Abstract
Background:
Autologous blood is a novel, high-efficacy sclerosant for treatment of malignant pleural effusion (MPE), similar to tetracycline. There has been no comparative data between autologous blood and a worldwide sclerosant like talc. We aimed to compare the effectiveness of autologous blood versus talc pleurodesis.
Methods:
A prospective study was conducted at Songklanagarind Hospital, Songkhla, Thailand. A total of 123 symptomatic MPE cases were randomized to receive autologous blood pleurodesis (ABP) versus pleurodesis with talc slurry. In the ABP group, 100 ml of autologous venous blood was instilled through a chest drain, followed by 50 ml of sterile normal saline (NSS). In the talc group, 20 ml of 1% lidocaine diluted in 30 ml NSS was instilled, followed by 4 g of sterile talc (Steritalc®, a non-small particle size talc) suspended in 100 ml of NSS. A 30-day pleurodesis efficacy (according to Paladine’s criteria), along with the adverse events, was evaluated.
Results:
Fifty-six cases in the ABP, and 54 cases in the talc group completed the study. There was no difference between the two groups in the demographic data. The overall pleurodesis success rate at 30 days was 82.0% in the ABP group, comparable to the talc pleurodesis group (87.0%, p = 0.12). The percentage of fever (9.0% versus 28.0%, p = 0.04), amount of acetaminophen required by each participant (2.2 ± 0.7 versus 4.6 ± 0.9 tablets, p = 0.03), pain score and percentage of cases who needed opioids (9.0% versus 26.0%, p = 0.02) and hospital stay (10.2 ± 2.7 versus 12.8 ± 3.4, p = 0.04) were significantly lower in the ABP group; no infectious or serious events occurred.
Conclusions:
ABP had an equivalent efficacy compared to talc pleurodesis for MPE treatment. ABP offered less fever and pain and could shorten hospital stays, and neither produced means ABP did not produce clotted drainage, pulmonary or systemic adverse events.
Introduction
Malignant pleural effusion (MPE) is a frequent complication of malignancies, and represents the advanced stage of underlying primary cancers. This condition is defined as an abnormal accumulation of pleural fluid containing neoplastic cells, confirmed either by cytological analysis or pleural biopsy.1,2 In general, lung and breast cancers are the most common causes of MPE. 2 Once MPE occurs it confers a worse prognosis, with a median survival of only 3–12 months.3,4 Three-quarters of untreated MPE cases will develop symptoms, such as dyspnoea, cough, chest discomfort or even respiratory and circulatory system failures; therefore, early diagnosis with prompt therapeutic interventions, when possible, are essential. 3 Systemic chemotherapy has been proven to be effective as a first-line treatment in MPE patients with a good performance status; however, most underlying tumours will become chemo-resistant, and a number of cases are not fit for chemotherapy. Thus, other useful palliative therapeutic options, such as chemical pleurodesis, thoracoscopy with pleurodesis or placement of an indwelling pleural catheter should be considered for recurrent symptomatic MPE.1,3,5
Chemical pleurodesis is a universally accepted procedure that obliterates the pleural space by instillation of sclerosant into the pleural cavity, which induces pleural inflammation – fibrosis resulting in extensive adhesion between the visceral and parietal pleura. 6 A variety of sclerosing agents have been used to treat MPE (for example, talc, tetracycline and bleomycin), with a clinical success rate ranging from 54% to 100%; these results relate to the prevention of recurrence of MPE, and improvement in patients’ quality of life.1,5,7–12 Among these efficacious sclerosants, talc showed higher success rates (70–100%), and has been generally advocated for treating MPE.5,7,8,11,13,14 However, in real clinical practice, pleurodesis with talc is significantly associated with both minor (fever, chest pain) and major adverse events, such as hypotension, dysrhythmia, pneumonitis, acute respiratory distress syndrome (ARDS, 0.7–9%), acute respiratory failure (ARF) and death (0–2.3%), raising issues regarding the toxicity of talc.1,5,14–24 As such, a new, safer sclerosing agent has been sought.11,14
Autologous blood is a human endogenous sclerosing substance that can induce pleural inflammation, fibrosis leading to pleural adhesions.25,26 Its application in creating pleurodesis was first described by Robinson in 1987, who used 50 ml of autologous venous blood to treat chronic, spontaneous pneumothorax, and showed an 85% clinical success rate. 27 Subsequently, data established that autologous blood pleurodesis (ABP, at doses of 50–120 ml) has high efficacy in terminating pneumothorax and persistent air leaks (PALs), with an overall success rate of 92%, comparable to talc and tetracycline.28,29 In the context of MPE, a prospective, randomized controlled trial (RCT) was conducted by the author and colleagues in 2011. 30 We decided to perform ABP by using 100 ml of peripheral venous blood without intrapleural lidocaine, and compared this to a 1 g of tetracycline pleurodesis plus lidocaine injection in 48 symptomatic MPE patients. The results demonstrated ABP was equally as effective as tetracycline for the treatment of MPE, with a 30-day pleurodesis success rate of 83.4% (versus 87.5% in the tetracycline group, p = 0.36). In addition, pleurodesis coupled with autologous blood produced significantly less fever, a lower percentage of pain requiring analgesia (4.2% versus 75%, p < 0.001) in addition to a shorter hospital stay (8.2 ± 4.6 versus 9.8 ± 5.5 days, p = 0.04) than pleurodesis with tetracycline, without any pulmonary and systemic toxicity. However, there have been limited data about the usefulness of ABP in MPE as well as a lack of comparative data between ABP and worldwide sclerosing agents. Therefore, the present study was conducted to compare the efficacy, and safety profile between ABP versus talc pleurodesis in patients with symptomatic MPE.
Materials and methods
A prospective, randomized, comparative study was conducted from June 2015 to June 2016 at Songklanagarind Hospital, Songkhla, Thailand. The selection criteria were patients at least 18 years of age, presenting with recurrent symptomatic MPE (cytologically or histologically confirmed), and a predicted life expectancy of greater than 1 month (ECOG performance status 0–2 and without severe comorbidities), who agreed to receive chemical pleurodesis and signed written informed consent. The exclusion criteria were as follows: (1) active pleural or systemic infection, (2) serum haematocrit <25% or hemodynamic instability, (3) haemothorax, chylothorax or a multiple aetiology of pleural effusion, (4) history of previous chemical pleurodesis, allergy to talc or lidocaine, (5) chest X-ray after chest tube drainage showing a trapped lung on the affected side and (6) pregnancy. The clinical trial was conducted in accordance with the World Medical Association Declaration of Helsinki Good Clinical Practice guidelines and ethical approval was obtained from the Ethics Committee of the institution (IRB#58-078-14-1). This study was registered at www.clinicaltrials.in.th (Thai Clinical Trials Registry, study ID: TCTR20150717001).
After obtaining the baseline characteristics, a small-bore chest tube [percutaneous drainage (PCD) 8–10 Fr], or a wide-bore chest drain [intercostal drainage, (ICD) 20–32 Fr] was inserted into the pleural cavity through the fifth or sixth intercostal space under local anaesthesia (LA). Although the success rates of pleurodesis were found to be similar regardless of the type of chest tubes, the PCD (which could shorten hospital stay) was selected as a priority, unless it had an impending respiratory failure, or the interventionist were unavailable. 31 Then, the pleural fluid was examined and drained by gravity until the output was less than 150 ml/day, 32 as well as a chest X-ray showing full inflation. Then a block randomization using a web-based central randomization system with variable block size was generated (the investigators were blinded to the block size) and participants were allocated into two groups, so as to perform either an ABP or talc pleurodesis.
Pleurodesis techniques
For patients in the ABP group, 100 ml (~2 ml/kg of predicted patient’s weight) of autologous peripheral venous blood without anticoagulant was instilled through the pleural space, with aseptic techniques (without intrapleural lidocaine injection), followed by 50 ml of sterile normal saline (NSS) for prevention of chest tube clogging.26,30 By contrast, in the talc slurry pleurodesis group, intrapleural lidocaine solution (20 ml of 1% lidocaine, made up to 50 ml with NSS) was injected through the chest tube; after waiting 5 min, 4 g of sterile talc (Steritalc®, a non-small particle size talc with an average particle size of 25 um manufactured by Novatech, La Ciotat, France) suspended in 100 ml of NSS was slowly instilled over 5–10 min. 8 Next, the drainage tube was clamped for 2 h without changing the patient’s position. 32 Hereafter, the chest tube was reconnected to the water sealed system, and continuous thoracic suction with −20 cm water pressure was generated in all cases. When post-sclerotherapy drainage was below 150 ml within 8 h of thoracic suction, the chest X-ray was repeated within 24 h and after fully re-expansion was documented the chest tube was immediately removed.
The effectiveness of chemical pleurodesis was assessed 30 days later by blinded chest physicians, using Paladine’s criteria as follows: 33
Complete response (CR): pleural fluids do not accumulate.
Partial response (PR): recurrence of small amounts of effusion, which does not need to be drained.
No response: recurrence of effusion, which requires a repeat drainage.
CR and PR would be considered as the successfulness of pleurodesis (primary endpoint). The length of hospital stay (LOS), post-pleurodesis hospital stay (duration between initiation of a sclerosant until hospital discharge), pleurodesis-related complications and cardiopulmonary adverse events, which occurred during hospitalization, and after the next 15 and 30 days were recorded by the unblinded primary care physicians under the supervision of a pulmonologist, and defined as secondary endpoints.34–36 Following sclerosing injection, a body temperature (BT) of at least 38°C was defined as pleurodesis-related fever, and a septic workup was initiated unless fever was resolved within 48 h. Acetaminophen (500 mg), one tablet, was given if BT was greater than 38.5°C, at 6-hour intervals. Pleurodesis-related pain was scored by patients using a numeric rating scale from 0 to 10 (0 = no pain, 1–3 = mild, 4–6 = moderate, 7–9 = severe, 10 = worst) at intervals of 5 min before, and then after 5 min, 6 h and 24 h of sclerosant. Additional intravenous opioids were administered by the primary physician according to pain intensity (moderately to worst pain). In cases of ABP non-response, repeated pleurodesis with talc was decided upon, while another sclerosing agent (except autologous blood) would be offered in talc non-responder cases, respectively.
Statistical analysis
The sample size was calculated using RCT for binary data formula. Estimated pleurodesis efficacy of talc and ABP were 98% 37 and 83%, 30 respectively. A power of 80% was set to determine the efficacy, with the alpha level at 0.05. The calculated sample size was 59 cases in each group. However, we enrolled more patients to account for a potential 5% drop-out, and therefore the total adjusted sample size was 124 cases. Proportion (%) was used to describe qualitative variables. Mean and standard deviation (SD) were used to describe quantitative variables. Chi-square test along with Student’s t test were used to analyse the qualitative and quantitative characteristics, respectively. The outcomes of interests were analysed by SPSS version 16 software, and a two-tailed test with a p value < 0.05 being considered statistically significant.
Results
A total of 123 consecutive patients with newly diagnosed MPE were enrolled. The majority aetiology of MPE was lung and gynaecologic cancers, which accounted for 82.1% of cases, followed by breast and gastrointestinal malignancies (13.0%); 4.9% had cancer of an unknown primary. After randomization, by using a block design, all participants were randomized into two groups: 62 patients were allocated to receive ABP, while 61 patients received talc pleurodesis, respectively. In total, six patients were lost to follow up and seven patients died from their primary cancers within 30 days, following pleurodesis. Hence, 110 patients were analysed at the end of the study (56 in the ABP group, and 54 in the talc group; Figure 1). The mean age of patients was 62 years in both groups. Dyspnoea, chest discomfort and cough were predominant presenting symptoms. Chest roentgenography demonstrated moderate-to-large pleural effusion. Pleural fluid on the affected side was characterized as an exudative profile, and cytological examination showed positive for malignant cells in a majority of cases. There was no statistically significant difference between the two groups regarding gender, primary cancer type, size of chest drain and the number of patients receiving systemic chemotherapy within 1 month before or after pleurodesis (Table 1).

Consort patient flow diagram.
Patient demographic data and clinical characteristics.

ICD, intercostal drainage; PCD, percutaneous drainage; SD, standard deviation. *Cytological negative but pleural biopsy showed malignant histology.
The CR and PR of pleurodesis, by using autologous blood in this study, was achieved in 22 cases (39.2%) and 24 cases (42.8%). While, pleurodesis by talc slurry was able to achieve CR in 22 cases (40.7%) and PR in 25 cases (46.3%), respectively. The overall pleurodesis success rate at 30 days (defined as a percentage of complete and partial responders) was 82.0% in the ABP group, compared to 87.0% in the talc pleurodesis group; however, there was insignificant difference between the two groups with respect to efficacy (p = 0.12; Table 2). In PR cases, there was no statistically significant difference between the two groups in the time to the first pleural drainage (after pleurodesis) among symptomatic cases (17.8 ± 4.4 weeks in the ABP versus 16.4 ± 4.9 weeks in the talc group, p = 0.09). In consideration of the type of chest tube, the total response rate was similar between the small-bore and large-bore catheter group, and the overall pleurodesis success rates did not show a statistically significant difference (ABP as successful in 85.1% in the PCD group versus 79.3% in the ICD group, p = 0.21; together talc pleurodesis success rates were 87.5% in the PCD group versus 86.7% in the ICD group, p = 0.18, respectively).
Pleurodesis efficacy.
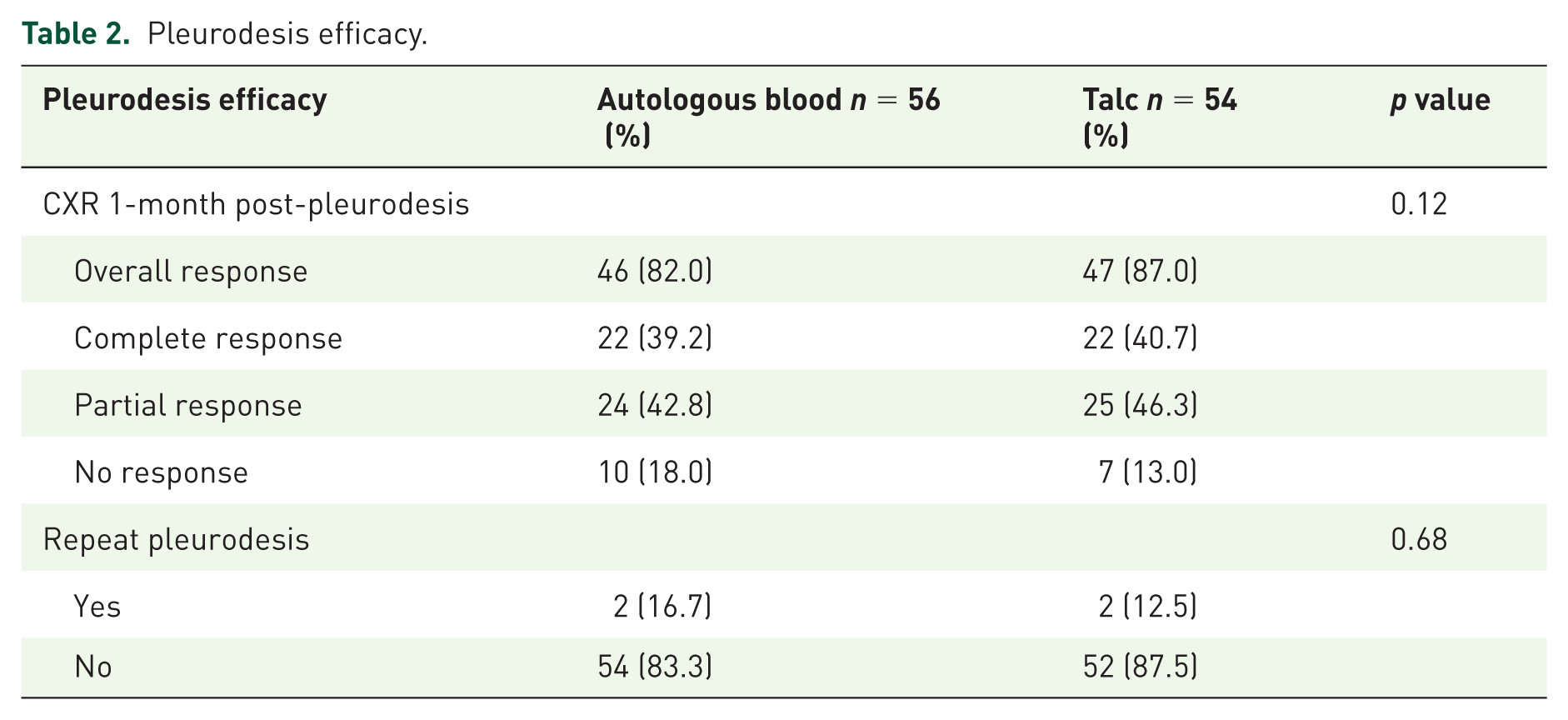
Chest pain and fever were the most frequent complaints as well as immediate complications. However, the average pain intensity, represented by the pain score within 24 h, was significantly lower in the ABP group, and there was a smaller percentage of patients who needed adjunctive intravenous analgesic in the group with ABP (9.0% versus 26.0%, p = 0.02; Figure 2; Table 3). In addition, the percentage of fever and the average amount of acetaminophen required by each participant (tablet), were also significantly lower in the ABP group (9.0% versus 28.0%, p = 0.04 and 2.2 ± 0.7 versus 4.6 ± 0.9, p = 0.03, respectively; Table 3), and all fevers were resolved within 48 h. No clotted drainage occurred in the group with ABP, but this developed in one patient of the talc group. Aside from this, the mean LOS as well as mean post-pleurodesis hospital stay in the ABP group were significantly shorter than those of the pleurodesis with talc (10.2 ± 2.7 versus 12.8 ± 3.4, p = 0.04 and 2.8 ± 0.9 versus 3.6 ± 1.8, p = 0.04, respectively; Table 3). There were no infectious complications, cardiopulmonary events or pleurodesis-related deaths in either study group within a 30-day follow-up period.
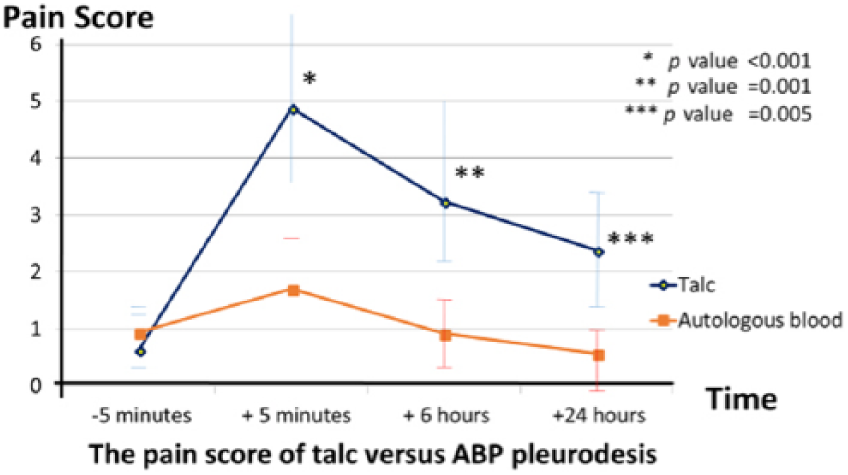
Pain score during the 24 h after the pleurodesis.
Pleurodesis-related adverse reactions.

SD, standard deviation
In the cases of ABP non-responders, repeated pleurodesis with talc was performed in 2 of 10 patients; the results demonstrated success in one. On the other hand, tetracycline pleurodesis proceeded in two of seven talc non-responder cases and showed success in one of these (Table 2).
Discussion
Pleurodesis with sclerosing agents has been established as a cost-effective palliative procedure for controlling MPE.12,38,39 The current study showed pleurodesis with autologous peripheral venous blood was not different to talc in terms of efficacy, with both treatments exhibiting equivalent benefits in their effectiveness. In addition, pleurodesis-related adverse events occurred less frequently with ABP than with talc.
As far as we know, the ideal sclerosant for treating MPE does not exist. Pleurodesis using talc, whether by slurry or poudrage, has significantly higher levels of efficacy (70–100%),7,8,14 and is more generally accepted than other sclerosants.5,11 Unfortunately, its use has been associated with many serious complications including hypotension, dysrhythmia, empyema, pneumonitis, ARDS (0.7–9%), ARF and mortality (0–2.3%).1,5,15–22 These unique respiratory and systemic toxicities are correlate with a dose much higher than 5 g,36,38 or when a small particle size talc (particle sizes <10 um),22,39–41 or talc poudrage under general anesthesia (GA) were used. 13 Thus, there is ongoing concern regarding the safety of talc administration, and a new safer sclerosing agent, which maintains a high efficacy, is now being sought.23,24
Autologous blood was discovered as a sclerosant a few decades ago. It could produce both microscopic and macroscopic pleural adhesions of a dose-dependent effect.25–27 It was first used as a chemical pleurodesis for spontaneous pneumothorax in 1987, and subsequently has been established as a well-tolerated and efficient sclerosant for terminating pneumothorax and PAL.27–29 In terms of MPE, we and our colleagues conducted a prospective RCT in 2011 to compare the efficacy and safety profile between ABP (100 ml) without intrapleural lidocaine versus 1 g of tetracycline pleurodesis plus lidocaine injection in 48 symptomatic patients with MPE. 30 The results showed ABP had a high pleurodesis success rate that was similar to tetracycline (83.4% versus 87.5%, p = 0.36), but significantly produced less fever, fewer cases requiring adjunctive opioids and a shorter hospital stay than pleurodesis with tetracycline, without any systemic adverse events. This present RCT trial was designed to compare the efficacy and safety profile between 100 ml of ABP without intrapleural lidocaine versus talc (4 g of talc slurry pleurodesis with intrapleural lidocaine injection), via a bedside chest tube, under LA in 110 symptomatic MPE patients. The dramatic results demonstrated that ABP provided high efficacy for the treatment of MPE, which was consistent with a previous study. 30 Eighty-two percent of cases in the ABP group achieved a successful pleurodesis at 30 days, which is comparable to pleurodesis with talc (82.0% versus 87.0%, p = 0.12; Table 2). Moreover, the results clearly revealed ABP did not create any infections or serious complications. Furthermore, the prevalence of pleurodesis-related fever (9.0% versus 28.0%, p = 0.04; Table 3) and intensity of sclerosant-related pain were also significantly less in the ABP group (Figure 2). We found that ABP could significantly reduce the amount of acetaminophen required by each participant (2.2 ± 0.7 versus 4.6 ± 0.9, p = 0.03) and decrease the percentage of pain requiring analgesia almost threefold compared to pleurodesis with talc (9.0% versus 26.0%, p = 0.02; Table 3). Aside from this, the mean LOS and post-pleurodesis hospital stay were significantly shorter in a group with ABP (10.2 ± 2.7 versus 12.8 ± 3.4 and 2.8 ± 0.9 versus 3.6 ± 1.8, p = 0.04, respectively; Table 3). No clotted drainage incurred in ABP, despite a lack of anticoagulation; however, this developed in one patient in the talc group. According to the findings of our two studies, ABP appears to have outstanding safety profiles; probably the best explanation for this may be from the autologous blood having its own property as a human endogenous substance, which causes less injury to the pleura, unlike some exogenous substances like talc and tetracycline.
Regarding talc pleurodesis, a recent meta-analysis demonstrated that both talc slurry under LA and talc poudrage via thoracoscopy under GA offer a similar rate of pleurodesis efficacy. Meanwhile, talc slurry is a minimally invasive technique that carries less risk of respiratory complications; moreover, it is offered at a low cost, is generally available and is more convenient to perform.12,13 Thus, talc slurry instillation was selected to be used instead of thoracoscopic talc insufflation in this study.
In regard to pleurodesis-related complications, there had been neither cardiopulmonary nor systemic toxicity reported in our talc group. It is possibly that this was due to the high dose and small particle size talc, which meant a much higher number of lung and systemic toxicities being precluded from the study. This was as we were concerned with patient safety and ethical issues.22,40–44
The advantages of our study are that: ABP profiles were compared to a worldwide high-efficacy sclerosant like talc. The significance levels of pleurodesis-related fever along with pain were well classified before antipyretic and opioids were administered. Likewise, pleurodesis-efficacy assessment was determined by blinded chest physicians. However, our study had several limitations. First, only the short-term efficacy of ABP was evaluated. Second, there is a lack of consensus concerning the optimal blood volume to be instilled. Third, there is no beneficial data of ABP on talc non-responder cases. Fourth, pleurodesis-related complications were reported by the unblinded primary physicians. Lastly, the per-protocol analysis was selected for usage, instead of an intention-to-treat analysis as some patients had dropped out of the study. Therefore, further studies are warranted to confirm long-term efficacy, the optimal blood volume and the benefit of ABP for the talc non-responder group.
In conclusion, ABP has an equivalent efficacy compared to talc pleurodesis for the treatment of MPE. ABP causes less fever and pain and could shorten hospital stay, compared to talc. Pleurodesis using autologous blood offers a simple, readily available and well-tolerated method of treatment, causing neither allergy nor pulmonary and systemic adverse events. This would suggest it is worthwhile to consider ABP as a good treatment option for MPE, especially when being concerned about talc or lidocaine-induced systemic toxicity, or in cases where talc is unavailable.
Supplemental Material
CONSORT_2010_Checklist – Supplemental material for Efficacy and safety profile of autologous blood versus talc pleurodesis for malignant pleural effusion: a randomized controlled trial
Supplemental material, CONSORT_2010_Checklist for Efficacy and safety profile of autologous blood versus talc pleurodesis for malignant pleural effusion: a randomized controlled trial by Warangkana Keeratichananont, Apichat Kaewdech and Suriya Keeratichananont in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The authors would like to acknowledge the Royal College of Physicians of Thailand (RCPT) for providing a Clinical Research Award in 2016.
Authors’ contributions
Keeratichananont: designed, conducted, supervised, reviewed the manuscript and coordinated. Kaewdech: conducted, the pleurodesis and statistical analysis. Keeratichananont: writing and review of the manuscript. All authors read and approved the final manuscript.
Authors’ note
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
