Abstract
Background:
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes coronavirus disease 2019 (COVID-19), a pandemic that has resulted in millions of deaths worldwide. Critically ill COVID-19 patients who require intubation and develop nosocomial pneumonia, commonly caused by gram-negative bacilli, have a higher mortality rate than those without nosocomial pneumonia.
Objectives:
The aim of this study is to compare the clinical characteristics and outcomes and associated risk factors of Alpha and Omicron SARS-CoV-2 variants in critically ill patients on mechanical ventilation (MV) with nosocomial pneumonia.
Design:
This is a retrospective single-center cohort study.
Methods:
This observational study was conducted at Taipei Veterans General Hospital, Taiwan from May 2021 to September 2022. Critically ill patients who had confirmed SARS-CoV-2 infection and intubated on a MV with bacterial pneumonia were enrolled. Demographic data, laboratory results, and treatment information were collected and analyzed. In addition, clinical outcomes among different SARS-CoV-2 variants were examined.
Results:
This study included 94 critically ill COVID-19 patients who required intubation and intensive care unit (ICU) admission. The Alpha group had a longer duration of SARS-CoV-2 viral shedding, MV days, and ICU stay, while the Omicron group had older age, more comorbidities, higher APACHE II scores, and higher in-hospital mortality (47.0% versus 25.0%, p = 0.047). However, independent risk factors for in-hospital mortality included malignancy, lower serum albumin levels, and lack of Remdesivir treatment, except for the SARS-CoV-2 variant.
Conclusion:
Our study discovered a higher in-hospital mortality rate in severe COVID-19 patients with MV and secondary pneumonia infected with the Omicron variant compared to the Alpha variant; however, real independent risk factors for in-hospital mortality are malignancy, lower serum albumin level, and lack of Remdesivir treatment.
Keywords
Background
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the causative agent of coronavirus disease 2019 (COVID-19), which was declared a pandemic by the World Health Organization on 11th March 2020. COVID-19 has resulted in millions of deaths worldwide. Taiwan experienced two waves of the COVID-19 pandemic involving the Alpha and the Omicron variants. The Alpha variant mainly prevailed from May to August 2021, while the Omicron variant was more prevalent from April to August 2022.1,2 Real-time reverse transcriptase polymerase chain reaction (RT-PCR) is a widely used diagnostic tool for identifying SARS-CoV-2 infection by detecting viral ribonucleic acid (RNA). The cycle threshold (Ct) value in PCR testing indicates the number of cycles required for the detection of viral RNA, with a Ct value of 40 or higher suggesting a negative result for SARS-CoV-2. 3 However, a positive PCR result does not suggest that the person is contagious, and a Ct value of 30 may indicate non-infectiousness, according to some studies. 4 Prior research suggests that advanced age, female gender, and co-infection with secondary infection are associated with prolonged viral shedding.5 –7 However, there is limited research comparing viral shedding across different SARS-CoV-2 variants, particularly among critically ill patients who require intubation and develop nosocomial pneumonia. Nosocomial pneumonia includes hospital-acquired pneumonia (HAP) and ventilator-associated pneumonia (VAP), and both can result in significant mortality rates and increased medical costs. 8 Gram-negative bacilli are commonly isolated from the airways of patients with nosocomial pneumonia. These bacteria, which include Pseudomonas aeruginosa, Acinetobacter species, Enterobacteriaceae, and Stenotrophomonas maltophilia, are frequently isolated pathogens in the airway of these patients. 9 A multicenter cohort study has revealed that co-infection with COVID-19 results in a higher risk of ICU admission, mechanical ventilation (MV), and in-hospital mortality compared to infection with COVID-19 alone, regardless of the SARS-CoV-2 variants involved. 10 However, there is still a lack of research on the clinical outcomes of critically ill patients who require intubation and develop secondary bacterial pneumonia while infected with different SARS-CoV-2 variants. Therefore, the aim of our study is to compare the viral kinetics and clinical outcomes of Alpha and Omicron SARS-CoV-2 variants in critically ill patients on MV with secondary pneumonia.
Methods
Study design, setting, and patients
This retrospective observational study was conducted at Taipei Veterans General Hospital in Taipei, Taiwan, which is a medical center with 2800 beds. The study period was from May 2021 to September 2022, which were the periods of the Alpha and Omicron variants outbreak in Taiwan. The study enrolled critically ill patients who had confirmed SARS-CoV-2 infection through RT-PCR and who were intubated on MV with subsequent bacterial pneumonia. The following exclusion criteria were applied: lack of RT-PCR confirmation for SARS-CoV-2, no admission to the ICU, age under 20 years, concurrent human immunodeficiency virus infection, patients using continuous positive airway pressure (CPAP) at home, patients without secondary pneumonia or pneumonia diagnosed prior to COVID-19 infection, and patients who did not receive appropriate antibiotic treatment.
Data collection and measurements
This study collected demographic data, such as age, gender, underlying diseases, laboratory results, and Acute Physiologic Assessment and Chronic Health Evaluation (APACHE) II scores 11 upon ICU admission or the day of respiratory failure. Treatment information, including the use of Tocilizumab, Remdesivir, and enoxaparin, was also recorded. Corticosteroid exposure was measured in dexamethasone equivalents, and the cumulative dosage of dexamethasone in milligrams (mg) from admission to discharge or death was recorded. The study also monitored clinical course and outcomes, such as the use of MV and extracorporeal membrane oxygenation, gastrointestinal bleeding, length of ICU stay, duration of SARS-CoV-2 viral shedding, MV days, ICU mortality, and in-hospital mortality.
Nosocomial pneumonia definitions
The diagnosis of bacterial pneumonia was based on the Infectious Diseases Society of America and the American Thoracic Society 2016 HAP/VAP guidelines, which required the presence of new or progressive pulmonary infiltration, consolidation, or cavitation on radiographic imaging, as well as clinical evidence such as unstable vital signs, new onset of thick, yellow or greenish sputum, changes in the character of sputum, increased respiratory secretions, increased requirements for suctioning, and deteriorating gas exchange. In terms of laboratory evidence, elevated levels of white blood cell counts, C-reactive protein, and procalcitonin were considered. Pathogens were obtained from respiratory specimens via endotracheal aspiration or sputum induction and were cultured for at least 48 h after hospitalization. HAP was defined as pneumonia that occurred two or more days after admission, while VAP was defined as pneumonia that developed 48 h after initiation of MV. 12 The patients diagnosed with nosocomial pneumonia in this study all received a course of appropriate antibiotics lasting 5–7 days, based on culture data confirmed by a detailed chart review.
SARS-CoV-2 RT-PCR
The RT-PCR for SARS-CoV-2 was conducted using the Roche Cobas 6800 system from Roche Diagnostics in Rotkreuz, Switzerland. Target nucleic acids from the samples were selectively amplified using specific forward and reverse primers for the ORF1aboratory non-structural region, which is unique to SARS-CoV-2. In addition, a conserved region in the structural protein envelope E-gene was selected for the detection of pan-Sarbecovirus. If the Ct value was equal to or greater than 40, it was considered negative in the RT-PCR results. The duration of SARS-CoV-2 shedding was determined as the period from the onset of symptoms until the day the Ct value exceeded 30, as several studies have considered a value of 30 or higher to be non-infectious, and samples with a Ct value over 30 did not yield any virus cultures. 4 In addition, a Ct value of at least 30 is the threshold for isolation release set by the Taiwan Center for Disease Control. 13 Patients who did not have a Ct value greater than 30 throughout their hospitalization were censored at their last recorded Ct value. All the patients were followed up from admission to death or discharge.
Statistical analysis
The study results are expressed as medians with interquartile ranges or numbers with percentages, as appropriate. The Mann–Whitney U test was utilized to compare continuous variables that did not follow a normal distribution. Pearson’s chi-squared test or Fisher’s exact test was utilized to compare categorical variables. Variables with a p value less than 0.1 between groups were included in the univariate and multivariate Cox proportional hazards models to identify factors that independently predict in-hospital mortality. Hazard ratio (HR) and 95% confidence intervals (CIs) were calculated using Cox proportional hazards models. Statistical significance was determined with a p value <0.05. The duration of SARS-CoV-2 shedding was assessed by constructing a Kaplan–Meier curve and performing a log-rank test. Two-tailed tests were utilized, and p values <0.05 were considered statistically significant. All statistical analyses were performed using IBM SPSS Statistics for Windows/Macintosh, Version 25.0 (IBM Corp., Armonk, NY, USA).
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental STROBE Checklist).
Results
Patient characteristics
A total of 291 patients with severe COVID-19 infection were enrolled during the study period. After excluding 1 patient under the age of 20, 3 patients who used CPAP at home, 160 patients who did not develop nosocomial pneumonia, 24 patients who were not intubated, 7 patients who were diagnosed with bacterial pneumonia before COVID-19 infection, and 2 patients who did not receive appropriate antibiotic treatment, the analysis included 94 critically ill COVID-19 patients on MV with subsequently bacterial pneumonia. Of these, 28 patients were from the Alpha wave while 66 patients were from the Omicron wave (Figure 1).
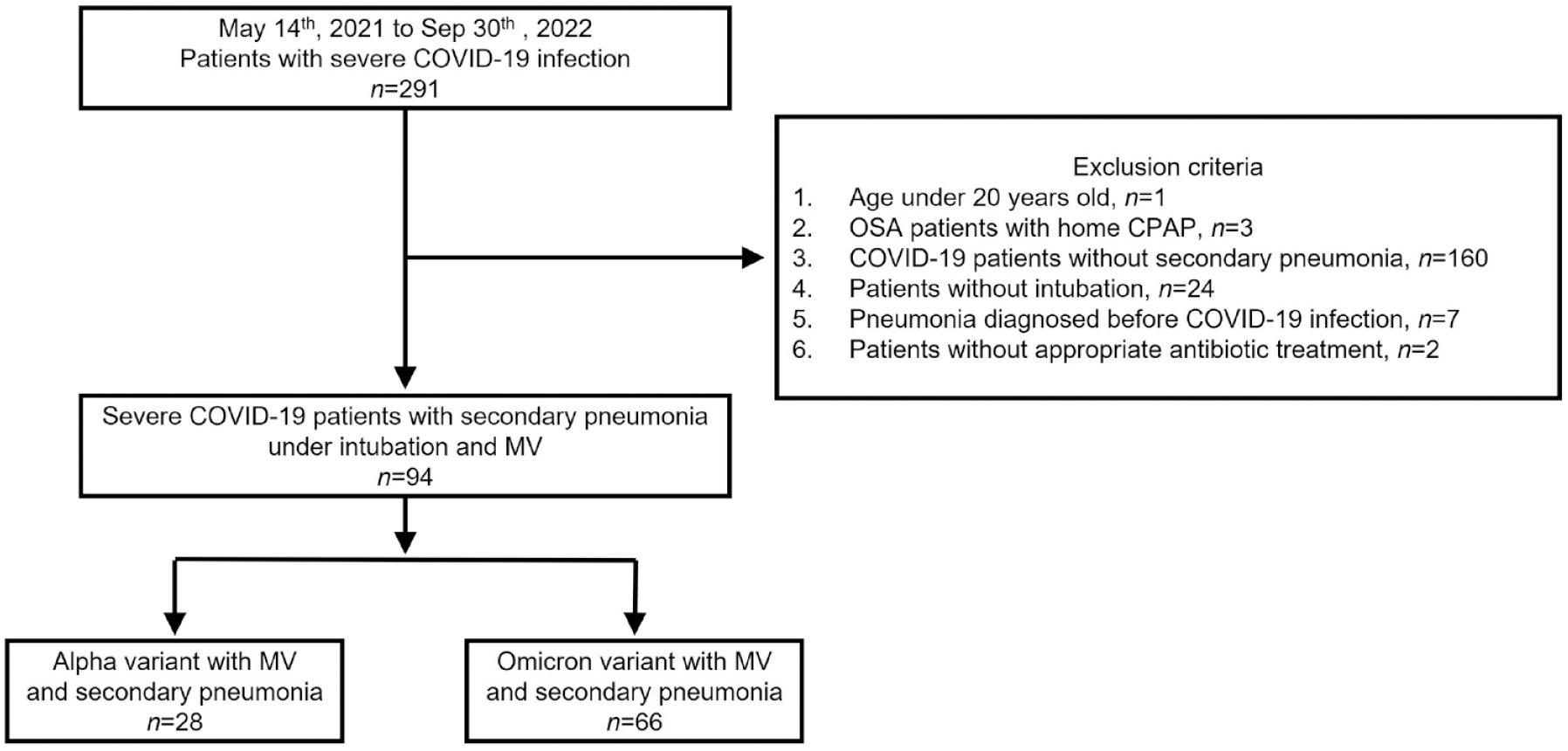
Study flowchart.
Table 1 presents the demographic characteristics and clinical features of the patients who were included in the study.
Clinical characteristics of patients according to different variant phenotypes.
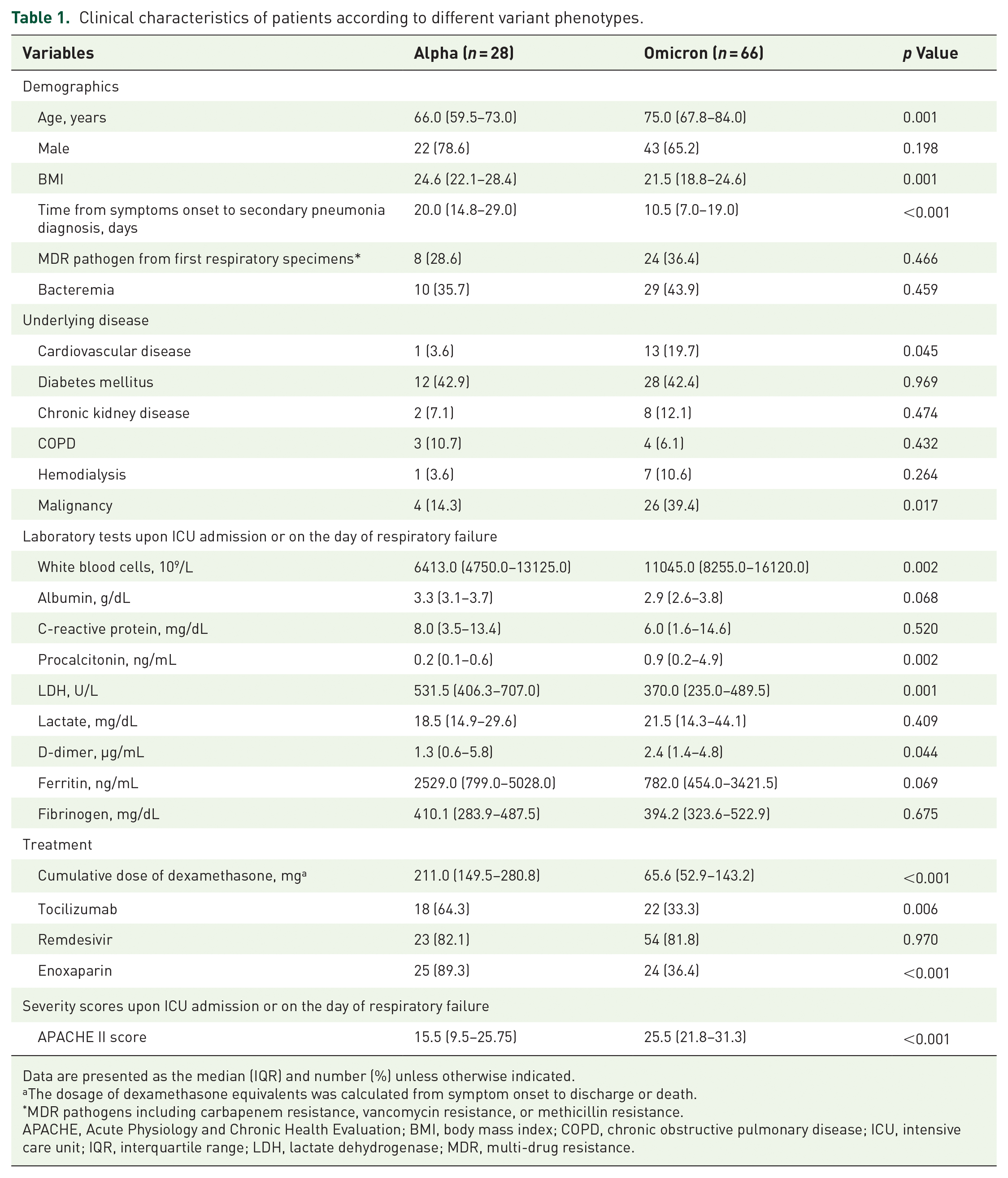
Data are presented as the median (IQR) and number (%) unless otherwise indicated.
The dosage of dexamethasone equivalents was calculated from symptom onset to discharge or death.
MDR pathogens including carbapenem resistance, vancomycin resistance, or methicillin resistance.
APACHE, Acute Physiology and Chronic Health Evaluation; BMI, body mass index; COPD, chronic obstructive pulmonary disease; ICU, intensive care unit; IQR, interquartile range; LDH, lactate dehydrogenase; MDR, multi-drug resistance.
According to Table 1, the demographic characteristics and underlying diseases of the Alpha cohort and Omicron cohort were compared. Overall, the duration from symptom onset to secondary pneumonia diagnosis was significantly shorter in the Omicron group than in the Alpha group (10.5 versus 20.0 days, p < 0.001). A total of 139 bacterial cultures were obtained from respiratory specimens, including sputum and endotracheal aspirate, in the Alpha variant group, with 59 being Stenotrophomonas species (42%), 21 being Acinetobacter species (15%), and 16 being Pseudomonas species (12%). In comparison, 239 positive cultures were collected in the Omicron group, with 50 being Stenotrophomonas species (21%), 50 being Acinetobacter species (21%), and 36 being Klebsiella species (15%) (Figure 2). The proportion of multi-drug resistance in the first respiratory specimens was similar between the two groups. Severe COVID-19 patients infected with the Omicron variant who were on MV with secondary pneumonia were found to have a significantly older age (75.0 versus 66.0, p = 0.001) and lower body mass index (BMI) (21.5 versus 24.6, p = 0.001) compared to those infected with the Alpha variant. Regarding underlying diseases, there was a higher prevalence of cardiovascular disease (19.7% versus 3.6%, p = 0.045) and uncured malignancy (39.4% versus 14.3%, p = 0.017) in the Omicron cohort. About treatment, patients with the Omicron variant received lower doses of steroids (65.6 mg versus 211.0 mg, p < 0.001), fewer Tocilizumab users (33.3% versus 64.3%, p = 0.006), and fewer enoxaparin users (36.4% versus 89.3%, p < 0.001) than those with the Alpha variant. Besides, the APACHE II score upon ICU admission or on the day of respiratory failure was significantly higher in the Omicron group (25.5 versus 15.5, p < 0.001). Patients with coexisting bacteremia were comparable between the two groups.
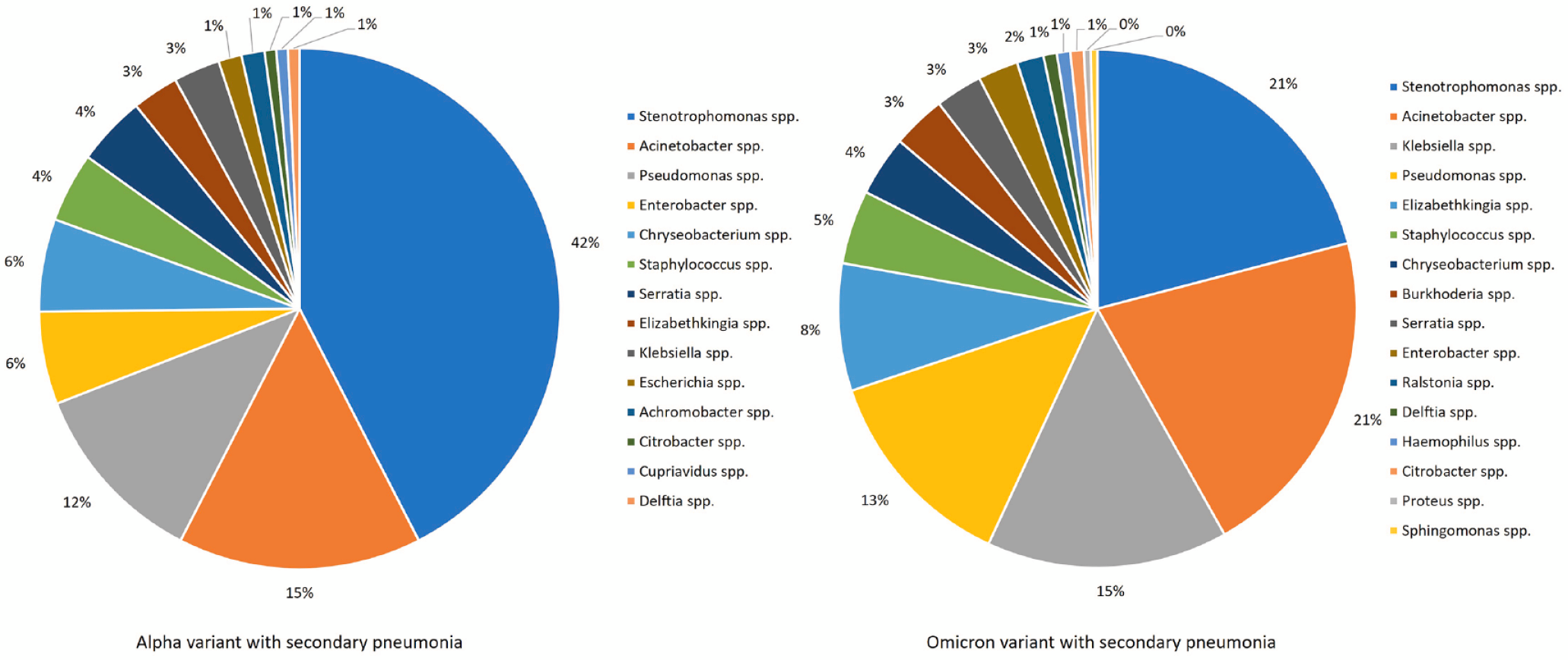
Distribution of bacterial cultures obtained from respiratory specimens, including sputum and endotracheal aspirate. There were 139 instances of positive culture in the Alpha group and 239 instances of positive culture in the Omicron group.
We subsequently examined the clinical outcomes between the two variants. Patients infected with the Omicron variant had a shorter length of stay in the ICU and shorter duration on MV. The in-hospital mortality rate was significantly higher in intubated patients with Omicron infection and secondary pneumonia than in the Alpha variant group (47.0% versus 25.0%, p = 0.047) (Table 2). We found that most Alpha-infected patients (5 out of 7, 71%) expired due to sepsis with organ failure. One patient died from cardiogenic shock with arrhythmia, and another from ischemic colitis.
Clinical outcomes of patients according to different variant phenotypes.

Data are presented as the median (IQR) and number (%) unless otherwise indicated.
ECMO, extracorporeal membrane oxygenation; GI, gastrointestinal; ICU, intensive care unit; IQR, interquartile range.
On the other hand, in the Omicron cohort, 25 out of 31 patients (81%) expired due to sepsis with organ failure. Among them, two patients were also diagnosed with acute myocardial infarction, one with combined pulmonary embolism, and one with stroke. Two patients died due to subdural hemorrhage, three from respiratory acidosis, and one from acute myocardial infarction. The most common cause of death in both groups was sepsis.
Moreover, severe Alpha variant-infected patients on MV with secondary pneumonia had a significantly longer duration of viral shedding compared to those with the Omicron variant.
Among the enrolled patients, 56 patients (60%) were alive at discharge. Both survivors and non-survivors had similar APACHE II scores at ICU admission or on the day of respiratory failure and the same proportions of bacteremia during admission. Of the patients who expired during hospitalization, 81% were infected with the Omicron variant. In addition, non-survivors had a significantly higher prevalence of malignancy (44.7% versus 23.2%, p = 0.028). In terms of laboratory tests conducted upon admission to the ICU or on the day of respiratory failure, non-survivors had lower serum levels of albumin and fibrinogen. Regarding treatment, there was no significant difference between both groups in terms of corticosteroid, Tocilizumab, and enoxaparin administration. However, in the mortality group, fewer patients received Remdesivir treatment (Supplemental Table 1).
To identify possible independent risk factors for in-hospital mortality, univariate and multivariate Cox proportional hazards models were performed (Table 3).
Association of in-hospital mortality by univariate and multivariate Cox regression analyses.

HR and 95% CI were derived from Cox proportional hazards models.
Albumin level was categorized by the median level of the total cohort.
CI, confidence interval; HR, hazard ratio.
In the univariate Cox proportional hazards models, we found that infection with the Omicron variant, presence of malignancy, lower serum albumin levels, and absence of Remdesivir administration were associated with in-hospital mortality. In the multivariate analysis, the presence of malignancy (HR, 3.321; 95% CI, 1.494–7.384; p = 0.003), lower serum albumin levels (HR, 0.417; 95% CI, 0.189–0.921; p = 0.030), and absence of Remdesivir administration (HR, 0.305; 95% CI, 0.132–0.705; p = 0.005) (Figure 3) were independent risk factors for further in-hospital mortality. We conducted propensity score matching, including significant factors such as age, BMI, cardiovascular diseases, and malignancy, to compare the Omicron and Alpha groups. After propensity score matching, the in-hospital mortality remained significantly higher in the Omicron group (23.1% versus 61.5%, p = 0.047) (Supplemental Tables 2 and 3).
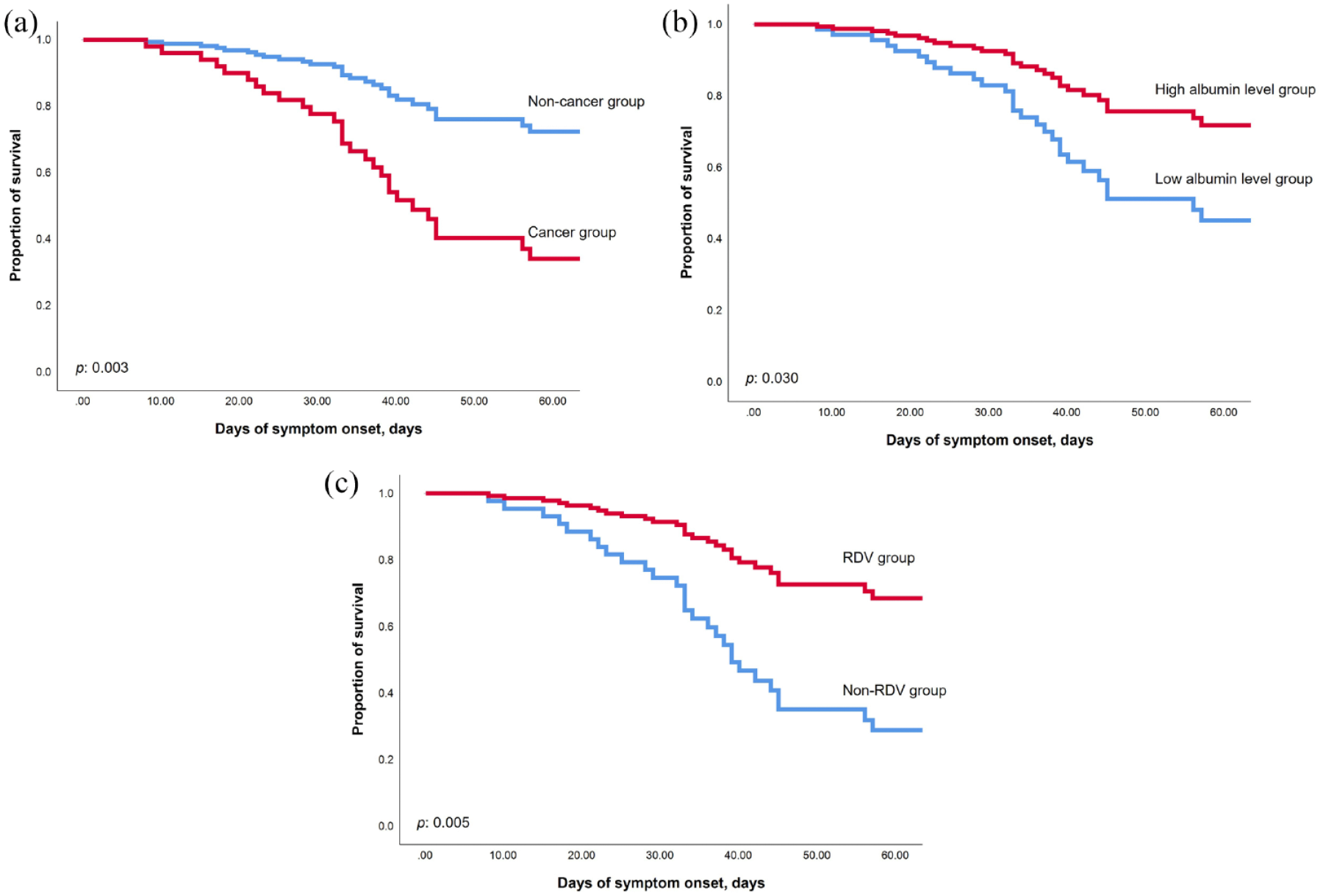
The multivariate Cox proportional hazards models between each independent risk factor. (a) The cancer group has a significantly higher in-hospital mortality rate (p = 0.003). (b) Patients with lower serum level of albumin has higher in-hospital mortality (p = 0.030). (c) The administration of RDV has significantly lower in-hospital mortality (p = 0.005).
Discussion
This study investigated the clinical outcomes and bacterial distribution of severe COVID-19 patients with intubation and subsequent bacterial pneumonia between the Alpha variant and the Omicron variant in Taiwan. We observed that the duration from symptom onset to the diagnosis of secondary pneumonia was significantly shorter in the Omicron cohort. Furthermore, the bacterial distribution was different between the two groups. The most encountered pathogens in the Alpha group were Stenotrophomonas species, while in the Omicron group, Stenotrophomonas species and Acinetobacter species were the most frequently cultured bacteria (Figure 2). Previous studies suggest that old age, the presence of cardiovascular diseases, and malnutrition may be risk factors for more severe forms of COVID-19 infection.14,15 We found that patients in the Alpha group had a longer duration of SARS-CoV-2 shedding, as well as longer MV days and ICU stay compared to the Omicron group. On the other hand, patients infected with the Omicron variant tended to be older and presented with higher APACHE II scores at admission. In addition, they exhibited lower BMI and lower serum levels of albumin, which may suggest a status of malnutrition. Furthermore, these patients experienced a higher in-hospital mortality rate. We also noticed that the comorbidity of malignancy, lower serum level of albumin, and absence of Remdesivir administration are independent risk factors for in-hospital mortality.
Previous studies have shown that several factors, including old age, and coexisting bacterial or fungal infections, may lead to prolongation of viral clearance.6,7,16 Currently, research on viral shedding between different variants is relatively scarce. Previous data suggest that higher viral loads may result in delayed viral clearance, and higher viral loads have been detected in patients infected with the Delta variant compared to those infected with the Omicron variant. 17 This may imply that the duration of viral shedding in Delta variant cases could be longer than that in Omicron variant cases. Another study investigated the peak exhaled breath and nasopharyngeal shedding between Alpha and Omicron and revealed no difference between the two groups. 18 Currently, there is limited research exploring the association between the duration of viral shedding between Alpha and Omicron variants in critical illness patients on MVs with secondary pneumonia. Further study with larger sample sizes is necessary to provide a better understanding.
Current evidence suggests that COVID-19 patients with secondary infections have a higher mortality rate compared to the overall number of hospitalized COVID-19 patients. 19 Our cohort study found a mortality rate of 40.4%, which was lower than the previously reported rate of 50.47% in severe COVID-19 patients with co-infections. 20 Previous studies did not find notable differences in hospitalization, ICU admission, or mortality rates among patients with Alpha, Gamma, and Delta variants. However, patients infected with the Omicron variant showed lower hospitalization rates, ICU admission rates, and oxygen requirements compared to other variants. 21 Few research specifically focuses on severe COVID-19 patients who are intubated and experience secondary pneumonia in the ICU. Our study tries to fill this gap and found that the mortality rate was higher in Omicron compared to Alpha in severe COVID-19 patients with intubation and nosocomial pneumonia. Besides, more non-survivors had a history of cancer and lower serum levels of albumin and fibrinogen. Regarding treatment, we found no significant differences in the doses of corticosteroids, administration of Tocilizumab, or enoxaparin between the two groups, except for a lower mortality rate among patients who received Remdesivir treatment during admission. A previous retrospective study demonstrated a significant reduction in overall inpatient mortality among COVID-19 patients treated with Remdesivir. 22 However, the exact mechanism by which Remdesivir lowers mortality remains unclear. Previous research has indicated that viral and bacterial co-infection can lead to excessive inflammation, which, in turn, can disrupt immune regulation and result in worse outcomes.23,24 Remdesivir, by reducing the SARS-CoV-2 viral load, may potentially minimize tissue damage and inflammation response. 25 This could partially explain why Remdesivir is associated with improved clinical outcomes in severe COVID-19 patients with secondary pneumonia. To our knowledge, our study is the first to evaluate the clinical efficacy of Remdesivir in severe COVID-19 patients on MV complicated with bacterial pneumonia.
Our study has several limitations. First, it is a retrospective cohort study with a small sample size conducted in a single center. Although we observed a longer duration of viral shedding in the Alpha group, the relationship between prolonged viral shedding in different SARS-CoV-2 variants, critical illness, and bacterial-viral co-infection remains uncertain. A larger prospective observational study is needed to further investigate this issue. Second, our hospital had a variable deisolation policy throughout the study period, which resulted in a lack of protocol in the process of checking Ct values from respiratory specimens. Nonetheless, this variability may reflect the real-world situation in which hospital policies and practices can vary over time. Finally, due to the isolation policy, certain examinations were not fully accessible, such as cardioechographic data or hemodynamic monitoring systems, which were limited.
To gain a better understanding of clinical outcomes in severe COVID-19 patients on MV with secondary pneumonia across different SARS-CoV-2 variants, a well-designed prospective clinical study is required.
Conclusion
In severe COVID-19 patients with MV and secondary pneumonia, our study discovered that the Omicron variant had a higher in-hospital mortality rate than the Alpha variant. However, malignancy, lower serum levels of albumin, and lack of Remdesivir treatment, except for the SARS-CoV-2 variant, are real independent risk factors for in-hospital mortality.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231213642 – Supplemental material for Comparison of clinical outcomes in critically ill COVID-19 patients on mechanical ventilation with nosocomial pneumonia between Alpha and Omicron variants
Supplemental material, sj-docx-1-tar-10.1177_17534666231213642 for Comparison of clinical outcomes in critically ill COVID-19 patients on mechanical ventilation with nosocomial pneumonia between Alpha and Omicron variants by Chuan-Yen Sun, Jhong-Ru Huang, Hsiao-Chin Shen, Ying-Ting Liao, Hung-Jui Ko, Chih-Jung Chang, Yuh-Min Chen, Jia-Yih Feng, Wei-Chih Chen and Kuang-Yao Yang in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666231213642 – Supplemental material for Comparison of clinical outcomes in critically ill COVID-19 patients on mechanical ventilation with nosocomial pneumonia between Alpha and Omicron variants
Supplemental material, sj-docx-2-tar-10.1177_17534666231213642 for Comparison of clinical outcomes in critically ill COVID-19 patients on mechanical ventilation with nosocomial pneumonia between Alpha and Omicron variants by Chuan-Yen Sun, Jhong-Ru Huang, Hsiao-Chin Shen, Ying-Ting Liao, Hung-Jui Ko, Chih-Jung Chang, Yuh-Min Chen, Jia-Yih Feng, Wei-Chih Chen and Kuang-Yao Yang in Therapeutic Advances in Respiratory Disease
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
