Abstract
A 53-year-old patient who experienced recurring upper abdominal pain and discomfort for 4 years was admitted to our hospital. Gastroscopy was performed to identify the location of the pain and evaluate the characteristics of a mass in the abdomen. Endoscopic ultrasonography (EUS) and abdominal computed tomography (CT) revealed a space-occupying lesion in the gastric fundus, suggestive of a submucosal tumor and highly likely of stromal origin. Surgical resection of the lesion was performed for identification; however, postoperative histopathological examination of the lesion revealed gastric fundus tuberculosis (TB). Gastric TB is relatively rare; therefore, clinicians should be highly suspicious of patients with abdominal symptoms from regions with a high incidence of TB to prevent treatment delay caused by misdiagnosis.
Keywords
Introduction
The gastrointestinal tract, particularly the ileocecal region, is the sixth most common site of extrapulmonary tuberculosis (TB). 1 In previous routine autopsy reports, the incidence of gastric TB ranged from 0.03% to 0.21%. 2 The relatively low incidence of gastric TB may be owing to several factors, namely the loss of gastric mucosal lymphoid tissue, rapid gastric emptying, the acidic environment, and integrity of the gastric mucosa. 3 Because the clinical manifestations in gastric TB are often nonspecific, surgery plays an important role in its diagnosis. Here, we report a patient with a space-occupying lesion in the gastric fundus that was considered a stromal tumor. The patient’s history, physical examination and laboratory examination findings were not indicative of TB, but postoperative histopathological examination revealed gastric fundus TB. To date, there have been only a few reported cases of patients with gastric fundus TB. Reporting these cases may help physicians in their diagnosis, leading to better treatment strategies.
Case report
A 53-year-old woman presented to our hospital because of “upper abdominal pain and discomfort for 4 years”; she was subsequently admitted. Four years prior to admission, no other signs and symptoms that would ascertain the etiology of the chief complaint were noted, such as pain under the xiphoid process, belching, fatigue, anorexia, fever, night sweats, throat discomfort, dizziness, dyspnea, hematemesis, melena, diarrhea, or weight loss. Furthermore, no precipitating or relieving factors for the pain were noted. Additional history indicated that the patient had no prior history of TB infection, nor was there exposure from family members. Abdominal examination revealed a flat and soft abdomen with deep tenderness under the xiphoid. No rebound tenderness or abdominal rigidity were elicited, and no mass was palpable in the abdomen. Gastroscopy revealed a submucosal mass on the posterior wall of the gastric fundus, approximately 1.0 cm × 1.5 cm in size, with a smooth surface showing mucosal bridge formation (Figure 1), and scattered erosions in the body of the stomach. The admitting diagnosis was a space-occupying lesion in the gastric fundus that was considered a stromal tumor, and non-atrophic gastritis.

Gastroscopy showing a submucosal mass in the gastric fundus.
After admission, the patient’s medical history was further probed, and she was administered oral anti-ulcer medication intermittently; however, the medication provided no relief. Ultrasonographic gastroscopy was performed to further analyze the lesion in the gastric fundus. Endoscopic examination showed an increase in the diameter of the gastric fundus to approximately 3.0 cm owing to the submucosal mass; its surface was smooth, with ulceration. Endoscopic ultrasonography (EUS) revealed a hypoechoic lesion (approximately 1.85 cm × 2.76 cm) originating from the gastric submucosa; the internal echogenicity was uniform, with clear borders and no nearby enlarged lymph nodes (Figure 2).With these findings, we suspected that the gastric submucosal mass was a stromal tumor. Abdominal non-contrast and contrast-enhanced computed tomography (CT) showed a soft tissue nodule with clear borders measuring approximately 2.3 cm × 2.0 cm protruding into the gastric cavity, with obvious enhancement; however, the local gastric wall was not significantly enhanced, and no abnormally enlarged lymph nodes were observed in the abdominal cavity (Figure 3). A diagnosis of stromal tumor, leiomyoma, or neurogenic tumor was more likely considering that the mass occupied the gastric fundus. Additionally, findings in routine blood examination, hepatitis panel, human immunodeficiency virus (HIV) and Treponema pallidum particle agglutination (TPPA) serology, liver and kidney function tests, blood coagulation tests, and the tumor markers, alpha-fetoprotein, carcinoembryonic antigen, carbohydrate antigen (CA)-153, CA-125, and CA-19-9 were unremarkable. Chest X-ray, electrocardiography, and pulmonary function test results were also unremarkable.

a: Endoscopy showing a gastric fundus submucosal bulge; b: Endoscopic ultrasonography (EUS) showing a fundic hypoechoic lesion originating from the submucosa.
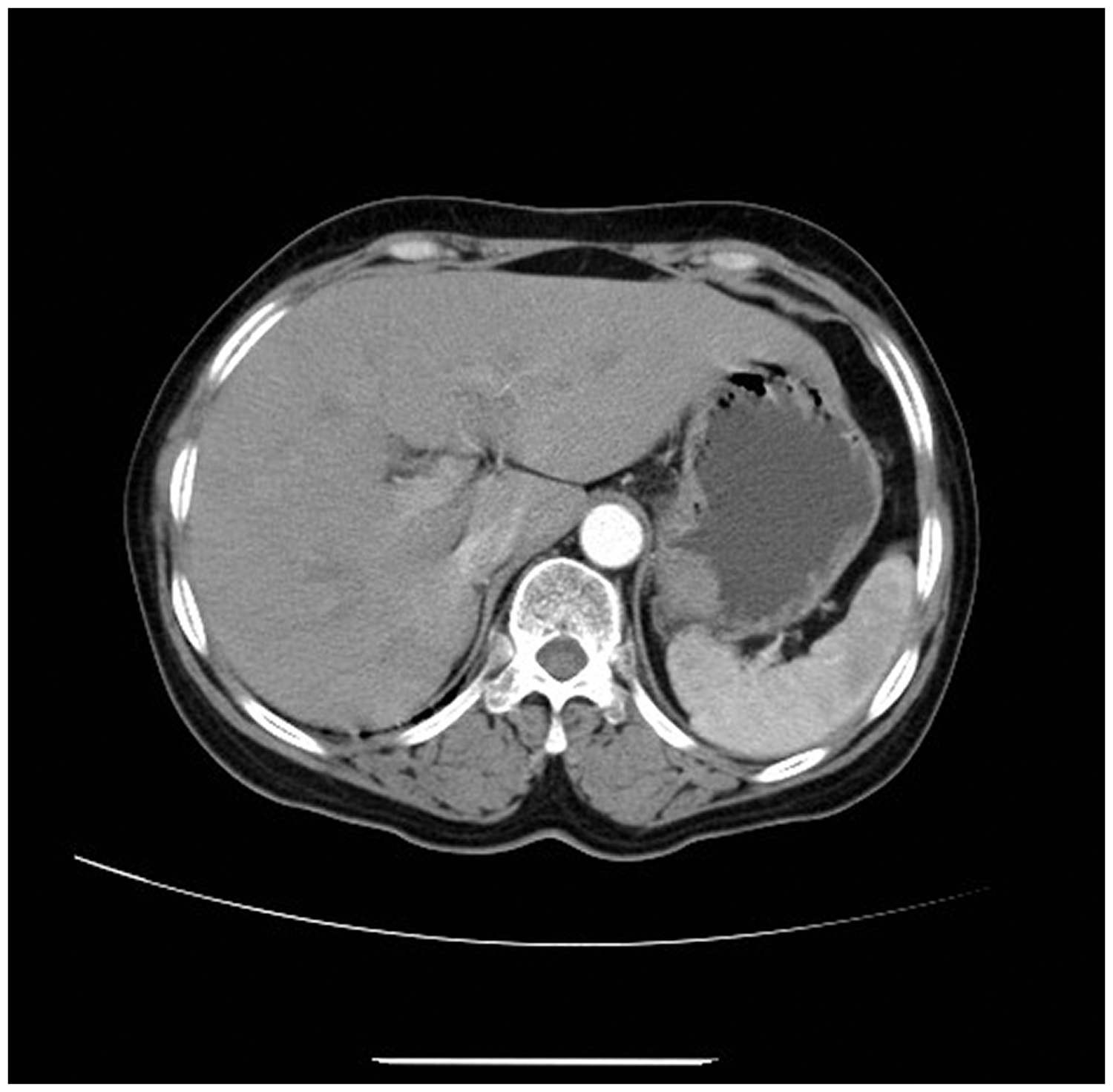
Abdominal computed tomography (CT) showing a soft tissue nodule with obvious contrast enhancement.
Considering the patient’s presentation and suspecting that the fundic mass may have deteriorated, laparoscopic surgery was performed. After exploring the abdomen, the stomach was opened, and the mass was identified and resected. Histological analysis of the resected mass (Figure 4) revealed granulomatous inflammation. Immunohistochemical staining (Figure 5) revealed that the tumor was cluster of differentiation (CD)68 (phosphoglucomutase 1, PGM1) (+), CD117 (−), CD34 (−), and DOG-1 (−). However, acid-fast staining revealed negative results. Repeated GeneXpert MTB/RIF (Cepheid, Sunnyvale, CA, USA) testing of the resected tissue confirmed the presence of Mycobacterium tuberculosis (MTB) (Figure 6) and sensitivity to rifampicin (RIF). We finally diagnosed gastric fundus TB.
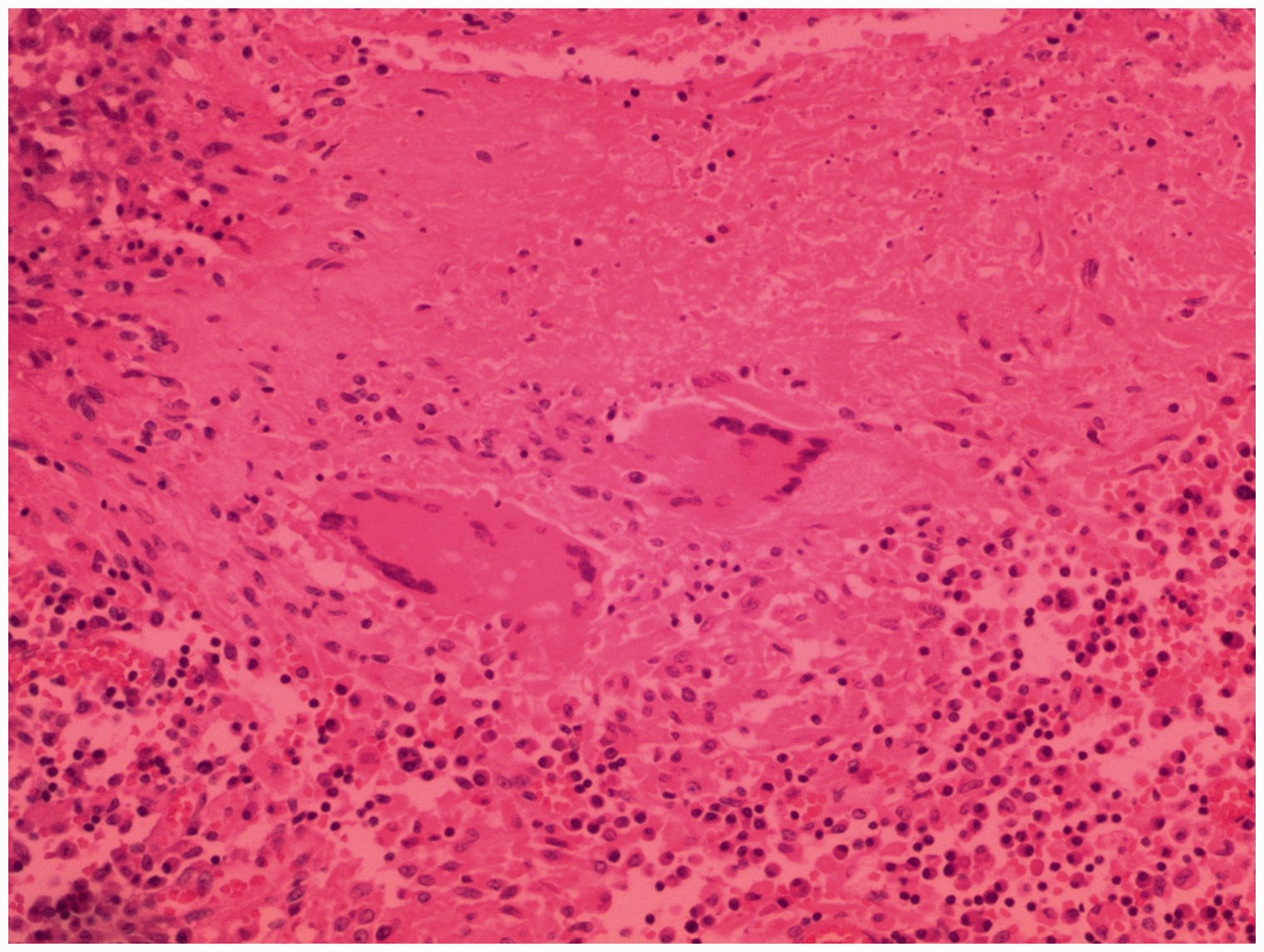
Histopathological examination findings (hematoxylin and eosin (H&E) staining × 20) showing chronic granulomatous inflammation.

Immunohistochemical staining, which was cluster of differentiation (CD)68 (phosphoglucomutase 1 (PGM1)) (+).

GeneXpert MTB/RIF testing revealed the specific amplification of five probes.
Owing to the difficulty of anesthetic resuscitation, the patient was transferred to the intensive care unit (ICU) for postoperative care. After smooth extubation and weaning, she was transferred back to our department the next day. Further treatment comprised anti-inflammatory medications, nutritional support, fluid and electrolyte balance maintenance, and symptomatic treatment. She recovered and was discharged from the hospital on the 10th postoperative day. Follow-up and continuous consultation for TB were recommended, and no postoperative complications occurred. Abdominal CT (Figure 7) and gastroscopy (Figure 8) were performed at the 3-month follow-up.

Abdominal computed tomography (CT) 3 months after the operation to re-evaluate the gastric fundus.

Gastroscopy of the gastric fundus 3 months after the operation.
Discussion
TB is an infectious disease caused by the bacteria, Mycobacterium tuberculosis (MTB). TB can cause significant harm to a person’s health and is a main cause of mortality worldwide. 4 Economically underdeveloped areas are more affected by this disease; in China, the Guizhou province has a high incidence of TB. 5 The bacteria can spread to different organs, including the gastrointestinal tract, where the ileocecal region is more commonly affected. In addition to the ileocecal region, gastric TB commonly occurs in the antrum and the prepyloric area near the lesser curvature of the stomach. These sites are rich in lymphoid follicles and are commonly involved in peptic ulcer disease. Currently, only a few cases of patients with TB involving the gastric fundus have been reported. 6
Gastric TB is often secondary to pulmonary TB or other extrapulmonary TB; however, the mechanism of infection is not fully understood. Four possible routes of infection have been proposed: 7 (1) direct invasion of the gastric mucosa by swallowing MTB; (2) hematogenous; (3) lymphatic; and (4) direct spread of TB in the gastric lymph nodes or peritoneum. Among these routes, the first may be more common; this is supported according to the use of gastric lavage to diagnose TB in children. 8 In the current report, the patient had no history of comorbid diseases; however, she lived in regions with a high incidence of TB for a significant amount of time. which may have led to exposure to the infection. The direct invasion of the bacteria by swallowing contaminated products, along with gastric atrophy with erosion, led to the colonization and growth of MTB at the base of the stomach, and gastric TB.
Ulcerative lesions and hypertrophic lesions are common pathological types of gastric TB. 9 As stated earlier, the clinical manifestations of gastric TB are nonspecific; however, abdominal pain or discomfort is a common symptom. Other manifestations are fever, symptoms of food poisoning, massive gastrointestinal bleeding, 10 , 11 and pyloric obstruction. 12 The diagnosis of gastric TB is challenging, and endoscopy often plays an important role. Gastroscopy usually reveals submucosal eminence or ulcer formation. Lesions found using EUS are described as hypoechoic lesions in the lamina propria, making its differential diagnosis from gastric stromal tumors difficult. Ultrasound-guided fine needle aspiration, EUS-guided Tru-Cut biopsy, and EUS-guided fine needle biopsy can aid in the diagnosis of TB because the lesions are often located in the submucosa; however, the diagnostic accuracy of biopsy in diagnosing gastric TB is low. 13 Polymerase chain reaction (PCR) of gastric aspirates has been used in the diagnosis of gastric TB, but the specificity is only approximately 85%. 14 Preoperatively in our patient, we considered that the mass in the gastric fundus was likely a stromal tumor according to the patient’s medical history and physical and auxiliary examination findings.
Because the clinical diagnosis of gastric TB is difficult, most patients undergo surgical intervention and are diagnosed postoperatively using pathological examinations. Our patient’s medical history, and physical and routine auxiliary examination findings did not indicate a diagnosis of TB. Therefore, to clarify the nature of the space-occupying lesion in the gastric fundus, laparoscopic exploration was warranted after communicating with the patient and other physicians. Laparoscopic exploration was performed to identify the gastric fundus tumor, which was found near the greater curvature. We subsequently resected the lesion, and postoperative pathological examination indicated that the lesion was caused by TB. Therefore, laparoscopy combined with transabdominal resection of gastric tumors may be more beneficial to patients with tumors with gastric submucosal eminence.
Conclusion
Although TB in the gastric fundus is rare, clinicians should suspect this possibility in patients with abdominal symptoms from regions with a high incidence of TB. A high clinical suspicion may aid in timely and accurate diagnosis and lead to appropriate treatment strategies, which ultimately benefits the patients and their families.
Footnotes
Acknowledgment
The authors acknowledge the patient’s participation in this study.
Ethics statement
This case report was approved by the Ethical Committee of the Affiliated Hospital of Zunyi Medical University, People’s Republic of China, in January, 2020. Written informed consent was obtained from the patient to undergo the procedures in this report and to publish this report. Our case report complied with the CARE guidelines (![]() ).
).
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by the Science Foundation of Guizhou province (No: 2020-1Y301).
Author contributions
JW and JL were responsible for managing the patient’s care. FY, XY, HL, and YC contributed to data collection and analysis. FY drafted the manuscript. JL designed and strictly revised the manuscript. All authors critically revised the manuscript and agreed to be fully accountable for the integrity and accuracy of the work. All authors have read and approved the final manuscript.
