Abstract
Objectives:
Autologous blood pleurodesis (ABP) is used for the treatment of pneumothorax with a good efficacy. The aim of this study is to determine the efficacy and safety of ABP in the treatment of malignant pleural effusion (MPE).
Methods:
A prospective study was conducted at Songklanagarind Hospital, Thailand. Symptomatic MPE patients were randomized to receive pleurodesis with either autologous blood or tetracycline. In the ABP group, 100 ml of autologous venous blood was instilled via chest tube followed by 50 ml of sterile normal saline (NSS). In the tetracycline group, 20 ml of 1% lidocaine diluted in 30 ml NSS was instilled followed by 1 g of tetracycline diluted in 100 ml of NSS. The chest tube was clamped for 2 hours, then reconnected to suction and removed. Pleurodesis effectiveness was evaluated according to Paladine’s criteria and adverse events were recorded.
Results:
A total of 48 symptomatic MPE patients were recruited. Of these, 24 cases were randomized to receive ABP and 24 cases received tetracycline. There was no difference between the two groups in baseline characteristics. The overall success rate of pleurodesis was the same in both the autologous blood group and the tetracycline group (83.4% versus 87.5%, p = 0.36). In the ABP group, the pain score and fever were significantly lower (8.3% versus 29.1%, p = 0.003) and there was a small percentage of cases that needed analgesia (4.2% versus 75%, p < 0.001); no serious events occurred.
Conclusions:
ABP was as effective as tetracycline in the treatment of MPE. ABP produced less pain and fever, and could shorten the hospital stay.
Introduction
Malignant pleural effusion (MPE) is a common complication of advanced malignancy, defined as an abnormal accumulation of fluid containing malignant cells in the pleural space, which is a pathological process that highlights the failure in the defense mechanism within the pleura and the abnormal mesothelial function [Kastelik, 2013; Stathopoulo and Kalomenidis, 2012]. The estimated annual incidence of MPE in the United States is between 150,000 and 175,000 cases [Bennett and Maskell, 2005; Marel et al. 1993]. Clinical symptoms vary from progressive dyspnea, cough and pleuritic chest pain to respiratory failure, thus affecting quality of life, shortening survival time and leading to poor prognosis [Burrows et al. 2000; van de Molengraft and Vooijs, 1989].
Systemic chemotherapy is the first-line treatment for symptomatic MPE patients with good performance status aimed at tumor shrinkage and pleural fluid absorption. However, most causative cancers become chemoresistant and many patients are not fit for chemotherapy. Therefore, one of the following palliative procedures is used to relieve symptoms, improve quality of life and reduce recurrence of effusion: pleural aspiration or chemical pleurodesis (intercostal tube drainage and instillation of sclerosant into the pleural cavity) or thoracoscopy with pleurodesis or placement of an indwelling pleural catheter [Kastelik, 2013; Zahid et al. 2011; Roberts et al. 2010; American Thoracic Society, 2000]. Repeated pleural aspiration alone or intercostal tube drainage without instillation of sclerosant are associated with a high recurrence rate and increased risk of iatrogenic pneumothorax and empyema [Boland et al. 1998; Sorensen et al. 1984]. Therefore, these procedures are used only for patients with a survival expectancy of less than 1 month or in case of a trapped lung (apposition of the parietal and visceral pleura cannot be achieved). The alternative is thoracoscopy with pleurodesis or placement of an indwelling pleural catheter; these procedures have their own limitations such as invasiveness (required sedation or general anesthesia), discomfort, cost, complications, and the availability of institutional and local expertise.
Therefore, chemical pleurodesis is the most common modality of therapy for patients with recurrent symptomatic MPE. Various sclerosants such as sterile talc, tetracycline, doxycycline, bleomycin, iodine and silver nitrate have been used to cause chemical irritation, leading to pleural inflammation, fibrosis and obliteration of the pleural space. The success rate for chemical pleurodesis ranges from 58 to 90%, with a higher efficacy in talc [Gondker et al. 2011; Mohsen et al. 2011; Terra et al. 2011; Roberts et al. 2010; Tettey et al. 2005; Shaw and Agarwal, 2004; Kuzdzal et al. 2003; Gravelyn et al. 1987; Fentiman et al. 1986]. However, Chemical Pleurodesis often occurs in sclerosant-related complications such as pleuritic chest pain (needs preprocedural intrapleural lidocaine with or without additional intravenous analgesic), fever, empyema and myelosuppression (bleomycin), and some develop tachycardia, acute pneumonitis impaired pulmonary function test (PFT), acute respiratory distress syndrome (ARDS) and respiratory failure, predominantly in talc [Xia et al. 2014; Kuzniar and Mutlu, 2004]; thus to date no ideal sclerosant exists.
In 1987, Robinson used autologous blood as the sclerosant to induce pleurodesis in patients with chronic spontaneous pneumothorax with a persistent air leak (PAL); this showed an 85% success rate without any complications [Robinson, 1987]. From animal experimental studies [Ozpolat et al. 2010; Mitchem et al. 1999], autologous blood had dual mechanism effects on PAL and created pleurodesis. Firstly, the formation of a clot (blood patch effect) and subsequently induced pleural irritation and inflammation, leading to the formation of pleural adhesions (fibrinous pleuritis).
Over time, there have been many published studies comparing 1–2 ml/kg of autologous blood (without instillation of lidocaine) versus other sclerosant-induced pleurodesis for termination of PAL and pneumothorax. The results showed that the overall success rate was the same as, or superior to, talc and tetracycline (75–92%), but significantly lower in pleuritic chest pain, dyspnea, impaired PFT, fever and empyema (0–18%). In addition, autologous blood pleurodesis (ABP) is a cheaper, simpler, no allergies, well-tolerated and readily available treatment, suggesting it is worthy of consideration for the first-line treatment of pneumothorax with PAL [Cao et al. 2012; Manley et al. 2012; Aihara et al. 2011; Chambers et al. 2010; Cobanoglu et al. 2009]. However, there has been no report concerning the efficacy of ABP for the treatment of MPE. Therefore, this study was conducted to determine the efficacy and safety profile of ABP in symptomatic MPE patients.
Methods
We conducted a pilot study in 2010 when ABP without intrapleural lidocaine was performed in 10 cases with symptomatic MPE at Songklanagarind Hospital, Songkhla, Thailand. The success rate was 83% without any serious adverse events. Subsequently a prospective, randomized, comparative study was conducted from September 2011 to November 2012. Inclusion criteria were patients aged 18 and older with symptomatic MPE (cytologically or histologically confirmed), life expectancy of more than 3 months and agreement to receive chemical pleurodesis for treatment of MPE. Patients diagnosed with active pleural or systemic infection, hemodynamic instability, hemothorax (pleural fluid over serum hematocrit > 50%), serum hematocrit < 25%, chylothorax, a combination of causes of pleural effusion, history of tetracycline or lidocaine allergy, chest X-ray showing trapped lung after chest tube drainage, and pregnancy were excluded from the study.
After signing informed consent forms, patients were randomized into two groups (using block randomization) to receive pleurodesis with autologous blood or tetracycline. After chest tube insertion with either the small catheter [percutaneous drainage (PCD) 8–10 French] or large bore catheter [intercostal drainage, (ICD) 20–32 French], pleural fluid was examined and completely drained until the drainage fell below 100 ml/day and chest X-ray showed the full expansion of the affected lung. Thereafter, in the ABP group, 100 ml of autologous venous blood without anticoagulant was obtained from each patient’s forearm using two 50 ml syringes. As fast as possible, the blood was instilled through the pleural cavity via the chest tube using sterile techniques (without intrapleural lidocaine injection) and followed by 50 ml of sterile normal saline (NSS) for preventing blood clots in the chest tube. In contrast, in the tetracycline pleurodesis group, 20 ml of 1% lidocaine, made up to 50 ml with NSS was instilled in the chest tube; after waiting for 5 minutes; it was followed by a low injection of 1 g of tetracycline diluted in 100 ml NSS within 5–10 minutes. After instillation of the sclerosant, the chest tube was clamped for 2 hours, during which time the patient did not change position. Then the clamp was released, the chest tube was reconnected back to the water-sealed system, and continuous thoracic suction with -20 cm water pressure was applied in both groups. Within the next 24 hours, chest X-ray was repeated in all cases and the chest tube was removed when the lung had fully re-expanded and drainage had fallen below 150 ml.
Pleurodesis effectiveness was evaluated 30 days after the procedure according to Paladine’s criteria [Tettey et al. 2005], which are as follows:
Complete response: no pleural effusion was detected.
Partial response: minimal pleural fluid was detected after pleurodesis without the need for repeat thoracentesis or drainage.
Nonresponse: massive amount of effusion was detected after pleurodesis or there was a need for repeat thoracentesis or pleurodesis.
The complete and partial responses were defined as the success of pleurodesis. Patient characteristics, length of stay in hospital and sclerosant-related complications within 48 hours, 15 days and 30 days were also recorded. Pleurodesis-related pain was scored using a numeric rating scale from 0 to10 (0 = no pain, 1–3 = mild, 4–6 = moderate, 7–9 = severe and 10 = worst) at interval of 5 minutes before, 5 minutes after, and 6 and 24 hours after sclerosant injection. Pain intensity and additional intravenous analgesics were scored and given by the primary physician. Nonresponse cases to ABP were repeated with tetracycline. Together, other sclerosants such as iodine or talc were used in nonresponse cases to tetracycline.
The study protocol was approved by the Ethics Committee of the institution (IRB#54-345-14-3-3) and was conducted in accordance with the World Medical Association Declaration of Helsinki and Good Clinical Practice guidelines 2013.
Statistical analysis
The mean and standard deviation (SD) were used to describe continuous data. Proportion (%) was used to describe categorical data. Student’s t-test and the chi-square test were used to analyze the continuous and categorical characteristics, respectively. The outcomes of interests were analyzed with SPSS version 16 software and the results were considered as statistical significant if p < 0.05.
Results
In the study, 24 cases received ABP and 24 cases received pleurodesis with tetracycline. The mean age was 62 years-old in both groups. Lung cancer was the most common primary site (70–79%) followed by ovarian, breast and sarcoma. Dyspnea with or without respiratory distress was the presenting symptom and chest X-ray showed moderately to massive pleural effusion in most cases. Two patients had bilateral pleural effusion, but the amount of effusion detected on the other side was small and thus drainage was required only in the hemithorax. A total of 17 cases (71%) in the tetracycline and nine cases (37%) in the autologous blood group received chemotherapy 2 weeks after pleurodesis (p = 0.02). There was no difference between the two groups in terms of gender, pleural fluid characteristics and type of chest tube (Table 1).
Baseline characteristics of patients in tetracycline and autologous blood groups.
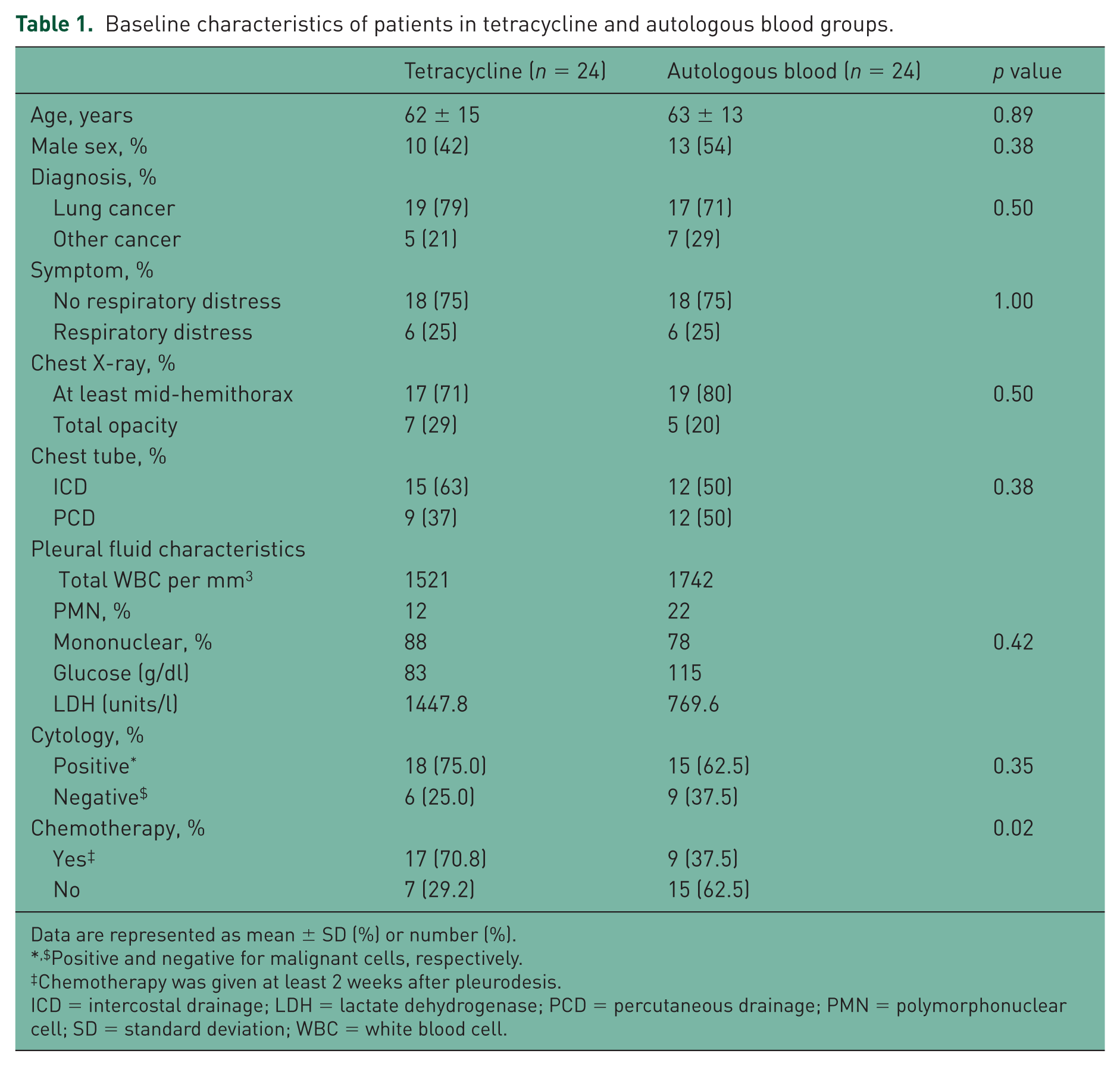
Data are represented as mean ± SD (%) or number (%).
Positive and negative for malignant cells, respectively.
Chemotherapy was given at least 2 weeks after pleurodesis.
ICD = intercostal drainage; LDH = lactate dehydrogenase; PCD = percutaneous drainage; PMN = polymorphonuclear cell; SD = standard deviation; WBC = white blood cell.
By the time of the evaluation after 30 days, the pleurodesis efficacy in the autologous blood group showed: complete response 41.7%; partial response 41.7%; and nonresponse 16.6%. The tetracycline group showed: complete response 37.5%; partial response 50%; and nonresponse 12.5%. The overall success rate of pleurodesis at 1 month (complete and partial response) was achieved in 83.4% of the autologous blood group patients and 87.5% in the tetracycline group (p = 0.36).
There was no statistically significant difference in the efficacy of pleurodesis between the two groups (Table 2). However, less sclerosant-related pain, fever and shorter hospital lengths of stay occurred in the autologous blood group compared with the tetracycline group (Table 3, Figure 1). The pain score was significantly lower at every time point and there was a small percentage of cases that needed adjunctive intravenous analgesics in the autologous blood group (4.2% versus 75%, p < 0.001). Fever occurred less in the autologous blood group (8.3% versus 29.1%, p = 0.003) and, in addition, hospital length of stay was significantly shorter in the autologous blood group (8.2 ± 4.6 versus 9.8 ± 5.5, p = 0.04). No clotted drainage, empyema or serious side effects were found in either group. Four cases failed to respond to ABP. In two of these cases pleurodesis with tetracycline was repeated, the results showed success in one case. In the other cases, the patients refused to undergo chemical pleurodesis again.
Pleurodesis efficacy.
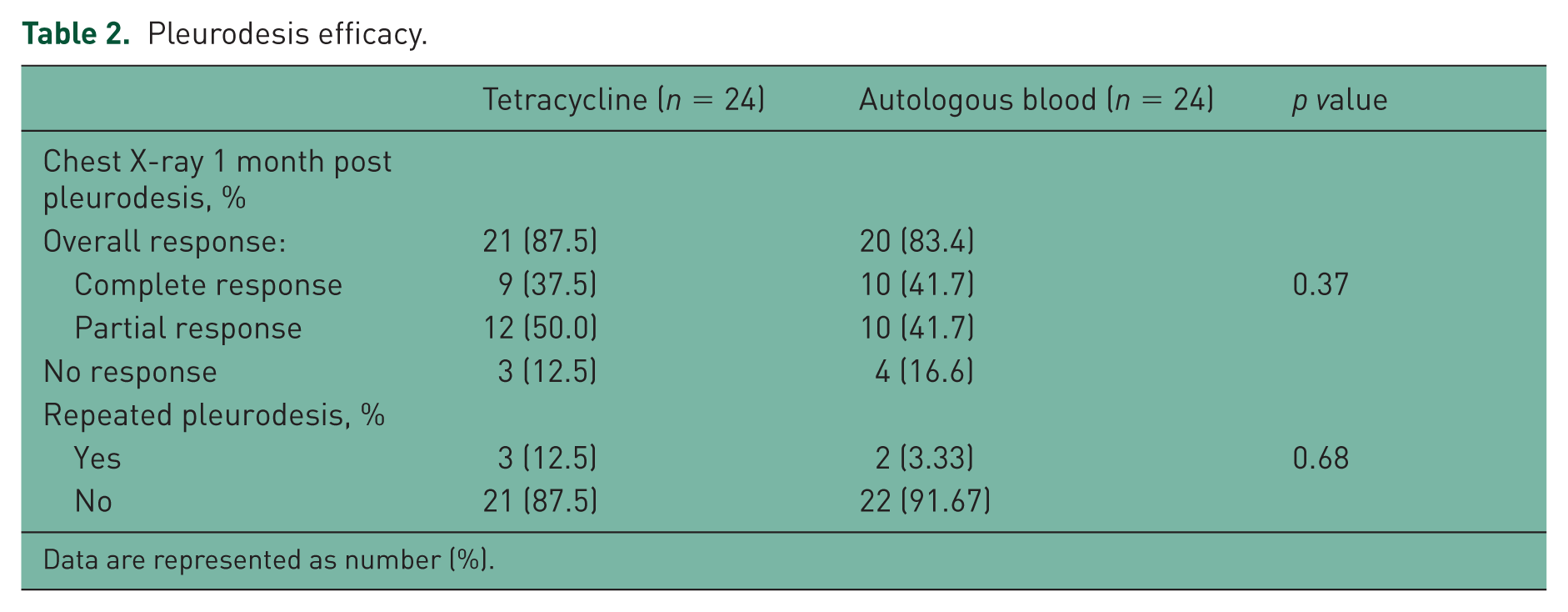
Data are represented as number (%).
Sclerosants’ related side effects and hospital lengths of stay.

Data are represented as mean ± SD (%) or number (%).
Pain score ranges from 0 to 10.
Before and immediately of pleurodesis 5 minutes, respectively.
6 and 24 hours after pleurodesis, respectively.
Sclerosant related fever defined as body temperature at least or greater than 38 degrees Celsius within 48 hours of pleurodesis and no evidence of empyema, organs specific and systemic infections.
SD = standard deviation.
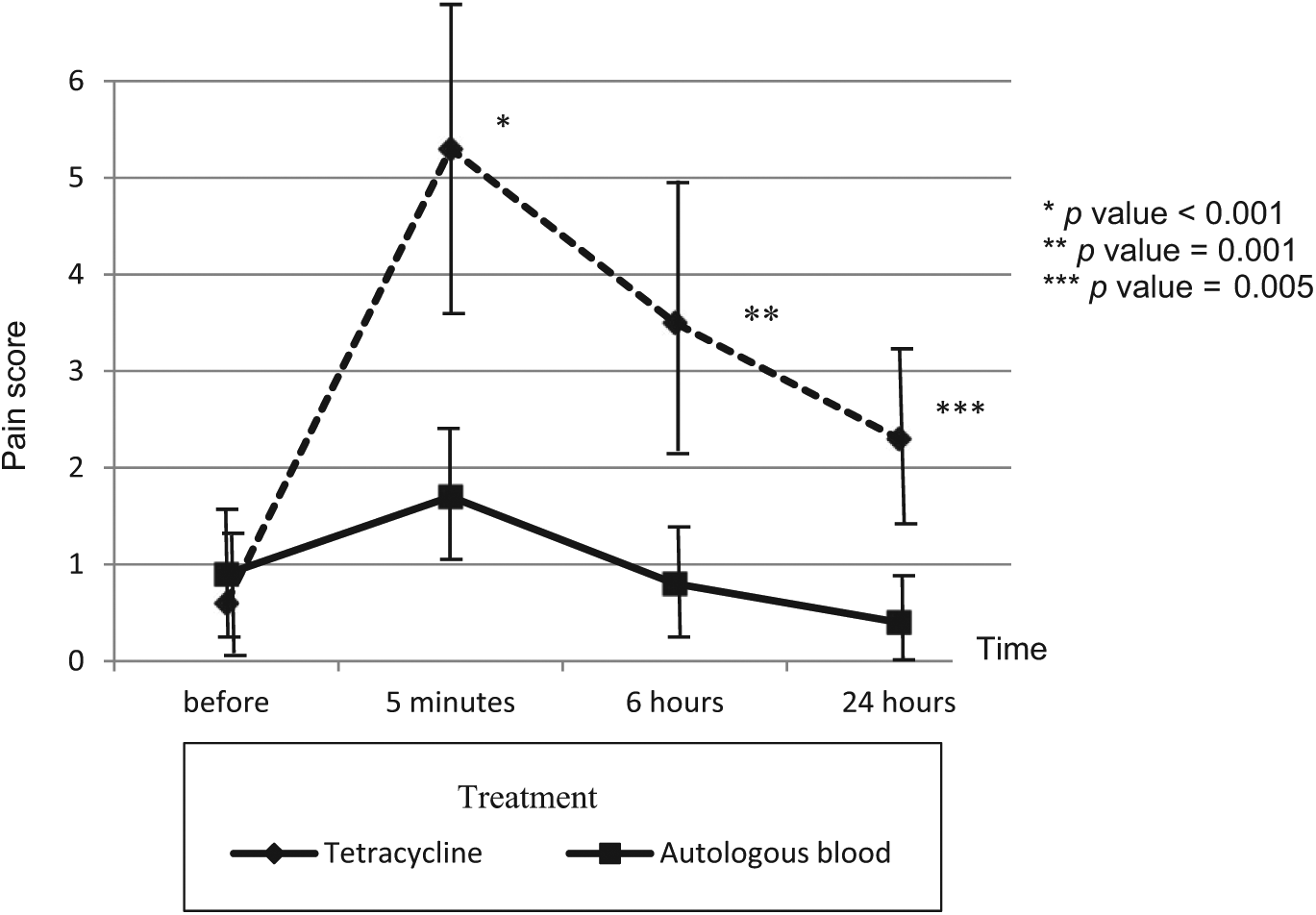
Sclerosant related pain score. Pleurodesis with autologous blood produces less pain at every time points after the pleurodesis compared with tetracycline group.
Discussion
To the best of our knowledge, this is the first study comparing autologous blood with tetracycline for pleurodesis in symptomatic MPE. The results showed autologous blood offered equal benefits in the efficacy as tetracycline and also fewer side effects occurred.
To date, there has been no the ideal sclerosant for the treatment of MPE. Talc is the most effective available sclerosant, but many associated serious systemic side effects have been observed [Kuzniar and Mutlu, 2004; Robinson, 1987]. Tetracycline is a safer, cheaper, more available sclerosant in many countries, including Thailand. From the historical data, tetracycline pleurodesis is equally as effective as doxycycline and bleomycin. Pleuritic chest pain and fever are, however, the major drawback of tetracycline and all the other available sclerosants [Roberts et al. 2010; American Thoracic Society, 2000]. Thus, a new sclerosant that provides both a high efficacy and a high safety profile is required. We know that ABP without the instillation of lidocaine gives a high efficacy in the treatment of pneumothorax with PAL (by induce fibrinous pleuritis) and also results in fewer episodes of pleuritic chest pain and fever compared with tetracycline [Cao et al. 2012; Ozpolat et al. 2010]. In our study, we used 100 ml of autologous blood without the instillation of lidocaine compared with tetracycline for the treatment of MPE. The study showed ABP had a high overall success rate that was similar to tetracycline (83.4% versus 87.5%, p = 0.36), no serious side effects occurred, significantly fewer with pleuritic chest pain needed adjuvant analgesics (4.2% versus 75%, p < 0.001), less fever (8.3% versus 29.1%, p = 0.003) and shorter hospital lengths of stay (8.2 ± 4.6 versus 9.8 ± 5.5,p = 0.04) compared with tetracycline.
Autologous blood is a human endogenous substance and thus it does not produce serious systemic toxicity, like some sclerosants. Therefore, pleurodesis with autologous blood is a safe procedure. However, our study has some limitations. Firstly, there is a lack of consensus concerning the optimal volume of blood to be instilled. Secondly, there are no data on the usefulness of repeated pleurodesis with autologous blood in nonresponder cases. Lastly, comparative data are limited to only autologous blood versus tetracycline. Therefore, further studies are needed to confirm the optimal blood volume for ABP, the efficacy and safety profile of ABP compared with other sclerosants and the usefulness of repeated ABP in nonresponder cases.
In conclusion, chemical pleurodesis using autologous blood was as effective as tetracycline. Pain and fever occurred less with ABP, there were no reported serious systemic side effects and ABP could shorten the hospital stay. Therefore, ABP is a good alternative choice in the treatment of symptomatic MPE, especially in patients with a history of tetracycline or lidocaine allergy.
Footnotes
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
Funding
The authors would like to acknowledge the Thoracic Society of Thailand for giving us the Clinical Fellowship Research Award in 2013.
