Abstract
Background:
The tuberculin-purified protein derivative (PPD) test is commonly used as a screening tool for tuberculosis (TB). However, the traditional judgment standard of the PPD test is influenced by subjective factors, which can lead to less accurate and intuitive test results.
Objectives:
To evaluate the accuracy of ultrasonography as a novel auxiliary judgment method for the tuberculin-PPD test and its clinical application.
Design:
This study was designed as a comparative study following the STROBE guidance.
Methods:
From February to May 2022, 208 patients with active tuberculosis infection were enrolled. Manual judgment and ultrasonography were employed in a double-blind-utilized manner, and the PPD examination results were recorded. Kappa statistic was performed to measure the concordance between the two diagnostic methods. Fisher’s exact test was used for the analyses of the PPD test results of all 208 active tuberculosis infection patients’ PPD results.
Results:
There was a significant difference between the two methods in the PPD result judgment (p < 0.001), particularly in the positive ratio of the PPD test results, (p < 0.05). Overall, 50 patients were determined as PPD positive based on manual judgment. However, only 24 patients’ PPD test results were determined as positive via ultrasonography. The remaining 26 patients should have been classified as strong positive but were misclassified as positive. The misdiagnosis ratio was 52% (26/50).
Conclusion:
Ultrasonography has superior accuracy to traditional manual judgment. Moreover, it does not rely on sophisticated clinical experience or training and can reveal subtle changes of the skin corresponding to each PPD test result providing intuitive results. In conclusion, ultrasonography can be used as an auxiliary interpretive approach for PPD test and has a promising future for clinical application.
Keywords
Introduction
Skin testing is a widely known diagnostic method used in allergy and infectious diseases, involving the intradermal injection of a small amount of antigen to assess either immediate IgE-mediated allergic or antigen-specific T-cell-mediated delayed-type hypersensitivity responses. 1 Tuberculosis (TB) is an airborne infectious disease caused by organisms of the Mycobacterium tuberculosis (Mtb) complex. 2 TB is severely harmful to human health because of its concealed onset and delayed healing syndromes and potential to cause multi-organ diseases.3,4 Besides, it continues to be a major cause of morbidity and mortality, primarily in low- and middle-income countries.2,5
For decades, tuberculin skin test, also known as tuberculin-purified protein derivative (PPD) skin test, has been one of the screening tools for TB in clinical practice. PPD is a substance produced by first steaming cultures of Mtb followed by repeated precipitation with ammonium sulfate to purify the proteins while reducing polysaccharide, nucleic acid, and lipid content. 1 Recently, Mosavari et al. utilized gamma radiation in the manufacturing procedure of PPD, which can eliminate the bacterium completely and can be considered as a novel and safe production technique. 6 PPD is a tool highly favored by clinicians for screening patients with suspected TB infection, as clinicians may lack laboratory experimental knowledge and it does not require experimental skills.1,7 The advantages of the PPD test are experimental stability, reaction accuracy, and less nonspecific reaction. Traditionally, the PPD test result is determined through artificial touching, visual observation, and size-measuring of skin induration. Manual judgment, however, is influenced by various subjective factors, such as the experience of the examiners and the skin condition of different patients, and may cause misdiagnosis. Hence, identifying an objective method for determining the PPD test result test has become an urgent clinical problem.
With the rapid development of the ultrasound apparatus, ultrasonography has been widely applied in the diagnosis of skin lesions,8–10 assessment of organ or tissue stiffness performance,11–13 and in other clinical areas.14–17 In this study, we aimed to apply ultrasonography and manual judgment for the determination of the PPD test result test. Briefly, the PPD test was performed on the patients. The two methods of result determination were carried out in a double-blind manner to evaluate the PPD test results. Furthermore, the ultrasonographic image of the PPD test induration were obtained to intuitively clarify the skin characteristics of the patients.
Participants and methods
Study design
This study was design as a comparative study. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement.
Research participants
Between February and May 2022, 389 patients with suspected TB-related symptoms (persistent cough, cough blood sputum, etc.) who underwent the conventional PPD test were included in the study. Ultimately, their thoracic imaging results, Mtb examination (sputum specimen), clinical symptoms, and PPD test results were examined to determine whether they had an active tuberculosis infection (ATBI), latent tuberculosis infection (LTBI), or were not infected with TB.
Among the patients, 147 were not infected with TB. Of the remaining 242 patients with TB infection, 208 and 34 were diagnosed with ATBI and LTBI, respectively. Generally, patients with LTBI do not need special treatment but need a close follow-up. Hence, the patients with LTBI were excluded from this study. Therefore, only 208 patients with ATBI (123 men and 85 women), with median age of 42.74 ± 18.06 (range, 12–82) years, were included. The screening procedure is illustrated in Figure 1.

The patient screening procedure.
Instruments and reagents
The GE-VVE 9 ultrasonic diagnostic instrument and a high-frequency probe with a frequency of 15–20 MHz were utilized in this study. Tuberculin-PPD (50 IU/mL) was obtained from Beijing Sanroad Biological Products Co. (Beijing, China).
PPD Test procedure
One-third of the middle and upper parts of the right front arm of the patient were selected, and intradermal PPD 0.1 mL (5 IU) was injected after routine disinfection. Patients were advised to avoid local heating, scratching, and contact with irritants at the test spot to not adversely affect the PPD test results. After 72 h, two diagnostic approaches, manual judgment and ultrasonography, were carried out in a double-blind manner to determine the PPD test results of all patients.
Manual judgment procedure
A ball-point pen was used to delimit the edges of the induration. The longitudinal and transverse diameters of the induration were measured using calipers. Then, the mean diameter value A (VA) was calculated, as shown in equation (1).
The classification criteria for the PPD test results were based on The People’s Republic of China Health Industry Standard – Diagnosis of Pulmonary Tuberculosis (WS288-2017), presented in Table 1.
Criteria used for the manual judgment of the PPD test results.

PPD, purified protein derivative.
Ultrasonography progress
The ultrasonography procedure was performed by two independent experienced doctors (Yi Huang and Ying Lei), with at least 5 years of experience with ultrasound diagnosis.
Patient’s forearm was laid flat on the examination bed to fully expose the PPD test area. The high-frequency probe was used to fully scan the area, obtaining the ultrasonographic features of the PPD test area and surrounding tissues, including the skin layer, superficial fascia layer, fat layer, deep fascia layer, and muscle layer (Collecting data such as tissue thickness, echo characteristics, and local blood flow distribution.) The longitudinal and transverse diameters of the PPD test area were measured and recorded with the mean diameter value B (VB), as shown in equation (2).
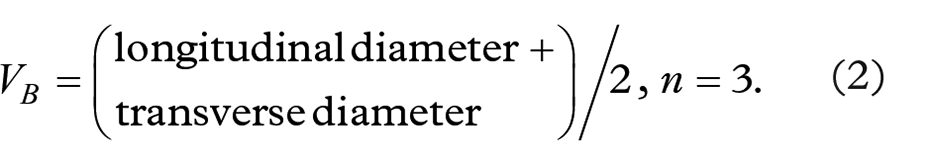
Similarly, the criteria for the determination of the PPD test results using ultrasonography is shown in Table 2.
Criteria for determination of the PPD test results using ultrasonography.

PPD, purified protein derivative.
Definition of negative/positive/strong positive ratio
The number of patients with each PPD result (negative, positive, strong positive) was calculated. Subsequently, the negative/positive/strong positive ratio was calculated by dividing the number of cases for each result by the total study sample (N = 208). For example, the positive ratio was calculated using equation (3).

Sample size calculation
This was a comparative study and the main evaluation index was the difference in PPD values measured by the two methods: manual interpretation and ultrasonography.
Refer to the method as Chow and Wang reported, 18 the p1 for ultrasonography was set to 0.9, the p2 for manual interpretation was set to 0.7, significance level α was set to 0.05, the power of 1-β was set to 0.9, and the ratio of sample size between the test group and the control group was set to 1:1. Applying the R language to calculate the sample size, the sample size of ultrasonography and manual interpretation was 79 for both methods. Considering a 10% loss to follow-up and refusal to follow-up, a sample of 176 patients was required. Hence, a total of 242 patients were included in our study, which meets the sample size requirement.
Statistical analysis
GraphPad Prism software (version 8.0.2, San Diego, CA, USA) and SPSS 26.0 were utilized for statistical analysis. Kappa statistic was applied to measure concordance between the two diagnostic methods. Fisher’s exact test was applied to analyze the difference in the determination of the PPD test results between the two diagnostic approaches. Multiple comparisons were performed using adjusted Bonferroni multiple comparison test. Statistical significance was set at p < 0.05 (two tailed). The detailed information on the two tests for the same person can be found in the Supplemental Documents.
Results
PPD test result test negative
Ultimately, 23 patients were negative via manual determination and the negative ratio was 11.06% (23/208). The color of dermatome was normal under manual observation, the insertion needle eyes were clearly visible, redness and swelling were not observed, and the obvious swollen tissue boundary was not touched by hand [Figure 2(a)]. In contrast, 19 patients were diagnosed as negative by ultrasonography and the negative ratio was 9.13% (19/208). The ultrasound images manifested no echogenic changes in the epidermal layer, dermis, and subcutaneous fat layer or epimysium, with only slightly increased echo in epidermal layer and subcutaneous fat layer with an enhancement range <5 mm [Figure 2(b) and (c)].

Diagram of a negative tuberculin-PPD test induration: (a) a PPD test-negative patient; (b and c) the epidermis, dermis and subcutaneous fat layer of skin are clear and complete with no obvious swelling.
PPD test result test positive
In total, 50 patients were diagnosed as positive via manual judgment and the positive ratio was 24.03% (50/208). The epidermis was red and swollen under observation and the edge of the swollen tissue could be touched by hand. No blisters, double circles, ulcerations, or lymphangitis were observed [Figure 3(a)]. In contrast, 24 patients tested positive using ultrasonography and the positive ratio was 11.54% (24/208). The ultrasound images showed that the epidermal layer, dermis layer, and subcutaneous fat layer were significantly thickened, and the echo was enhanced. The inner line-like structure disappeared and the epimysium layer was slightly thickened or not thickened [Figure 3(b) and (c)]. In addition, color Doppler flow imaging (CDFI) revealed blood flow in the subcutaneous fat layer [Figure 3(d)–(f)].

Diagram of a positive tuberculin PPD test induration: (a) PPD test positive patient; (b and c) the dermis and subcutaneous fat layer of the epidermis were swollen and the adventitia of the muscular layer was compromised (circle); (d–f) the dot-like blood flow signals in the subcutaneous fat layer could be observed through CDFI (arrow).
PPD test result test strong positive
In total, 135 patients were showed strong positive PPD test results by manual judgment and the strong positive ratio was 64.9% (135/208). After manual observation, the skin colliculus was found to be red and swollen, the edge of swollen tissue could be touched, the diameters of positive induration were greater than or <15 mm; however, blisters, double circles, rupture, and lymphangitis could be clearly witnessed [Figure 4(a) and (b)].

Diagram of a strong positive tuberculin PPD test induration: (a and b) skin with redness and blisters after PPD injection using manual judgment; (c and d) ultrasound image shows that the dermis is not continuous and the echo of the subcutaneous fat layer is uneven; the irregular low echo can be observed, the edema of the subcutaneous fat layer is obvious and the adventitia is obviously thickened (circle); (e and f) blood flow signals in the subcutaneous fat layer observed through CDFI (arrow).
Overall, 165 patients were determined to have strong positive PPD test results using ultrasonography and the positive ratio was 79.32% (165/208). The ultrasound images showed that the epidermis, dermis, and subcutaneous fat layer were significantly thickened, and the echo was enhanced. Besides, the linear structure in the fat layer disappeared, and the adventitia became thicker, suggesting that the PPD test induration was necrotic. Meanwhile, the images also revealed that the degree of edema in each layer of the colliculus and the echo of the subcutaneous fat layer were enhanced. A band or grid-like low echo perpendicular to the skin could be observed [Figure 4(c) and (d)]. CDFI suggested blood flow in the subcutaneous fat layer [Figure 4(e) and (f)].
Data from all study sample
Figure 5 demonstrates the diameter of the PPD test induration measured among the 208 patients with ATBI. Notably, the dispersion degree of the results in the ultrasonography is obviously higher than that in the results in the manual judgment. This is because ultrasonography not only measures the value of the PPD test results but also reflects the changes on the superficial skin directly, resulting in a higher degree of numerical dispersion in the results.
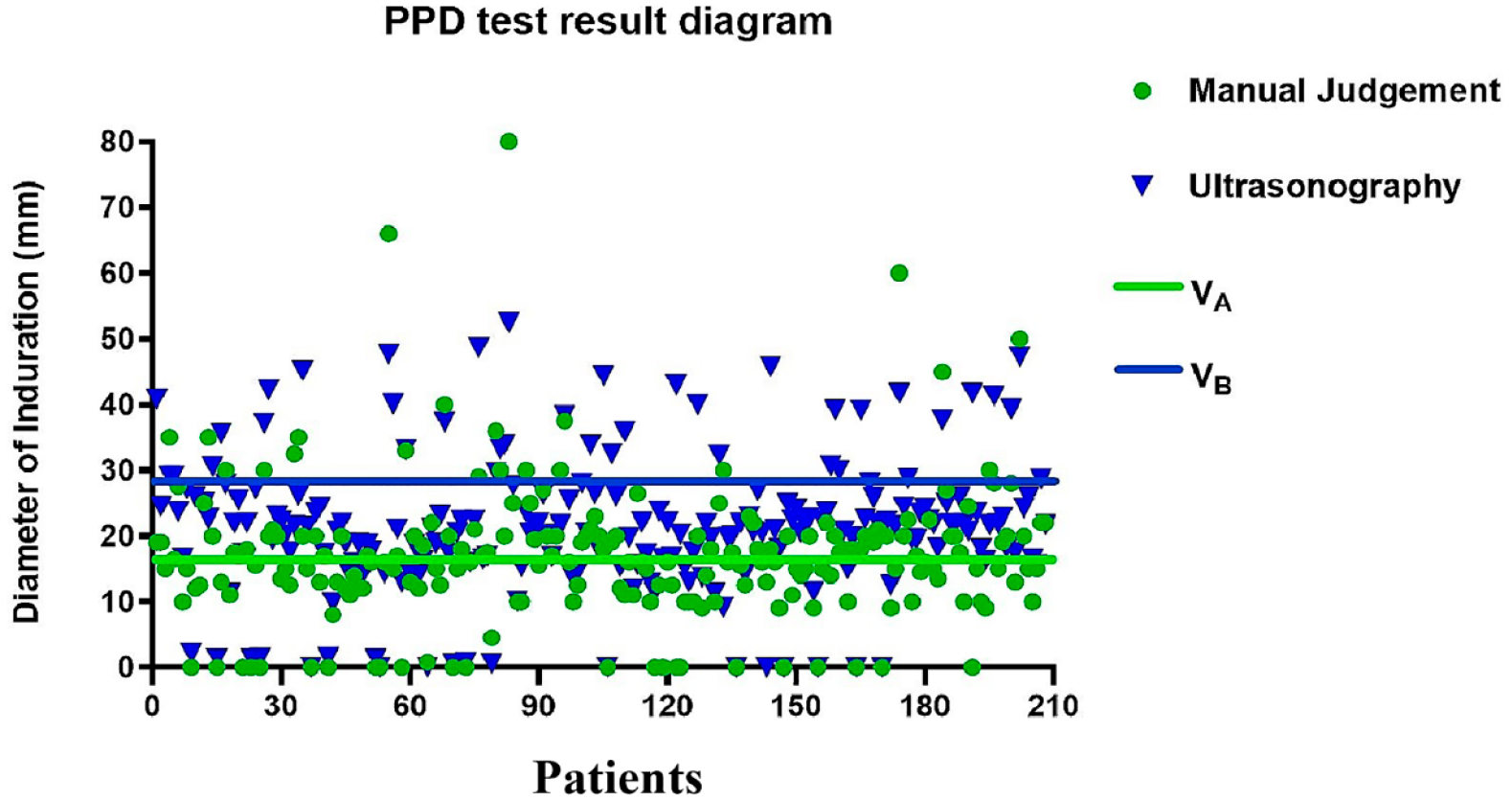
Diagram of measurements of the tuberculin PPD test induration mean diameter: (VA) mean diameter obtained by manual judgment; (VB) mean diameter obtained by ultrasonography.
As for the evaluation for the concordance between the two diagnostic methods, the Kappa statistic value was 0.635, which should be between 0.6 and 0.85 for the results to be considered relatively consistent across the two methods. In the evaluation of the PPD test results of patients with ATBI, there was a significant difference between the two methods (p < 0.001). Specifically, in the positive ratio of the PPD result judgment, a significant difference existed between the two methods (p < 0.05).
Discussion
PPD test results are necessary for guiding TB treatment. For PPD-negative patients, it is often indicated that they have not been infected by Mtb or that they have been infected but the skin allergic reaction had not occurred. Therefore, it is necessary to make a comprehensive judgment by analyzing other clinical symptoms, such as whether the patient suffered from infectious diseases, tumors, and long-term use of immunosuppressants, since PPD is also present in nontuberculous mycobacteria and in the Bacillus Calmette-Guerin (BCG) vaccine strains. 19 Accordingly, for PPD-positive and strong positive patients, the first step is to confirm whether they have been recently vaccinated (BCG vaccine) or not. Next, a series of treatment approaches, such as anti-tuberculosis drugs, are selected according to their TB symptoms.
Recently, a growing number of studies have reported on the application of ultrasonography to assess anatomic changes in dermatologic entities, not only for skin conditions but also for fungal and scalp afflictions.20–22 For skin conditions, cutaneous sonographic examination can qualify and quantify abnormalities within the skin layers and surrounding structures, making it a reliable adjunctive tool not only in the diagnosis phase but also in the assessment of the activity and severity of cutaneous diseases. 23 Furthermore, ultrasonography allows accurate tracking of inflammatory and fibrotic skin injuries in localized scleroderma, considering that the changes occur in the thickness of the dermis.24–26 The skin in the affected regions shows comparatively greater thickening compared to that in healthy regions.27,28 In fact, these research basics provided an inspiration for our study.
Compared to the traditional PPD test results method, ultrasonography can display the depth and scope of skin allergy at the site of tuberculin reaction, especially the sonographic changes of the epidermis, subcutaneous fat layer, and epimysium of induration caused by skin edema. Furthermore, it can reveal the strong echo signals in each layer, with measurement accuracy of up to 0.01 mm, making the result more precise and more compelling. In addition, the operation of ultrasonography is simple, the result determination process is rapid and does not to rely on clinical experience.
From Table 3, it can be noted that the two methods do not show any difference for PPD-negative patients with PPD negative. Nevertheless, among the 50 patients whose PPD results were manually judged as positive, only 24 were diagnosed as positive via ultrasonography. The remaining 26 patients should have been classified as strong positive but were misdiagnosed as positive. The misdiagnosis ratio was 52% (26/50). Such misclassification is strongly likely to delay the disease period and provision of treatment. As for the strong positive ratio, it can be noted that ultrasonography showed a significantly higher ratio than manual judgment (165 versus 135). From Figure 5, it can be noted that the dispersion of results obtained by manual judgment is lower than that in the results obtained by ultrasonography. Nevertheless, this is not a favorable scenario. The manual judgment method only measures the numerical value of the test result or simply observes it with the naked eye. Hence, such artificial operation may easily neglect the subtle changes of subcutaneous skin. As a result, the measured data are relatively small, resulting in a higher degree of data concentration.
Differences between the two methods in diagnosing PPD test results (N = 208).

Compared with positive results obtained by manual judgment, p < 0.05.
PPD, purified protein derivative.
Nonetheless, there were some limitations in the present study. Initially, as an immunological examination tool, the PPD test is inevitably affected by the patient’s immunity. Therefore, there is a certain probability of false-positive results being present. Hence, it is useful to combine other examination methods (e.g. interferon-gamma release assay) with ultrasonography to improve the efficiency of the PPD test. Moreover, the PPD test results were only measured 72 h later. Therefore, it is necessary to evaluate multiple time points to measure the degree of change in PPD values, to check whether the value reaches a plateau or still increases. In addition, our study was a single-center comparative study. Therefore, multicenter prospective studies should be carried out in the future to further verify the applicability and generalizability of ultrasonography for the PPD test.
Conclusion
The ultrasonography diagnosis process and examination result for the PPD test are rapid and precise, respectively. Compared with manual judgment, ultrasonography does not rely on sophisticated clinical experience or training. In conclusion, ultrasonography can be used as a distinctive approach for determining the PPD test result and it can serve as an auxiliary method for the traditional PPD test judgment approach, which has promising application in clinical practice.
Supplemental Material
sj-xlsx-1-tar-10.1177_17534666231213638 – Supplemental material for Ultrasonography – a novel auxiliary interpretive approach for tuberculin-purified pure protein derivative skin test: a comparative study
Supplemental material, sj-xlsx-1-tar-10.1177_17534666231213638 for Ultrasonography – a novel auxiliary interpretive approach for tuberculin-purified pure protein derivative skin test: a comparative study by Weijie Jiao, Yi Huang, Ying Lei, Jun Zhang and Ming Yu in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

