26.5% of patients with asthma and 29.6% of patients with COPD, which is considerably higher than the 12% of patients reported in NOVELTY. 20 This may reflect the implementation of different diagnostic criteria in each study.
Interpretation of novel findings
Our analysis of this heterogenous patient population from routine clinical practice indicated strong relationships between lower FEV1, FVC, and patient-reported symptoms using the SGRQ, mMRC Dyspnoea Scale, and RSQ across physician-assigned diagnoses of asthma and/or COPD. Lower FEV1 and FVC were significantly associated with worse symptoms of breathlessness and frequent productive cough across diagnoses of asthma and/or COPD in this single cohort and expand on previous observational findings in COPD alone indicating associations between decreased FEV1 and breathlessness, and between FVC decline and sputum production in individuals who smoke and those with a history of smoking. 21
A significant correlation was also found between lower spirometric parameters and nocturnal awakening in this real-world patient population with asthma and/or COPD. Separate population-based studies have shown a relationship between FEV1 decline and nightly respiratory symptoms in patients with airflow limitation (FEV1/FVC ratio less than the lower limit of normal) 22 and a significant association between a higher risk of obstructive sleep apnea (measured by the minimal apnea prediction index) and lower FEV1 in patients with a physician-assigned diagnosis of asthma. 23
Data for correlations between spirometric assessment of lung function and episodes of disease worsening across COPD and asthma are scarce. In this analysis, the associations between lower spirometric assessment of lung function and exacerbations and symptom worsening were significant across patients diagnosed with asthma and/or COPD; furthermore, there were some differences between the strength of these relationships in asthma and in COPD. To the best of our knowledge, these differences have not been shown previously in the same patient cohort; findings from a registry study focused on patients with COPD only demonstrated a correlation between more rapid decline in post-BD FEV1 and higher exacerbation frequency. 24
In this analysis, patient-reported worsenings of their asthma and/or COPD were reported every 3 months through a structured questionnaire collecting frequency of these events, their management, and related healthcare utilization. Events of patient-reported worsenings may reflect respiratory symptom variability. If these are treated by oral corticosteroids and/or antibiotics, they can possibly be considered as exacerbations. It is accepted that a substantive proportion of exacerbations of asthma and/or COPD are under-reported or under-recognized. In addition to symptom variability, a subset of patient-reported worsenings can approximate to milder exacerbations not captured by study physicians. In this international study, reporting of these events is likely to be less dependent on healthcare system access and local healthcare practice.
In NOVELTY, patients were included if they had a physician-assigned diagnostic label of asthma and/or COPD; therefore, the NOVELTY population reflects patients who present in clinical practice. To determine whether differences could be seen between patients who had been classified according to strict diagnostic criteria and patients who had a physician-assigned diagnostic label, we conducted a sensitivity analysis of FEV1 %pred in patients with a high likelihood of asthma versus physician-assigned asthma and in patients with a high likelihood of COPD versus physician-assigned COPD. Results from this analysis demonstrated that the correlation between FEV1 %pred and symptoms and exacerbations was similar regardless of the diagnostic method.
Whether patients included in NOVELTY were diagnosed by a practitioner or a specialist is unknown. However, patients recruited from primary care settings may be less likely to have specialist-assigned diagnoses when compared with those from secondary care. To determine whether differences could be seen between these populations, we conducted a sensitivity analysis of FEV1 %pred in patients from primary care and patients not from primary care. Results from this analysis demonstrated that the correlation between FEV1 %pred and symptoms and exacerbations was similar regardless of the recruitment setting. These findings are completely novel.
Future studies
Given the heterogeneity of asthma and COPD, as well as important differences in the treatment recommendations, 7 there is considerable interest in finding biomarkers to better phenotype patients across diagnostic labels. The Advanced Diagnostic Profiling substudy of NOVELTY is using novel imaging techniques, advanced pulmonary function tests, and metabolic modalities to examine structural and functional changes in the lungs and identify early markers and phenotypes of lung disease. 25 The study aims to identify clinically meaningful patient profiles that may enable a move toward a precision medicine-based approach to patient care in the future. 25 Further analyses of NOVELTY will continue to build on the results presented in this paper, and aim to identify treatable traits (beyond airflow limitation) that are present across diagnoses of obstructive lung disease in order to drive a personalized approach to patient care going forward.
Strengths and potential limitations
The diverse, international patient cohort of NOVELTY is recruited from routine clinical care and provides a unique opportunity for observational analyses in a single population and, for the first time, the direct comparison of outcomes using the same methodological approach in patients with asthma and/or COPD in a real-life setting without the stringent criteria required for many clinical trials. Given that as little as 5% of patients with asthma and/or COPD meet these criteria, 6 the external validity of their results comes into question. Therefore, advances in our knowledge of obstructive lung disease, particularly the identification of biomarkers that are associated with outcomes and could potentially change the treatment paradigm, are reliant on observational studies that do not discriminate by such criteria. The main strength of this analysis is that the findings are generalizable to a heterogenous population of patients with obstructive lung disease who present in clinical practice.
Furthermore, it is well known that there are limitations with using spirometry to diagnose patients with COPD. As previously noted, single measures of airflow limitation do not reflect the heterogeneity of disease. 15 Additionally, airflow obstruction becomes more prevalent with increasing age; therefore, using an FEV1/FVC <0.7 fixed ratio cut-off leads to significant over-diagnosis among older patients.18,26 The use of physician-assigned diagnosis in NOVELTY enables evaluation of patients by the diagnostic labels that they have received in the real world.
A potential limitation is that baseline variables, such as exacerbation history or episodes of symptom worsening, may have been subject to recall bias. Additionally, as NOVELTY is an observational study, patients were already on treatment at the time of analysis, so results may differ from those at the time of diagnosis before treatment had been initiated.
Conclusion
In this large cohort of patients with asthma and/or COPD in routine clinical care, lower FEV1 and FVC were significantly associated with a wide range of respiratory and other patient symptoms across physician-assigned diagnoses of asthma, asthma + COPD, and COPD. The association between lower FEV1 and physician-reported exacerbations was stronger in patients with COPD only than in those with asthma. These findings expand existing knowledge of the interplay between spirometry variables, symptoms, and exacerbations in real-world patients with asthma and/or COPD.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241254212 – Supplemental material for Relationships between symptoms and lung function in asthma and/or chronic obstructive pulmonary disease in a real-life setting: the NOVEL observational longiTudinal studY
Supplemental material, sj-docx-1-tar-10.1177_17534666241254212 for Relationships between symptoms and lung function in asthma and/or chronic obstructive pulmonary disease in a real-life setting: the NOVEL observational longiTudinal studY by Alberto Papi, Rod Hughes, Ricardo del Olmo, Alvar Agusti, Bradley E. Chipps, Barry Make, Erin Tomaszewski, Keith Peres Da Costa, Divyansh Srivastava, Jørgen Vestbo, Christer Janson, Pierre-Régis Burgel and David Price in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666241254212 – Supplemental material for Relationships between symptoms and lung function in asthma and/or chronic obstructive pulmonary disease in a real-life setting: the NOVEL observational longiTudinal studY
Supplemental material, sj-docx-2-tar-10.1177_17534666241254212 for Relationships between symptoms and lung function in asthma and/or chronic obstructive pulmonary disease in a real-life setting: the NOVEL observational longiTudinal studY by Alberto Papi, Rod Hughes, Ricardo del Olmo, Alvar Agusti, Bradley E. Chipps, Barry Make, Erin Tomaszewski, Keith Peres Da Costa, Divyansh Srivastava, Jørgen Vestbo, Christer Janson, Pierre-Régis Burgel and David Price in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-3-tar-10.1177_17534666241254212 – Supplemental material for Relationships between symptoms and lung function in asthma and/or chronic obstructive pulmonary disease in a real-life setting: the NOVEL observational longiTudinal studY
Supplemental material, sj-docx-3-tar-10.1177_17534666241254212 for Relationships between symptoms and lung function in asthma and/or chronic obstructive pulmonary disease in a real-life setting: the NOVEL observational longiTudinal studY by Alberto Papi, Rod Hughes, Ricardo del Olmo, Alvar Agusti, Bradley E. Chipps, Barry Make, Erin Tomaszewski, Keith Peres Da Costa, Divyansh Srivastava, Jørgen Vestbo, Christer Janson, Pierre-Régis Burgel and David Price in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-jpg-4-tar-10.1177_17534666241254212 – Supplemental material for Relationships between symptoms and lung function in asthma and/or chronic obstructive pulmonary disease in a real-life setting: the NOVEL observational longiTudinal studY
Supplemental material, sj-jpg-4-tar-10.1177_17534666241254212 for Relationships between symptoms and lung function in asthma and/or chronic obstructive pulmonary disease in a real-life setting: the NOVEL observational longiTudinal studY by Alberto Papi, Rod Hughes, Ricardo del Olmo, Alvar Agusti, Bradley E. Chipps, Barry Make, Erin Tomaszewski, Keith Peres Da Costa, Divyansh Srivastava, Jørgen Vestbo, Christer Janson, Pierre-Régis Burgel and David Price in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The authors would like to thank the patients who participated in this study, the NOVELTY Scientific Community, and the NOVELTY study investigators who are listed in full in Supplemental Tables S4 and S5. Medical writing support was provided by Linda Brown, BSc, Katie Webster, BSc, and Matt Brownsword, PhD, and editorial support was provided by Joe Alling, BSc, Sharmin Saleque, MSc, and Jess Galbraith, BSc, all of Core (a division of Prime), London, UK, in accordance with Good Publication Practice guidelines (![]() ), and funded by AstraZeneca. All authors contributed to the study design or the analysis and/or interpretation of the data. All authors were responsible for the manuscript content and editorial decisions and approved the final version. The authors received no honoraria related to the development of this publication.
), and funded by AstraZeneca. All authors contributed to the study design or the analysis and/or interpretation of the data. All authors were responsible for the manuscript content and editorial decisions and approved the final version. The authors received no honoraria related to the development of this publication.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
