Abstract
Background:
Epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitors (TKIs) are standard treatments for advanced EGFR-mutated non–small cell lung cancer (NSCLC) patients. Osimertinib is an effective therapy for NSCLC patients with acquired resistance due to T790M mutation after first- and second-generation EGFR-TKI treatment. This study aimed to analyze the clinical outcomes of sequential therapy following first-line EGFR-TKIs and the predictive factors of an acquired T790M mutation.
Methods:
Between January 2014 and December 2018, data from 2190 advanced NSCLC patients with common EGFR mutations (exon 19 deletion and L858R) receiving first- and second-generation EGFR-TKIs in Linkou, Kaohsiung, Chiayi and Keelung Chang Gung Memorial Hospitals were retrospectively retrieved and analyzed.
Results:
Until August 2021, among 1943 patients who experienced progressive disease, 526 underwent T790M mutation tests, and their T790M-positive rate was 53.6%. Exon 19 deletion mutation and progression-free survival (PFS) of >12 months were positively associated with secondary T790M mutation. Different first-line first- and second-generation EGFR-TKI therapies did not affect the appearance of acquired T790M mutations. The median overall survival (OS) was 58.3 [95% confidence interval (CI): 49.0–67.5] months among the patients with T790M mutation who received second-line osimertinib therapy compared with 31.0 (95% CI: 27.5–34.5) months among the patients without T790M mutation who received chemotherapy alone. The multivariate analysis showed that a poor performance status (score: >2), nonadenocarcinoma histology, stage IV cancer, liver metastasis, brain metastasis, PFS while on first-line EGFR-TKIs, and subsequent chemotherapy without third-generation EGFR-TKIs were significant independent unfavorable prognostic factors for OS.
Conclusion:
This study demonstrated the efficacy of first-line EGFR-TKIs and sequential osimertinib therapy. The results of our study suggest that T790M mutation tests are important for the use of subsequent osimertinib, which yielded favorable survival outcomes.
Keywords
Introduction
In 2022 in the United States, the estimated number of new lung cancer cases ranked second, following prostate cancer in men and breast cancer in women; meanwhile, the associated death ranked first for both sexes. 1 In contrast to small cell lung cancer, non–small cell lung cancer (NSCLC) accounts for more than 80% of cases of lung cancer, 2 and multimodal treatment, including surgery, radiotherapy, and systemic treatment, is the main therapeutic strategy based on the clinical stage of the tumors and the feasibility based on the underlying patient-related factors.3,4
Before the era of targeted therapy, platinum-based doublet chemotherapy was the standard first-line treatment strategy, which was associated with an average progression-free survival (PFS) of 4 months and an overall survival (OS) of 8 months.5,6 With the development of targeted therapy, the most commonly used and studied targeted agents are epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitors (TKIs), such as gefitinib and erlotinib [first-generation (1G)],7–10 afatinib and dacomitinib [second-generation (2G)],11–14 and osimertinib [third-generation (3G)].15,16
The FLAURA study showed a significant improvement in OS among patients with NSCLC who harbored EGFR mutations and were treated with osimertinib as first-line therapy [median OS: 38.6 months, 95% confidence interval (CI): 34.5–41.8 months] compared with that among patients who received standard-of-care treatment using other EGFR-TKIs, such as gefitinib or erlotinib (median OS: 31.8 months, 95% CI: 26.6–36.0 months). 15 However, the improvement in OS was marginal [hazard ratio (HR): 0.8, p = 0.046] and may be limited among non-Asian populations and patients with exon 19 deletion.
Sequential treatment after progression on first-line 1G or 2G EGFR-TKIs is typically based on whether T790M resistance mutation is acquired during progression. 17 Acquisition of T790M mutation accounts for greater than 50% of cases that progress following treatment with 1G or 2G EGFR-TKIs. 17 In the prospective clinical trial AURA3, osimertinib was shown to have a significantly higher response rate and longer PFS than chemotherapy among acquired T790M mutation in advanced NSCLC patients who experienced progressive disease while on first-line EGFR-TKI treatment. Based on these results, osimertinib has been suggested as a standard therapy for advanced NSCLC patients with acquired T790M mutation rather than conventional chemotherapy. 18
Although sequential osimertinib in advanced NSCLC patients with acquired T790M mutation previously treated with 1G or 2G EGFR-TKIs has been widely administered in clinical practice, the clinical OS and outcome of these patients have rarely been reported. A global and observational study (GioTag study) demonstrated that EGFR-mutated advanced NSCLC patients receiving sequential afatinib and osimertinib had a median chemotherapy-free treatment time of 27.7 months and a median OS of 37.6 months.19,20 The study patients recruited in the GioTag study all acquired T790M mutation, and all received afatinib as first-line therapy.19,20 However, the survival outcomes among patients with NSCLC that progress on 1G or 2G EGFR-TKIs without an acquired T790M mutation or with an unknown T790M status on progression remain unclear. This retrospective study aimed to evaluate the outcomes of sequential treatment following first-line EGFR-TKI therapy and the factors predicting the acquisition of T790M mutation.
Materials and methods
Data collection
Data for all patients included in this study were obtained from the Chang Gung Research Database; 21 and patient data were obtained from the cancer registries associated with the Linkou, Kaohsiung, Keelung, and Chiayi branches of Chang Gung Memorial Hospital (CGMH) from June 2014 to March 2018.22,23
Patient selection and clinicopathological features
Patients who were diagnosed with advanced NSCLC harboring common EGFR mutations (exon 19 deletion or L858R mutation) and treated with gefitinib, erlotinib, or afatinib as first-line treatment, without prior systemic treatment for advanced NSCLC, were enrolled in this study. Patients with de novo T790M mutation before beginning any treatment were excluded from this study and reported separately. 24 Dacomitinib was not available during the study period. Clinicopathological features, including age, sex, smoking history, performance status (PS), tumor histology, tumor involvement, EGFR mutation (exon 19 deletion or L858R mutation), tumor response, and subsequent treatment, were evaluated. All patients underwent tissue or liquid biopsy after progression while on EGFR-TKIs. A positive outcome for T790M mutation on either tissue or liquid biopsy was considered to indicate T790M positivity. The majority of tissue biopsy samples were assessed using the amplification-refractory mutation system, and a minority were assessed using next-generation sequencing. The last follow-up time point in the study was August 2021.
Tumor response, survival, and statistical analysis
The tumor response was evaluated in accordance with the Response Evaluation Criteria in Solid Tumors (version 1.1). The detailed definitions of tumor response, PFS, and OS have been described in our previous study. 25
Categorical variables were compared using Pearson’s chi-square test or Fisher’s exact test, and continuous variables were compared using a t test. A univariate analysis was performed to evaluate the potential predictors for acquired T790M mutation, including age (>65 versus ⩽65 years), sex, Eastern Cooperative Oncology Group (ECOG) PS score (0 versus 1 versus ⩾2), smoking history, histology (adenocarcinoma versus nonadenocarcinoma), location of metastases, first-line EGFR-TKIs, and PFS while on first-line EGFR-TKIs. A multivariate analysis was performed to identify the independent predictors.
PFS and OS were estimated using the Kaplan–Meier method and compared using the log-rank test. Univariate analysis was performed to evaluate the potential prognostic factors, while multivariate analysis was performed by including all variables with p < 0.05 in the univariate analysis to evaluate the independent prognostic factors. The results are presented as HRs and 95% CIs in the Cox regression analyses. IBM SPSS Statistics for Windows (version 23.0; Armonk, NY, USA) was used to perform all statistical analyses, and p < 0.05 was considered significant. Survival curves were also plotted using R software.
Results
Sequential treatment after 1G/2G EGFR-TKIs
This study enrolled 2190 patients with EGFR-mutated NSCLC treated with first-line EGFR-TKIs, including 635 treated with afatinib, 496 with erlotinib, and 1060 with gefitinib. By the end of August 2021, 1943 patients experienced progressive disease or switched to other systemic treatment owing to intolerable adverse events, and 526 patients were evaluated for T790M mutation via either tissue or liquid biopsy or both.
According to various subsequent treatments, the patients were divided into three groups: osimertinib (regardless of chemotherapy), 3G EGFR-TKIs other than osimertinib (regardless of chemotherapy), and chemotherapy without 3G EGFR-TKIs. The classification of the sequential treatment strategies and the number of patients in each group are summarized in Figure 1.

Summary of subsequent treatment following EGFR-TKI treatment.
Clinical factors associated with acquired T790M mutation after first-line EGFR-TKI treatment
Among the 526 patients who had received T790M mutation tests (tissue or liquid biopsy or both), 282 patients (53.6%) were positive for T790M mutation. The T790M positivity rates were 54.3% (169/311) on tissue biopsy and 42.9% (112/261) on liquid biopsy. Forty-six patients were evaluated via both tissue and liquid biopsies, and 43 of the 46 patients had negative T790M on either liquid or tissue biopsy. The concordance between tissue and liquid biopsy was not assessed as only a highly selected subgroup of patients (less than 10% of the whole cohort and most patients received testing sequentially rather than concurrently) underwent both tissue and liquid biopsy in this study, which possibly led to an inadequate assessment. Although tissue biopsy showed a higher T790M positivity rate than liquid biopsy, the performance of additional liquid biopsy likely resulted in an increased overall number of detected cases of T790M mutations, particularly in the patients who were not suitable for tissue rebiopsy or had insufficient tissue.
The clinical factors associated with acquired T790M mutation after EGFR-TKI treatment were evaluated (Table 1). The patients with exon 19 deletion had higher rates of T790M mutation than those with L858R mutation (59.6% versus 46.5%, p = 0.003, Figure 2(a)). In addition, a longer PFS while on first-line EGFR-TKI treatment was associated with a higher rate of T790M mutation (44.9% for <6 months, 44.4% for 6–12 months, 62.7% for 12–18 months, 60.0% for 18–24 months, and 57.7% for > 24 months, p = 0.011, Figure 2(b)). Clearly, an adequate cutoff PFS for the prediction of T790M mutation might be 12 months, implying that the patients with a PFS of >12 months had a greater change in T790M positivity (60.1%) than the patients with a PFS of <12 months (44.5%). For the different first-line EGFR-TKIs, no significant difference was found in the T790M mutation rate (afatinib: 54.2%, erlotinib: 55.3%, gefitinib: 51.2%, p = 0.757, Figure 2(c)).
Univariate and multivariate analysis for predictors of acquired T790M mutation (n = 526).

CI, confidence interval; l9del, exon 19 deletion; PFS, progression-free survival; TKI, tyrosine kinase inhibitor.
Adjusted by mutation only.
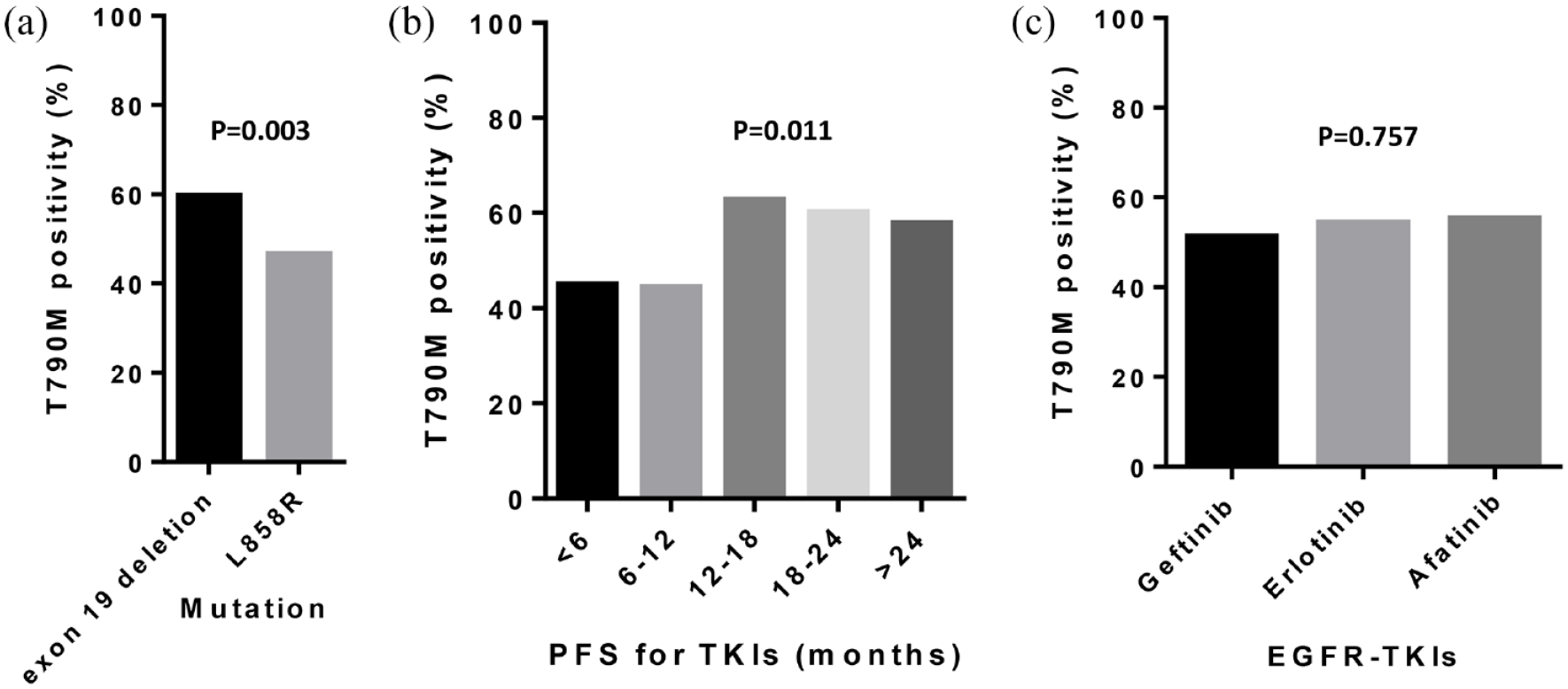
(a) T790M positivity based on EGFR mutation, (b) PFS while on TKIs, and (c) first-line treatment. (a) The patients with exon 19 deletion had a higher positivity rate for T790M mutation than those with L858R mutation (p = 0.003). (b) The patients who had a longer PFS while on afatinib had a higher positivity rate for T790M mutation than those who had a shorter PFS while on afatinib (p = 0.011). (c) The patients treated with the different frontline EGFR-TKIs had similar positivity rates for T790M mutation (p = 0.757).
The multivariate analysis showed that exon 19 deletion (versus L858R mutation, odds ratio: 1.65, 95% CI: 1.16–2.35, p = 0.005) and PFS while on EGFR-TKIs were significant factors associated with T790M mutation.
In terms of the numbers of metastatic or specific sites, no association was found (Supplementary Table S1).
Treatment outcomes of second-line osimertinib based on the T790M mutation status
A total of 172 patients received second-line osimertinib therapy following first-line EGFR-TKIs, regardless of the presence of T790M mutation. Of the 147 patients who received osimertinib therapy with a known T790M mutation status, 135 had T790M positivity, and 12 had T790M negativity. The clinical tumor response was significantly different between those with T790M positivity and those with T790M negativity (p = 0.034). The overall objective response rate (ORR) of osimertinib was 52.6% for cases positive for T790M mutation, which was higher than the ORR for cases negative for T790M mutation (25.0%, p = 0.067). The disease control rate (DCR) was 79.3% among the patients with a confirmed T790M mutation, which was higher than that among the patients without T790M mutation (41.7%, p = 0.008, Supplementary Table S2).
The median PFS while on osimertinib among the patients with a positive T790M mutation was 12.6 (95% CI: 8.2–17.0) months, which was higher than that among those with a negative T790M mutation, 3.1 (95% CI: 0.9–5.3) months (log-rank p = 0.001, Figure 3(a)). The median OS estimated from the initiation of first-line EGFR-TKIs among the patients with T790M positivity and negativity was 58.3 (95% CI: 49.0–67.5) and 38.1 (95% CI: 25.5–50.8) months, respectively, with no significant differences between the groups (log-rank p = 0.196, Figure 3(b)).

Treatment outcomes of second-line osimertinib based on the T790M mutation status. (a) The median PFS while on osimertinib among the patients with T790M mutation was 12.6 (95% CI: 8.2–17.0) months, which was higher than that among those without T790M mutation, 3.1 (95% CI: 0.9–5.3) months (log-rank p = 0.001). (b) The median OS estimated from the initiation of first-line TKIs among the patients with T790M positivity and negativity was 58.3 (95% CI: 49.0–67.5) and 38.1 (95% CI: 25.5–50.8) months, respectively, with no significant differences between the groups (log-rank p = 0.196).
Treatment outcomes of second-line osimertinib based on first-line EGFR-TKIs
We further evaluated whether specific first-line EGFR-TKIs influenced the efficacy of second-line osimertinib among patients with T790M mutation. Among the 135 patients with a positive T790M mutation who received second-line osimertinib therapy following first-line EGFR-TKI treatment, 54, 41, and 40 were administered afatinib, erlotinib, and gefitinib, respectively. The clinical tumor response was not significantly different between various EGFR-TKIs (p = 0.603). The ORR of osimertinib was 46.3% among those treated with afatinib, 53.7% with erlotinib, and 60.0% with gefitinib, with no significant difference found (p = 0.415). The DCR was 81.5% among the patients treated with afatinib, 75.6% with erlotinib, and 80.0% with gefitinib (p = 0.776) (Supplementary Table S3).
The median PFS on osimertinib among the patients administered first-line afatinib, erlotinib, or gefitinib was 11.6 (95% CI: 6.9–16.3), 15.3 (95% CI: 6.9–23.7), and 12.6 (95% CI: 5.5–19.7) months, respectively, and no significant difference was found between various EGFR-TKIs (log-rank p = 0.645, Figure 4(a)). The median OS estimated from the initiation of first-line EGFR-TKIs among the patients who were treated with first-line afatinib, erlotinib, and gefitinib was 61.8 (95% CI: not evaluable), 53.9 (95% CI: 31.8–76.0), and 54.8 (95% CI: 43.2–66.4) months, respectively, with no significant differences found between groups (log-rank p = 0.674, Figure 4(b)).
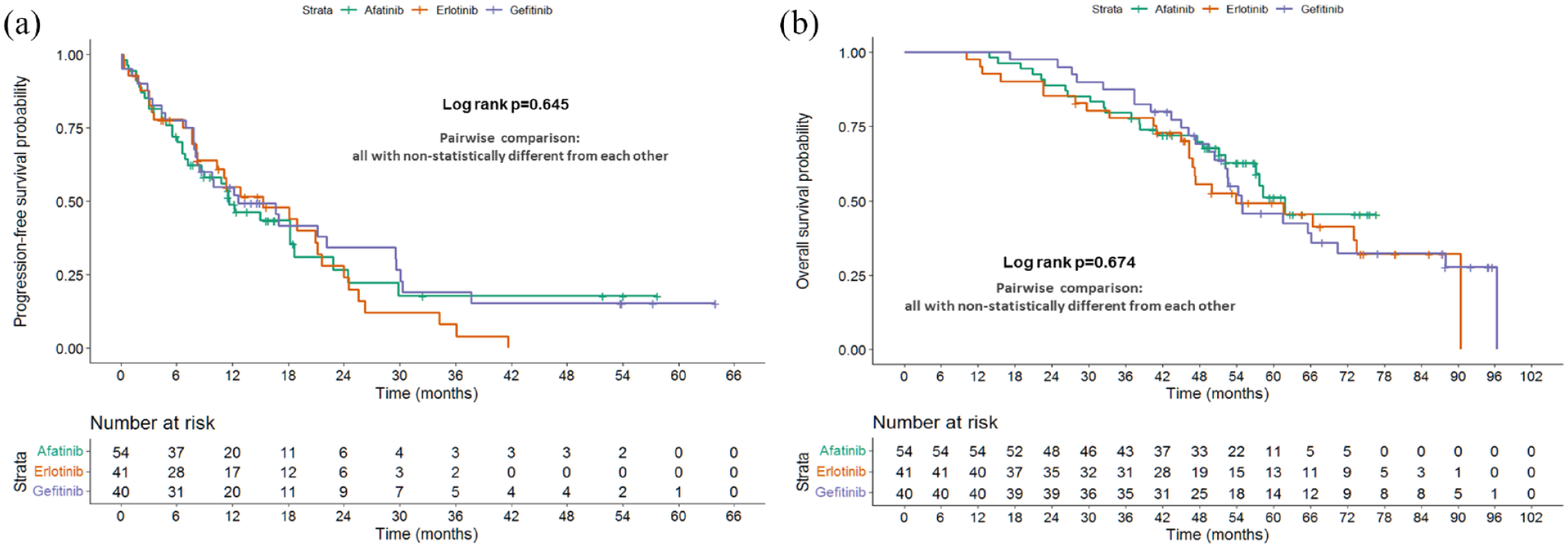
Treatment outcomes of second-line osimertinib based on first-line TKIs. (a) The median PFS while on osimertinib among the patients administered first-line afatinib, erlotinib, or gefitinib was 11.6, 15.3, and 12.6 months, respectively, and no significant difference was found between various TKIs (log-rank p = 0.645). (b) The median OS from the initiation of first-line TKIs among the patients who were treated with afatinib, erlotinib, or gefitinib was 61.8, 53.9, and 54.8 months, respectively, with no significant differences between the groups (log-rank p = 0.674).
Treatment outcomes of second-line chemotherapy following first-line EGFR-TKI treatment
Among the patients who received chemotherapy without 3G EGFR-TKIs following progression while on EGFR-TKIs, no significant difference in OS was observed between the patients with T790M positivity and those with T790M negativity (median OS: 30.3 months among those with T790M positivity versus 31.0 months among those with T790M negativity, p = 0.940, Figure 5(a)). However, the median OS was 16.2 months and significantly shorter among the patients with an unknown T790M mutation status than among patients with positive (p = 0.001) and negative T790M mutations (p < 0.0001) (Figure 5(a)) Interestingly, the patients treated with first-line afatinib followed by chemotherapy without 3G EGFR-TKIs had a significantly longer median OS (24.8 months) than the patients initially treated with erlotinib (21.8 months, p = 0.048) or gefitinib (22.3 months, p = 0.022) (Figure 5(b)).

(a) Treatment outcomes of second-line chemotherapy based on the T790M mutation status and (b) first-line TKIs. Among the patients who received chemotherapy without 3G EGFR-TKIs following progression while on TKIs, no significant difference in OS was observed between the patients with T790M positivity and negativity (median OS: 30.3 versus 31.0 months, p = 0.940); their OS was significantly longer than that of the patients with an unknown T790M mutation status (median OS: 16.2 months, p = 0.001) and negative T790M mutation (p < 0.0001). (b) The patients treated with first-line afatinib had a significantly longer median OS (24.8 months) than the patients treated with erlotinib (21.8 months, p = 0.048) and gefitinib (22.3 months, p = 0.022).
Comparison between osimertinib and chemotherapy
T790M genetic testing should be the standard of care among patients with conditions that progress while on EGFR-TKIs, and subsequent treatment should be chosen based on the T790M mutation status. The use of 3G EGFR-TKIs should be applied for patients with T790M positivity owing to the minimal benefit of 3G EGFR-TKI treatment in those without T790M mutations (Figure 3), whereas chemotherapy is the standard treatment for patients with T790M negativity. The median OS was 58.3, 30.3, and 31.0 months among the patients with T790M positivity treated with osimertinib, osimertinib, and chemotherapy and T790M negativity treated with chemotherapy, respectively (Supplementary Figure S1). As expected, the patients with T790M positivity treated with osimertinib achieved the best OS, and no difference was found between those with and without T790M mutations who received chemotherapy without 3G EGFR-TKIs.
Among the 370 patients from the three abovementioned groups, the univariate analysis showed that the PS, adenocarcinoma (versus nonadenocarcinoma) histology, stage IV (versus stage IIIB) cancer, lung metastasis, liver metastasis, brain metastasis, bone metastasis, adrenal metastasis, pericardial metastasis, number of metastatic sites, frontline TKI response, PFS while on first-line TKI, T790M mutation status, and second-line treatment (osimertinib or chemotherapy without 3G EGFR-TKIs) were the prognostic factors associated with OS (Supplementary Figure S1). Meanwhile, the multivariate analysis showed that poor PS (score of ⩾2 versus 0, HR: 2.40, 95% CI: 1.42–4.08, p < 0.001), nonadenocarcinoma histology (HR: 4.35, 95% CI: 1.68–11.27, p = 0.002), stage IV cancer (HR: 1.94, 95% CI: 1.09–3.45, p = 0.024), liver metastasis (HR: 1.62, 95% CI: 1.09–2.41, p = 0.016), brain metastasis (HR: 1.42, 95% CI: 1.06–1.90, p = 0.017), PFS while on first-line EGFR-TKIs, and subsequent chemotherapy without 3G EGFR-TKIs (versus osimertinib, HR: 3.30, 95% CI: 2.17–5.02, p < 0.001) were significant independent unfavorable prognostic factors for OS (Table 2).
Univariate and multivariate analysis of prognostic factors of overall survivals in patients treated with EGFR-TKIs following by osimertinib or chemotherapy.

CI, confidence interval; 19del, exon 19 deletion; CR, complete response; NA, not assessed; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease; TKI, tyrosine kinase inhibitor.
Discussion
The results of our study provide important clinical information for clinicians dealing with sequential 1G/2G and 3G EGFR-TKI therapy for advanced EGFR-mutated NSCLC patients. The overall positive rate of acquired T790M mutation after 1G/2G EGFR-TKI treatment in patients with NSCLC harboring common EGFR mutations was 53.6%, and tissue biopsy had a higher positive rate for T790M mutation (54.5%) than did liquid biopsy (42.9%). The patients with exon 19 deletion and PFS of >12 months who received first-line EGFR-TKI treatment experienced increased T790M positivity compared with the patients with L858R mutation and PFS of <12 months who received EGFR-TKI treatment. First-line 1G/2G EGFR-TKIs did not have an impact on T790M positivity, and sequential osimertinib therapy was more effective (higher ORR, higher DCR, and longer PFS) for patients with T790M mutation than for those without. The patients with T790M mutation who received osimertinib therapy following first-line EGFR-TKI treatment had a median OS close to 5 years (58.3 months), and the choice of first-line EGFR-TKIs did not affect the efficacy of subsequent osimertinib regarding response, PFS, or OS. Finally, the patients who received chemotherapy without 3G EGFR-TKIs following first-line EGFR-TKIs, regardless of the presence of T790M mutation, had a median OS less than 3 years (30.3 months for those with T790M positivity, 31.0 months for those with T790M negativity, and 16.2 months for those with an unknown T790M mutation status).
Previous studies have shown that secondary T790M mutation is an important biomarker for subsequent osimertinib therapy in advanced EGFR-mutated NSCLC patients who have progressive disease after first-line 1G/2G EGFR-TKI treatment because osimertinib prominently benefits the ORR and PFS of patients with secondary T790M mutation.17,26,27 In previous studies, including prospective studies (e.g. the AURA Extension and AURA2 trials) and real-world clinical studies, the acquired T790M detection rates ranged from 50% to 60%.17,23–25 The positive rate of acquired T790M mutation in this study was 52.5%, which is consistent with previous studies.
After understanding the occurrence rate of secondary T90M mutation among patients with acquired resistance to 1G/2G EGFR-TKIs treatment, the issue of identifying the potential predictive factors for acquired T790M mutation emerges. In the analysis of our study, the baseline exon 19 deletion and duration of PFS while on first-line EGFR-TKIs (>12 months) were identified as independent predictive factors associated with acquired T790M mutation. Similar to our study, two previous real-world studies showed that a first EGFR-TKI treatment duration longer than 12 months was a predictive factor for the appearance of the secondary T790M mutation.17,26 In the same two previous studies, baseline exon 19 deletion had a trend of a higher secondary T790M mutation rate than baseline L858R, but no statistical significance was achieved in either study.17,26 The patient number may contribute to this difference because more than 500 patients in our study had secondary T790M mutation tests, but the patient numbers in the studies of Wu et al. and Chiang et al. were 407 and 240, respectively.17,26 These two previous studies also reported that baseline exon 19 deletion and longer first-line treatment were positively associated with secondary T790M mutation rate.26,27 The use of different EGFR-TKIs did not influence the occurrence of the acquired T790M mutation, which is compatible with most reports.26,27 Taken together, these results suggest that secondary T790M mutation tests could be more useful for those with baseline exon 19 deletion mutations or first-line EGFR-TKI treatment durations longer than 12 months, and repeated tissue biopsies and multiple detection tests (e.g. next-generation sequencing and liquid biopsy) may be appropriate. In the report by Chiang et al., 26 repeated biopsy was found to increase the rate of T790M mutation detection. In addition, either tissue-based or plasma-based sequencing alone was insufficient to completely detect driver mutations, so using both techniques for detection should be complementary.28,29 The abovementioned findings are compatible with our findings and suggest that repeated sequencing using tissue or liquid biopsies should be considered, particularly for patients with baseline exon 19 deletion or long periods on first-line EGFR-TKI therapy, but their first biopsies show no evidence of T790M mutations. Repeated sequencing via tissue or liquid biopsies may help identify more patients who are candidates for osimertinib treatment.
Our study results showed that different first-line 1G/2G EGFR-TKI treatments did not impact the PFS of subsequent second-line osimertinib therapy. In addition, the median OS of 1G/2G EGFR-TKIs and osimertinib therapy sequential therapy was 58.1 months in our study, and there was no statistical significance for the different sequential combination therapies (median OS: afatinib–osimertinib 61.8 months; erlotinib–osimertinib 53.9 months; and gefitinib–osimertinib 54.8 months). Similar findings were reported in the other two previous studies; no significant differences were found among the different first-line EGFR-TKIs in terms of PFS or OS during subsequent osimertinib therapy.26,30 Although one study reported that afatinib therapy followed by osimertinib therapy might be superior to 1G EGFR-TKI treatment, the benefit was limited in terms of ORR and DCR but not PFS or OS. 31 In the subgroup analysis of previous studies that investigated the outcome of EGFR-mutated NSCLC patients receiving sequential afatinib and osimertinib therapy, the median OS of these studies was between 4 and 5 years in the Asian population.20,28,32 In contrast, the patients with T790M mutation who received chemotherapy without 3G EGFR-TKIs had an OS similar to those without T790M mutation, which was significantly shorter than the OS of the patients with T790M mutation who received osimertinib treatment.
In our study, 40%–50% of the patients had no acquired T790M mutation after 1G/2G EGFR-TKI treatment and were not considered suitable for osimertinib treatment in clinical practice. Osimertinib demonstrated very limited efficacy among patients without the acquired T790M mutation, which is likely due to an alternative resistance mechanism in this population. Next-generation sequencing is suggested to identify possible resistance targets; 29 otherwise, chemotherapy (with or without immunotherapy) is the standard treatment for patients without T790M mutation. 33 The T790M-mutated patients treated with subsequent chemotherapy alone had a similar median OS to those without T790M treated by chemotherapy in our analysis, and this result aligns with that in previous reports.26,32 These results in our and previous studies indicate that chemotherapy plays a role in treating advanced EGFR-mutated NSCLC patients, and the timing of chemotherapy administration in these patients may depend on whether the acquired T790M mutation appears.
Although rebiopsy has been recommended as a routine practice in EGFR-mutated patients who experience progressive disease after first-line EGFR-TKI treatment, some patients receive subsequent therapy directly without rebiopsy. The main reasons why patients did not undergo rebiopsy include personal acceptance, older age, poor performance, tumor site unapproachable by a procedure, and a tiny central nervous system (CNS) metastasis.34–36 Older age, poor performance status, and CNS metastasis are known unfavorable clinical factors associated with survival and outcome in advanced EGFR-mutated patients.37,38 Among the patients with unknown T790M status in our study, 62 patients still received osimertinib (n = 25) and other 3G EGFR-TKIs (n = 37). Chemotherapy is currently recommended as second-line therapy for EGFR-mutated patients without secondary T790M after 1G/2G EGFR-TKI therapy because the genetic alterations in T790M-negative patients are heterogeneous and resistant to osimertinib or other 3G EGFR-TKIs.39,40 Taken together, these findings explain why patients with unknown T790M status (median OS 16 months) had shorter survival than those with known T790M-positive (median 31 months) and T790M-negative (median 30 months) status in this study.
Some limitations exist in our retrospective study. First, T790M mutation was detected using different methods, including direct sequencing, polymerase chain reaction, or next-generation sequencing, primarily based on the affordability of the testing. Although different sensitivities should be considered among various tests, the overall positivity for T790M mutation in this study is compatible with previous reports. False-negative testing results might have existed, which could explain why some patients (3/12, 25%) without T790M mutation still responded to osimertinib. Second, we did not analyze the impact of subsequent 1G/2G EGFR-TKI treatment and immunotherapy. The efficacy of subsequent 1G/2G EGFR-TKI treatment after progression while on first-line 1G/2G EGFR-TKIs is limited,41,42 so the strategy of switching to alternative 1G/2G EGFR-TKIs after progression is not widely used in clinical practice. Immunotherapy is generally considered inactive for EGFR-mutated NSCLC unless it is used in combination with chemotherapy, which is currently being clinically studied. Finally, for the 3G EGFR-TKIs other than osimertinib, patients may be enrolled in different clinical trials with other 3G EGFR-TKIs that are not approved by the US Food and Drug Administration (FDA).43,44 Some patients enrolled in the clinical trials conducted in our institutions, and their clinical data, such as treatment efficacy, are not currently available. There were some patients who obtained osimertinib outside of our institution, in some cases even outside of the country, and their information on osimertinib administration is uncertain. Therefore, the results of patients taking 3G EGFR-TKIs other than osimertinib were unreliable, so we did not analyze the clinical outcomes of this patient group.
Conclusion
Our study comprehensively evaluated sequential treatment with 1G/2G EGFR-TKIs followed by osimertinib or chemotherapy in patients with NSCLC harboring EGFR mutations. Real-world experience highlighted the importance of T790M mutation testing and the use of osimertinib, which yielded favorable survival outcomes.
Supplemental Material
sj-docx-2-tar-10.1177_17534666221132731 – Supplemental material for Sequential treatment in advanced non–small cell lung cancer harboring EGFR mutations
Supplemental material, sj-docx-2-tar-10.1177_17534666221132731 for Sequential treatment in advanced non–small cell lung cancer harboring EGFR mutations by Ping-Chih Hsu, John Wen-Cheng Chang, Ching-Fu Chang, Chen-Yang Huang, Cheng-Ta Yang, Chih-Hsi Scott Kuo, Yueh-Fu Fang and Chiao-En Wu in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-tif-1-tar-10.1177_17534666221132731 – Supplemental material for Sequential treatment in advanced non–small cell lung cancer harboring EGFR mutations
Supplemental material, sj-tif-1-tar-10.1177_17534666221132731 for Sequential treatment in advanced non–small cell lung cancer harboring EGFR mutations by Ping-Chih Hsu, John Wen-Cheng Chang, Ching-Fu Chang, Chen-Yang Huang, Cheng-Ta Yang, Chih-Hsi Scott Kuo, Yueh-Fu Fang and Chiao-En Wu in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
