Abstract
Objective
We built a prediction model of mortality risk in patients the with Acinetobacter baumannii (AB)-caused hospital-acquired (HAP) and ventilator-associated pneumonia (VAP).
Methods
In this retrospective study, 164 patients with AB lower respiratory tract infection were admitted to the respiratory intensive care unit (RICU) from January 2019 to August 2021 (29 with HAP, 135 with VAP) and grouped randomly into a training cohort (n = 115) and a validation cohort (n = 49). Least absolute shrinkage and selection operator regression and multivariate Cox regression were used to identify risk factors of 90-day mortality. We built a nomogram prediction model and evaluated model discrimination and calibration using the area under the receiver operating characteristic curve (AUC) and calibration curves, respectively.
Results
Four predictors (days in intensive care unit, infection with carbapenem-resistant AB, days of carbapenem use within 90 days of isolating AB, and septic shock) were used to build the nomogram. The AUC of the two groups was 0.922 and 0.823, respectively. The predictive model was well-calibrated; decision curve analysis showed the proposed nomogram would obtain a net benefit with threshold probability between 1% and 100%.
Conclusions
The nomogram model showed good performance, making it useful in managing patients with AB-caused HAP and VAP.
Keywords
Introduction
The respiratory intensive care unit (RICU) provides centralized treatment for patients with respiratory-related critical illness. The presence of drug-resistant pathogen strains as well as patient mortality are at high levels in the RICU. As the most common pathogen leading to nosocomial infection, Acinetobacter baumannii (AB) often exhibits strong acquired resistance and environmental adaptability and dissemination. In recent years, the prevalence of carbapenem-resistant A. baumannii (CRAB) has increased considerably, as well as that of multidrug-resistant A. baumannii (MDRAB), extensively drug-resistant A. baumannii (XDRAB), and pan-drug resistant A. baumannii (PDRAB), together representing a global health threat. 1 The respiratory tract, blood, urinary tract, and wounds are common sites of AB infection. In the intensive care unit (ICU), AB-related infections account for 7.9% of ventilator-associated pneumonia (VAP) cases and 5.7% to 15.7% of bloodstream infections.2–3 Studies show that mortality owing to AB-related infection among patients in the ICU ranges from 28.3% to 84.3%. Moreover, the independent predictors of mortality vary by country and region; these are associated with initial empirical antimicrobial therapy, immunosuppression, sepsis, septic shock, and the availability or use of medical devices, among others.4–6 To facilitate timely medical intervention and improve patient prognosis, it is important to identify patients with AB-caused hospital-acquired pneumonia (HAP) and VAP in the RICU with a high mortality risk. However, few studies related to the prognosis of these patients have been carried out. The nomogram is a graphical, mathematical model that has been used to predict prognosis by estimating clinical events and integrating relevant prognostic factors in numerous diseases.7–8 The objective of the present study was to validate the prognostic factors of AB-caused HAP and VAP in terms of 90-day mortality among patients in the RICU and to develop a predictive nomogram to individually predict the probability of 90-day mortality among patients with AB-caused HAP and VAP in the RICU.
Methods
This study was retrospective and was conducted following the Declaration of Helsinki (as revised in 2013). The reporting of this study conforms to the TRIPOD (Transparent Reporting of a multivariable model for Individual Prognosis or Diagnosis) statement. 9 The Medical Ethics Committee of the Second Affiliated Hospital of Suzhou University approved the study on 2 January 2019 (No. JD-LK-2019-013-02), and informed consent was provided by all living patients. We de-identified patients' details such that they cannot be identified in any way. For patients who died, written consent for publication was acquired from a family member.
Patient selection and specimens
We retrospectively enrolled patients with AB lower respiratory tract infection (AB-LRTI) admitted to the RICU of The Second Affiliated Hospital of Soochow University from January 2019 to August 2021, including patients with HAP as well as those with VAP. These patients were randomly grouped into a training cohort (n = 115) and a validation cohort (n = 49). Available specimens included sputum, tracheal aspirate, or bronchoalveolar lavage. Sputum was collected in the morning to reduce contamination by common oral bacteria. Sterile normal saline was used to rinse the mouth before sampling, and the deeply coughed sputum was collected as the specimen. For patients who produced little sputum, a 4% sodium chloride solution was atomized and inhaled to induce expectoration, and the sputum was immediately sent for examination. The criteria for sputum specimens were sputum smear microscopy per low-power field showing <10 squamous epithelial cells, >25 multinucleated leukocytes or squamous epithelial cells/leukocytes ≤1:2.5. Samples of lower respiratory tract aspirate or bronchoalveolar lavage fluid obtained using a bronchoscope under strict aseptic conditions were placed in a sterile culture bottle and immediately sent for culture. Respiratory samples were inoculated on blood agar plates and plates containing chocolate agar selective medium for the detection of Haemophilus spp., and then placed in a 5% to 10% CO 2 incubator for 18–24 hours at 35°C. The bacteria were isolated, purified, and then identified using a VITEK 2 Compact automatic microbial analyzer (bioMérieux, Craponne, France). Using the MIC method to assess levels of drug sensitivity, we judged the results according to guidelines of the Clinical & Laboratory Standards Institute (2014). Once AB was isolated, we evaluated whether the patient had AB colonization or infection. Some scholars define respiratory bacterial colonization as the isolation of bacteria from respiratory samples without substantial laboratory or clinical alterations supporting the diagnosis of infection. 10 In China, it is suggested 11 that when patients are infected with AB, apart from the general manifestations of bacterial infection (such as fever, elevated white blood cell or neutrophil counts, and increased C-reactive protein), the following should also be considered: 1) new, continuous, or aggravated pulmonary imaging findings, clinical symptoms, and signs consistent with pneumonia; 2) the patient’s internal factors, including accompanying diseases, autoimmunity, prior use of antibiotics, and other related risk factors, such as duration of mechanical ventilation; 3) treatment with antibiotics leads to temporary improvement followed by worsen, consistent with the AB infection; 4) the clinical value of positive cultures is evaluated according to the methods used and quality of samples, bacterial concentration (quantitative or semi-quantitative culture), and results of bacterial smear; 5) sputum cultures show a pure or predominant culture of AB more than twice.
After identifying whether patients had AB colonization or AB infection, we enrolled those in whom we could isolate pure AB without other pathogens or other gram-negative bacteria, using the same semi-quantitative culture methods. These patients were all consistent with LRTI, which met the criteria of the European Respiratory Association and European Association of Clinical Microbiology and Infectious Diseases, 12 with an acute illness course (≤21 days), a common symptom of cough, and at least one symptom of LRTI such as expectoration, dyspnea, wheezing, or chest pain, not related to other causes such as rhinitis or bronchial asthma. All patients also conformed to the criteria for hospital-acquired infection; 13 infection, including HAP and VAP, occurred ≥48 hours after the patient was admitted to the hospital but did not exist or was not in the latent stage when admitted to the hospital. The definitions of HAP and VAP met the applicable criteria according to the guidelines of the American Thoracic Society and Infectious Diseases Society Of America. 14 The clinical data of these patients were reviewed and analyzed.
Inclusion and exclusion criteria
The inclusion criteria were as follows: 1) patients over 18 years old; 2) the available specimens included sputum, tracheal aspirate, or bronchoalveolar lavage; 3) patients met the criteria for AB-LRTI and hospital-acquired infection, including HAP and VAP; and 4) patients with the first AB infection.
The exclusion criteria were: 1) contaminated samples or duplicate strains isolated from the same patient; 2) non-AB-infected or AB-colonized patients; 3) co-infection (i.e., simultaneous infection with AB and other pathogens); and 4) patients with missing data.
Co-infection here refers to AB and other gram-negative pathogenic bacteria having the same semi-quantitative culture results in which anti-AB treatment alone is ineffective and targeted combined anti-infection treatment based on the drug sensitivity characteristics of both pathogens is effective. With AB and other pathogenic non-gram-negative bacteria, such as gram-positive bacteria, fungi, or atypical pathogens, anti-AB treatment alone is ineffective; however, treatment targeting A. baumannii and gram-positive bacteria, fungi, or atypical pathogens is effective. After 72 hours of anti-infection treatment, the treatment efficacy was assessed. We defined clinical treatment effectiveness as a decrease in the expected heat peak, the patient’s symptoms and signs are alleviated, inflammatory indicators are decreased, and imaging shows greater absorption of lung lesions than previously.
Data collection
Clinical data included the patients' medical history during hospitalization and in follow-up visits. The following data were collected: 1) patient characteristics, such as age and sex; 2) complications, such as chronic pulmonary diseases, cerebrovascular diseases, cardiovascular diseases, diabetes mellitus, and immunosuppression; 3) use of antibiotics within 90 days prior to isolation of AB, such as carbapenems, β-lactams/β-lactamase inhibitors, or cephalosporins; 4) clinical manifestations, such as fever, yellow phlegm, septic shock, white blood cell count >10 × 109/L or <4 × 109/L, neutrophil-lymphocyte ratio, platelets <100 × 109/L, serum albumin <30 g/L, or high-sensitivity C-reactive protein >10 mg/L; and 5) other, including receiving invasive mechanical ventilation before isolation of AB, prolonged invasive mechanical ventilation, days in the ICU, Acute Physiology and Chronic Health Evaluation (APACHE) II score, AB-caused HAP, AB-caused VAP, infection with CRAB, ≥4 invasive operations, and ≥3 bronchoscopies before isolation of AB. Mortality was analyzed 90 days after the first AB-caused HAP or VAP.
Statistical analysis
We used EpiData 3.1 to process the raw data and established the database after logical error correction, double-entry, and a consistency check. We used R software (R 4.0.1) for the statistical analysis (www.r-project.org). Continuous variables are expressed as mean ± standard deviation, and comparisons between groups were conducted using a t-test. Enumeration data are expressed as n (%), and comparisons between groups were performed using Pearson's chi-square test. We randomly divided the data into training and validation cohorts in a 7:3 ratio with the R “sampling” package. We also used the “glmnet” package in R to perform least absolute shrinkage and selection operator (LASSO) regression, the “rms” package to draw the nomogram, and the “pROC” package to plot the receiver operating characteristic (ROC) curve. The 27 variables mentioned above were taken as independent variables, and the probability that patients would die within 90 days was considered a dependent variable. In the training cohort, we used LASSO regression to screen potential prognostic factors. We analyzed the selected elements in a multivariate Cox proportional hazard model to identify significant prognostic factors associated with 90-day mortality of AB-caused HAP and VAP among patients in the RICU. We constructed the nomogram prediction model based on the results. In repeated sampling 1000 times using the bootstrap method, the nomogram was internally verified, and the validation cohort was verified as external validation. The concordance index (C-index), ROC, and area under the ROC curve (AUC) were used to assess model discrimination. A calibration curve and decision curve analysis (DCA) were used to evaluate the efficiency and practicability of the nomogram in both the training and validation cohorts. p-values less than 0.05 indicated statistical significance.
Results
Clinical characteristics
A total of 164 patients were included in this study, 29 patients with HAP and 135 with VAP. The average age of these patients was 71.7 ± 14.8 years, and 78.0% (128/164) were men. The training cohort comprised 115 patients, and the validation cohort included 49 patients. The comparison of baseline data showed no significant difference between the training and validation cohorts (Table 1), including for age, sex, complications, use of antibiotics within 90 days of isolating AB, clinical manifestations, and other factors (receiving invasive mechanical ventilation before AB isolation, prolonged invasive mechanical ventilation, AB-caused HAP, AB-caused VAP, days in the ICU, APACHE II score, CRAB, ≥4 invasive operations, ≥3 bronchoscopies before isolating AB).
Demographic and clinical features of patients in the training and validation cohorts.
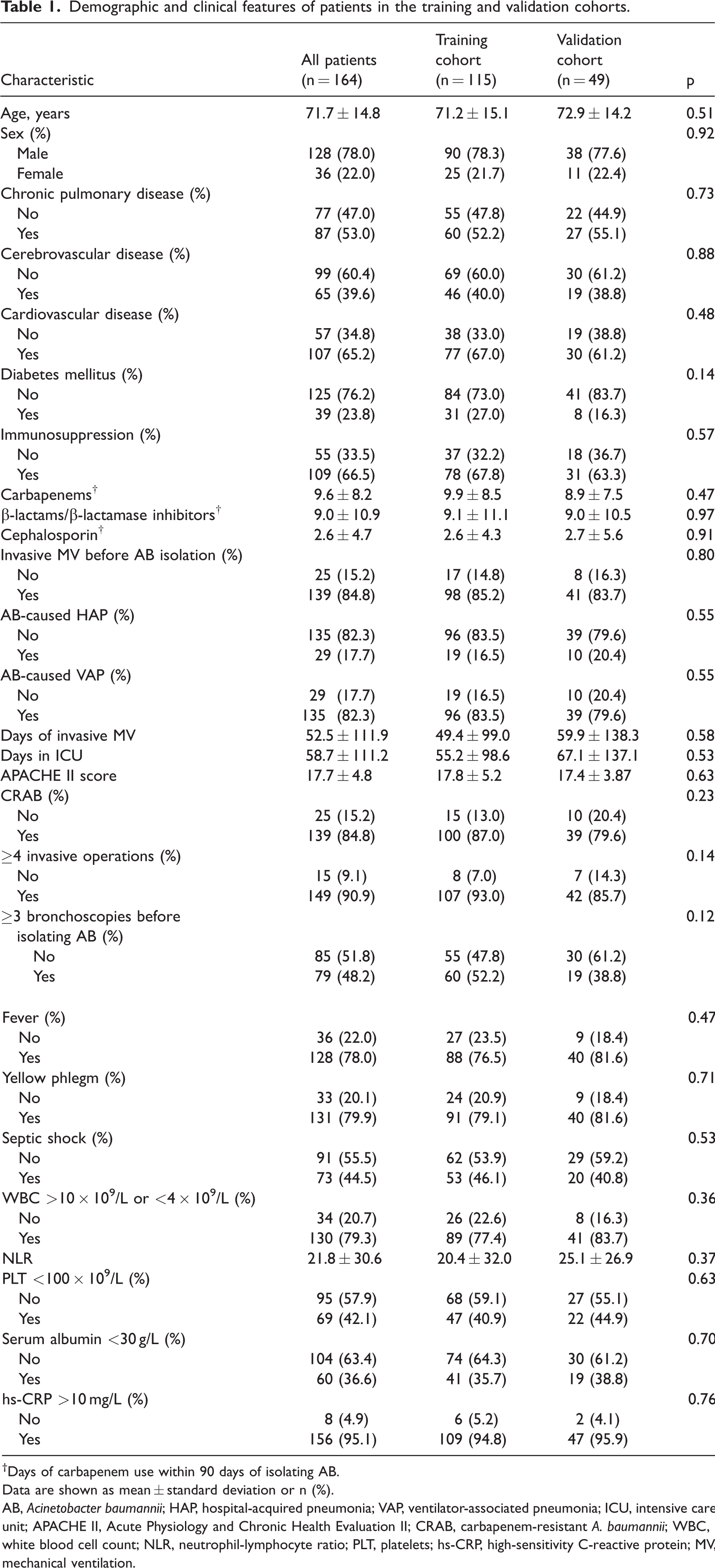
†Days of carbapenem use within 90 days of isolating AB.
Data are shown as mean ± standard deviation or n (%).
AB, Acinetobacter baumannii; HAP, hospital-acquired pneumonia; VAP, ventilator-associated pneumonia; ICU, intensive care unit; APACHE II, Acute Physiology and Chronic Health Evaluation II; CRAB, carbapenem-resistant A. baumannii; WBC, white blood cell count; NLR, neutrophil-lymphocyte ratio; PLT, platelets; hs-CRP, high-sensitivity C-reactive protein; MV, mechanical ventilation.
We initially found that the 90-day mortality of AB-caused HAP and VAP was 56.7 (93/164), and that of VAP alone was 51.2% (84/164). The 90-day mortality of CRAB was 57.9% (95/164). In 87.6% (64/73) of patients with septic shock, this was owing to AB-caused VAP (Table 1).
Prognostic factors of 90-day mortality in patients with AB-LRTI in the RICU
The 27 variables were screened in the training cohort using LASSO regression, with 10-fold cross-validation to select the optimal lambda parameter. When the minimum cross-validation error was taken as the optimal lambda value of the model, we counted the corresponding number of variables of the non-zero regression coefficient (Figure 1a–1b). LASSO regression suggested that days in the ICU, CRAB, days of carbapenem use within 90 days of isolating AB, and septic shock were prognostic variables of 90-day mortality among patients in the RICU with AB-caused HAP and VAP (Table 2). Cox regression was used to test these four variables in the training cohort. The occurrence of 90-day mortality after AB-caused HAP and VAP in the RICU was taken as the dependent variable (occurrence = yes, non-occurrence =no). The assignment of independent variables was as follows. The original values of days in the ICU and days of carbapenem use within 90 days of isolating AB were entered, and the presence of CRAB and septic shock (occurrence = yes, non-occurrence = no) was assessed. Cox regression analysis showed that the three variables CRAB, septic shock, and days of carbapenem use within 90 days of isolating AB were independent risk factors for 90-day mortality among patients in the RICU with AB-caused HAP and VAP (odds ratio [OR]: 16.432, 7.230, and 1.131, respectively; all p < 0.05). Days in the ICU was somewhat predictive of 90-day mortality owing to AB-caused HAP and VAP among patients in the RICU (OR: 0.963, p < 0.05) (Table 2).
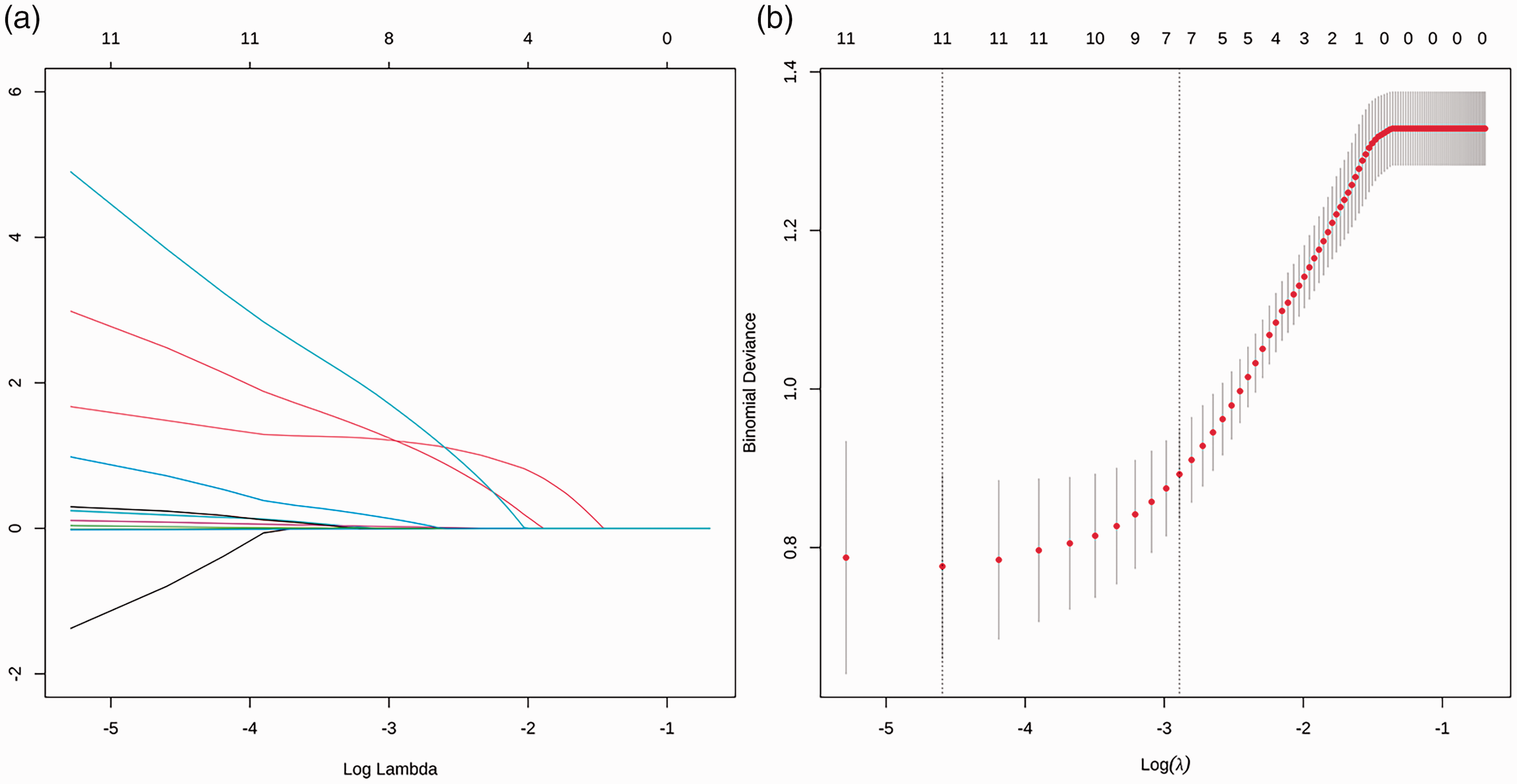
Twenty-seven coefficient penalty plots of 90-day death-related risk factors, number of 90-day mortality-related risk factors in LASSO regression to obtain optimal lambda parameter values. (a) Lambda = 0.05556 and (b) number of variables with a non-zero regression coefficient was seven
Univariate and multivariate logistic regression models in the training cohort.

ICU, intensive care unit; CRAB, carbapenem-resistant Acinetobacter baumannii; OR, odds ratio; CI, confidence interval.
†Days of carbapenem use within 90 days of isolating AB.
Nomogram construction and validation
The four independent prognostic factors (days in the ICU, CRAB infection, days of carbapenem use within 90 days of isolating AB, septic shock) were incorporated to establish a model for predicting the risk of 90-day mortality among patients in the RICU with AB-caused HAP and VAP (Figure 2). In plotting the nomogram, the corresponding point index for each prediction was first obtained, and the total score was recorded. Then, the total risk score was predicted by calculating the risk of 90-day mortality owing to AB-caused HAP and VAP in RICU patients. For example, the cumulative score of various predictors in a patient who spent 25 days in the RICU (97.5 points), had CRAB infection (8.0 points), no septic shock (0 points), and 10 days of carbapenem use within 90 days of isolating AB (4.0 points) was 97.5 + 8 + 0 + 4 = 109.5 points. Thus, the patient's corresponding predicted risk of death was 0.65 (65%) (Figure 2). The predicted risk value could then be used, which may facilitate timely medical intervention and improve the prognosis of such patients (Figure 2). We used C-index, ROC, AUC, calibration plots, and DCA to assess the performance of this nomogram. The C-index of the predictive model in the training and validation cohorts was 0.922 (95% confidence interval [CI]: 0.873–0.970) and 0.823 (95% CI: 0.706–0.941) (Figure 3a–3b), respectively. The AUC of 90-day mortality was 0.922 (95% CI: 0.873, 0.971) and 0.823 (95% CI: 0.703, 0.943) among RICU patients in both cohorts, respectively (Figure 3c–3d), which demonstrated stable and favorable performance of the model in AB-caused HAP and VAP. The calibration curve indicated a high degree of calibration (Figure 3a–3b). Moreover, the DCA showed that with a threshold probability between 1% and 100%, using the proposed nomogram to predict 90-day mortality would obtain a net benefit for patients in the RICU with AB-caused HAP and VAP (Figure 3e–3f).
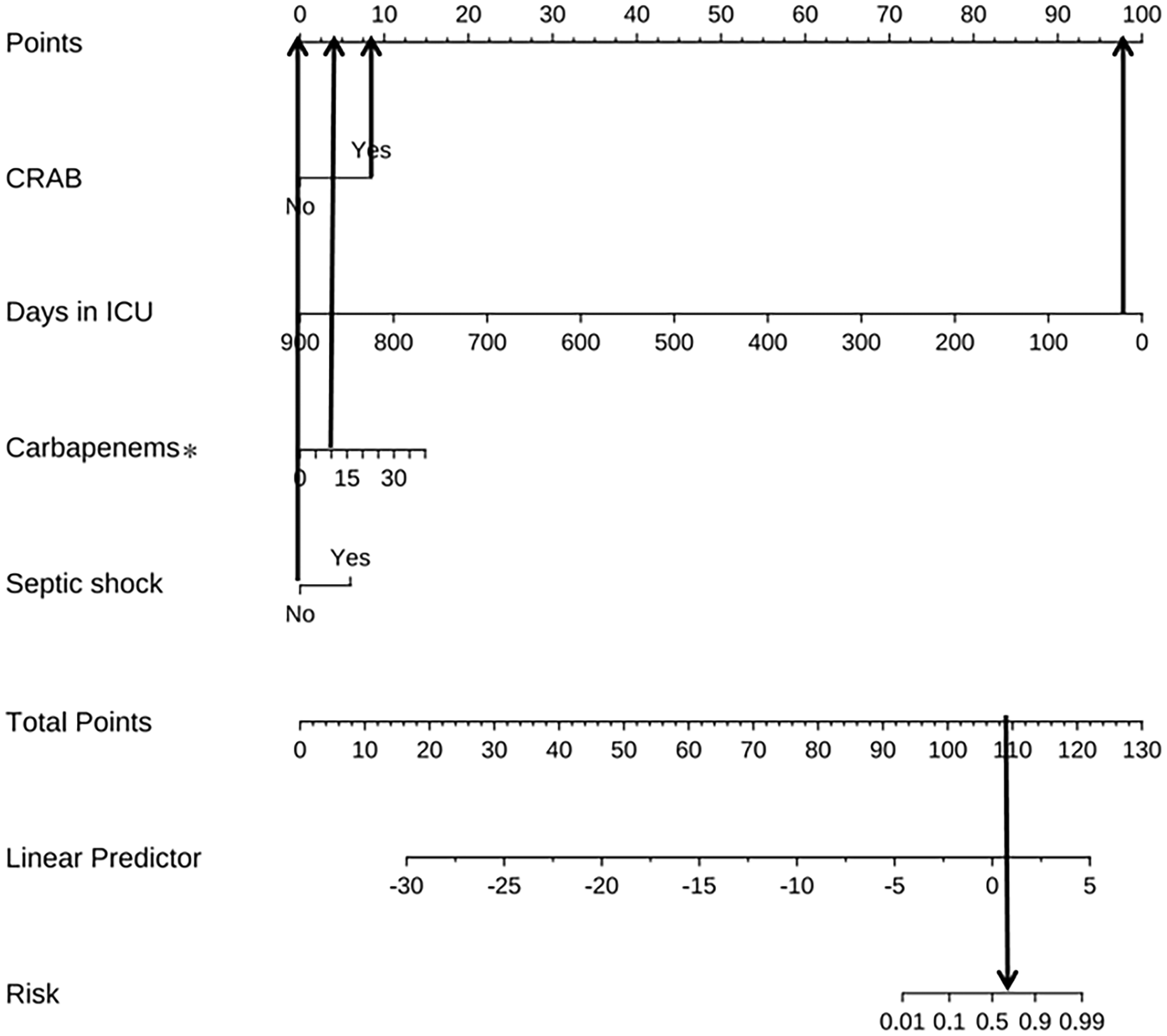
Example of prediction nomogram for the risk of 90-day mortality among patients in the RICU with AB-caused HAP and VAP.

Performance of the nomogram. Calibration curves for validating calibration of the nomogram. (a) Training cohort; (b) validation cohort. ROC curves for validating discrimination of the nomogram. (c) Training cohort; (d) validation cohort (AUC 0.922 vs 0.823). DCA curves for validating net income of the nomogram. (e) Training cohort and (f) validation cohort.
Discussion
In this study, the 90-day mortality of AB-caused HAP and VAP of patients in the RICU was 56.7% (93/164), consistent with previous studies reporting mortality of AB-related infections in the ICU ranging from 28.3% to 84.3%. These studies attempted to identify all predictors of mortality, including ineffective empirical antimicrobial therapy, immunosuppression, sepsis, septic shock, mechanical ventilation, ICU stay, malignancy, Pitt bacteremia score, and APACHE II score at onset of infection.4–6
An extended stay in the ICU is a risk factor for developing AB infection. 4 There is little research on the relationship between days in the ICU and the mortality of AB infection. In this study, we found a significant correlation between days in the ICU and 90-day mortality of AB-caused HAP and VAP. Figure 2 shows that the number of days in the ICU was a predictive factor for 90-day mortality among patients in the RICU with AB-caused HAP and VAP (OR: 0.963, p < 0.05), which may mean that patients who remain in the ICU for a long time have fewer points on the nomogram than those with shorter ICU stays. In our study, patients with a shorter stay in the ICU were usually critical. Most had ineffective empirical antimicrobial therapy, immunosuppression, sepsis, septic shock, and mechanical ventilation, which may lead to poor prognosis or death in a short time. AB may have a more subtle relationship in patients who have longer ICU stays. This mainly depends on treatment of the primary disease and the patient's immune status. AB can adhere to the respiratory tract or invasive pipeline in biofilm where it can survive and colonize the host, making these bacteria difficult to remove; thus, the presence of AB can exist in some patients for a long time. These patients are at risk for recurrent AB-LRTI. A 3-year study at three ICUs in a Thai tertiary care center used a multifaceted intervention aiming to reduce colonization and infection with PDRAB. The authors found that the rate of nosocomial AB infection in patients with AB colonization after admission was higher than that in patients without colonization. 15
Zhang H. et al. 16 found that CRAB was an essential factor predicting the risk of death from AB infection. We also found that CRAB infection was an independent risk factor for 90-day mortality owing to AB-caused HAP and VAP among RICU patients. However, there were some differences between that past study and ours. Zhang H. et al. covered different infection sources and there were no restrictions on the ward; our study focused on AB-LRTI in the RICU, which was more focused on patients with respiratory-related critical illness. The proportion of patients with CRAB infection in this study was 84.8% (139/164), consistent with reports of severe drug resistance worldwide. Data from the China Antimicrobial Surveillance Network show that the resistance rate of AB to carbapenems increased from 31% in 2005 to 78.1% in 2018. 17 The rate of hospital-isolated CRAB in Vietnam, Singapore, and other Asia-Pacific countries also rose from 46.7% in 2005 to 90% in 2016. 18 A study in Pakistan found that among 681 (41%) respiratory tract specimens of AB infection, 583 (85.5%) were carbapenem-resistant. 19 The high rate of CRAB worldwide has attracted widespread attention.17–20 For every finite treatment option, CRAB has become a serious clinical challenge.1,21 A previous systematic review and meta-analyses including 16 observational studies reported that mortality in patients infected with CRAB was twice as high as that in patients infected with carbapenem-susceptible A. baumannii. 22 Another systematic review and meta-analysis found that the global prevalence of multidrug-resistance among AB-caused HAP and VAP was 79.9% (range, 60% to 87%), pooled from 114 studies; the overall mortality estimate pooled from 27 studies was 42.6% (range, 38% to 48%). 23 A recent meta-analysis of 1706 patients infected with CRAB included 899 (52.7%) reported deaths. 24 Our study enrolled 164 patients with AB-LRTI, which included 17.7% (29/164) with HAP and 82.3% (135/164) with VAP; the proportion of CRAB infections was 84.8%, and all of them were MDRAB. The 90-day mortality of CRAB was 57.9% (95/164), which was consistent with previous research.
According to multiple logistic regression analysis, longer carbapenem use within 90 days of isolating AB was related to higher mortality of AB-caused HAP and VAP in the RICU (OR: 1.13; 95% CI: 1.029–1.243). In recent years, AB and carbapenems usually appear in a bundled mode; the extensive use of antibiotics in the clinic has exerted intense selection pressure on AB, especially with use of carbapenems and third-generation cephalosporins, which have promoted the evolution and spread of drug resistance of AB in the hospital. 25 Özgür et al. 4 found that carbapenem exposure may be involved in the gene regulation of XDRAB, increasing the virulence of sensitive strains and the number of drug-resistant strains. Recently, researchers have found that owing to selection by imipenem, the host genome of A. baumannii ATCC 17978 and the blaOXA-23-producing plasmid pAZJ221 have co-evolved to improve the adaptability of AB. 26 The carbapenemase-producing plasmid plays an essential role in the formation and transmission of CRAB, and blaOXA-23 is the most common carbapenemase-encoding gene in CRAB in China. 27 This could explain why longer carbapenem use within 90 days of isolating AB causes higher mortality owing to AB-induced HAP and VAP. Our study findings help to fill a research gap regarding the effect of the duration of carbapenem use before AB isolation on mortality among RICU patients with AB-caused HAP and VAP.
We further found that septic shock in AB-induced HAP and VAP was related to higher mortality (OR: 7.230; 95% CI: 2.078–25.154). The specimens in our study were sputum, tracheal aspirate, or bronchoalveolar lavage, excluding blood culture specimens, and we only analyzed nosocomial AB-LRTI; therefore, septic shock in our study was related to AB-induced HAP and VAP, and 87.6% (64/73) of cases of septic shock were related to AB-caused VAP. Septic shock is a manifestation of severe infection that often occurs in critically ill patients, who are more likely to have a poor prognosis. Dysregulation of the response to infection caused by sepsis and septic shock can be life-threatening and is associated with mortality rates above 30%, despite advances in organ support and antibiotic therapy. 28 In our study, the proportion of patients with CRAB infection was high, and all isolates were MDRAB. Busani 29 reported that patients with septic shock caused by MDRAB in the ICU had an increased risk of death and a mortality rate of 62.5%. A retrospective analysis in Italy found that ICU patients with septic shock owing to MDRAB infection had higher 30-day mortality than those with Klebsiella pneumoniae carbapenemase-producing K. pneumoniae (84.8% vs. 44.5%, p < 0.001). 30 Our study results were consistent with the above research.
The strengths of the present study are that, to our knowledge, this was the first nomogram for predicting 90-day mortality among patients in the RICU with AB-caused HAP and VAP. The characteristics of AB-caused HAP and VAP were comprehensively assessed; we also described the identification of AB colonization and infection in detail. The scope of this study was mainly the RICU; we constructed a prognostic signature and nomogram for predicting 90-day mortality risk owing to AB-caused HAP and VAP in the RICU. This model showed good performance and may be applied clinically to manage the high risk of death owing to AB-caused HAP and VAP, promote patient recovery, and relieve the pressure of ICU bed shortages. However, this study also has certain limitations. The retrospective nature of the study has inherent limitations of missing variables and avoiding selection bias. The included data were from a single center and were limited, affecting the universality of the results. Our model is unsuitable for other hospital departments and infection sites; further comparisons can be made by including non-ICU data and other infection sites.
Conclusions
In this study, we found that the number of days in the ICU, infection with CRAB, number of days of carbapenem use within 90 days prior to isolating AB, and septic shock were significantly associated with 90-day mortality among patients in the RICU with AB-caused HAP and VAP. The nomogram prediction model constructed using the above four indexes can be used for individualized and graphical prediction of the prognosis in patients with AB-caused HAP and VAP who are in the RICU. Effective and timely treatment can result in a good prognosis for these critically ill patients. However, this was a single-center retrospective study with a limited sample size. Therefore, our study results should be confirmed in a prospective multicenter, large-scale study.
Footnotes
Acknowledgements
We gratefully acknowledge the role of all our colleagues, nurses, and others involved in the care of patients.
Authors’ Contributions
Conception and design: Y Pei, Y Huang; Administrative support: T Zhou, M Shi; Provision of study materials or patients: Y Pei, Y Huang, Z Yao, C Chen; Collection and assembly of data: Y Pei, X Pan, A Zhong, Y Xing; Data analysis and interpretation: Y Pei, Y Huang, B Qian, T Zhou; Manuscript writing: All authors; Final approval of manuscript: All authors.
Data availability statement
Data will be provided upon reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Development Program of Suzhou, China (grant no. SYS2020025, SLJ202008, SLT201930).
