Abstract
Telavancin (TLV) is a lipoglycopeptide derivative of vancomycin (VAN), which has activity against Gram-positive aerobic bacteria, and is especially effective against methicillin-resistant Staphylococcus aureus (MRSA) and Gram-positive bacteria resistant to VAN. Comparative clinical studies of TLV have demonstrated noninferiority compared with VAN in the treatment of hospital-acquired Gram-positive pneumonia, with high cure rates for TLV-treated patients with monomicrobial S. aureus infection, including isolates with reduced VAN susceptibility. The results based on the patients’ clinical response were supported by supplemental post-hoc analyses of 28-day mortality. In Europe and the USA, TLV is approved as a useful alternative for patients with difficult-to-treat, hospital-acquired MRSA pneumonia when there are very few alternatives. The present article reviews TLV’s pharmacological characteristics and clinical efficacy resulting from clinical trials giving a detailed picture of its properties and position in the management of hospital-acquired pneumonia.
Introduction
Hospital-acquired pneumonia (HAP) is the second most common nosocomial infection. It accounts for up to 25% of all intensive care unit (ICU) infections and for more than 50% of antibacterial agent prescriptions, with mortality rates as high as 76% reported under some circumstances in ventilated patients [Vincent et al. 2009]. Factors commonly related to this increased mortality are inappropriateness of empiric antibiotic treatment, the existence of bacteraemia and the virulence of the microorganism [Inchai et al. 2015]. Treatment failure in HAP or ventilator-associated pneumonia (VAP) is often due to intrinsically drug-resistant organisms, such as methicillin-resistant Staphylococcus aureus (MRSA), Pseudomonas aeruginosa, Acinetobacter baumannii, Klebsiella spp. and Enterobacter spp., resulting in higher mortality [ATS/IDSA et al. 2005; Barbier et al. 2013].
Potential causative pathogens for HAP, according to the American Thoracic Society/Infectious Diseases Society of America (ATS/IDSA) 2005 guidelines are shown in Table 1.
Potential pathogens and recommended empirical antimicrobial therapy for the treatment of nosocomial pneumonia.

HAP, hospital-acquired pneumonia; MDR, multiple drug resistance; VAP, ventilator-associated pneumonia.
Historically, MRSA represents an important cause of HAP, the second most common bacterial aetiology in VAP in the USA, and in-hospital mortality rates in these MRSA-pulmonary infections range from 10% to 30%. Furthermore, recent reports have noted an increased prevalence in healthcare-associated MRSA pneumonia, such as community-associated MRSA (CA-MRSA) infections [Lewis et al. 2014], reflecting a rise in the nosocomial epidemiology of S. aureus; the USA300 clone has also been implicated as a cause of invasive infections among hospitalized patients [Seybold et al. 2006]. Although MRSA rates have declined rapidly in Europe, MRSA pneumonia continues to result in poor outcomes [Chastre et al. 2014].
For decades, vancomycin (VAN) was viewed by many as the ‘gold standard’ for the treatment of MRSA infections. However, during the 1990s, strains of S. aureus with reduced susceptibility to glycopeptides, such as VAN-intermediate S. aureus (VISA), heteroresistant VISA strains (hVISA) and extremely uncommon strains of S. aureus fully resistant to VAN (VRSA), have emerged in the clinical arena [Liapikou et al. 2012; Nannini and Stryjewski, 2008]. Recently, an outbreak of linezolid-resistant S. aureus (LRSA) was reported in an ICU in Madrid, Spain. In that report, 12 patients with LRSA were identified and 50% mortality was reported [Sánchez García et al. 2010].
As a result of the increased incidence and mortality of MRSA-nosocomial infections, there is an urgent need for new antimicrobials with specific activity against MRSA in empiric therapy, which should have good safety profiles, adequate tissue penetration and clinical efficacy.
Telavancin (TLV) is a new lipoglycopeptide antibiotic for the treatment of Gram-positive infections, including organisms with reduced susceptibility to VAN (e.g. VISA and VRSA), and is currently approved by the US Food and Drug Administration (FDA) for the treatment of adults with HAP and VAP caused by susceptible isolates of S. aureus when alternative treatments are not suitable [Wenzler and Rodvold, 2015]
Chemistry
TLV is a semisynthetic VAN derivative bearing both lipophilic and hydrophilic groups [Liapikou et al. 2012; Nannini and Stryjewski, 2008; Vibativ, 2015] (Figure 1). The hydrophilic (phosphomethyl) aminomethyl moiety is present at the 40 position of ring 7. The hydrophobic decylaminoethyl moiety is attached to the vancosamine sugar and is the reason for the classification of TLV as a lipoglycopeptide.

Chemical structure of telavancin.
In vitro, it has a rapid (within 10 min) bactericidal effect, which is thought to be due to its dual mechanism of action, disrupting bacterial cell-wall synthesis and membrane integrity. TLV binds with high affinity to a specific bacterial target called lipid II, which is present in the cell membrane [Higgins et al. 2005]. Fluorescent microscopy studies had demonstrated that TLV binds avidly to the division septum, the site of active cell-wall synthesis, as this site is rich in lipid II [Lunde et al. 2010].
It is postulated that the hydrophobic side chain of TLV amplifies the interaction with the bacterial cell wall, increasing binding for the terminal D-alanyl-D-alanine, which potentially improves the activity of TLV against MRSA and enterococci with the vanA gene, and would explain the 10-fold greater peptidoglycan synthesis inhibition of TLV when compared with VAN [Nannini and Stryjewski, 2008; Higgins et al. 2005].
Pharmacokinetics/pharmacodynamics
The activity of the drug in vitro is rapidly bactericidal and concentration dependent, with the ratio of area under the time concentration curve to minimum inhibitory concentration (AUC/MIC) as the best predictor of activity in animal models [Nannini and Stryjewski, 2008; Shaw et al. 2005]. The recommended dosing for TLV is 10 mg/kg administered over a 60-min period in patients 18 years of age or older by intravenous infusion once every 24 h for 7–21 days [Vibativ, 2015].
In a single dose of TLV at the FDA-approved adult dose (10 mg/kg), the mean Cmax was 87.5 μg/ml, AUC0–24 was 762 (± 81) μg h/ml, half-life was 7.5 (± 2.28) h, clearance was 11.8 (± 1.4) ml/h/kg and the volume of distribution was 115 (± 6) ml/kg [Vibativ, 2015; Shaw et al. 2005].
Preliminary results from an in vitro MRSA model suggest that free drug AUC/MIC ratios of 50–100 are associated with a 1- to 2-log decrease in bacterial counts and minimal resistance emergence, while maximal activity was observed at a total AUC/MIC of 404 [MacGowan et al. 2011].
TLV is extensively protein bound (> 90%) and its elimination half-life ranges from 6.1 h to 9.1 h at doses above 5 mg/kg, supporting once-daily dosing of 10 mg/kg over 60 min by intravenous infusion [Vibativ, 2015]. The postantibiotic effect of TLV against most Gram-positive organisms has been reported to range from 4 h to 6 h.
Penetration of TLV into possible sites of infection has been examined in healthy subjects. Based on plasma and epithelial lining fluid (ELF) pharmacokinetic data obtained from 20 healthy subjects, Lodise and colleagues used population pharmacokinetic modelling and Monte Carlo simulation techniques and found the mean AUCELF/free AUCplasma penetration ratio to be 1.01 (± 0.96) [Lodise et al. 2008]. The second study compared plasma, ELF and alveolar macrophage concentrations of TLV in 20 healthy subjects and demonstrated that TLV’s penetration into the ELF was considerable after 8 h, with a mean Cmax of 3.73 μg/l and was significant in alveolar macrophages with a Cmax of 45 μg/l after 12 h, achieving concentrations up to 8-fold and 85-fold, respectively, above TLV’s MIC90 of MRSA strains (0.5 μg/ml) [Gotfried et al. 2008]. Unlike daptomycin, the antibacterial activity of TLV is not affected by pulmonary surfactant.
The pharmacokinetic properties of TLV in paediatrics (under 18 years of age) and pregnant females have not been studied.
Biofilms on endotracheal tubes may complicate the treatment of VAP, therefore, biofilm penetration and bacterial inhibition are potentially important considerations in determining a treatment strategy (Sandrock and Shorr, 2015). Animal models evaluating TLV against biofilm-producing S. aureus (and the glycopeptide-intermediate Staphylococcus aureus strain), S. epidermidis and Enterococcus faecalis revealed that TLV performed better than glycopeptides (VAN and teicoplanin) and linezolid, with MICs for TLV 8–16 times lower than VAN [Gander et al. 2005; Chan et al. 2015].
In addition, in vitro studies show that TLV has a low propensity to select for resistant strains compared with the glycopeptide antimicrobials and linezolid [Kosowska-Shick et al. 2009].
The drug is excreted mainly via the kidneys, and 76% of the administered dose is extracted unchanged in urine. Table 2 lists recommended dosage adjustments for patients with renal insufficiency. Data from phase III studies support the dosing regimen of 10 mg/kg every 48 h given to patients receiving dialysis [Vibativ, 2015; Worboys et al. 2015].
Dosage adjustments of telavancin in patients with renal insufficiency.

Antimicrobial activity
TLV is active in vitro against broad-spectrum Gram-positive bacteria, such as S. pneumoniae, independent of penicillin susceptibility, staphylococci including MRSA, and organisms with reduced susceptibility to VAN (e.g. VISA, VRSA) and Enterococcus spp. [Vibativ, 2015; Karlowsky et al. 2015]. TLV has also shown activity against a number of anaerobes, including Actinomyces, Clostridium spp. including C. difficile, Eubacterium, Lactobacillus, Propionibacterium, Peptostreptococcus and Corynebacterium spp. TLV’s activity (MIC < 2 μg/ml) against Gram-positive anaerobes and Corynebacterium spp. was assessed in an in vitro model, where it inhibited 90% of the anaerobic isolates and 100% of the Corynebacterium isolates at concentrations of ⩽ 1 μg/ml [LaPlante and Mermel 2009; Finegold et al. 2009].
It is noteworthy that TLV was found to have the potential to kill nongrowing bacteria [Liapikou et al. 2012; Odenholt et al. 2007; Vibativ, 2015], and possesses bactericidal activity against intraphagocytic S. aureus, irrespective of resistance phenotypes (MRSA, VISA, VRSA) enabling the killing of intracellular bacteria [Barcia-Macay et al. 2006; Mendes et al. 2012]. Results from a European surveillance study showed that TLV MICs range between 0.06 μg/ml and 0.5 μg/ml for both methicillin-susceptible Staphylococcus aureus (MSSA) and MRSA, which was 2–4- fold lower than that for VAN, 4–80-fold lower than that for linezolid and 2-fold lower than that for daptomycin [Jansen et al. 2007].
TLV also has activity against Panton–Valentine leukocidin-producing and nonproducing CA-MRSA strains with a MIC90 of 0.5 μg/ml, which is the same for VAN [Nannini and Stryjewski, 2008; Saravolatz et al. 2007].
In an effort to minimize the variability in TLV MICs observed in surveillance studies, a revised TLV broth microdilution protocol has been developed. This new method is used for other lipoglycopeptides and provides more accurate MICs [CLSI, 2012]. At present, revised consensus breakpoints are under review by the European Committee on Antimicrobial Susceptibility Testing and have been approved by the FDA [Wenzler and Rodvold, 2015]. MICs for Gram-positive organisms utilizing this method have been found to be lower, suggesting greater efficacy than previously thought [Ross et al. 2014].
Consequently, the new susceptibility interpretive criteria for TLV approved by the FDA are as follows: S. aureus, ⩽ 0.12 μg/ml; Streptococcus pyogenes, ⩽ 0.12 μg/ml; Streptococcus agalactiae, ⩽ 0.12 μg/ml; Streptococcus anginosus group, ⩽ 0.06 μg/ml; E. faecalis (VAN susceptible), ⩽ 0.25 μg/ml [Karlowsky et al. 2015; Vibativ, 2015].
Utilizing this new broth microdilution method and new FDA breakpoints, a study of 10,920 nonduplicate isolates collected between 2011 and 2012 confirmed high-level activity against Staphylococcus, Streptococcus and VAN-susceptible Enterococcus. The study also confirmed that TLV has decreased activity against vanA strains of VAN-resistant enterococci [Mendes et al. 2015b].
The recent in vitro activity of TLV from two large surveillance programmes (2013 CANWARD and 2011–2013 SENTRY) is summarized in Tables 3 and 4.
Activity of telavancin and comparators against Gram-positive cocci (2013 CANWARD).Minimum inhibitory concentration (μg/ml).

MRSA, methicillin-resistant Staphylococcus aureus; NARSA, Network on Antimicrobial Resistance in Staphylococcus aureus.
Activity of telavancin and comparators against a worldwide collection of Gram-positive cocci (2011–2013 SENTRY Antimicrobial Surveillance Program).
Minimum inhibitory concentration (μg/ml).
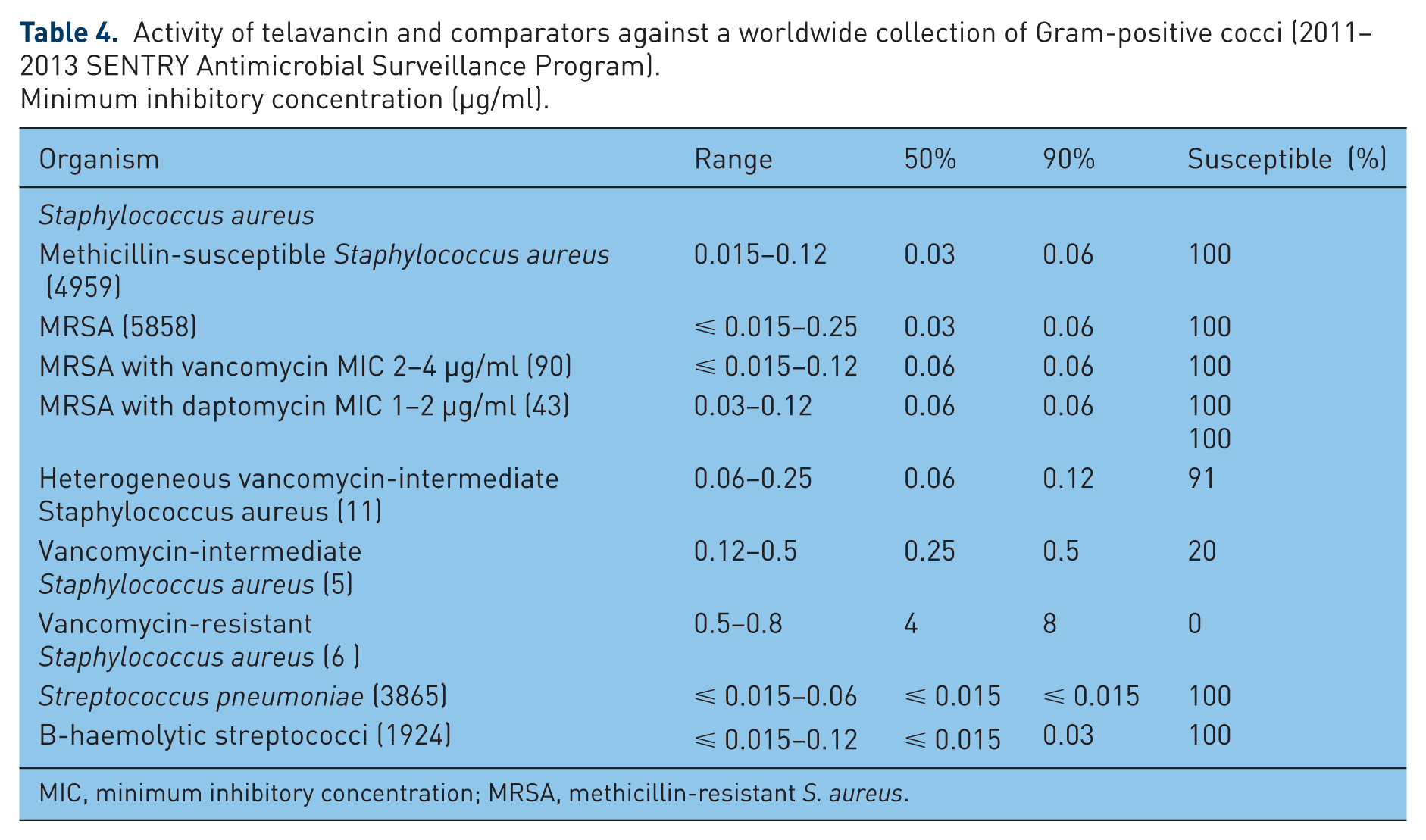
MIC, minimum inhibitory concentration; MRSA, methicillin-resistant S. aureus.
The efficacy of TLV against S. aureus isolates worldwide, regardless of methicillin susceptibility, was studied in the 2011–2013 SENTRY Antimicrobial Surveillance Program [Mendes et al. 2015c], where isolates were tested for susceptibility by broth microdilution following Clinical and Laboratory Standards Institute (CLSI) guidelines [Ross et al. 2014]. All S. aureus isolates were inhibited at a concentration <⩽ 0.12 μg/ml, with MIC50 and MIC90 values of 0.03 μg/ml and 0.06 μg/ml. When TLV activity was evaluated against S. aureus isolates with higher VAN MIC values (⩾ 2 μg/ml), including VISA isolates (MIC 4 μg/ml), a slight decrease in TLV potency (2-fold) was observed [Mendes et al. 2010], but TLV was more potent than VAN against hVISA (16–32-fold greater) and VISA (16-fold greater), but demonstrated reduced activity against VRSA (MIC5090, 4/8 μg/ml).
In the CANWARD study all S. aureus isolates tested demonstrated TLV MICs of ⩽ 0.12 μg/M ml [Zhanel et al. 2013]. TLV remained active against hVISA (median MIC/MIC90, 0.06/0.12 μg/ml), but exhibited reduced activity against the VISA (median MIC/MIC90, 0.12/0.25 μg/ml) and VRSA (median MIC/MIC90, 0.5/1.0 μg/ml) strains. In the same study TLV was the most potent agent tested against Streptococcus pneumoniae and inhibited all isolates at an MIC of ⩽ 0.03 μg/ml.
TLV was reported to have synergy when combined with all the examined antibiotics, but the highest synergy rates were observed at 24 h when subinhibitory concentrations of TLV were combined with clinically relevant, subinhibitory concentrations of gentamicin, ceftriaxone, meropenem and rifampicin [Lin et al. 2010].
Clinical efficacy in HAP
A fundamental aspect to take into account in the management of HAP is to ensure that initial treatment of HAP is appropriate and adequate, based on clinical presentation, time of onset relative to admission and the potential for multidrug-resistant pathogens. The treatment of HAP is complicated by the frequent involvement of MRSA. The therapeutic efficacy of VAN in the treatment of HAP caused by S. aureus (especially MRSA) has been questioned owing to concerns that inadequate dosing (several times because of toxicity or sepsis) leads to low concentrations of the drug in the ELF. TLV is the new alternative, approved in the USA and Europe for the treatment of HAP, especially for MRSA pneumonia.
A recent study demonstrated that TLV has equal or greater potency than the comparator antibiotics against 2279 Gram-positive pathogens implicated in nosocomial pneumonia [Pfaller et al. 2010]. TLV inhibited all staphylococci at less than 0.5 mg/l and had MIC90 values that were four-fold lower than those of VAN, teicoplanin, daptomycin, linezolid and quinupristin/dalfopristin against VAN-sensitive enterococci isolates, but was less potent than daptomycin and linezolid against VAN-resistant enterococci.
The Assessment of Telavancin for Treatment of Hospital-Acquired Pneumonia (ATTAIN) studies I and II were phase III trials that looked at the efficacy of TLV for treating HAP. The ATTAIN studies were two identical, international, double-blind, controlled trials comparing TLV (10 mg/kg/24 h) and VAN (1 mg/12 h) in the treatment of Gram-positive HAP (including VAP) for up to 21 days, in 1503 randomized patients. The first data from the ATTAIN trials were published in 2011 by Rubinstein and colleagues and met the criteria for noninferiority of TLV compared with VAN [Rubinstein et al. 2011]. A total of 1076 patients from ATTAIN were included in the all-treated (AT) population of the analysis. Cure rates in the pooled AT population were 58.9% for TLV and 59.5% for VAN (95% confidence interval [CI] for the difference, −5.6% to 4.3%), whereas in the pooled clinical evaluable (CE) patients, cure rates were 82.4% for TLV and 80.7% for VAN (95% CI for the difference, –4.3% to 7.7%).
The secondary objective of the trials was to perform a pooled analysis of the superiority of TLV over VAN in patients with a confirmed MRSA infection.
S. aureus was the most common pathogen isolated from the respiratory tract with approximately 60% (464 patients) MRSA. Clinical cure rates in patients who had S. aureus isolated at baseline (microbiologically evaluable population) were similar for TLV and VAN (78.1% and 75.2%, respectively), including MRSA (74.8% and 74.7%, respectively) subsets [Rubinstein et al. 2011]. The cure rate among patients with MRSA with reduced susceptibility to VAN (MIC ⩾ 10 ng/ml) was 87% in those who received TLV versus 74% in those who received VAN (95% CI 0.5–23.0).
It was observed that lower cure rates with TLV versus VAN occurred in patients with mixed (Gram-positive/Gram-negative) infections (66.2% versus 79.4%). However, in patients with mixed infections who received adequate Gram-negative coverage, cure rates were similar between the two groups [Corey et al. 2015].
Approximately 30% of the AT population in the ATTAIN studies had VAP. For VAP due to MRSA, clinical cure rates were much higher for TLV, 75.0% versus 57.6% for VAN (difference 17.8%, 95% CI −5.1% to 37.2%).
In addition, another post-hoc analysis of the ATTAIN study data was performed to examine the efficacy and safety of TLV for nonventilator-associated pneumonia (NV-HAP) [Rubinstein et al. 2014]. Clinical cure rates in the CE population were similar for patients with NV-HAP treated with TLV and VAN (83.1% versus 84.1%, respectively), while in the CE population of MRSA NV-HAP cure rates were 74.8% for TLV and 79.3% for VAN.
Bacteraemia is an important potential complication in HAP and may result in increased mortality [Siempos et al. 2010]. A subgroup analysis into the ATTAIN trials comparing the efficacy and safety of TLV with that of VAN in patients with HAP and S. aureus bactaeremia was performed [Stryjewski et al. 2013]. Cure rates for TLV and VAN were similar (41% versus 40%, respectively) for the 53 patients with S. aureus bacteraemic HAP, and the cure rate for MRSA infections favoured TLV (42% versus 33%), but this difference was not statistically significant.
These reports highlight the potential use of TLV as salvage monotherapy after standard therapy has failed in serious infections caused by S. aureus.
Another post-hoc analysis of the ATTAIN studies exploring the impact of TLV therapy according to the ATS/IDSA guidelines and with the primary endpoint the 28-day survival showed the noninferiority of TLV versus VAN, with similar cure rates in the ATS/IDSA-AT (59% versus 59%) and ATS/IDSA-CE (83% versus 80%) groups [Corey et al. 2014].
Only when S. aureus was the sole pathogen did cure rates favour TLV in ATS/IDSA-CE patients (86% versus 75%).
In a post-hoc analysis of data from two phase III ATTAIN trials [Torres et al. 2014], excluding those with severe renal impairment (creatinine clearance [CrCl] < 30 ml/min, including patients on haemodialysis) and patients with acute renal failure at baseline, the 28-day mortality rate in the TLV and VAN target treatment groups was similar (16.1% versus 16.9%, respectively) to the overall ATTAIN programme (20.0% versus 18.6%, respectively). Clinical cure and clinical cure plus 28-day survival rates favoured TLV over VAN in patients from whom only Gram-positive pathogens were recovered at baseline.
Safety
In clinical trials, TLV was well tolerated, with a low incidence of drug discontinuation due to adverse effects (AEs). The most common AEs that occurred in over 10% of TLV-treated patients in trials to date included taste disturbance, nausea, vomiting and foamy urine. Nephrotoxicity has been noted with TLV, with increased serum creatinine levels of up to 1.5 times baseline values reported, and was most likely to occur in older patients with underlying comorbidities that predisposed them to kidney dysfunction (e.g. pre-existing renal disease, diabetes mellitus, congestive heart failure or hypertension) or in patients receiving nephrotoxic medications.
Although the findings of the trials suggest otherwise, TLV should not be used in patients with congenital long QTc syndrome, prolongation of the QTc interval, uncompensated heart failure or severe left ventricular hypertrophy. Use of intravenous unfractionated heparin sodium is contraindicated with TLV administration because the activated partial thromboplastin time test results are expected to be artificially prolonged for 0–18 h following TLV administration [Vibativ, 2015].
In detail, in the ATTAIN trials the rates of treatment-emergent AEs leading to study discontinuation were higher in the TLV arm compared with the VAN arm (8% versus 5%). Acute renal failure was the most common AE leading to discontinuation of TLV (1.2%). Clinically significant renal function decline was also seen at a higher rate among the TLV arm compared with the VAN arm (16% versus 10%).
The ATTAIN studies showed that the safety profile of TLV influenced mortality [Corey et al. 2014]. Among patients with moderate to severe kidney dysfunction (CrCl ⩽ 50 ml/min), 28-day survival rates for TLV were lower than VAN (59% versus 70%, respectively), and in those with very severe kidney disease (CrCl < 30 ml/min), 28-day survival was again lower in the TLV compared with the VAN group (47% versus 62%, respectively).
Owing to the renal toxicity reported in the clinical development of TLV, both European and US labels for the antimicrobial contain boxed precautions for its use [Masterton et al. 2015]. In the European label TLV is contraindicated in patients with pre-existing acute renal failure and those with severe renal impairment (ClCr < 30 ml/min and patients undergoing haemodialysis). Consequently, it is advised that patients treated with TLV should have their renal function (serum Cr and urinary output) monitored daily for at least the first 3–5 days of therapy and every 48–72 h thereafter. If renal function is found to markedly decrease, then the continuation of therapy should be assessed [Samara et al. 2012].
History of approval of TLV in Europe and USA
European marketing approval for TLV was first granted in September 2011 for the treatment of adults with nosocomial pneumonia, including VAP, known or suspected to be caused by MRSA, for whom alternative treatments are not suitable [EMA, 2011].
In 2012, the European Medicines Agency (EMA) suspended a previously granted marketing authorization for TLV in the treatment of nosocomial pneumonia due to deficiencies in good manufacturing practices at the pharmaceutical supplier. A new manufacturing site was established in 2013, and EMA authorized marketing of TLV in March 2014 [Clinigen Healthcare Ltd, 2014; Clinigen Specialty Pharmaceuticals, 2014]. Afterwards, in July 2014, the National Institute for Health and Care Excellence (NICE) in the UK published advice on the appropriate use of TLV based on evidence of its efficacy and safety [NICE, 2014].
In the USA, in June 2013, after two rejections [Wenzler and Rodvold, 2015], the licensing of TLV was expanded to include the treatment of adult patients with HAP and VAP caused by susceptible isolates of S. aureus when alternative treatments are not suitable [Masterton et al. 2015]. The FDA has included a warning for TLV in patients with pre-existing moderate or severe renal impairment (CrCl ⩽ 50 ml/min). These patients who were treated with TLV for HAP/VAP had increased mortality compared with VAN [FDA, 2012, 2014a, 2014b]. In addition, TLV is approved in the USA for the treatment of patients with complicated skin and skin structure infections caused by susceptible isolates of Gram-positive bacteria, including S. aureus, both MSSA and MRSA.
In 2014, the CLSI revised its antimicrobial susceptibility testing method for TLV, resulting in MIC determinations that are more accurate and demonstrate greater in vitro potency. The CLSI testing method changes coincided with revised TLV MIC interpretive breakpoint criteria for susceptibility [Ross et al. 2014].
Conclusion
The treatment options for MRSA HAP/VAP are limited. Novel classes are clearly needed for MRSA because current drug classes exhibit treatment-limiting toxicities and emerging resistance. After VAN and linezolid, TLV is commercially available again after a long and winding road of historical hurdles involving regulatory approvals and manufacturing issues.
In two adequate and well-controlled clinical trials (ATTAIN trials), TLV has been shown to be an effective treatment of nosocomial pneumonia due to Gram-positive pathogens with demonstrated noninferiority to VAN based on the prespecified clinical response as well as a post-hoc secondary all-cause mortality (lower boundary of the 95% CI greater than -10% for both endpoints). However, among patients with severe renal impairment (CrCl < 30 ml/min), the mortality rates for the TLV group were higher than those of the VAN group. Therefore, clinicians need to consider the risk–benefit balance when choosing TLV in patients with severe renal insufficiency at baseline.
TLV’s spectrum of activity, its activity against VAN-resistant isolates of S. aureus (VRSA) and vanB VAN-resistant enterococci, does not require therapeutic drug-level monitoring as VAN does, and may have a comparable overall cost to VAN.
As scientific communities suggest, TLV should be considered for patients with HAP/VAP for whom other approved agents are not suitable, particularly those with MRSA strains exhibiting a VAN MIC ⩾ 1 μg/ml.
TLV has advantages over other glycopeptide antimicrobials in terms of its potency (at least eight-fold greater than comparators), dual mechanism of action, spectrum of activity, once-daily dosing and the lack of requirement for therapeutic drug monitoring.
Clinical outcomes with TLV as part of combination therapy are lacking despite high rates of in vitro synergy and improved bactericidal activity demonstrated with aminoglycosides, β-lactams and rifampicin [Leonard et al. 2013].
The evidence for TLV as empiric or definitive therapy for patients with known or suspected S. aureus bacteraemia has demonstrated promise [Corey et al. 2014], and suggests that TLV could potentially be used in place of combination therapy. The ongoing observational use registry (TOUR) and the phase III clinical trials in patients with complicated S. aureus bacteraemia and endocarditis should offer valuable information about real-world clinical experiences and potentially establish roles for TLV in the future. Additional clinical studies are needed to determine the role of TLV in clinical practice.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
