Abstract
Introduction:
Brain metastases (BM) are common in advanced non-small cell lung cancer (NSCLC), and the prognosis is poor with few therapeutic options. This study evaluated the efficacy of three epidermal growth factor receptor–tyrosine kinase inhibitors (EGFR-TKIs) in preventing and treating BM in patients with EGFR mutation-positive advanced NSCLC.
Methods:
Patients with EGFR mutation-positive advanced NSCLC who visited a tertiary referral center from 1 December 2013 to 30 November 2017 were analyzed retrospectively. They received gefitinib, erlotinib, or afatinib until disease progression, death, or intolerable adverse events. The cumulative incidence of subsequent BM of initial non-BM patients, progression-free survival (PFS), and overall survival (OS) of the BM and non-BM patients were estimated and compared using the Kaplan–Meier and log-rank tests.
Results:
306 NSCLC patients were enrolled, with 116, 75, and 115 receiving first-line gefitinib, erlotinib, and afatinib, respectively. The afatinib group had a better PFS [12.7
Conclusions:
Our data suggested that, compared with gefitinib, afatinib provided better benefits significantly in terms of PFS and OS. Both had the same effectiveness in preventing subsequent BM.
Introduction
Brain metastases (BM) have a poor prognosis and typically result in the deterioration of a patient’s quality of life, and about 25–45% of patients with lung cancer develop BM during the course of their disease. 1 According to our own recent study and another study, epidermal growth factor receptor (EGFR) mutation is one of the predictors for subsequent BM.2,3 The previous standard management for BM included irradiation (whole-brain radiation therapy and stereotactic radiosurgery) and surgical resection. Traditional cytotoxic agents usually do not penetrate the blood–brain barrier (BBB) and show a suboptimal intracranial response rate of 30% and a median overall survival (OS) of 7.7 months. 4 Owing to their good penetration through the BBB, the use of EGFR–tyrosine kinase inhibitors (TKIs) to treat BM in patients with EGFR mutation-positive non-small cell lung cancer (NSCLC) has drawn increasing attention. In a phase II study of EGFR mutation-positive NSCLC patients with BM, treatment with a first-line reversible EGFR-TKI, erlotinib or gefitinib, showed a disease control rate of 93%, while the median progression-free survival (PFS) and OS were 6.6 and 15.9 months, respectively. 5 Afatinib is a second-generation TKI that binds irreversibly to EGFR, and is the first TKI with significant overall survival benefits in comparison with traditional chemotherapy. Although afatinib is less validated for the treatment of BM than gefitinib or erlotinib, a combined analysis of the BM subgroups in the LUX-Lung 3 and LUX-Lung 6 trials revealed a better PFS for the afatinib group than the chemotherapy group. 6
Despite better survival outcomes in BM patients treated with EGFR-TKIs, brain recurrence after a good response to first-line EGFR-TKIs remains a major problem. Given the severe effect of BM on survival, the prevention of such metastases by an effective treatment is crucial. It has been reported that central nervous system (CNS) recurrence after an initial response to an EGFR-TKI was observed in 25–33% of patients treated with gefitinib and 1–8% of patients treated with erlotinib, respectively.7–10 Although another recent study also reported a difference between erlotinib and gefitinib with respect to the prevention of CNS recurrence and the treatment of CNS metastases, 11 there has been no report directly comparing all three first-line EGFR-TKIs, that is, gefitinib, erlotinib, and afatinib, in terms of their effectiveness in preventing and controlling BM in NSCLC patients harboring EGFR mutations. This retrospective study thus compared the effects of gefitinib, erlotinib, and afatinib in the prevention and control of BM in patients with EGFR mutation-positive advanced NSCLC. Moreover, we performed Cox proportional hazards regression for the predictors of subsequent BM and determinants of PFS and OS after BM.
Materials and methods
This study was reviewed and approved by the Review Board and Ethics Committee of National Cheng Kung University Hospital (NCKUH B-ER-106-212). All data were anonymized, and, given the retrospective nature of the study, the need for written informed consent was waived. This research was carried out in accordance with approved guidelines and the Declaration of Helsinki. All of the EGFR mutation-positive patients with newly diagnosed or recurrent advanced NSCLC who visited the National Cheng Kung University Hospital from 1 December 2013 to 30 November 2017 were enrolled in the study. The patients all underwent a chest computed tomography (CT) scan, bone scan, and brain imaging [CT or magnetic resonance imaging (MRI)] for staging, based on the tumor, node, metastasis (TNM) system proposed by the American Joint Committee on Cancer, 7th edition. Stage I–IIIA patients were excluded, leaving only stage IIIB–IV patients in the analysis set.
We recorded the baseline characteristics of these patients, including age, sex, mutation subtype, performance status, initial BM, and TNM staging. All of the patients took gefitinib, erlotinib, or afatinib at the discretion of the treating providers and underwent brain imaging at the initial diagnosis or the recognition of advanced disease. Follow-up brain MRI or CTs were arranged by the doctors according to CNS signs or symptoms. CNS metastases included parenchymal BM and radiographically diagnosed leptomeningeal disease. The treatment modalities, including TKIs and radiotherapy, were recorded. Disease progression was determined based on the radiographic evidence according to Response Evaluation Criteria in Solid Tumors version 1.1. 12
Epidermal growth factor receptor mutation analysis
Tumor tissues from primary lung tumors or metastatic lesions were obtained for EGFR mutation analysis. Tissue samples that consisted of >80% tumor content, as determined
Statistical analysis
The frequencies and descriptive statistics of the demographic and clinical variables were calculated. Categorical variables were compared using the Chi-square test or Fisher’s exact test, whereas continuous variables were compared using Student’s
Results
Characteristics of patients
A total of 306 patients who visited the hospital from 1 December 2013 to 30 November 2017 were enrolled. Of those patients, 263 had newly diagnosed and 43 had recurrent EGFR mutation-positive advanced NSCLC. Figure 1 details the inclusion of subjects for analysis. Of the included patients, 116 (37.9%) patients received gefitinib, 75 (24.5%) patients received erlotinib, and 115 (37.6%) received afatinib as first-line therapy. Higher proportions of the patients who received afatinib had a better performance status (Table 1) and exon 19 deletions. The proportion of patients with initial BM was higher among the patients who received erlotinib. The distributions of EGFR mutation subtypes among three TKI treatment groups are summarized in Supplementary Table 1.

Flow chart describing enrollment of patients in the study.
Demographic and clinical characteristics of patients.

BM, brain metastases; ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor; PS, performance status; NA, not applicable.
Progression-free survival and overall survival of all patients
A comparison of PFS and OS of all the patients stratified by the different EGFR-TKIs is shown in Figure 2. PFS was significantly longer in the patients who received afatinib compared with those who received erlotinib or gefitinib [log-rank test,
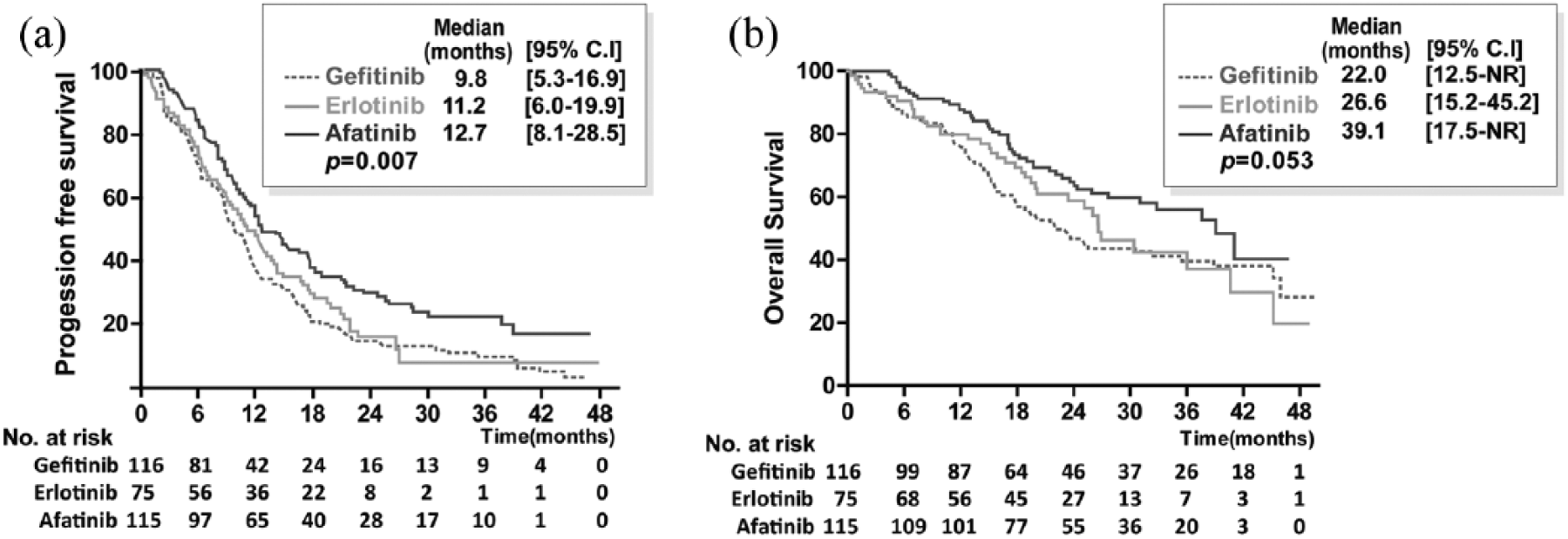
Progression-free survival (a) and overall survival (b) in patients with non-small cell lung cancer and epidermal growth factor receptor gene mutations treated with gefitinib, erlotinib, or afatinib.
Cox proportional hazards regression of all patients for progression-free survival and overall survival.

BM, brain metastases; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor; HR, hazard ratio; PS, performance status.
The prevention and treatment of brain metastases by the three first-line epidermal growth factor receptor–tyrosine kinase inhibitors in patients with epidermal growth factor receptor-mutated non-small cell lung cancer
At the initiation of treatment, there were 93, 41, and 85 patients in the gefitinib, erlotinib, and afatinib groups, respectively, who were free of BM. The cumulative incidences of subsequent BM at 6, 12, 24, and 36 months were 3.8%, 13.9%, 34.6%, and 53.6%, respectively, in the gefitinib group; 5.6%, 9.3%, 9.3%, and 60.3%, respectively, in the erlotinib group; and 0%, 2.8%, 28.3%, and 41.5%, respectively, in the afatinib group, indicating no significant difference [

The prevention and treatment for brain metastases by three first-line epidermal growth factor receptor–tyrosine kinase inhibitors in patients with epidermal growth factor receptor mutation-positive advanced non-small cell lung cancer.
The analysis results of PFS stratified by the initial diagnosis of BM are shown in Figure 3(b). Patients with initial BM were associated with a shorter median PFS than those without initial BM [8.9
Cox proportional hazards regression for progression-free survival and overall survival of patients with initial brain metastases.

ECOG, Eastern Cooperative Oncology Group; CI, confidence interval; EGFR, epidermal growth factor receptor; HR, hazard ratio; PS, performance status.
Discussion
To the best of our knowledge, this is the first real-world study evaluating and comparing the impact of three first-line EGFR-TKIs in the prevention and treatment of BM in patients with EGFR mutation-positive advanced NSCLC. The PFS and OS were better for afatinib than for gefitinib in all the patients, and among those without BM. Afatinib also provided the same effectiveness in preventing and treating BM as gefitinib and erlotinib, as shown in Figures 2 and 3(a). BM remains a major complication in lung cancer patients due to the limited penetration of the BBB by chemotherapy. Recent studies have demonstrated that EGFR-TKIs have better intracranial efficacy than chemotherapy.6,17 However, a real-world study comparing the efficacy of different EGFR-TKIs in preventing or treating BM has not previously been published. Li and colleagues showed that the time to neurological progression was effectively extended in pre-existing BM patients with EGFR-sensitizing mutations initially treated with erlotinib compared with gefitinib (30
Since many studies have found that lung cancer patients with EGFR mutations tend to develop BM, choosing a treatment that can help to prevent BM is also critically important. Heon and colleagues demonstrated that lower rates of CNS progression were noted in EGFR-mutant advanced NSCLC patients initially treated with an EGFR-TKI than in such patients treated with chemotherapy. 17 In that study, the cumulative incidences of subsequent BM at 6, 12, and 24 months were 1%, 6%, and 21%, respectively, in the EGFR-TKI group; findings that partially validate our results. Our study further disclosed that mediastinal lymph node involvement was a poor prognostic factor for subsequent BM, a finding which also corroborates the results of other studies.14,22,23
Unlike many retrospective studies reporting that erlotinib is more effective than gefitinib in preventing and treating BM,11,24–26 our study showed similar effects among the three EGFR-TKIs. The mechanism underlying a difference in the treatment effects of erlotinib and gefitinib has been considered attributable to the difference in maximum tolerated dose (MTD); the MTD of gefitinib is one third that of erlotinib.
27
Therefore, the concentration of erlotinib in the CNS would be relatively higher than that of gefitinib. In addition, several investigations have also shown that the concentration of erlotinib in cerebrospinal fluid (CSF) is higher than that of gefitinib. However, the integrity of the BBB has been reported to be disrupted by the tumors themselves, usually at the later stages of the disease.
28
In addition, the tumors themselves facilitate angiogenesis without a normal BBB. These findings can be radiologically observed in terms of brain edema around BM and
Several limitations of the current study must be acknowledged. First, it was a single-center retrospective study, and there were significant differences in the characteristics of the three groups. The participants who received afatinib included higher proportions with better performance and exon 19 deletions, whereas those who received erlotinib included a higher proportion with BM. These differences have also been noted in some real-world studies,
21
and one possible explanation is that physicians usually prescribe afatinib as a first-line treatment based on the favorable OS of patients with exon 19 deletions.
33
Furthermore, because many studies have reported that erlotinib is more effective in treating BM than gefitinib,11,24–26 physicians may tend to prescribe erlotinib for patients with initial BM.
11
Although we had tried to control for possible confounders using the Cox model, a subgroup analysis comparing three groups of patients with exon 19 deletions and Eastern Cooperative Oncology Group performance statuses of 0–1 showed that the PFS and OS were similar among patients receiving different kinds of EGFR-TKIs (Supplementary Figure 2 and Supplementary Table 5). Given that there were more BM in the erlotinib group, no statistical difference in the proportion of patients receiving brain radiotherapy (
In conclusion, our study revealed a better PFS in patients treated with afatinib in comparison with patients treated with gefitinib. Furthermore, compared with the other two EGFR-TKIs, afatinib provided similar intracranial efficacy in patients with or without pre-existing BM. This is the first study to directly compare first- and second-generation TKIs in terms of their effectiveness in preventing and treating CNS metastases. Prospective studies with patients of matched characteristics and regular brain images would be worthwhile for future research.
Supplemental Material
Supp_Figure_1 – Supplemental material for Preventing and treating brain metastases with three first-line EGFR-tyrosine kinase inhibitors in patients with EGFR mutation-positive advanced non-small cell lung cancer
Supplemental material, Supp_Figure_1 for Preventing and treating brain metastases with three first-line EGFR-tyrosine kinase inhibitors in patients with EGFR mutation-positive advanced non-small cell lung cancer by Po-Lan Su, Yi-Lin Wu, Wei-Yuan Chang, Chung-Liang Ho, Yau-Lin Tseng, Wu-Wei Lai, Wu-Chou Su, Chien-Chung Lin and Szu-Chun Yang in Therapeutic Advances in Medical Oncology
Supplemental Material
Supp_Figure_2AB – Supplemental material for Preventing and treating brain metastases with three first-line EGFR-tyrosine kinase inhibitors in patients with EGFR mutation-positive advanced non-small cell lung cancer
Supplemental material, Supp_Figure_2AB for Preventing and treating brain metastases with three first-line EGFR-tyrosine kinase inhibitors in patients with EGFR mutation-positive advanced non-small cell lung cancer by Po-Lan Su, Yi-Lin Wu, Wei-Yuan Chang, Chung-Liang Ho, Yau-Lin Tseng, Wu-Wei Lai, Wu-Chou Su, Chien-Chung Lin and Szu-Chun Yang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Po-Lan Su and Yi-Lin Wu contributed equally to this work. We are indebted to Yi-Ting Yen, Wen-Ping Su, Shang-Yin Wu, Yu-Ming Yeh, and Cheng-Hung Lee for their generous support with the recruitment of subjects. This study is based in part on data from the Cancer Data Bank of National Cheng Kung University Hospital.
Funding
This study was supported by the grant MOHW106-TDU-B-211-144-004 from the Ministry of Health and Welfare, Taipei, Taiwan; the grants MOST 104-2314-B-006-046-MY3 and MOST105-2314-B-076- MY2 from the Ministry of Science and Technology, Taipei, Taiwan; and the grant NCKUH-10503002 from National Cheng Kung University Hospital, Tainan, Taiwan.
Conflict of interest statement
The authors declare that there is no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
