Abstract
Inhaled iloprost is a well-established medication to treat pulmonary arterial hypertension (PAH), a serious and potentially fatal disease of the pulmonary resistance vessels. The therapeutic administration of iloprost requires six to nine inhalations per day, due to the short biological half-life of this prostacyclin analogue. The I-NebTM AADTM, introduced in 2006, is the most commonly used nebulizer for delivering iloprost, requiring at least 6.5 min for an inhaled dose of 5 µg. In order to reduce inhalation time, a portable nebulizer based on modern-device technology was developed. The acute safety and tolerability of rapid iloprost inhalation
Pulmonary arterial hypertension
Pulmonary arterial hypertension (PAH) is an infrequent, yet serious and potentially fatal disease of the pulmonary vasculature with persistent vasoconstriction and remodelling of the resistance arterioles as pathophysiological hallmarks. PAH represents classified in an important subgroup within the common and diverse disease entity pulmonary hypertension (PH). The definition of PH is merely based on haemodynamic characteristics, with an increase in mean pulmonary artery pressure (mPAP) ≥ 25 mmHg at rest, as determined by right-heart catheterization. 1 The complex and multifactorial nature of this disease, however, results in a clinical classification comprising five groups with similar pathophysiology, haemodynamics, clinical picture and treatment options:1,2 group 1: PAH; group 2: PH due to left-heart disease; group 3: PH due to lung diseases and/or hypoxia; group 4: chronic thromboembolic PH (CTEPH) and other pulmonary artery obstructions; group 5: PH with unclear and/or multifactorial mechanisms. Group 1 PH itself comprises an array of disorders sharing similar pulmonary vascular pathophysiological changes and clinical characteristics. Haemodynamically, PAH represents a precapillary PH with a pulmonary artery wedge pressure ⩽ 15 mmHg and a pulmonary vascular resistance > 3 Wood units, in addition to the elevated mPAP.1,3 Histopatho-logically, PAH is characterized by specific structural changes of the pulmonary arterioles, comprising medial smooth muscle hyperplasia, thickening and fibrosis of intima and adventitia, formation of neointima and plexiform lesions, vascular pruning and perivascular inflammation.4,5 To be classified as PAH, absence of specific causes of precapillary PH such as lung diseases, CTEPH or other rare disorders is required. 1 PAH may be idiopathic, heritable, drug or toxin induced, or associated with other disorders such as connective tissue disease, human immunodeficiency virus infection, congenital heart disease, portal hypertension and schistosomiasis. PAH is a rare disease; incidence ranges from 2.0 to 7.6 annual cases per million and prevalence from 11 to 52 per million of adult inhabitants.6–8 Clinical symptoms of this chronic disease comprise dyspnoea on exertion, fatigue, chest pain, oedema and syncope; death is closely linked to progressive failure of the right ventricle resulting from rising afterload due to increased pulmonary vascular resistance. Deciphering the pathophysiological background of PAH has facilitated the development of specific PAH medication over the last 3 decades. Imbalances between vasoconstrictive 9 and vasodilatory endogenous mediators10,11 have been identified as important factors for persistent vasoconstriction and remodelling of the pulmonary arterioles. 12 In particular, the three principal signalling pathways of pulmonary vasoregulation, namely the prostacyclin, the nitric oxide and the endothelin pathway, provide the mechanistic basis for the currently approved PAH-specific medicines.1,13 The development of these mainly vasodilatory drugs has considerably improved therapy of PAH; amelioration of clinical symptoms, deceleration of disease progression and prolonged survival have been documented.14,15
Therapy of pulmonary arterial hypertension with inhaled iloprost
Intravenous prostacyclin (epoprostenol) was the first drug approved for PAH therapy in 1995 based on clinical and haemodynamic benefits, demonstrated
In numerous clinical trials with PAH patients, iloprost aerosol therapy has demonstrated safety and efficacy, as well as in monotherapy30–40 and in combination with other specific drugs.41–43 Following a successful pivotal phase III study, 44 inhaled iloprost was approved in many countries for aerosol therapy of severe PAH. Inhaled iloprost is currently recommended as class I monotherapy in patients with PAH in World Health Organization (WHO) functional class III and as class IIb monotherapy in WHO functional class IV. Furthermore, inhaled iloprost can be added to pre-existing oral bosentan in sequential combination therapy (WHO functional class II to IV patients, class IIb). 1 According to the prescribing information, Ventavis® (Bayer AG, Leverkusen, Germany) is administered by a suitable inhalation device six to nine times per day with a single inhaled iloprost dose of 2.5 μg or 5.0 µg. 45
In the first clinical studies, iloprost was diluted in physiological saline (maximal iloprost concentration of 10 µg/ml) and delivered by a provisional inhalation system comprising a continuous-output jet nebulizer, reservoir and filter.
30
The output and efficiency of this inhalation system were limited, resulting in a duration of inhalation of 15 min for the delivery of an effective dose of approximately 2.8 µg iloprost. In the course of the development of inhaled iloprost, three different jet nebulizers were compared in a crossover trial with 12 PH patients.
46
An iloprost dose of 5 µg inhaled within approximately 10 min caused nearly superimposable pharmacodynamic and pharmacokinetic effects. Subsequently, a different technique for the nebulization of iloprost was validated using an efficient ultrasonic device.
47
In the pivotal phase III trial, the jet nebulizer HaloLiteTM (Respironics Inc., PA, US) was employed to deliver precise doses of iloprost (2.5 and 5 µg).
44
This device was breath actuated and produced aerosol only during the inspiration phase of the breathing cycle, while continuously monitoring and adapting aerosol delivery to the patient’s breathing pattern.
48
Soon after approval of inhaled iloprost, however, the HaloLiteTM, as well as the second-generation adaptive aerosol-delivery (AADTM) device ProdoseTM (Respironics Inc., PA, US) were no longer available for administration of Ventavis®. After demonstration of comparable
Efficient therapy with inhaled iloprost requires six to nine inhalations per day during waking hours, owing to the short duration of drug action. The administration of a single 5.0 μg iloprost dose nominally takes 6.5 to 10 min, depending on the type of nebulizer. In clinical studies, however, prolonged inhalation times were observed in some patients, in particular when using the I-NebTM AADTM device.50,51 In consideration of the frequency and length of each inhalation, the use of inhaled iloprost is very time consuming and laborious for the patients, with risk of nonadherence. Therefore, there have been several attempts to reduce duration of iloprost inhalation. Our group considered cutting down the inhalation time from 12 to 2 min for the delivery of a dose of 2.8 µg iloprost by the use of an ultrasonic nebulizer, but abandoned this plan after preliminary catheter investigations revealed systemic side effects such as decreases in systemic blood pressure and vascular resistance, increase in heart rate, flush and jaw pain. 47 Since then, shorter inhalation times for iloprost have been regarded unrealizable; iloprost bolus inhalation has no longer been proposed and pursued. In contrast, rapid inhalation of treprostinil, a second stable analogue of prostacyclin, was shown to cause selective beneficial haemodynamic effects in the pulmonary circulation without significant systemic side effects.52–54 Therefore, treprostinil bolus inhalation by a metered dose or soft-mist inhaler delivery has always been regarded as a viable therapeutic strategy, although not realized and available for patients until now.
Based on significant advances in aerosol-device technology,55,56 the concept of rapid iloprost inhalation (bolus inhalation) for improving the established iloprost aerosol therapy was again investigated. A new vibrating mesh nebulizer with high aerosol output in combination with control of the inhalation manoeuvre was developed, offering patient-adaptive breath-triggered bolus inhalation. Control of the inspiratory flow rate and inspiratory volume, together with the timing of aerosol generation during the inspiration cycle allow for rapid inhalation, precise dosing and drug targeting to specific sites within the respiratory tract. 56
Pilot study with rapid iloprost inhalation (bolus inhalation)
In preparation for a pilot study investigating iloprost bolus inhalation in patients with PH, the inhalation system AKITA
2
Apixneb® (Activaero GmbH, Gemünden, Germany) was characterized
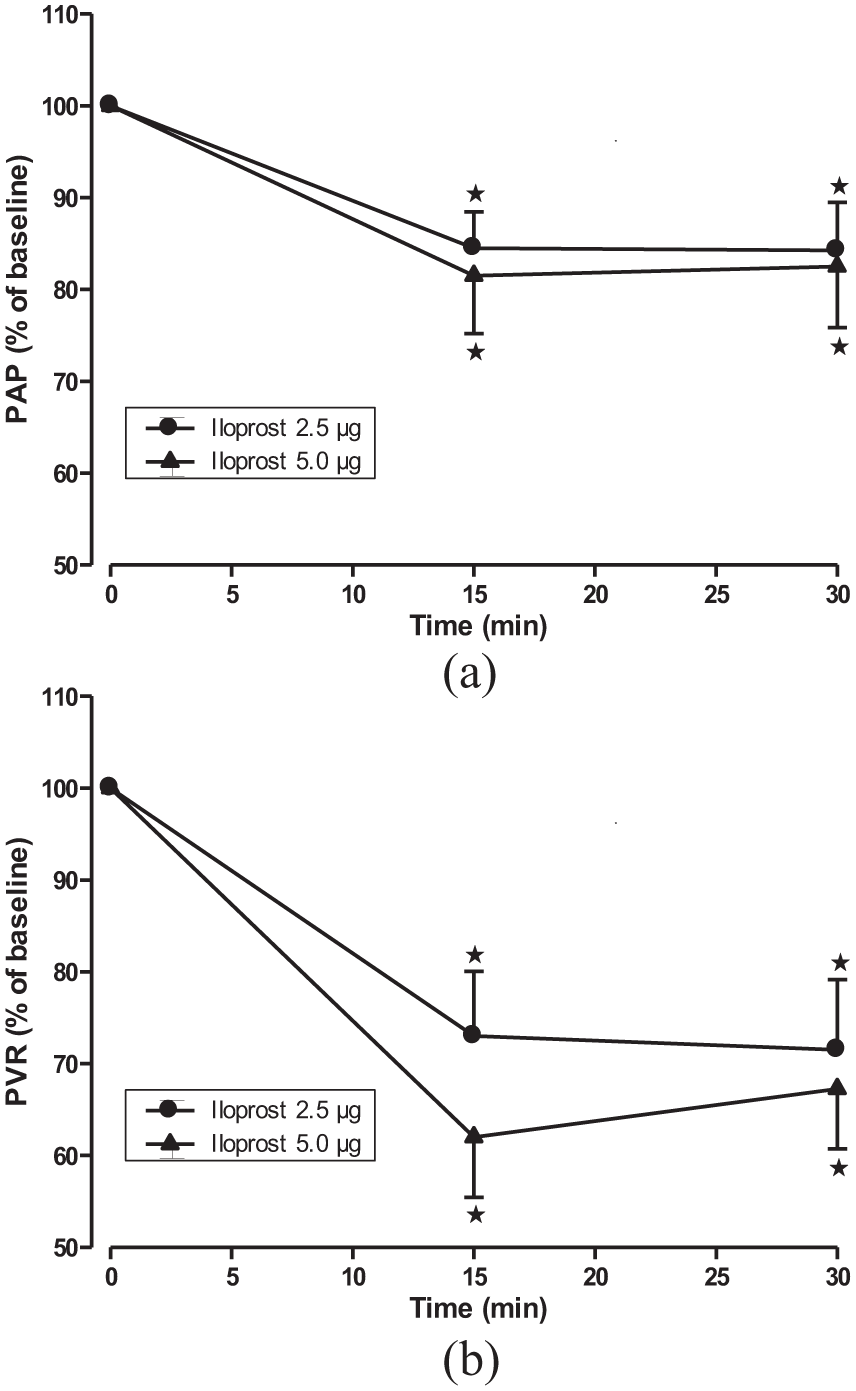
Pulmonary vascular effects of iloprost bolus inhalation.

Systemic haemodynamic side effects of iloprost bolus inhalation.
The surprising feasibility of iloprost bolus inhalation is most likely due to advances in aerosol-device technology. Breath actuation, control of inspiratory volume and flow, as well as defined aerosol–air–bolus inhalation facilitate short treatment times, reproducible dosing and targeted pulmonary drug deposition.56,58 In particular, this technology helps to selectively reach the peripheral pulmonary regions, with high alveolar deposition of up to 60% of the inhaled drug dose and significant reduction of extrathoracic and central drug deposition.57,59,60 This specific deposition pattern not only prevents local adverse effects in the upper respiratory tract, but may also reduce systemic side effects through decreased drug absorption across airway mucosa. In fact, after conventional jet nebulization of iloprost, approximately 80% of inhaled iloprost entered the vascular compartment, suggesting rapid iloprost mucosal absorption in the airways is responsible for systemic side effects. 46
In this regard, a study comparing inhaled treprostinil delivered by an advanced soft-mist inhaler (AERxTM, Aradigm Corporation, CA, US) with a conventional ultrasonic nebulizer (OptinebTM, Nebu-Tec GmbH, Elsenfeld, Germany) is of particular interest. Cipolla and colleagues demonstrated that deep-lung pulmonary delivery of aerosolized treprostinil resulted in delayed systemic absorption of treprostinil (lower maximum serum concentration, delayed time of maximum serum concentration with comparable area-under-the-serum-level–time curve). 61 Therefore, the therapeutic pulmonary effects, absence of relevant side effects and excellent tolerability of rapid iloprost inhalation might well be attributed to the specific delivery of the drug to the alveolar region by use of an advanced aerosol technique.
Safety and tolerability of iloprost delivered via the BREELIBTM nebulizer
Following the successful proof-of-concept pilot study with iloprost bolus inhalation, the portable piezoelectrical nebulizer BREELIBTM (Vectura Group plc, Chippenham, UK), specific for iloprost inhalation, was developed. This device features a powerful vibrating mesh aerosol generator, as well as electronic and mechanical equipment to control the inhalation manoeuvre. The generation of the aerosol pulse is breath triggered, with an aerosol bolus of 2 s followed by aerosol-free air for 1 s. The inhaling patient is guided by light-emitting diode feedback until the target inhalation volume is reached. Additionally, the inspiratory speed is restricted by a mechanical flow-limitation valve. These features diminish the influence of the patient’s breathing pattern on pulmonary drug deposition, thereby ensuring delivery of an exact and reproducible iloprost dose. The BREELIBTM enables the inhalation of a dose of 5 µg of iloprost within approximately 2 min, a duration significantly shorter compared with the nominal 6.5–10 min designated in the Ventavis® prescribing information for other nebulizers.
In order to demonstrate safety of rapid iloprost inhalation, a multicentre, open-label, randomized crossover phase I/II study in 27 PAH patients was performed. 62 The four-part trial compared acute tolerability of iloprost inhalation using the BREELIBTM in comparison with the I-NebTM AADTM nebulizer. The primary safety endpoint was the proportion of patients with a meaningful maximum increase in heart rate (⩾25%) or a meaningful maximum decrease in systolic blood pressure (⩾20%) within 30 min after completion of inhalation. Additional safety variables were systolic, diastolic and mean blood pressure, heart rate, oxygen saturation, AEs and laboratory variables. Assessment of pharmacokinetic parameters was included in the first two parts of the study in order to verify adequate iloprost exposure when inhaling with the two devices.
Male or female adult patients with confirmed diagnoses of PAH and pre-existing Ventavis® therapy (5 µg
Safety and tolerability
The first 11 patients in part 1 tolerated the BREELIBTM iloprost inhalations of 1.25 µg and 2.5 µg with only mild and transient adverse effects. According to the protocol, the inhalation of 1.25 µg was then omitted and subsequent patients received directly 2.5 µg iloprost. The primary safety outcome, defined as proportion of patients with a maximum increase in heart rate ⩾ 25% or a maximum decrease in systolic blood pressure (SBP) ⩾ 20% within 30 min after inhalation compared with pretreatment values, was analysed from the data obtained in part 2 of the study. All five observed haemodynamic events were changes in SBP with three more BREELIBTM patients (four patients, 15%) compared with I-NebTM AADTM (one patient, 4%). Based on data from the pivotal phase III trial,
44
this result was within the expected range; a difference of up to three events was calculated to occur randomly when comparing both treatments in 24 patients. Secondary haemodynamic safety variables, for example, mean maximum increase in heart rate or mean maximum decrease in SBP showed no statistical difference. Systemic hypotension, defined as SBP ⩽ 90 mmHg irrespective of symptoms, was recorded in four patients after inhalation with the BREELIBTM
In the 2-week crossover part 3 of the study, more AEs were documented with the use of BREELIBTM (52%) compared with the I-NebTM AADTM (27%). Reported AEs comprised local irritation (e.g. cough, oropharyngeal pain) and typical side effects of prostanoid therapy (e.g. flush, headache). However, there was no difference in serious AEs (SAEs) between the two modes of iloprost delivery, with two events in each group.
The AEs reported in parts 1–3 of the study were all mild and transient, not requiring medical intervention or leading to discontinuation of the treatment. No SAEs were seen in parts 1 and 2; the two SAEs with both nebulizers in part 3 were evaluated as not related to the study drug (Table 1).
Adverse events reported in parts 1–3 of the BREELIBTM study (adapted from Gessler et al. 62 ).

Hypotension was predefined as SBP ⩽ 90 mmHg, irrespective of hypotensive symptoms.
Patients can have experienced more than one AE.
AE, adverse event; HR, heart rate; I°, ; SBP, systolic blood pressure.
Nearly all patients completing part 3 of the study (25 of 26 patients) decided to switch to the new nebulizer BREELIBTM for continuation of their pre-existing inhaled iloprost therapy within the optional long-term extension (part 4). This is of importance, as all patients entering into the study were accustomed to use the I-NebTM AADTM nebulizer for iloprost delivery. By the time of preparing the published manuscript of the study, median treatment duration in the long-term extension phase was 330 days, with a total of 8287 days of drug exposure. Seven patients (28%) withdrew from the study: two patients died due to right-heart failure from PAH progression; three patients discontinued due to lung transplantation; one due to worsening of PAH; and one due to lack of efficacy. Nearly all patients experienced adverse effects; the drug-related events in six patients were nonserious and mostly in accordance with the established safety and side-effect profile of inhaled iloprost and the nature of PAH. None of the reported SAEs in 13 patients were attributable to the inhalation of the study drug.
Pharmacokinetics
The pharmacokinetic variables (PKs) collected in parts 1 and 2 of the study demonstrated consistent and efficient delivery of iloprost
Duration of inhalation
As expected, durations of inhalation were drastically shorter with BREELIBTM compared with the I-NebTM AADTM. Median inhalation time with the I-NebTM AADTM was 10.9 min (range 4.3– 22.1 min) and thus significantly longer than the designated 6.5 min given in the Ventavis® prescribing information. This result is in good accordance with observations in the daily routine and in other clinical studies with prolonged inhalation times for the I-NebTM AADTM.45,50,51 In contrast, median inhalation time with the BREELIBTM was reduced by 76% to only 2.6 min (range 1.6– 3.4 min). This reduction was consistent in all patients, with no overlap in the ranges for median inhalation times. As a result, net daily time required for six to nine iloprost inhalations is significantly reduced from 65 to 98 min with I-NebTM AADTM to only 16–23 min with the BREELIBTM.
Conclusion and perspectives
The vibrating mesh nebulizer BREELIBTM was developed to markedly reduce inhalation times of iloprost aerosol PAH therapy. The safety and tolerability of this approach was demonstrated in a four-part clinical study in comparison with the standard nebulizer I-NebTM AADTM. As a result, the duration of inhalation was cut down from 10.9 to 2.6 min using the BREELIBTM, with good tolerability and absence of SAEs.
Following the successful phase I/II clinical trial, the nebulizer BREELIBTM was approved in several European countries to administer Ventavis® in PAH. Based on the experiences and data from the trial, starting with a dose of 2.5 µg is recommended when initiating or switching from other nebulizers to iloprost therapy with BREELIBTM. If there is good tolerance, the dose should be increased to 5.0 µg. In view of the ongoing long-term extension phase of the study and the clinical availability of the BREELIBTM, further data on safety, tolerability and efficacy of iloprost aerosol therapy with the new device is forthcoming.
The significant reduction of inhalation times with a modern and compact nebulizer raises expectations of improving patient acceptance and adherence to iloprost aerosol therapy. One of the structural disadvantages, prolonged inhalation times, of inhaled iloprost compared with treprostinil, the second inhalative prostanoid for PAH therapy, is thus abrogated. Since 2015, a further drug addressing the prostacyclin pathway has been approved for therapy of PAH. The oral selective IP prostacyclin-receptor agonist selexipag was investigated in a randomized, double-blind, placebo-controlled phase III trial with 1156 patients suffering from PAH. 63 The primary endpoint of this event-driven study, a composite of death from any cause or a complication related to PAH, significantly turned out in favour of selexipag compared with placebo. Premature discontinuation due to AEs occurred twice as often for the active compound (14.3%) than for placebo (7.1%); no significant difference regarding mortality was observed between the two groups. In the current guidelines, selexipag has a class I recommendation for monotherapy of PAH patients in WHO functional class II and III. Additionally, selexipag can be added to pre-existing oral medication with endothelin receptor antagonists and/or phosphodiesterase-5 inhibitors in sequential combination therapy. 1 Most PH experts and drug committees recommend starting with oral phosphodiesterase-5 inhibitors and/or oral endothelin receptor antagonists as initial treatment for adult PAH patients in functional class II and III. Prostacyclin analogues and prostacyclin-receptor agonists are mainly used as add-on therapy when such oral medication has inadequate clinical results or fails to prevent disease progression or clinical deterioration. Against this background, the established prostanoids, including the two aerosol therapies iloprost and treprostinil, compete with oral selexipag when a drug addressing the prostacyclin pathway is desired or necessary for PAH therapy. While data on efficacy and safety of long-term selexipag treatment as well as comparative studies with the prostacyclin analogues are lacking, comfort and convenience of drug administration may play a decisive role for the selection of a prostanoid drug. In this regard, there is room for improvement of iloprost aerosol therapy, even with the introduction of BREELIBTM. Patients still have to fill the nebulizer with Ventavis® solution from a separate ampulla and clean the device after each inhalation. In view of the possibility of administering iloprost effectively and safely within a few breaths, the concept of metered dose or soft-mist delivery by portable, multidose devices is feasible. Such approaches show promise in making inhalative delivery of iloprost in PAH as easy and convenient as inhalative delivery of bronchodilators or corticosteroids in asthma or chronic obstructive pulmonary disease. Modern aerosol therapy may contribute to improving convenience and compliance for patients, hopefully resulting in broader acceptance and improved efficacy of iloprost aerosol therapy in PAH.
Supplemental Material
Author_response_v1 – Supplemental material for lloprost delivered via the BREELIBTM nebulizer: a review of the clinical evidence for efficacy and safety
Supplemental material, Author_response_v1 for lloprost delivered via the BREELIBTM nebulizer: a review of the clinical evidence for efficacy and safety by Tobias Gessler in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_1_v1 – Supplemental material for lloprost delivered via the BREELIBTM nebulizer: a review of the clinical evidence for efficacy and safety
Supplemental material, Reviewer_1_v1 for lloprost delivered via the BREELIBTM nebulizer: a review of the clinical evidence for efficacy and safety by Tobias Gessler in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_2_v1 – Supplemental material for lloprost delivered via the BREELIBTM nebulizer: a review of the clinical evidence for efficacy and safety
Supplemental material, Reviewer_2_v1 for lloprost delivered via the BREELIBTM nebulizer: a review of the clinical evidence for efficacy and safety by Tobias Gessler in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_2_v2 – Supplemental material for lloprost delivered via the BREELIBTM nebulizer: a review of the clinical evidence for efficacy and safety
Supplemental material, Reviewer_2_v2 for lloprost delivered via the BREELIBTM nebulizer: a review of the clinical evidence for efficacy and safety by Tobias Gessler in Therapeutic Advances in Respiratory Disease
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Tobias Gessler is co-inventor of patent applications/patents in the field of pulmonary hypertension partly licensed to United Therapeutics and Vectura. He received lecture honoraria and consulting fees from Bayer and was investigator in trials involving Bayer and United Therapeutics.
Supplementary Material
There are supplementary data available for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
