Abstract
Background:
Biological therapies have revolutionized the treatment of severe asthma with type 2 inflammation. Although such treatments are very effective in reducing exacerbation and the dose of oral steroids, little is known about the persistence of symptoms in severe asthma patients treated with biologics.
Purpose:
We aim to describe asthma control and healthcare consumption of severe asthma patients treated with biologics.
Design:
The Second Souffle study is a real-life prospective observational study endorsed by the Clinical Research Initiative in Severe Asthma: a Lever for Innovation & Science Network.
Methods:
Adults with a confirmed diagnosis of severe asthma for at least 12 months’ duration were enrolled in the study. A self-administered questionnaire including the Asthma Control Questionnaire (ACQ), Asthma Quality of Life Questionnaire (AQLQ) and a compliance evaluation test was given to the patients. Healthcare consumption within 12 months prior to enrolment was documented. In patients receiving biologics, doctors indicated whether the patients were biologic responders or non-responders.
Results:
The characteristics of 431 patients with severe asthma were analysed. Among them, 409 patients (94.9%) presented asthma with type 2 inflammation (T2 high) profile, and 297 (72.6%) patients with a T2 high phenotype were treated with a biologic. Physicians estimated that 88.2% of patients receiving biologics were responders. However, asthma control was only achieved in 25.3% of those patients (ACQ > 0.75). A high proportion of patients (77.8%) identified as responders to biologics were not controlled according to the ACQ score. About 50% of patients continue to use oral corticosteroids either daily (25.2%) or more than three times a year for at least three consecutive days (25.6%). Gastro-oesophageal Reflux Disease (GERD) and Obstructive Sleep Apnoea syndrome (OSA) were identified as independent factors associated with uncontrolled asthma.
Conclusion:
Although a high proportion of severe asthma patients respond to biologics, only 25.3% have controlled asthma. GERD and OSA are independent factors of uncontrolled asthma.
Introduction
Asthma is a chronic heterogeneous disease affecting more than 330 million people worldwide. A significant socio-economic burden is associated with asthma, especially in its severe uncontrolled form. 1
Severe asthma is defined as asthma requiring high doses of inhaled steroids and another controller or long-term oral corticosteroids (OCSs). 2 Like asthma, severe asthma is a heterogeneous disease characterized by different features over time such as frequency and severity of symptoms, exacerbation rate, bronchial obstruction or dependence on OCSs. It is a highly disabling disease resulting in a limitation of physical activities, an alteration in the quality of life, frequent emergency department visits and professional or school absenteeism. 3 In 2019, severe asthma was estimated to affect between 4% and 6% of asthmatic patients. In France, severe asthma is estimated to affect between 0.18% and 0.51% of the French adult population. 4
Recent studies have shown that severe asthma can be divided into two phenotypes: asthma with type 2 inflammation (T2 high) and asthma with no characteristics of type 2 inflammation (T2 low). Blood eosinophil count (>0.15 G/L), rise in the exhaled fraction of nitric oxide (FeNO > 20 ppb), sputum eosinophilia (>2%) and the presence of an atopy marker (perennial aeroallergens positive skin tests and/or presence of specific IgE) are typical markers of type 2 inflammation.5,6
In the last decade, several biologics have been approved for use in patients with uncontrolled severe asthma. In large clinical trials, those biologics are associated with a significant reduction in exacerbation or OCS doses in addition to improved asthma control.7,8 These therapeutic effects were more marked in patients with elevated type 2 biomarkers. 9 This has led the GINA to recommend the use of biologics, instead of maintenance OCS therapy, in patients with T2 high severe asthma (GINA Step 5). 10
As previously mentioned, data on the use of biologics in severe asthma were obtained through clinical trials with highly selected patients.11,12 However, severe asthma patients managed in everyday clinical practice could exhibit different characteristics compared to those participating in clinical trials. Consequently, real-life studies are warranted to give broader insights into different aspects related to biologics per se and to the disease. 12
Altogether, pragmatic phase IV clinical trials, prospective studies based on collecting data (survey, cohort) and studies on healthcare databases are needed to provide robust data on biologic agents for severe asthma in real life. 12
With several novel therapies currently used and developed for asthma, it is an appropriate time to consider whether remission in asthma is now an achievable treatment target. To date, asthma remission has only been described as the spontaneous cessation of asthma disease activity (e.g. due to the transition from childhood to adulthood) and not as a therapeutic target. Asthma remission is no longer a pipe dream but could become a goal with the use of biologics. 13
In this context, we conducted a real-life national observational study based on a questionnaire to describe the characteristics of severe asthma patients. A specific analysis was performed in T2 high patients receiving biologics. The purpose of this work was first to describe the phenotypic profile distribution of severe asthma patients and second the change over time of patients treated with biologics. Care pathways, asthma control and quality of life have also been analysed. The real-life impact of biologics was evaluated according to objective measurements of asthma control and practitioners’ and patients’ perceptions.
Methods
The Second Souffle study is an observational study conducted between July 2020 and July 2021 by the CRISALIS/F-CRIN network. CRISALIS is the French Clinical Investigation Network dedicated to the Severe Asthma Labeled Excellence Research Network by the French Clinical Research Infrastructure Network (F-CRIN). Toulouse University Hospital sponsored this study with the institutional financial support of AstraZeneca. Public study data are available on the ClinicalTrial.gov database (NCT 04438408). Informed patients were recruited by their investigating pulmonologist after having given their informed oral consent pursuant to French legislation.
Eligibility criteria and patient’s recruitment
The inclusion criteria were age ⩾18 years and a confirmed diagnosis of severe asthma of at least 12 months of duration by a pulmonologist according to ATS/ERS criteria. 14
The non-inclusion criteria were participation in a therapeutic clinical trial or refusal to participate in this study, pregnant or breast-feeding women and reading and comprehension difficulties preventing the patient from completing the questionnaire.
Patients were recruited from 31 active sites in France. The investigational sites included 12 CRISALIS Network Centres and 19 other asthma sites working in collaboration with the network (either public or private hospitals). At the end of the follow-up consultation, a self-administered questionnaire was given to severe asthma patients. The pulmonologist completed in advance the medical data questionnaire required for stratification according to phenotype and biologic.
Questionnaire
The questionnaire was subdivided into seven parts as follows: part 1 was completed by the pulmonologist, and parts 2–7 by patients only. The questionnaire included 61 demographic and clinical items including standard of care self-questionnaires such as the Asthma Control Questionnaire (ACQ),15,16 Asthma Quality of Life Questionnaire (Mini-AQLQ) 16 and compliance evaluation test. 17
The ACQ was historically the first questionnaire specifically developed and validated to measure asthma control. The Mini-AQLQ was based on the AQLQ, which was developed to measure the functional impairments that are most troublesome to adult asthma patients. The Mini-AQLQ is shorter and simpler, displays strong evaluative and discriminative measurement properties, and can be used with confidence in both longitudinal (clinical trials) and cross-sectional (surveys) studies. 16 The more asthma is controlled, the lower the ACQ score: well controlled (0–0.75), partially controlled (0.75–1.5) and uncontrolled (>1.5). Conversely, the higher the AQLQ score, the better the quality of life (scores ranging from 1 to 7). Minimal clinically significant differences for both scores are reflected by a change in score of 0.5.
The compliance evaluation test questionnaire provides an assessment of treatment compliance through scoring: 0–2 corresponds to acceptable compliance (0 = good compliance, 1–2 = minor compliance problem) and ⩾3 = poor compliance.
The entire self-questionnaire was tested and validated by a panel of voluntary patients (the French Severe Asthma Patients’ Association).
Definition of subgroups
Severe asthma patients were divided into two phenotypes: asthma associated with type 2 inflammation (T2 high) and asthma with no characteristics of type 2 inflammation (T2 low). Type 2 high inflammation was defined according to one of the following criteria: systemic eosinophilia (>0.15 G/L), a rise in the exhaled (FeNO > 20 ppb), sputum eosinophilia (>2%) and the presence of an atopy marker (perennial aeroallergens positive skin tests and/or presence of specific IgE).5,6
In patients receiving biologics, doctors were invited to assess the patient as a responder or non-responder (‘subjective measure’) based on clinical, biological and functional data. In addition, an asthma control evaluation was obtained from the ACQ (‘objective measure’). Healthcare consumption within 12 months prior to enrolment and patient satisfaction were also documented.
In France, asthma patients can have long-term illness (LTI) coverage, which provides full reimbursement of asthma-related healthcare costs by the National Health Insurance scheme. The LTI status was also noted.
Statistical analysis plan and data collection
The endpoints for asthma control and patient quality of life were based on the validated scores (ACQ and mini-AQLQ). Data were available for the overall population and, subsequently, for T2 high patients receiving biologics compared to T2 high patients not receiving biologics. Patients were defined as receiving biologics if they had been treated for at least 90 days. Super-responders in the biologic group were defined as follows: ACQ < 0.75 and FEV1 > 80%, no exacerbation in the last 12 months and no OCSs in the last 12 months. 13
A bilateral confidence interval of 10% was estimated for 55% of a cohort of 400 patients [i.e. a confidence interval (CI) of (50%; 60%)].18,19 All data were collected and centralized to create the database. Descriptive variables were expressed as occurrence and missing values, mean and standard deviation. Percentages were calculated from the number of data observed. A bilateral 95% CI was calculated if considered relevant (Clopper–Pearson Method). Missing values were not replaced. Patients with missing values for stratification were excluded from the analysis (n = 6). Categorical data and continuous data were compared using Fisher’s exact test and t-test, respectively. Univariate analysis with the regression logistic model based on asthma control was first performed on all clinical variables available. Variables with a p < 0.20 were selected for subsequent multivariate analysis. p value, odds ratio (OR) and 95% CI were determined. A p < 0.05 was considered statistically significant. Moreover, in response to the question ‘factors associated with asthma control’, if the OR values were <1, the factors compared were inversely correlated. Data were analysed using SAS software version 9.4, Copyright © SAS Institute Inc., Cary, NC, USA.
Results
Patients characteristics
A total of 437 severe asthma patients were enrolled in this study between July 2020 and July 2021. Data were missing for six patients. In total, 431 patients were available for the final analysis. The overall cohort comprised mostly women (62.9%) and overweight/obese patients (59.0%) (Table 1). Severe asthma was diagnosed before 18 years of age in 6.1% of patients only. In this cohort, 25.0% had an interval of more than 10 years between the onset of asthma symptoms and the first visit to a pulmonologist. Around one-quarter of patients (25.5%) received maintenance OCS. Non-compliance with treatments was observed in 10.6% of patients.
Sociodemographic characteristics and clinical data of the population enrolled in the study and analysed (N = 431). Continuous data are expressed as mean ± standard deviation and categorical data are expressed in numbers (percentages).

ACQ, asthma control questionnaire; AQLQ, asthma quality of life questionnaire; BMI, body mass index; ICS, inhaled corticosteroid; LTI, long-term illnesses with 100% of patient’s healthcare costs being covered by the main French Health Insurance System; NA, number of patients with missing data; OCS, oral corticosteroid.
The LTI status with 100% of patients’ healthcare costs being covered by the primary French Health Insurance system was granted to the vast majority of patients (87.7%). The ACQ and Mini-AQLQ scores were significantly correlated (n = 413, Pearson correlation − 0.791, p < 0.001) (Supplemental Table 1), indicating a strong link between asthma control and quality of life. In the overall population, 88.4% of patients had at least one comorbidity (Figure 1).

Comorbidity characteristics of severe asthma patients (n = 431).
Characteristics of the population stratified by T2 high phenotype and biologic
Among the 431 patients enrolled in the study, 409 patients (94.9%) presented a T2 high profile and 297 (72.6%) patients with a T2 high phenotype were treated with a biologic (Figure 2). The mean duration of biologic treatment was 34.6 ± 35.4 months. In T2 high patients, both those receiving/not receiving biologics displayed similar characteristics in terms of gender, age and smoking status (Table 2). LTI status was more common in patients receiving biologics (96.6%) versus patients not receiving biologics (76.9%) (p < 0.001). The proportion of patients on maintenance OCS therapy was similar among patients receiving (25.2%) and not receiving biologics (26.3%). Compliance with maintenance therapy was deemed acceptable in 91.5% and 84.4% of T2 high patients receiving and not receiving biologics, respectively.

Study design and overall population analysed.
Sociodemographic characteristics and clinical data of the T2 high phenotype patient subgroup (N = 409). Continuous data are expressed as mean ± standard deviation and categorical data are expressed in numbers (percentages).
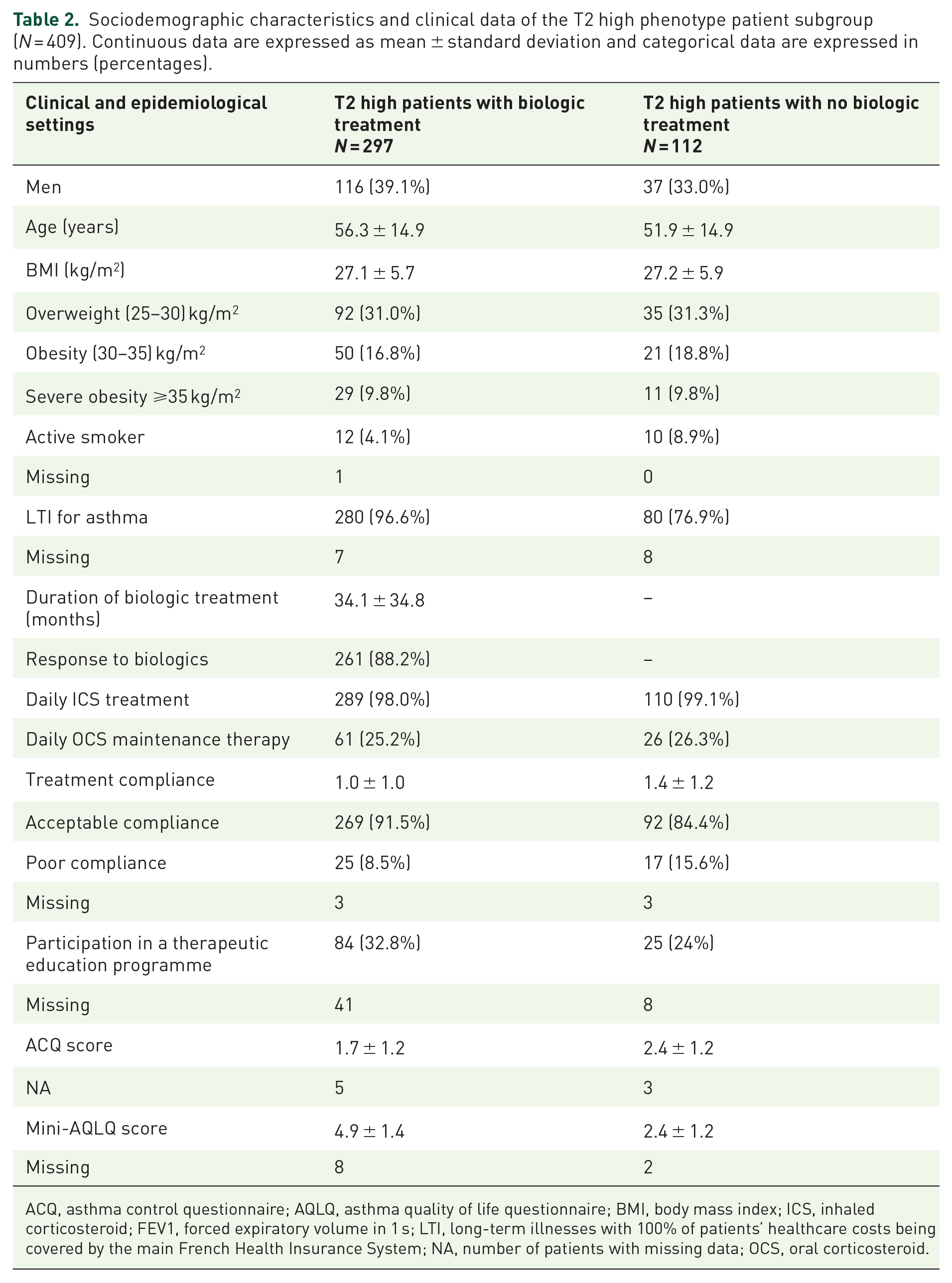
ACQ, asthma control questionnaire; AQLQ, asthma quality of life questionnaire; BMI, body mass index; ICS, inhaled corticosteroid; FEV1, forced expiratory volume in 1 s; LTI, long-term illnesses with 100% of patients’ healthcare costs being covered by the main French Health Insurance System; NA, number of patients with missing data; OCS, oral corticosteroid.
In the T2 high population, the proportion of patients participating in a therapeutic education programme was 32.8% for patients receiving biologics versus 24.0% for patients not receiving biologics (p = 0.13) (Table 2). The proportion of comorbidities was higher in T2 high patients treated with biologics compared to those not receiving biologics (87.5% versus 61.3%) (Supplemental Figure 1).
Impact of biologics on asthma control in T2 high patients
Doctors estimated that 88.2% of T2 high patients receiving biologics responded to those treatments [Figure 3(a)]. However, asthma control was only achieved in 25.3% of those patients [Figure 3(b)]. A high proportion of patients (74.6%) with uncontrolled asthma according to the ACQ score were defined as responders to biologics by doctors (Supplemental Figure 2). The ACQ and Mini-AQLQ scores were significantly correlated in T2 high patients receiving biologics (n = 286, Pearson correlation − 0.803, p < 0.001) (Supplemental Table 1), confirming a strong link between asthma control and quality of life. Despite receiving biologics, more than half of patients continued to use OCS either daily (25.2%) or more than three times a year for at least three consecutive days (25.6%) [Figure 3(c)]. In T2 high patients receiving biological therapy, 87.2% were satisfied with their treatment (taking those totally satisfied and satisfied into consideration) [Figure 3(d)]. In patients receiving biologics within 12 months prior to enrolment, 5.4 medical consultations, 1.4 exacerbations, 0.5 hospital admissions and 0.6 emergency visits were documented [Figure 3(e)].

Impact of biologic treatment among T2 patients receiving biologics based on therapeutic response defined by a pulmonologist (a), asthma control score (b), OCS consumption (c), patient satisfaction with treatment (d), healthcare consumption (e) and the proportion of super-responders among responders to biologics (f).
In T2 high patients receiving biologics, 13.5% were considered super-responders [Figure 3(f)].
Asthma control and quality of life in T2 high patients whether or not receiving biologics
A significant improvement in the ACQ score was observed in T2 high patients receiving biologics compared to those not receiving biologics (1.7 ± 1.2 versus 2.4 ± 1.2; p < 0.0001). This difference was noted for all ACQ score items apart from FEV1 [Figure 4(a)].

Asthma control and quality of life scores for T2 high patients: comparison between patients receiving (n = 297) and not receiving biologics (n = 112). Radar graphs illustrate comparison between patients groups for (a) asthma control using ACQ scores and (b) quality of life using Mini-AQLQ score
A comparison of quality of life measured through the Mini-AQLQ in T2 high patients receiving biologics compared to those not receiving biologics shows a significant improvement in emotional function, symptoms indicative of limited physical activity (p < 0.0001, respectively) and environmental stimuli (p < 0.0007) [Figure 4(b)].
Noticeably, all quality of life parameters improved in T2 high patients receiving biologics. This improvement is correlated to asthma control.
Factors correlated to asthma control in T2 high patients receiving biologics
The analysis of factors associated with asthma control (reflected by the ACQ score) in T2 high patients receiving biologics shows that two comorbidities namely gastroesophageal reflux disease (GERD) or sleep apnoea syndrome significantly decrease the probability of asthma control in univariate and multivariate models (Table 3). Other factors were considered for analysis, but did not impact the probability of asthma control in this population: age at diagnosis, BMI, duration of biologic treatment, nasal polyposis, gender, age at baseline, smoking status and chronic bronchitis.
Univariate and multivariate analyses.

OR, odds ratio.
Bold values correspond to statistical significant value (p-value < 0.05 or OR < 1).
corresponds to statistical significant values both in univariate and multivariate analysis (p-value < 0.05).
Discussion
In this study, although the proportion of severe asthmatic patients responding to biologics is as high as 88.2% accompanied by a significant improvement in the ACQ score, asthma control was only achieved in 25.3% of patients. More than 50% of those patients continue to use OCS in the short or long term. Despite receiving biological therapy within 12 months prior to enrolment, our patients still had a mean of 5.4 medical consultations, 1.4 exacerbations, 0.5 hospital admissions and 0.6 emergency visits.
The prevalence of severe asthma patients with low type 2 asthma usually ranges from 15% to 30%.20 –22 In our study, this prevalence was much lower (i.e. 5.1%) because the criteria for low type 2 asthma were more restrictive. Indeed, contrary to previous studies, we used a blood eosinophilia cut-off of 0.15 G/L and a FeNO cut-off of 20 ppb. As recently described by the Global Initiative for Asthma (GINA) guide, those criteria are more reflective of type 2 inflammation. Given the large predominance of T2 high phenotype (94.9%) among severe asthma patients recruited for the study and the large proportion of patients receiving biologics, we focused the analysis on the group of T2 high patients treated with those medicinal products (n = 297). Our study is close to other observational studies in terms of other parameters. In fact, the majority of our patients were women and subjects were mostly overweight or obese. The mean age at severe asthma diagnosis was 39.5 ± 18.1 years; only 13.4% of patients had severe asthma onset before the age of 18.
Regarding the response rate of biological therapies in our patients, 88.2% were considered responders to those treatments according to doctors. This number is in line with the results of large randomized, controlled trials because the response rate ranged between 76% and 83% in those trials. 23 However, although biological therapies are effective, more than 50% of our patients still took OCS either daily (25.2%) or at least three times a year as short-course therapy (25.6%) in our study. In the PONENTE trial, which assessed the rate of OCS elimination after introducing benralizumab for patients receiving long-term oral steroids, 37.12% were still given oral steroids at 1 year. 24 Interestingly, 20.5% had biological adrenal insufficiency during the follow-up. 24 In this trial, the dose reduction in oral steroids was standardized. Our real-life study and the PONENTE trial show that OCS elimination is not achieved in all patients despite the use of biologics. Further studies are required to better understand the reason for OCS continuation. Indeed, we did not assess the reason for continued OCS administration in our patients. Indeed, it would have been interesting to establish whether OCS was continued for medical reasons (adrenal insufficiency), patient choice or doctor preference.
Although we observed a high response rate and patient satisfaction with biologics, asthma control evaluated by the ACQ (‘objective’ evaluation) (ACQ ⩽ 0.75) was achieved in only 25.3% of our patients. Hence, a significant proportion of patients defined as responders to biologics by the doctor (74.6%) is not controlled. This is mostly due to the fact that the primary criteria for evaluating the response to biologics are based on the reduction in asthma exacerbation and/or the dose of oral steroids. Moreover, the ACQ score focused on asthma symptoms within the last 7 days, whereas doctors evaluate treatment response over a longer period. Other symptoms such as dyspnoea, cough and limitations in activities are not taken into account in assessing the response to biologics despite the fact that these may impact asthma control. Our study shows that much remains to be done to normalize the quality of life of severe asthma patients treated with biologics.
The most impactful symptoms despite the use of biological therapy seem to be breathlessness and limitations in activities. Individuals with severe asthma perform fewer moderate and vigorous activities than their controls. 25 Interestingly, higher levels of activity and lower levels of sedentary time are associated with better exercise capacity and asthma control. A long history of severe asthma may have led to exercise limitation, which could explain, at least in part, the persistent breathlessness in our patients treated with biological therapies. In our study, we did not assess the number of patients with pulmonary rehabilitation, which is shown to be effective in severe asthma. 26 The low asthma control rate could also be due to the presence of comorbidities impacting quality of life. In our study, 32% had nasal polyposis and 9.5% presented cardiovascular disease. These comorbidities could have impacted the ACQ and breathlessness. Moreover, since dysfunctional breathing is an independent risk factor for poor asthma control and quality of life, 27 it may also explain the low proportion of patients with controlled asthma in our study. However, this comorbidity was not documented. Finally, we observed that FEV1 did not change despite the use of biologics. Airflow limitation may also be associated with a change in the ACQ.
We performed a multivariate analysis to establish which factors are associated with a lack of asthma control. We found that GERD and OSA decrease the probability of asthma control. As shown in previous studies, 28 severe asthma patients have a high incidence of OSA, which is confirmed in the present study (prevalence 19.3%). Moreover, a bidirectional relationship between OSA and asthma is suggested. In particular, accumulating evidence suggests that OSA contributes to poor asthma control. 28 Asthmatics with coexisting OSA experience an improved quality of life with Continuous Positive Airway Pressure treatment. 29 This effect appears more pronounced in severe OSA or poorly controlled asthma. However, we have no data regarding either the treatment or the severity of OSA in our study. Regarding GERD, we found a high prevalence (29.5%) of GERD in our severe asthma patients, which is consistent with previous data. It is generally accepted that GERD is more common in patients with ‘difficult-to-control’ asthma than in patients with well-controlled asthma.30,31 Conversely, patients with GERD are reported to have significantly worse asthma control (based on the Asthma control test) than patients without GERD. It has previously been suggested that obesity, OSA and GERD are collinear comorbidities in severe asthma. 32 However, contrary to GERD and OSA, obesity was not found as an independent factor of poor asthma control in our study. It is important to notice that GERD and OSA were among the most prevalent comorbidities in our cohort. However, we cannot exclude that other comorbidities with a smaller sample size play a role in the lack of asthma control.
Our study has several limitations. Firstly, we did not document the type of biological therapy used in the study. However, this study was not designed to compare biological therapies per se. The main aim was to assess the effect of these medicinal products in patients with severe asthma. Biological therapies were used according to the manufacturers’ recommendations and international guidelines on severe asthma. Secondly, we initially planned to compare the characteristics of patients with T2 high and T2 low asthma. However, only 5.1% of patients with T2 low asthma were enrolled in the study. This is because our study was a real-life study. It is interesting to see that, in a real-life study, the proportion of patients with T2 low asthma is not as high as previously described. Finally, we did not assess the patients over time. However, we were able to collect robust clinical data from a large cohort of patients with severe asthma in a real-life study.
Conclusion
To conclude, although a high proportion (88.2%) of patients with severe asthma responded to biological therapies, our study highlights that only 25.3% achieved asthma control at the time of evaluation. Uncontrolled asthma in patients responding to biological therapies is likely to be multifactorial. However, GERD and OSA seem to be independent factors associated with uncontrolled asthma. Further studies are warranted for a better understanding of uncontrolled asthma in patients treated with biological therapies. Although the latter have revolutionized the way severe asthma patients are treated, much remains to be done to further improve the quality of life of these patients.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231202749 – Supplemental material for Improvement in severe asthma patients receiving biologics and factors associated with persistent insufficient control: a real-life national study
Supplemental material, sj-docx-1-tar-10.1177_17534666231202749 for Improvement in severe asthma patients receiving biologics and factors associated with persistent insufficient control: a real-life national study by Laurent Guilleminault, Claire Camus, Chantal Raherison-Semjen, Audrey Capdepon, Arnaud Bourdin, Philippe Bonniaud, Stéphanie Fry, Gilles Devouassoux, François-Xavier Blanc, Christophe Pison, Clairelyne Dupin, Naji Khayath, Joelle Courdeau, Judith Valcke-Brossollet, Cécilia Nocent-Ejnaini, Fabien Rolland, Carmen Lamandi, Alain Proust, Anaig Ozier, Laurent Portel, Wanda Gaspard, Pauline Roux-Claude, Antoine Beurnier, Stéphanie Martinez, Jean-Marc Dot, Florence Hennegrave, Guillaume Vignal, Etienne Auvray, Nicolas Paleiron, Nicolas Just, Jean Miltgen, Maud Russier, Cécile Olivier, Camille Taillé and Alain Didier in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
We wish to thank CARELY for providing support in carrying out the study including logistics, data entry, data analysis and statistics. We are grateful to CRISALIS, Clinical Research Initiative in Severe Asthma: a Lever for Innovation & Science Centres, for its technical support and to all clinical research technicians for their helpful contribution to this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
