Abstract

Introduction
Severe asthma is characterised by ongoing requirements of high-dose inhaled glucocorticoids and long-acting bronchodilators and the recurrent use of oral corticosteroids to achieve adequate control. 1 Patients with severe asthma suffer from increased hospitalisations, frequent exposure to oral corticosteroids (OCS) and reduced quality of life. 2 Anti-interleukin (IL)-5 biologics have had a pivotal role in improving asthma control in patients with severe eosinophilic asthma. Mepolizumab and reslizumab are monthly therapies that target IL-5, while benralizumab targets IL-5R and is initially administered monthly for three doses and every 2 months thereafter.
The COLUMA (Open-label Long Term Extension Safety Study of Mepolizumab in Asthmatic Subjects) 3 and the ANDHI studies 4 (which investigated the safety and efficacy of Benrazliumab) have shown that targeting IL-5 reduces asthma exacerbations, reduces oral corticosteroid use and improves lung function in severe asthma. Anti-IL-5 therapies are well tolerated with a low incidence of adverse drug reactions. 5 Raised creatine kinase (CK) has been reported in severe asthma during exacerbations; however, the mechanisms are not well understood. 6 A previous trial of reslizumab reported myalgia in 1% of patients receiving the drug, while elevated CK levels which were transient were seen in 0.8% of patients who received the drug; however, they had no clinical significance and did not result in the discontinuation of the drug. 7 However, the differing effects of different anti-IL-5 therapies on CK levels and any clinical significance are not well described.
Methods
We performed a real-world analysis of 53 patients who received mepolizumab, reslizumab or benralizumab between 2017 and 2020 in our severe asthma clinic and examined the impact IL-5 biological therapy had on patient CK levels. Baseline demographics were recorded from medical records. CK levels were recorded pre-commencement of IL-5 therapy and then 0–3 months, 3–6 months, 6–9 months and 9–12 months post initiation of therapy. The upper limit of CK level in our institution is 180 U/L. A clinically significantly CK rise was defined as a CK level of 1.5 times the upper limit of normal (ULN) after commencing anti-IL-5 therapy. 8 All patients provided informed consent, and clinical information was extracted from patient charts under protocols approved by the Research Ethics Committee of the Cork Teaching Hospital. Statistical analysis was performed using SPSS 27 (IBM Corp, Armonk, NY, USA) and Prism 9 (GraphPad Software Inc., San Diego, California, USA).
Results
In this study, 53 patients were administered a biologic: 16 prescribed mepolizumab, 21 reslizumab and 16 benralizumab. Both mepolizumab and benralizumab were administrated via the subcutaneous route while reslizumab was administered intravenously. Demographic characteristics are described in Table 1. There was no statistically significant difference in CK levels between anti-IL-5 groups prior to commencement of therapy. Following initiation of anti IL-5 therapy, there was no statistically significant difference across the three therapies except at 3–6 months, where this was a trend towards significance (p = 0.06) (Figure 1(a)–(e)). The effect at each time point demonstrates a significant difference between mepolizumab and benralizumab at 3–6 months (p = 0.04) (Figure 1(c)).
Baseline characteristics.

ANOVA, analysis of variance; CK, creatine kinase.
Data are presented as no. (%), mean ± standard deviation and median (interquartile range). Parametric variables were compared using chi-square test for independence and one-way ANOVA. Nonparametric variables were compared using the Kruskall–Wallis test.

Creatine kinase (CK) levels at (a) baseline, (b) 0–3 months, (c) 3–6 months, (d) 6–9 months and (e) 9–12 months after starting anti-IL-5 therapy. Dashed line indicates upper limit of normal CK value in an adult (180 U/L). There was no significant difference across all three anti-IL-5 biological therapies at each timepoint as shown, except between mepolizumab and benralizumab at 3–6 months. Data presented as median with IQR and analysed using Mann–Whitney U test.
When we examined the effect of each anti-IL-5 therapy on CK levels, there was a significant rise in the mepolizumab group at 3–6 months (p = 0.01) and 6–9 months (p = 0.03) post-treatment initiation (Figure 2(a)). In the reslizumab group, there was a significant rise at 6–9 months (p = 0.03) (Figure 2(b)), with no significant change at any time point in the benralizumab group (Figure 2(c)). At 12 months across all groups, there is no statistical difference in CK levels from baseline (Figure 2(a)–(c)). This indicates that while CK levels are elevated in the initial months of therapy, this is transient and levels plateau at 12 months (Figure 3).
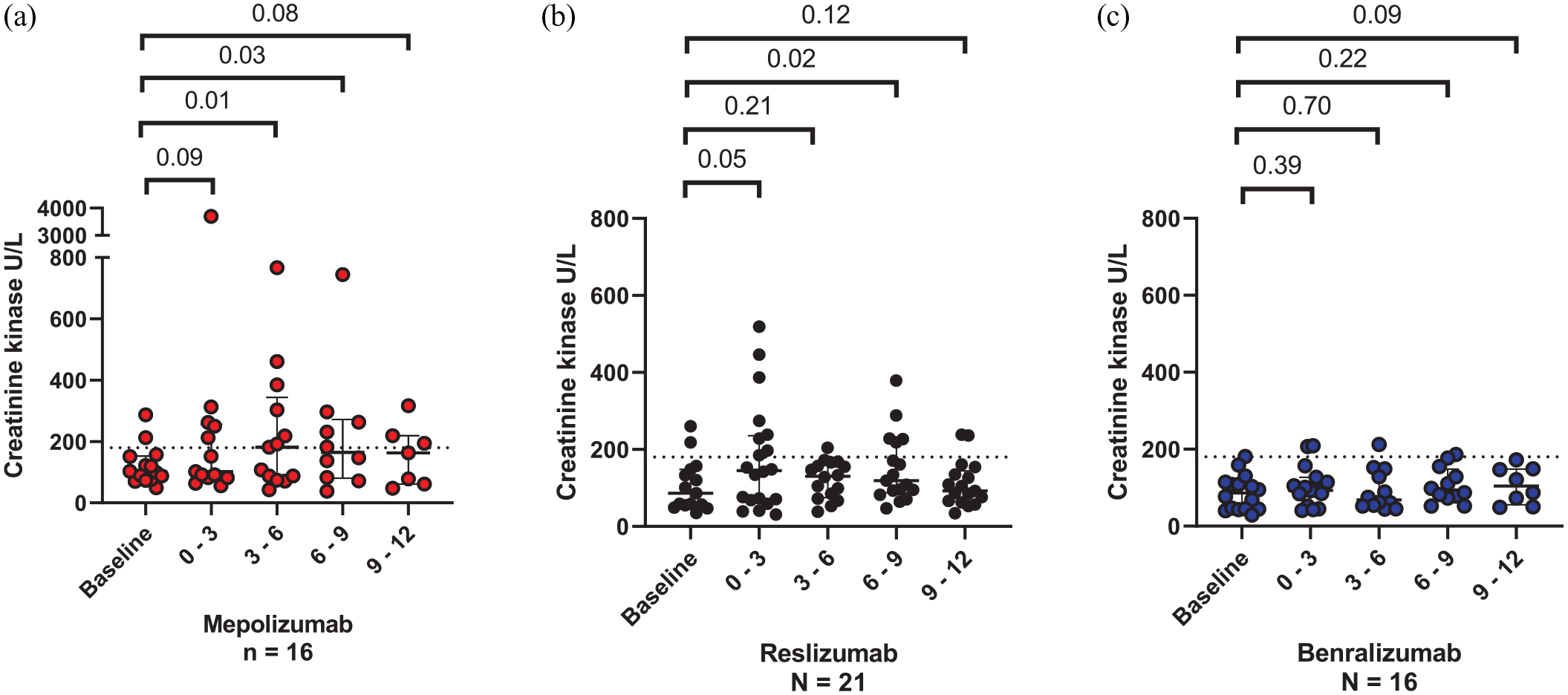
Effect of each anti-IL-5 therapy on creatine kinase (CK) up to 12 months post treatment initiation. Dashed line indicates upper limit of normal CK value in an adult (180 U/L). Within the mepolizumab group, there was a significant increase in CK compared to baseline at 3–6 months and 6–9 months post treatment, with a significant increase in CK within the reslizumab group at 6–9 months post treatment (p = 0.03), and a trend towards significance at 0–3 months post treatment (p = 0.051). There was no significant change in CK levels from baseline up to 12 months after initiation of benralizumab treatment. Data presented as median with interquartile ranges and analysed using Wilcoxon signed-rank test.

Change in creatine kinase (CK) levels up to 12 months post anti-IL-5 therapy. Data are presented as mean and error bars indicate standard error of the mean.
There was no significant correlation between age and total change in CK from baseline (r = 0.18, p = 0.24). Overall, 9 patients (17.0%) had a clinically significant CK rise as defined by 1.5 increase from ULN: 5 patients on mepolizumab, 4 patients on reslizumab and 0 patients on benralizumab. Clinically significant CK rise was significantly associated with anti-IL-5 group (p = 0.04); however, it was not associated with sex (p = 0.65) or age (p = 0.90).
In our cohort – one patient in the mepolizumab group did appear to be affected by rise in CK level. The patient was a 63-year-old gentleman who had a diagnosis of asthma for approximately 30 years and previously trialled omalizumab, an anti-immunoglobulin E biological therapy, which did not improve his symptoms or asthma control. Prior to anti-IL-5 therapy commencement, he was treated with maintenance OCS for 5 years. On commencement of mepolizumab, his baseline CK level was 287 U/L. At 3–6 and 6–9 months, the CK level rose to 767 and 1496 U/L, respectively. He experienced muscle stiffness, cramps and generalised fatigue. The frequency of mepolizumab administrated was reduced from 4 weekly to 8 weekly. At 9–12 months, his CK level had reduced to 385 U/L, and it was hypothesised that the initiation of IL-5 therapy and subsequent reduction in OCS dose revealed an underlying inflammatory myositis disorder. However, when we remove the above patient from the data set, we still observed a significant rise in the mepolizumab group at 3–6 months (p = 0.03); however, again, this was transient and there was no significant difference at later timepoints. Previous studies in the literature have reported increased CK levels in association with reslizumab 9 and benralizumab. 10 Our results showed no significant increase in CK with benralizumab over 12 months of treatment, with an increase in CK levels following reslizumab treatment only at 6–9 months post commencing treatment. Any increase in CK therefore also appears to be independent of the route of administration of the drug.
Discussion
The above data demonstrates a transient elevation in CK levels at the beginning of anti IL-5 therapy which is not sustained at 12 months. Elevated CK levels have been reported with numerous medications including immunotherapies. There have been case reports of elevated CK levels and muscle-related complaints in inflammatory bowel disease (IBD) patients receiving infliximab [anti-tumour necrosis factor (TNF)-α]. In their exploration of the impact of infliximab on CK levels in IBD patients, Theodoraki hypothesised that the elevated levels could be due to a proposed impact of TNF-α inhibition on inflammatory and metabolic pathways – and that this led to a series of events which allowed calcium ions to enter muscle cells. 11
Some studies in pharmacotherapy have suggested a genetic component. As many as 5% of patients taking statin therapy develop elevated CK levels. The SEARCH (Study of the Effectiveness of Additional Reductions in Cholesterol and Homocysteine) Collaborative group identified a single-nucleotide polymorphism in the SLCO1B1 gene on chromosome 12 that was associated with a higher risk of statin-induced myopathy. 12 Furthermore, the discovery of a mutation in the DAG1 gene associated with asymptomatic elevated CK levels has been found significantly associated with ulcerative colitis. 13 This may indicate that there are other genetic factors which predispose patients to developing elevated CK levels following the introduction of immunotherapies.
Randomised controlled trials investigating the novel therapies such as anti-thymic stromal lymphopoietin and anti IL-4/IL-13 have not demonstrated clinically significant CK elevation.14,15
Our data provide reassurance that anti IL-5 therapy is not generally associated with a clinically significant rise in CK levels, and although CK levels increase during initial months of therapy, this is temporary. The case identified of an unmasking of an underlying myositis is noteworthy and clinicians should be aware of this potential outcome.
