Abstract
Epidermal growth factor receptor (EGFR) mutation is one of the key oncogenic mutations in non-small-cell lung cancer with adenocarcinoma histology. Exon 19 deletions and exon 21 L858R substitutions account for 90%, while EGFR exon 20 insertions constitute 4–10% of EGFR mutations and are the third most prevalent activating EGFR mutations. EGFR exon 20 insertions are associated with decreased sensitivity to EGFR tyrosine kinase inhibitors and, until recently, effective targeted therapy against these tumours remained an unmet clinical need and chemotherapy was the only treatment of choice available. The approval of amivantamab and mobocertinib for patients who have progressed after chemotherapy represents an important step forward in the management of these patients. Here in this review, we summarize the epidemiology, structure and the tumour microenvironment of EGFR exon 20 insertion and also review the systemic treatments, including targeted therapies and ongoing clinical trials in EGFR exon 20 insertion mutations, as well as detection methods for EGFR exon 20 insertion. Lastly, resistant mechanisms and future directions are addressed.
Keywords
Introduction
Epidermal growth factor receptor (EGFR) mutations represent a key oncogenic driver alteration in non-small-cell-lung cancer (NSCLC) with a frequency ranging from 10–15% and up to 50% in Caucasians and East Asians, respectively. 1 Of the EGFR mutations, exon 19 deletions and exon 21 Leu858Arg point mutation (L858R), also known as common mutations, account for 90% of all EGFR mutations. 2 These mutations are sensitive to EGFR tyrosine kinase inhibitors (TKIs), including first-generation EGFR TKIs gefitinib and erlotinib, second-generation afatinib and dacomitinib and third-generation osimertinib.3–8 Uncommon mutations, including exon 18 G719X, exon 20 S768I, exon 21 L861Q, exon 20 insertions and complex mutations are resistant to first-generation EGFR TKIs. 9 Thus, afatinib remains the preferred regimen for uncommon EGFR mutations such as G719X, S768I and L861Q. Patients with NSCLC with EGFR exon 20 insertion mutations are usually resistant to EGFR TKIs, and until recently, chemotherapy was the only treatment of choice available. 10 The recent approval of two targeted agents, amivantamab 11 and mobocertinib 12 for patients with advanced NSCLC harbouring EGFR exon 20 insertion mutations who have progressed after chemotherapy (Figure 1) confirms this molecular alteration is an actionable target that warrants further exploration.

Timeline of development of targeted therapies for EGFR exon 20 mutations.
Herein, the epidemiology, structure, and genomic and tumour microenvironment of EGFR exon 20 insertion mutations are covered. The review highlights the systemic treatments, including targeted therapies and ongoing clinical trials in EGFR exon 20 insertion mutations, as well as detection methods for EGFR exon 20 insertion mutations. Lastly, resistant mechanisms and future directions are addressed.
Epidemiology of EGFR exon 20 insertion mutations
When considered as separate molecular subset, EGFR exon 20 insertion mutations constitute about 1–2% of all NSCLC cases, but among EGFR mutations, 4–10% of all observed EGFR mutations in NSCLC.10,13,14 In addition to lung adenocarcinoma, other solid tumours such as glioblastoma, urothelial carcinoma and endometrial adenocarcinoma harbour EGFR exon 20 insertion mutations, and overall, EGFR exon 20 insertion mutations comprise of 0.35% of identified mutations in AACR project GENIE. 15 Similar to other oncogenic molecular alterations in NSCLC, EGFR exon 20 insertion mutations are usually mutually exclusive with other genetic driver alterations, and have similar clinical characteristics with the classical activating EGFR mutations, which are enriched in non-smokers, Asian and adenocarcinoma histology. 16 A recent literature studied the frequency of mutation according to population ethnicity or global region. 17 EGFR exon 20 insertion mutations accounted for 1–12% of EGFR mutations, and the frequency was 0.1–4% for all NSCLC cases. Comprehensive genomic profiling of NSCLC also revealed that EGFR exon 20 insertion mutation was identified in 12% of all EGFR-mutant NSCLC, with various spectrum of EGFR exon 20 insertion variants. 18 The most common variants were D770_N771 > ASVDN and N771_P772 > SVDNP, which account for 21% and 20% of all EGFR exon 20 insertion mutations, respectively. The frequency of EGFR exon 20 insertion mutations was as high as 4% in Asia Pacific, whereas it was reported 1.3% of all NSCLCs in Europe. 17 It is difficult to conclude that there is geographic variation, because the frequency of EGFR exon 20 insertion mutations may be underestimated due to insufficient testing methods that may only detect common EGFR mutations. 17
Structure of EGFR exon 20 insertion mutations
EGFR exon 20 insertion mutations can be largely classified as inframe insertions or duplications of 3–21 base pairs typically occurring between AA761 and AA775.13,18 EGFR exon 20 insertion mutations contains two essential regions: the regulatory C-helix domain (AA762-766) and the adjacent loop (AA767-774) following C-helix. The most frequent site of mutations identified in EGFR exon 20 insertion mutation is in the loop following C-helix, specifically between exons 767 and 774. More than 100 unique activating EGFR exon 20 insertion mutations have been identified, unlike the homogeneous mutations in common EGFR mutations. The location and frequency of each EGFR 20 insertion mutation is described in Figure 2.13,18,19

Location of EGFR exon 20 insertion mutations (a) and various spectrum of EGFR exon 20 insertion (b) 13,18,19.
The unique mechanism of EGFR exon 20 insertion mutations is associated with the altered structure upon mutations. As reported by Yasuda et al., the crystal structure of D770_N771insNPG revealed a shift of the C-helix, creating an inward position that keeps the active state of EGFR in the absence of ligand binding. 13 Thus, this shift in conformation enables continuous activation of EGFR with markedly hinderance in TKI binding, resulting in resistance to EGFR TKIs used in the treatment of common sensitizing EGFR mutations. 19
Due to their heterogeneous location of the insertions, the in vitro sensitivity to EGFR inhibitors may be variable (Figure 3).13,20–22 A classic example of EGFR exon 20 insertion mutation is D770_N771insNPG, which activates but has no affinity for first-generation EGFR TKIs. In contrast, A762_Y764insFQEA insertions may respond to first-generation EGFR TKIs, and have shown 10-fold higher binding affinity than other EGFR exon 20 insertion mutations. 13 Isolated case reports have been published on the sensitivity of H773dup, H773_V774insNPH and N771delinsKG mutations to afatinib, but the clear mechanism of response needs to be further investigated in prospective studies.20–22

Heat map representation of IC50 values for proliferation of therapeutic agents in EGFR exon 20 insertion. The colours mark the sensitivity to different agents: sensitive (green, <10), intermediate (yellow, 10–99) and resistant (red, >100).
Genomic and tumour microenvironment characteristics
The characterization of genomic and immune microenvironment of EGFR exon 20 insertion mutant NSCLC-tumours currently remains scarce. NSCLC harbouring EGFR exon 20 insertion mutation is associated with co-occurring genomic alterations associated with tumour suppressor and cell cycle alterations, notably with TP53 mutations, cyclin-dependent kinase inhibitor 2A/2B (CDKN2A/2B) genes, as well as NK2 homebox 1 (NKX2-1), and RB transcriptional co-repressor 1 (RB1) genes. 18 These alterations were also noted in other sensitizing EGFR mutations such as exon 19 del and L858R at comparable frequency. Similarly, TP53 and CDKN2A were identified as co-occurring mutations in addition with PIK3CA and EGFR amplifications. 23
With regards to programmed death-ligand 1 (PD-L1) tumour expression, in one study, 76% (107/141) of tumour samples were negative for PD-L1 expression. The tumour mutational burden (TMB) have tended to be low, with reports ranging between 3.4 and 4.6 mutations/Mb. 24 The low TMB observed in tumours with EGFR exon 20 insertion mutation is similar to NSCLC with common EGFR exon 19 del and L858R mutations. 18 Taken together, the data suggest the activity of immune checkpoint inhibitors (ICIs) is likely to be blunted in NSCLC harbouring EGFR exon 20 insertion mutations and the immune phenotype of these tumours should be further evaluated.
Systemic therapies
Currently, the first-line treatment for patients with EGFR exon 20 insertion mutations is platinum-based chemotherapy, and amivantamab or mobocertinib in second-line setting. In this section, we discuss on the role of chemotherapy ± immunotherapy, and the development of novel targeted agents
Chemotherapy ± immunotherapy
In patients with advanced NSCLC with EGFR exon 20 insertion mutations, treatment with a platinum chemotherapy was associated with a longer time to treatment discontinuation (TTD) of 7 months, and a median overall survival (OS) of 20 months, whereas in patients without molecular alterations the TTD and OS was 4 and 12 months, respectively. 25 Other retrospective studies have reported an overall response rate (ORR) of 11–39% and median progression-free survival (PFS) of 6.4–8.9 months with platinum-based chemotherapy.26,27 These results are in sharp contrast to treatment with EGFR TKIs, with an ORR of 13%, and median PFS and OS of 3.4 and 9.5 months respectively.28–30 While chemotherapy is the standard of treatment for patients with EGFR exon 20 insertion mutations, clinical trials with targeted therapies for EGFR exon 20 insertion mutations are strongly recommended due to the limited activity and toxicity of platinum-based chemotherapy. 31
As alluded previously, EGFR exon 20 insertion mutations have reduced efficacy with ICIs.32,33 In patients treated with ICIs as monotherapy as first-line treatment, the ORR was 9.1% and a median PFS and OS of 3.1 and 11 months, respectively. 34 A meta-analysis on EGFR exon 20 insertion mutations reported similar outcomes. 35 When treated with ICIs, EGFR exon 20 insertion mutations demonstrate higher disease control rate of 6 and 12 months of 36% and 11% compared with classic EGFR mutations of 16% and 0%, respectively. 32 The addition of platinum-based chemotherapy to ICIs have reported an ORR of 18.8% with a median PFS and OS of 4.5 and 11.3 months.
Targeted therapies
Until recently, effective targeted therapy against NSCLC tumours with EGFR exon 20 insertion mutations remained an unmet clinical need. The tyrosine kinase (TK) domain of EGFR induces activation of signalling pathway such as phosphatidylinositol 3-kinase (PI3K)/AKT, Janus kinase 2/signal transducer and activator of transcription 3, and Ras/mitogen-activated protein kinase (MAPK) and stimulates downstream components involved in cell proliferation, cell cycle progression, survival and motility.36–39 Gefitinib and erlotinib reversibly bind to EGFR while afatinib and dacomitinib irreversibly bind to EGFR covalently. 40 However, these first- and second-generation EGFR TKIs have shown disappointing clinical against most EGFR exon 20 insertion mutations 41 due to steric hindrance, except for EGFR A763_Y764insFQEA, occurring in 6% of EGFR exon 20 insertion mutations NSCLC, which has been reported as being sensitive to a first-generation EGFR TKI. 13 The use of the third-generation TKI osimertinib in EGFR exon 20 insertion mutations is described below. None of these EGFR TKIs are approved by United States Food and Drug Administration (FDA) for use in patients with EGFR exon 20 insertion mutations.
A key challenge with EGFR exon 20 insertion mutations is the diverse mutational landscape. However in the past 5–10 years, several emergent therapies and clinical trials have been specifically developed for this unique molecular subgroup 14 (Figure 1). With a variety of mechanisms of action, these novel treatments represent an important step forward in the management of patients with EGFR exon 20 insertion mutations NSCLC. Results of these trials are summarized in Table 1. The frequency of treatment-related toxicites of key agents are summarized in Supplementary Figure 1.
Clinical trials in patients with EGFR exon 20 insertion mutations.

ALT, alanine aminotransferase; CI, confidence interval; DoR, duration of response; EGFR, epidermal growth factor receptor; G3, grade 3; NR, not reported; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; TRAEs, treatment-related adverse events; TRDR, treatment-related dose reduction; TTD, time to treatment discontinuation.
Mobocertinib
Mobocertinib (TAK-788) is a first-in-class, potent, oral, irreversible TKI. Mobocertinib and its two active metabolites, AP32960 and AP32914, are approximately equally potent in inhibiting EGFR. Its isopropyl ester domain targets proteins in the vicinity of the alpha C-helix, a binding site not exploited by osimertinib.57–59 The covalent interaction and irreversible binding with EGFR cysteine 797 also lead to increased potency via higher affinity binding, more sustained EGFR kinase activity inhibition and greater overall selectivity. Preclinical in vitro studies demonstrated selectivity of mutant EGFR over wildtype (WT) EGFR, with more potent inhibition of Ba/F3 cells expressing EGFR exon 20 insertion than WT EGFR (IC50 4.3–22.5 nmol/L and 34.5 nmol/L, respectively). There is also in vivo anti-tumour efficacy in patient-derived models and murine orthoptic models. 58 Based on this, mobocertinib was evaluated in a phase I/II dose-escalation/expansion trial which identified 160 mg/day as the recommended phase II dose. 43
In September 2021, mobocertinib 160 mg/day was granted FDA approval. This is the first approval of an oral targeted therapy for patients with EGFR exon 20 insertion mutations. Results were promising with ORR 28% and median PFS 7.3 months in the platinum pre-treated cohort. There was high incidence of EGFR-driven toxicities. Common adverse events (AE) included diarrhoea, rash, paronychia, decreased appetite, nausea, dry skin, vomiting and stomatitis which occurred in >20% of patients. However, most gastrointestinal and skin events were grade 1–2 in severity except for diarrhoea which is the only grade 3–4 treatment-related AE reported in greater than 10% of patients. One patient died from heart failure that was considered treatment related and its product labelling includes a boxed warning for QTc prolongation and Torsades de Points. 44
Amivantamab
Amivantamab is a bispecific antibody targeting MET and EGFR with three distinct mechanisms of actions: inhibition of ligand binding to its target receptors; degradation of these receptor via the lysosome pathway; and Fc-dependent phagocytosis by M1/M2 macrophages and antibody-dependent cellular toxicity by natural killer cells60,61 (Table 1). The phase I CHRYSALIS study is a dose-escalation, dose-expansion study which included a population with EGFR exon 20 insertion mutations (N = 187). In the efficacy population (N = 81) of post platinum population treated with amivantamab, a response rate of 40% and PFS of 8.3 months was reported. Safety profile was manageable. 45 AE associated with EGFR inhibition included rash, paronychia, stomatitis, pruritis and diarrhoea while that associated with MET inhibition included hypoalbuminemia and peripheral oedema with most being grade 1–2. Interstitial lung disease was reported in 4% of patients in the CHRYSALIS study. These findings are clinically meaningful considering the population of interest has relapsed metastatic or unresectable NSCLC with a 5-year survival rate of less than 10%. 62 Based on these results, amivantamab received accelerated approval from the FDA in May 2021 for patients with advanced NSCLC harbouring EGFR exon 20 insertion mutations with progression on or after platinum-based chemotherapy.
In both Study 101 and CHRYSALIS, a variety of EGFR exon 20 insertion mutations were identified and there does not appear to be a clear association of likelihood or depth of response with mutation variants. 55 Although the response rate with amivantamab was numerically greater than mobocertinib (40% versus 28%, respectively), this should be interpreted with caution given cross-trial comparisons. It should be highlighted the OS and PFS appeared similar at 22.8 months and 24 months, 8.3 and 7.3 months for amivantamab and mobocertinib, respectively. There are several differences between amivantamab and mobocertinib. Amivantamab is administered intravenously, initially weekly, then biweekly while mobocertinib is administered orally daily. While both have on-target EGFR toxicities, amivantamab more commonly caused skin rash yet has much lower rates of diarrhoea than mobocertinib. Mobocertinib also had higher rates of treatment-related discontinuations. All grade infusion reactions occurred in 65% of patients receiving amivantamab although the incidence of grade 3 or more infusion reactions was only 3%. Amivantamab’s unique mechanism of action as an antibody to both EGFR and cMet, as well as low fructose backbone with high affinity for FcYRIIIa/CD16a 63 may account for increased selectivity and efficacy with decreased toxicity when compared to other targeted therapies for EGFR exon 20 insertion mutations NSCLC. 62
Other TKIs targeting EGFR exon 20 insertion mutations are under development and are discussed in the following section.
Poziotinib
Poziotinib is an irreversible pan-human epidermal growth factor receptor (HER) inhibitor with activity against mutations or insertions of HER1, HER2 and HER4. Owing to its small size and flexibility and increased halogenation, poziotinib can circumvent steric changes of the EGFR exon 20 insertion mutations drug binding pocket. In vitro testing demonstrated that poziotinib had an average IC50 value of 1.0 nM in Ba/F3 cell lines with an EGFR exon 20 insertions, making it approximately 100 times more potent than osimertinib and 40 times more potent than afatinib. 19
Given its preclinical activity, ZENITH20 study was initiated to evaluate poziotinib in patients with EGFR exon 20 insertion mutations. 64 The 16 mg/day dose was chosen as it was the highest daily dose without dose-limiting toxicity in a phase I study involving HER2 amplified breast cancer. 65 Furthermore, 16 mg/day dosing was also used in a single centre phase II clinical trial enrolling previously treated patients with NSCLC harbouring exon 20 insertions which demonstrated a promising 8-week unconfirmed ORR of 58%. 66 Unfortunately, while the results of the ZENITH20 study 64 reported clinically meaningful activity with an ORR of 14.8% as well as PFS of 4.2 months for previously treated EGFR exon 20 NSCLC, it was tempered by significant side effects due to inhibition of WT EGFR. 28% and 26% of patients had grade 3 or more treatment related rash and diarrhoea, respectively. The incidence of treatment related pneumonitis was 4% although some cases may have been confounded by prior treatment with checkpoint inhibitors.
Pharmacokinetic (PK) simulation demonstrated that twice a day (BID) dosing could reduce toxicity while preserving activity compared to once a day (QD) dosing due to the relatively short half-life (T1/2). 67 The ZENITH20-5 randomized patients to either 10, 12, 16 mg OD or 6, 8 mg BID of poziotinib. Randomized cohorts of QD versus BID dosing (16 mg QD versus 8 mg BID; 12 mg QD versus 6 mg BID) indicate that incidence of ⩾grade 3 AE of rash, diarrhoea and stomatitis was lower with BID dosing (31% versus 21% and 27% versus 16%, respectively) in cycle 1. BID dosing versus QD schedules also resulted in the relative reduction in dose interruptions by 38% and 52%, respectively.67,68
While poziotinib is effective against EGFR exon 20 insertion mutations, heterogeneity in responses may be observed depending on the location of EGFR exon 20 insertion mutation. This is due to marked conformations of distinct regions of receptor known to effect drug binding. Near loop region mutations proximal to the C-helix (AA767-772) influences the orientation of distinct residues of the P-loop which helps to stabilize poziotinib and increase drug binding affinity, which is distinct to insertions occurring at the distal far loop region (AA773-775) (PMID: 34526717). The response rate of poziotinib for near and far loop region mutations was 46% and 0%, respectively (p = 0.0015). In vitro testing has also demonstrated a positive correlation between drug sensitivity and mutation location (R = 0.67, p = 0.0003). 69
CLN 081 (TAS6417)
CLN 081 is another promising novel oral irreversible EGFR TKI with a unique pyrrolopyrimidine scaffold, with potent, broad-spectrum activity against EGFR mutations.70,71 A unique feature of CLN-081 is its potency and selectivity for inhibition of EGFR exon 20 insertion mutations versus WT EGFR. Cell line models harbouring various EGFR exon 20 insertion mutations demonstrated that CLN-081 has a WT to mutant half-maximal inhibitory concentration (IC50) ratio of 134-fold for A763_Y764insFQEA, 17.4-fold for D770_N771insSVD, 17.2-fold for D770_N771insG, 6.37-fold for V769_D770insASV, 4.55-fold for H773_V774insPH and 4.51-fold for H773_V774insNPHA 14 This is in contrast to the first- and second-generation EGFR TKIs erlotinib and afatinib which shows minimal selectivity for mutant receptors. 71
A phase I/II dose-escalation/dose-expansion trial is ongoing with preliminary results reported at ASCO 2022. 53 In all, 73 patients were enrolled across doses ranging from 30 to 150 mg BID. Enrolment at 150 mg BID was stopped after 11 patients based on toxicity. In a heavily pretreated cohort with 66% patients receiving ⩾2 prior lines of treatment and 36% of patients with prior EGFR TKI, the ORR and PFS was 38.4% and 10 months, respectively. There is also a manageable safety profile. Most AEs were grade 1 and 2, dose reductions and dose discontinuations due to AE were uncommon at doses below 150 mg BID at 11% and 6%, respectively, with no ⩾grade 3 rash observed at doses <150 mg. Pneumonitis were observed in four patients, but cases were asymptomatic or confounded by comorbid medical illness. 72
Osimertinib ± necitumumab
While the third-generation EGFR TKI osimertinib has activity against both canonical activating and T790M mutant forms of EGFR, there significant overlap in terms of conformation between EGFR exon 20 insertion mutations and WT EGFR in the ATP-binding pocket. Pre-clinical studies have reported that osimertinib was active in EGFR exon 20 insertion mutant cell lines and tumour xenografts with a wide therapeutic window.73–75 This translates to variable clinical responses for EGFR exon 20 insertion mutations variants. For instance, responses have been reported in patients with A767_V769dup, A763_Y764insFQEA, H773_V774insAH, S768_D770dup and D770_N771insG.76,77
While pre-clinical data proposed a potential activity of osimertinib, there has been conflicting reports of efficacy. Retrospective studies have reported an ORR of 16.7–67.7%. 76 However, in a prospective study of 14 patients treated with osimertinib 80 mg OD, none experienced an objective response. 78
Studies have reported an association between area under the curve (AUC)/IC50 and PFS, indicating the concentration dependent efficacy of osimertinib in EGFR exon 20 insertion mutations. Coupled with data from pre-clinical studies showing a IC50 approximately 10–100 higher for EGFR exon 20 insertion mutations than for EGFR exon 19 deletion, exon 21 L858R and T790M mutations,74,75 this provides a rationale for increase dose intensity of osimertinib.77–79 Furthermore, there is also known safety profile of 160 mg daily for patients with central nervous system (CNS) metastasis.80–83 Based on this rationale, osimertinib at 160 mg OM has been evaluated in two phase II trials ECOG-ACRIN 5162 and POSITION20.46–48 The reported ORR and PFS was 25% and 27%: 9.7 and 5.5 months, respectively, as well as AEs consistent with other reports of this regimen.
Necitumumab is a fully human lgG1 monoclonal antibody targeting EGFR. As a fully human monoclonal antibody, risks of hypersensitivity are expected to be lower. 84 It works by binding to EGFR with higher affinity and specificity than EGF, blocking ligand induced phosphorylation of EGFR and downstream pathway activation. In addition, the Fc portion can also induce antibody-dependent cell-mediated cytotoxicity (ADCC), an important mechanism of antitumour activity related to complement activation and triggering of immune effector cells. 85 The combination of necitumumab and osimertinib was tested in select settings of EGFR TKI resistance including EGFR Exon 20 insertion mutations in the phase I ETCTN California Cancer Consortium study. In this EGFR exon 20 insertion population pretreated with platinum chemotherapy, RR was 22% (4/18) with a PFS of 6.9 months. Rash was the most common grade 3 AE, occurring in 13% of patients.49,50
Sunvozertinib (DZD9008)
Sunvozertinib is an oral, potent, irreversible and selective TKI targeting EGFR exon 20 insertion mutations as well as EGFR sensitizing, T790M and uncommon EGFR mutations with weak activity against WT EGFR. It has 1.4- to 9.6-fold selectivity on EGFR exon 20 insertion mutations compared with EGFR WT. Oral administration of sunvozertinib also demonstrated profound anti-tumour efficacy in a dose-dependent manner in patient-derived xenografts model. 51
Two ongoing phase I trials for sunvozertinib, WU-KONG 1 and WU-KONG 2, are being conducted for metastatic NSCLC harbouring EGFR or HER2 mutations. A pooled analysis was performed to assess safety, PK and antitumour efficacy. 52 In the 56 patients with EGFR exon 20 insertion mutations, the confirmed ORR was 50% across all dose levels. PFS rate at 6 months for 100-mg, 200-mg, 300-mg and 400-mg cohorts was 50%, 53.3%, 44.6% and 44.4%, respectively, and has not been reached. The drug appears tolerable. All grade diarrhoea and rash occurred in 53.9% and 40.2% of patients, respectively, but the incidence of ⩾grade 3 diarrhoea was only 4.9% and no patients experienced ⩾grade 3 rash. Based on safety and tolerability data in the dose-escalation cohorts, 400 mg was defined as the maximum tolerated dose (MTD), and 200 mg to 400 mg were selected for dose expansion.51,86
Furmonertinib (Alflutinib, AST2818)
Furmonertinib is a novel third-generation EGFR TKI targeting both EGFR sensitizing mutations, T790M and sparing WT EGFR. 87 It has shown superior efficacy compared with gefitinib as first-line therapy in patients with EGFR mutation positive (exon 19 deletion or exon 21 L858R) along with an acceptable safety profile. 88 Furmonertinib has also been approved in China on 3 March 2021, for the treatment of EGFR T790M mutant NSCLC based on a phase IIb study showing RR of 74% in patients with de novo or acquired EGFR T790M mutations. 89 There is also an encouraging antitumour activity in EGFR exon 20 insertion mutations NSCLC based on preclinical studies. Furmonertinib effectively inhibited BaF3 cells expressing EGFR exon 20 insertion mutations with mean IC50 of 11–20 nM and was active in patient-derived xenograft models harbouring EGFR exon 20 insertion mutations. In a phase Ib study of previously untreated patients with EGFR exon 20 insertion mutations NSCLC treated with furmonertinib, the ORR was 71% (5/7 patients). 54
Tarloxitinib
Tarloxitinib is a prodrug that harnesses tumour hypoxia to generate high levels of a potent, covalent pan-HER TKI, tarloxotinib-effector (tarloxotinib-E), within the tumour environment. This tumour-selective delivery mechanism was designed to minimize the dose-limiting toxicities of EGFR WT inhibition. 56 PK analysis also confirmed markedly higher levels of tarloxitinib-E in tumour tissue than plasma or skin. In vitro, tarloxitinib-E was demonstrably active, inhibiting cell signalling and proliferation in patient-derived cancer cell lines. In vivo, tarloxotinib induced tumour regression and growth inhibition in murine xenograft models. The RAIN-701 trial of tarloxotinib 150 mg/m2 IV weekly exhibits antitumour activity with stable disease in 6/11 (55%) of patients. Most common AEs included prolonged Qtc (All grade 60.9%, ⩾grade 3 34.8%), rash (All grade 43.5%, ⩾grade 3 4.3%) and diarrhoea (All grade 21.7%, ⩾grade 3 4.3%). Treatment-related dose reductions and discontinuations occurred in 21.7% and 4.3%, respectively. 56 Tarloxitinib is also approved in China for treatment of NSCLC after two lines of chemotherapy based on the ALTER 303 trial. 90
BDTX 189
BDTX 189 is an orally available, ATP competitive and irreversible small molecule TKI against families of allosteric HER2 and EGFR mutations while sparing WT EGFR. In the phase I/II Masterkey-01 trial of BDTX-189 in patients with advanced solid tumours harbouring any one of more than 48 oncogenic alterations in EGFR or HER2 oncogene, responses were seen in 1 out of 3 evaluable NSCLC patients. The most frequent AEs occurring in ⩾20% of patients were diarrhoea (All grade: 36%, ⩾G3: 8%), nausea (all grade: 28%, ⩾G3: 0%) and vomiting (all grade: 25%, ⩾G3: 3% G3). 91 Despite encouraging safety and efficacy of BDTX-189 as an inhibitor of oncogenic mutants of EGFR including EGFR exon 20 insertion mutations, its development has been discontinued by Black Diamond Therapeutics as part of a restructuring plan to prioritize the development of other drugs.
BAY-2476568
BAY-2476568 is a potent and selective, reversible inhibitor with 20-fold selectivity for EGFR insertion mutations compared to WT EGFR in EGFR-expressing Ba/F3 cells. 92 Activity has also been demonstrated in vivo in xenograft models. It also shows potent activity against the classical activating EGFR exon 19 deletions and exon 21 L858R substitutions and retained its potency in the presence of C797S, typically an acquired resistance mutation to osimertinib. 92
BLU 451
BLU 451 is a brain penetrant, EGFR WT sparing, covalent small molecular inhibitor of EGFR exon 20 insertion mutations, atypical EGFR mutations (G719C, G719S and L861Q) as well as the more common sensitizing mutations. Preclinical data have demonstrated antitumour activity in an intracranial xenograft model. BLU 451 is currently undergoing evaluation in a phase I/II global open label to determine the MTD, safety, tolerability as well as to evaluate the antitumour activity in patients with or without brain metastasis.93,94
ORIC-114
ORIC-114 is a brain penetrant, orally bioavailable, irreversible small molecular inhibitor was designed to target exon 20 insertions in EGFR and HER2. In preclinical biochemical assays, ORIC-114 sub-nanomolar IC50 potency and greater average fold selectivity for exon 20 mutations over EGFR WT compared with poziotinib, CLN-081 and BDTX-189. Promising responses at 3 mg/kg were observed with 9 out of 10 complete responses observed in patient-derived xenograft model. Furthermore, ORIC-114 also demonstrated high brain penetration with good brain to plasma unbound ratio in mice and efflux transporters that limit brain penetration seem to have minimal impact on ORIC-114. 95 In view of the promising preclinical efficacy, there is an ongoing phase I study to evaluate the safety and MTD of ORIC-114 in patients with advanced solid tumours with EGFR or HER2 exon 20 alternations or HER2 amplification and will allow enrolment of patients with asymptomatic treated or untreated CNS metastases.
Ongoing trials for EGFR exon 20 insertions are summarized in Table 2.
Ongoing trials for patients with EGFR exon 20 insertion mutations.
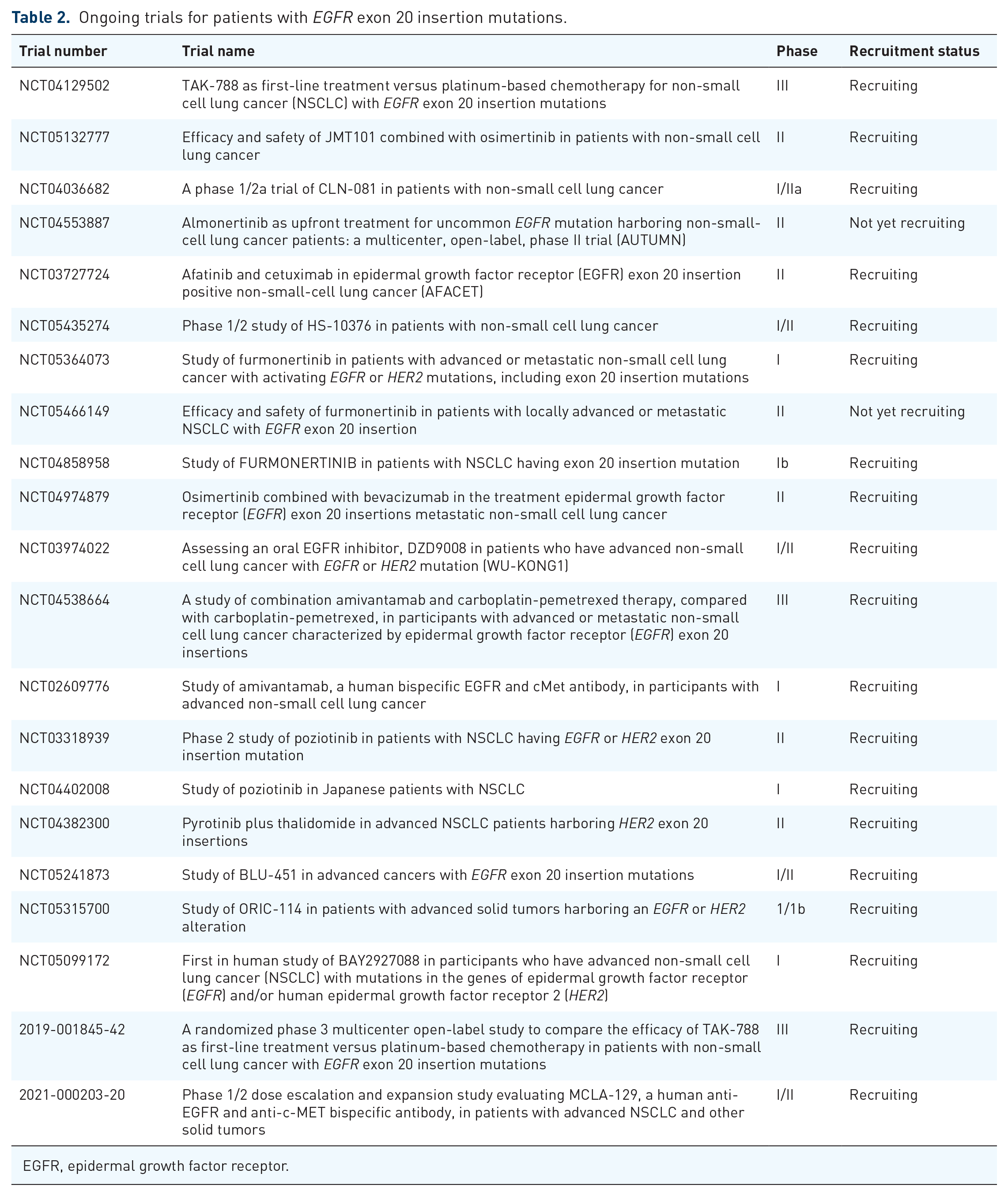
EGFR, epidermal growth factor receptor.
Detection of EGFR exon 20 insertion mutations
EGFR exon 20 insertion mutations can be detected through several methods such as polymerase chain reaction (PCR)-based or next-generation sequencing (NGS)-based technologies using tissue or liquid biopsy. 96 However, EGFR exon 20 insertion mutations are molecularly heterogeneous, and comprehensive identification of wide range of variants is challenging. 97 Using PCR-based methods may miss approximately 50% of cases with EGFR exon 20 insertion mutations and therefore the sole reliance on PCR methods to identify these subset of patients may result in underdiagnoses. 96 In contrast, NGS platforms are able to capture diverse array of EGFR exon 20 insertion mutations, including rare variants. As a result, the detection rate of EGFR exon 20 insertion mutations increased over the last decade due to the shift from PCR to NGS. The recent approval of targeted therapies for EGFR exon 20 insertion mutations, including amivantamab and mobercertinib, further supports the importance for NGS testing. 31 Currently, US FDA-approved companion diagnostics are Guardant360® CDx (Guardant, California, CA) and FoundationOne® Liquid CDx (Foundation Medicine, Cambridge, MA). 98 Other detection methods, such as Sanger sequencing, RNA sequencing, direct sequencing and mass spectroscopy, are alternative options that are less commonly used. 17
Mechanisms of acquired resistance
Although preliminary, several pre-clinical studies have shown that on-target secondary mutations, bypass pathway activation and epithelial–mesenchymal transition (EMT) confer resistance to therapies targeting EGFR exon 20 insertion mutations (Figure 4). 99 Identified on-target mechanisms to poziotinib 69 and tarloxotinib 100 include EGFR C797S and T790M mutations. Bypass pathway activations have been identified as acquired resistance to poziotinib, including reactivation of MAPK/phosphoinositide 3-kinase (MAPK/PI3K) pathway such as PIKCA E545K and MAP2K2 S94L, and amplifications in MET, EGFR and CDK6. 101 Furthermore, in vitro analysis shows that resistance to poziotinib is associated with EMT with increase in AXL expression and downregulation of E-cadherin. 69 In patients showing progression with osimertinib, EMT and histologic transformation to squamous cell carcinoma and small-cell carcinoma have been reported.102,103 Currently, the mechanisms of acquired resistance to osimertinib remain unknown.

Potential mechanism of resistance to therapeutic agents in EGFR exon 20 insertion. Various resistance mechanisms include (1) on target mechanism such as C797S and T790M after poziotinib and tarloxitinib treatment, (2) bypass pathway activation such as MAPK/PI3K activation and MET amplification and (3) EMT after treatment with poziotinib.
With emerging treatment options for EGFR exon 20 insertion mutations, including amivantamab and mobocertinib, and data on new targeted agents awaiting results, the potential resistance mechanisms of these agents remain to be elucidated. 14 More than 100 EGFR exon 20 insertion mutations subtypes have been identified with similar therapeutic sensitivity and depth of response to new targeted agents. 104 Whether these specific variants show unique resistance to the new targeted agents remains unknown. 14 Moving forward, identifying and characterizing the potential mechanisms of resistance will inform on future drug development and may pave way to overcome acquired resistance.
Upcoming therapies and future directions
The proportion of patients with EGFR exon 20 insertion mutations represents a substantial group. 13 Despite recent progress with multiple agents in clinical development and showing signs of activity for patients with EGFR exon 20 insertion mutations, further work is required to improve the care of patients with EGFR Exon 20 insertion mutations to determine the optimal sequencing of treatment, safety profile and treatment of patient with brain metastasis.
While both amivantamab and mobocertinib are FDA approved for the treatment of patients with EGFR exon 20 insertion mutations, these drugs are approved in the post-platinum setting. Platinum-based doublet remains the standard first-line regimen and trials are underway to move targeted therapy in EGFR exon 20 insertion mutations to the front-line setting. The randomized PAPILLON study, for example, is comparing amivantamab plus chemotherapy versus chemotherapy alone. 105 The optimal sequencing of currently available approved agents mobocertinib and amivantamab also deserves further investigation.
Given the high propensity for brain metastasis in lung cancer, 106 further research is needed to investigate the effects of drugs against untreated brain lesions. CHRYSALIS 45 and STUDY101 58 only included patients with treated asymptomatic brain metastasis. In the CHRYSALIS study, 38 out of 114 (33%) of patients had baseline brain metastasis. Intracranial only disease progression occurred in 11% of patients, and 12 out of 38 (32%) with baseline brain metastasis versus 5 out of 76 (6.6%) without baseline brain metastasis had intracranial progression. 107 In STUDY101, 25% of patients had intracranial disease progression. 108 WUKONG also allowed enrolment of patients with stable and asymptomatic brain metastasis. In all, 23 out of 56 (41%) had baseline brain metastasis and although intracranial lesions were not assessed as target lesions, encouraging antitumour activity was seen with a RR of 30%.51,52
To treat metastatic brain disease effectively, blood–brain barrier (BBB) permeability is essential. However, the EGFR-TKIs are also substrates for human BBB efflux transporters to varying degrees, with the P-glycoprotein and breast cancer resistance protein 109 playing a crucial role and this influences the distribution of the compound at equilibrium.110,111 Osimertinib, for instance, is a weak efflux transporter substrate (efflux ratio 3.2) and has the most BBB penetrance with a brain to free plasma ratio (Kp) value of 0.21 compared with other first-, second-generation TKIs as well as poziotinib which have a Kp value ⩽0.12. 112 BLU 451 and ORIC-114 are brain penetrant small molecular inhibitors with promising preclinical intracranial penetrance with ongoing clinical trials to evaluate its clinical efficacy in patient with brain metastasis.93–95 ORIC 114 has a low efflux ratio of 0.7, and this predicts for brain penetrance. ORIC 114 also exhibits high exposure in the brain at 1 and 4 h after administration in mice models, compared to poziotinib and BDTX-189, where brain exposure levels are below quantification limits. 95 In preclinical brain metastasis xenograft model carrying the T790M mutation, sunvozertinib-induced tumour regression was also observed at 25 mg/kg BID and 50 mg/kg BID. 51 These drugs have demonstrated promising preclinical activity and are candidates for development in patients with brain metastasis. An in vitro transporter assays as an early screen may also identify drugs with better CNS activity.
Conclusion
Patients with NSCLC with EGFR exon 20 insertion mutations have de novo resistance to EGFR TKIs due to steric hindrance affecting binding of TKIs used in common sensitizing EGFR mutations. The development of several novel small molecule compounds that selectively inhibit EGFR exon 20 insertion as well as the approval of amivantamab and mobocertinib for patients who have progressed after prior chemotherapy holds promise for effective therapeutic options for these group of patients. Several challenges lie ahead. The frequency of EGFR exon 20 insertion mutations may be underestimated due to insufficient testing methods and the diverse mutational landscape. There is also substantial unmet need for first-line treatment strategies. Finally, resistance mechanisms and strategies to overcome them as well as management of patients with CNS metastasis require further research. Nevertheless, new drugs in development beyond mobocertinib and amivantamab provides exciting prospect that novel treatments will provide a wider range of effective treatment options for patients with EGFR exon 20 insertion mutations.
Supplemental Material
sj-jpg-1-tam-10.1177_17588359221146131 – Supplemental material for Advances in the management of non-small-cell lung cancer harbouring EGFR exon 20 insertion mutations
Supplemental material, sj-jpg-1-tam-10.1177_17588359221146131 for Advances in the management of non-small-cell lung cancer harbouring EGFR exon 20 insertion mutations by Jia Li Low, Sun Min Lim, Jii Bum Lee, Byoung Chul Cho and Ross A Soo in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
