Abstract
Malignant pleural effusion (MPE) affects more than 1 million people globally. There is a dearth of evidence on the therapeutic approach to MPE, and not surprisingly a high degree of variability in the management thereof.
We aimed to provide practicing clinicians with an overview of the current evidence on the management of MPE, preferentially focusing on studies that report patient-related outcomes rather than pleurodesis alone, and to provide guidance on how to approach individual cases.
A pleural intervention for MPE will perforce be palliative in nature. A therapeutic thoracentesis provides immediate relief for most. It can be repeated, especially in patients with a slow rate of recurrence and a short anticipated survival. Definitive interventions, individualized according the patient’s wishes, performance status, prognosis and other considerations (including the ability of the lung to expand) should be offered to the remainder of patients. Chemical pleurodesis (achieved via intercostal drain or pleuroscopy) and indwelling pleural catheter (IPC) have equal impact on patient-based outcomes, although patients treated with IPC spend less time in hospital and have less need for repeat pleural drainage interventions. Talc slurry via IPC is an attractive recently validated option for patients who do not have a nonexpandable lung.
Introduction
The incidence of malignant pleural effusion (MPE) is 660 per million population, resulting in more than 1 million people affected globally and representing a growing healthcare burden.1,2 In the United States (US) alone, more than 125,000 hospital admissions per year are attributable to MPE, with an inpatient mortality of approximately 12% and an associated cost of over US$5 billion per year. 3
Lung cancer is the most common cause of MPE, accounting for almost half of all cases in some series. 4 Up to 15% of lung cancer patients will have an MPE at presentation, and up to 50% will develop an MPE during the course of the disease. 5 Other common causes are breast cancer and hematological malignancies, particularly lymphomas. 1 There is major geographical variation in the incidence of malignant pleural mesothelioma (MPM), which will be complicated by MPE in more than 90% of cases. 1
MPE is most often caused by direct or hematogenous spread of malignant cells to the visceral pleura with secondary seeding to the parietal pleura, but can also result from direct tumor invasion or hematogenous spread to the parietal pleura. 6 Malignancy can also cause a pleural effusion without direct pleural involvement. This condition, known as a paramalignant effusion, can arise from various mechanisms such as obstructive pneumopathy, pulmonary embolism, lymphatic mediastinal obstruction and superior vena cava syndrome. 7
Dyspnea is the most common symptom in patients with a pleural effusion, and the primary indication for an intervention. 8 Altered chest wall mechanics and caudal displacement of the diaphragm are considered to be the main mechanisms involved, but many other poorly defined processes are likely to contribute. 9 The degree of dyspnea is often disproportionate to the size of the effusion, as underlying lung cancer may cause both pulmonary collapse and pulmonary arterial infiltration, resulting in minimal ventilation: perfusion mismatching. Chest pain may not be a prominent symptom, unless chest wall invasion or MPM is present. 9
An intervention for the management of MPE will perforce be palliative in nature, as no procedure has yet been shown to prolong life in this setting.8,10 There is unfortunately a dearth of high-quality evidence on the therapeutic approach to MPE, and not surprisingly a high degree of variability in the management of the condition. 1 As more therapeutic options are developed, the disparities in practice increase, a trend which is particularly evident when surveys completed by pulmonary physicians are compared with their surgical counterparts. 11 Pulmonary physicians offer talc slurry pleurodesis or indwelling pleural catheters (IPCs) to the majority of cases where intervention is required and refer less than 20% for video-assisted thoracoscopic surgery (VATS), whereas almost 70% of cardiothoracic surgeons opt for VATS pleurodesis as first-line therapy. 12
Another major limiting factor of the existing literature on MPE is the fact that outcome measures were radiological endpoints and ‘successful’ pleurodesis was seen as surrogates for successful interventions. 1 Only recently have patient-related outcome measures, including time spent in hospital (or avoidance of hospitalization), palliation of symptoms and quality of life (QoL), been reported as main outcomes, as opposed to fluid reaccumulation. 1
The primary aims of this review are to provide the practicing clinician with an overview of the current evidence base on the management of MPE, and to provide practical guidance on how to approach individual cases. Ultimately management should be tailored around the patient’s individualized needs and wishes, and to an extent to local expertise. 1
Body
Prognosis
MPE generally signifies incurable disease with a poor prognosis.1,8 The median survival is around 3–12 months, but can vary significantly according to cell type, performance status of the patient, staging and whether a chemosensitive malignancy is present. 10 Assessing anticipated survival in patients with MPE is paramount, as it will aid in clinical decision making and impact on the intervention offered in many cases. 1 The best validated scoring system is the LENT score (Table 1), which utilizes the pleural fluid analysis, Eastern Cooperative Oncology Group (ECOG) performance status score and tumor type to predict survival. 13 A patient with metastatic lung cancer and a poor performance status will, for example, have a significantly worse anticipated survival than a patient with MPM, a good performance status and favorable pleural fluid parameters (median survival 1.5 versus 10.5 months).
The LENT score calculation (adapted from Clive and colleagues). 13
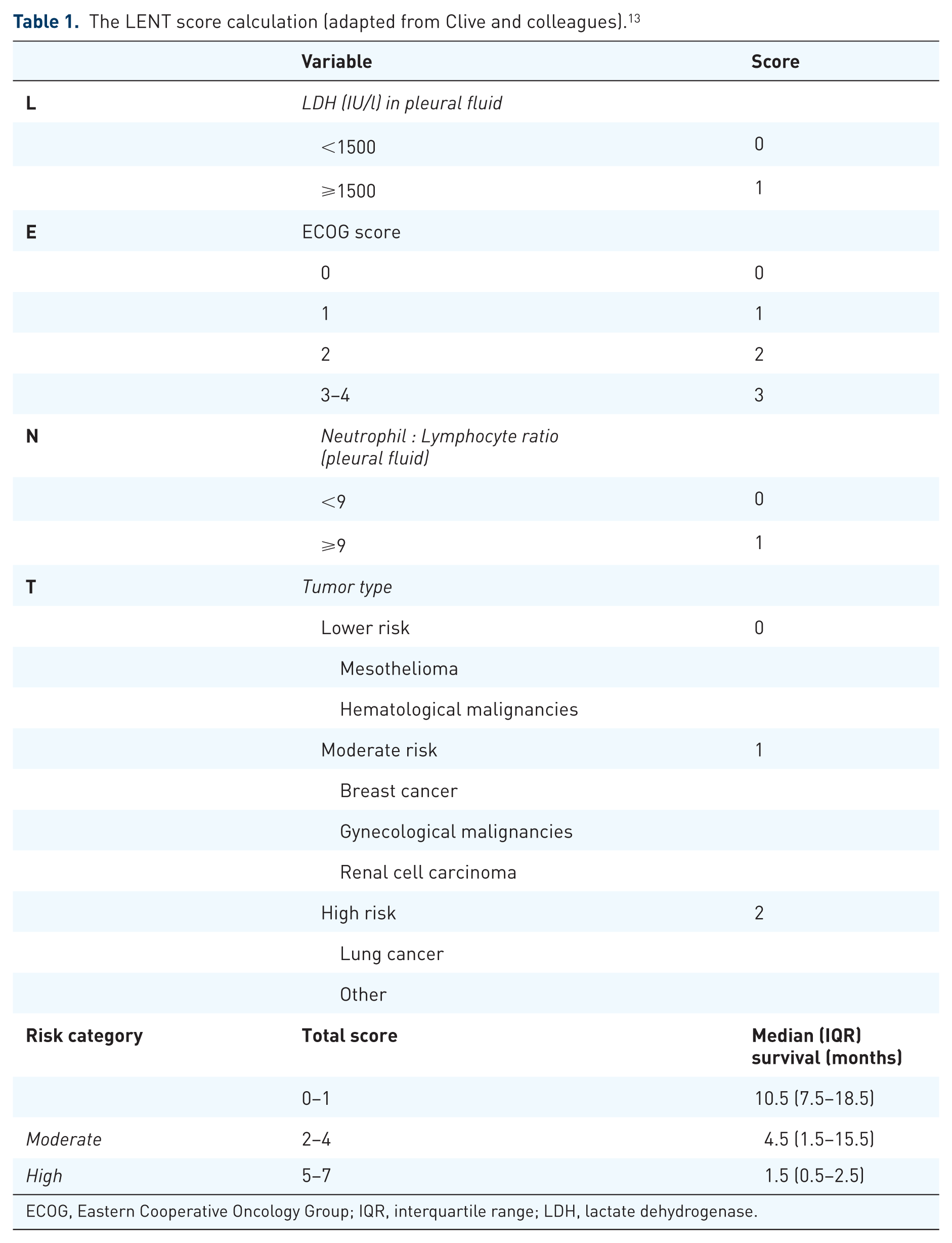
ECOG, Eastern Cooperative Oncology Group; IQR, interquartile range; LDH, lactate dehydrogenase.
General principles
Once the diagnosis of an MPE is confirmed, palliative measures should be considered, with the aims of improving QoL (minimizing dyspnea), limiting pleural procedures and avoiding the need for repeated hospital or doctor visits. 9 In general, only symptomatic patients should be offered interventions which should be tailored to their needs. 1 Many factors influence the choice of definitive management offered, including the prognosis, performance status, size/recurrence rate, whether or not the underlying lung will expand once the fluid is drained, whether or not therapeutic thoracentesis provided symptomatic relief, the chemosensitivity of the malignancy, local expertise and personal preferences.8,14
The treatment options for MPE are summarized in Table 2. A therapeutic thoracentesis provides immediate relief in most cases, provided other causes for dyspnea (e.g. pulmonary embolism) are not present. 9 Therapeutic thoracentesis can be repeated (as an outpatient), especially in patients with slow rate of recurrence, in patients who have a very short anticipated survival or a poor performance status. 8 However, a definitive intervention should be offered to patients with a long-anticipated survival, given the cumulative discomforts, risks and costs with repeated thoracentesis. 8 A recent retrospective cohort study found that guideline consistent care using definitive procedures (IPC or pleurodesis) compared with repeat thoracentesis was associated with fewer subsequent procedures and complications; however, pleurodesis resulted in more inpatient days. 15
Treatment options for malignant pleural effusions.

ICD, intercostal tube drainage; NEL, nonexpandable lung; IPC, indwelling pleural catheter; VATS, video-assisted thoracoscopic surgery.
Asymptomatic effusions, irrespective of size, generally require no specific interventions. 1 Several studies have shown that only around half of patients with MPE require or opt for definitive treatment during the course of their disease.16,17
Minimally invasive/nonsurgical interventions
Intercostal tube drainage with chemical pleurodesis
Chemical pleurodesis which is achieved by the instillation of a sclerosant via an intercostal drain (ICD) is still considered by many as the first-line intervention in cases without a nonexpandable lung (NEL), particularly in patients with an anticipated survival of >3 months.1,18 The improvement in QoL and other patient-related outcomes is comparable with other interventions, although patients spend more time in hospital, both initially and until death, when compared with IPCs.19,20
The use of talc as a pleural sclerosant was first described as early as 1935.21,22 In what is still the largest randomized controlled trial (RCT) to date on pleurodesis, the radiological success rate of talc pleurodesis was approximately 75% after 1 month. 23 The same study found that thoracoscopy with talc poudrage was not superior to talc slurry via ICD, although a post-hoc analysis suggested that a subgroup of lung and breast cancers tended to have a higher pleurodesis rate with talc poudrage. 23
Small size talc particles have been associated with the development of acute respiratory distress syndrome (ARDS), in all probability because of systemic absorption. 1 However, calibrated French large-particle talc is believed to have less systemic absorption and as such does not cause ARDS. 24 Numerous other agents have been used to achieve chemical pleurodesis, including bleomycin, tetracycline, doxycycline, iodopovidone, silver nitrate and even mistletoe.22,25,26 Many of these sclerosants have been associated with major systemic complications, such as hyponatremia, deranged liver enzymes and subclinical hypothyroidism with iodopovidone, pulmonary toxicity with bleomycin, and acute kidney injury and ARDS with silver nitrate. 8 At least two meta-analyses found that nonrecurrence of the effusion was more likely with talc than other sclerosants (except doxycycline), suggesting that there is little advantage in using other agents over large-particle talc.27,28
There is surprisingly little high-quality evidence regarding the practical aspects of pleurodesis via an ICD, and hence major differences in practice. 1 Current available evidence suggests that small bore tubes (12–14F) are as effective as large bore drains, with potentially fewer complications. 29 Pleurodesis should only be performed once pleural apposition is achieved, and generally only once <150 ml is drained over 24 h. 1 The traditional practice of rotating the patient is no longer advocated, as this may lead to dislodgement of the ICD. 18 Guidelines based on expert opinion suggest clamping for an hour. 18
IPCs
Tunneled IPCs are usually inserted as an outpatient procedure and home drainage is performed, offering an ambulatory alternative to ICD and pleurodesis.1,30 Although the primary aim is to intermittently drain the MPE in order to maintain adequate lung expansion and relieve dyspnea, spontaneous pleurodesis occurs in approximately 24–45% of all cases, usually within 7 weeks. 9
Catheter malfunction and tract metastases (in the case of MPM) are observed in 9–10% of cases treated with IPC. 31 Pleural infection (1–5%) and cellulitis (2–3%) are relatively uncommon. 32 Skin flora, including Staphylococcus aureus, accounts for the majority of infections, while Pseudomonas aeruginosa and Enterobacteriaceae responsible are also sometimes isolated. 32 Pleural infection normally occurs at least 6–8 weeks after insertion, suggesting that the procedure itself is unlikely the source. 33 The vast majority are mild and resolve with oral antibiotic treatment, and the removal of the IPC is not necessary unless the infection fails to respond. 33 The IPC also permits intrapleural tissue plasminogen activator/DNase therapy, which is sometimes employed to facilitate drainage. 32
Patients managed with IPC spend less time in hospital, both initially and during the course of their disease, compared with pleurodesis. 19 Early evidence suggested that both IPC and ICD with pleurodesis provided comparable improvement in shortness of breath and QoL, although some studies reported at least a trend towards superior improvements with IPC.19,34 Current evidence also suggests that patients treated with IPCs are much less likely to require further invasive pleural drainage procedures. 20
Cost comparisons are difficult and in all probability influenced by the healthcare system. Evidence using United Kingdom costings has suggested that the overall mean costs per year for IPC were not significantly different when compared with ICD with pleurodesis, although IPC became a significantly cheaper option in those who survived <14 weeks.35,36
The ASAP trial, which excluded all cases with NEL, was plagued by a high dropout rate and early deaths. 37 The investigators, however, concluded that ‘aggressive’ daily drainage (compared with alternative day drainage) significantly increased the rate of autopleurodesis (47% versus 24%, p = 0.003) and decreased the median time to autopleurodesis (54 versus 90 days) in patients. 37 QoL and patient satisfaction were similar between the groups. 37
The AMPLE study, a large open-label, multicenter, multinational clinical trial that included 146 patients randomized to either an IPC (n = 74) or talc pleurodesis (n = 72) was recently published. 20 Patients randomized to the IPC arm spent fewer days in hospital from treatment to death (10 versus 12 days, p = 0.03, translating to 6% versus 11% of patients’ remaining life span respectively). No significant between-group differences in improvements in breathlessness or QoL were observed. 20
The combination of IPC and talc pleurodesis has the potential advantage of facilitating early tube removal, thereby decreasing the inconvenience of regular drainage, risks of infection and mechanical failure and cost. 1 This approach was initially investigated on a small scale in two studies, one where an IPC was inserted at thoracoscopy following talc poudrage and another where talc slurry was instilled via the IPC. Both studies demonstrated 92% success rates at 6 months.38,39
In the very recently published IPC-Plus study all patients underwent drainage via an IPC regularly on an outpatient basis. 30 Patients with no evidence of NEL at day 10 were randomly assigned to receive either 4 g of talc slurry (n = 69) or placebo through the IPC (n = 70). At day 35, a total of 30 of 69 patients (43%) in the talc group had successful pleurodesis, compared with 16 of 70 (23%) in the placebo group (p = 0.008). 30 The fact that the study employed only intermittent IPC drainage after talc may have been the reason for the relatively modest pleurodesis rate compared with earlier studies. 20 No significant between-group differences in effusion size and complexity, number of inpatient days, mortality, or number of adverse events were identified. No significant excess of blockages of the IPC was noted in the talc group. QoL scores and symptom control were also shown to be superior in the talc arm of the study. 30
Yet another approach to combine pleurodesis and IPC is the silver nitrate-coated IPC. The SEAL-MPE study was the first in-human trial designed to evaluate the basic safety profile of the novel silver nitrate-coated IPC (SNCIPC) device in MPE. 40 The device was reasonably well tolerated, and pleurodesis after a median of 4 days was achieved in eight of nine patients with expandable lungs. 40
Finally, some centers routinely insert an IPC at the time of talc poudrage. It has the theoretical advantage of earlier discharge, and having the IPC as backup if pleurodesis fails. 15 However, pleurodesis failure tends to only occur after months, casting doubt on the merits of inserting a ‘prophylactic’ IPC in all cases. 23
Medical thoracoscopy/pleuroscopy
Physician-performed thoracoscopic treatment of MPE is a validated intervention performed in endoscopy units under local anesthesia and conscious sedation. 41 Pleuroscopy-assisted talc poudrage can offer excellent palliation to patients with longer expected survival, and can be performed at the time of a diagnostic procedure in patients with a high probability of MPE based on endoscopic findings. 1 It is more invasive and resource consuming when compared with ICD with pleurodesis or IPC, but may have superior rates of pleurodesis in lung and breast cancer patients. 23 Complications such as postoperative pneumonia and respiratory failure are more common after talc poudrage during pleuroscopy than talc slurry via ICD, although poudrage has been reported to be more comfortable. 23
With current advances in the cytological assessment of pleural fluid and refinement of image-guided parietal pleural biopsies and liquid biopsies (e.g. circulating tumor DNA) which are increasingly used in countries with high epithelial growth factor receptor mutations for lung cancer, relatively fewer patients will likely undergo diagnostic pleuroscopy in the future.1,42
Surgical interventions
VATS
VATS is generally considered as more invasive and resource consuming than the interventions discussed above. It is performed under general anesthesia via multiple access ports, usually with single-lung ventilation which is achieved by means of double lumen endotracheal intubation. 1 Advantages include the fact that it permits the complete deflation of a lung, hence superior visualization and access for interventions, while allowing assessment of the underlying lung’s ability to fully expand while being inflated with positive pressure. 1 The latter, if adequate, is often followed by pleurodesis. Other potential benefits are the ability to perform mechanical abrasion of the visceral and parietal surfaces, and even parietal pleurectomy.1,43 Some centers even perform decortication in order to expand a lung that is trapped by malignant infiltration of the visceral pleura, although this is associated with a higher complication rate including persistent air leak.44–46
While there is major heterogeneity in practice, most centers would only perform VATS in patients who are deemed fit for surgery. Most studies report a success rate of >90%, although this is highly dependent on the definition of successful pleurodesis and patient selection.44,47 VATS has the advantage of potential liberation from an MPE until death without the need for any further interventions. Although patients with poor lung functions are generally excluded from studies, VATS under sedation with local anesthesia (also known as ‘tubeless’) are increasingly being offered to poor surgical candidates. 48 Another relatively recent development is the use of thoracoscopes with a diameter of 2 mm or flexi-rigid pleuroscopes to perform ‘mini-VATS’. 49
Protagonists of VATS pleurodesis will highlight the seemingly obvious advantages: The ability to perform adhesiolysis and distribute talc evenly in the pleural space. 1 There is, however, no evidence to support the notion that VATS pleurodesis is superior to bedside ICD and instillation of a sclerosant like talc slurry. 1 The largest RCT of VATS in MPE to date failed to show a significant difference in the success rate of the interventions. 23 Three smaller studies also failed to demonstrate superiority in terms of pleurodesis success rate.50–52 Similarly, a meta-analysis and a more recent retrospective study of MPM patients also could not demonstrate any significant benefit of VATS over more conservative measures.16,53 Moreover, the median length of hospital stay for an uncomplicated VATS pleurodesis is 5.8 to 10.7 days. 44 It has an inhospital mortality rate of 1–8%.23,44,54 Up to a quarter of patients may experience some complications, both short-term (fever, atelectasis, pneumonia, and prolonged air leak) and long-term (post-VATS chest wall pain).23,44,55
Other surgical procedures
Pleuroperitoneal shunts that are tunneled under the skin from the chest to the abdomen can be inserted for patients with NEL.56,57 Fluid is manually pumped by means of a pumping chamber in the subcutaneous tissue overlying the costal margin. Due to the limited volume of the pump chamber, pumping may be required more than 100 times per 24 h, necessitating great compliance from the patient or carers. 1 A 95% successful palliation rate was reported in a retrospective study, but 15% developed complications, including technical failure, infection and shunt-fracture. 56 Concomitant ascites is a contraindication for pleuroperitoneal shunting.
Extrapleural pneumonectomy is a highly controversial procedure offered by some centers for the attempted cure of MPM. It carries significant morbidity and mortality. In fact, the MARS study found that the radical surgery approach significantly shortened survival by 5.1 months. 58 The subsequent MesoVATS study found no survival difference between VATS partial pleurectomy and talc pleurodesis (either slurry or poudrage). 59 Patients randomized to VATS partial pleurectomy did however spend more time in hospital and experienced more procedure-related complications. 59
Patient-centered approach
Despite the relatively small evidence base and the gross heterogeneity in the management of MPE, certain principles should be employed to guide the management in a particular patient. Interventions to manage MPE must be individualized and should generally only be offered to patients who have experienced symptomatic improvement following initial therapeutic thoracentesis or those in whom benefit is extremely likely (i.e. patients who are breathless with no other identifiable cause). Further management should be guided by practical aspects, for example, the ability of the underlying lung to expand, the performance status of the patient, their expected survival and most importantly, the patient’s preference.
An approach which is based on current evidence is summarized in Figure 1. Repeated therapeutic thoracenteses should realistically only be offered to patients with a very slow rate of reaccumulation and an extremely short anticipated survival or poor performance status. It should be avoided in most cases, given the cumulative risk, discomfort, and costs and frequency of pleural procedures. Definitive management should be offered to these patients, but can be deferred if no reaccumulation is observed, as is sometimes seen in patients with chemosensitive malignancies.
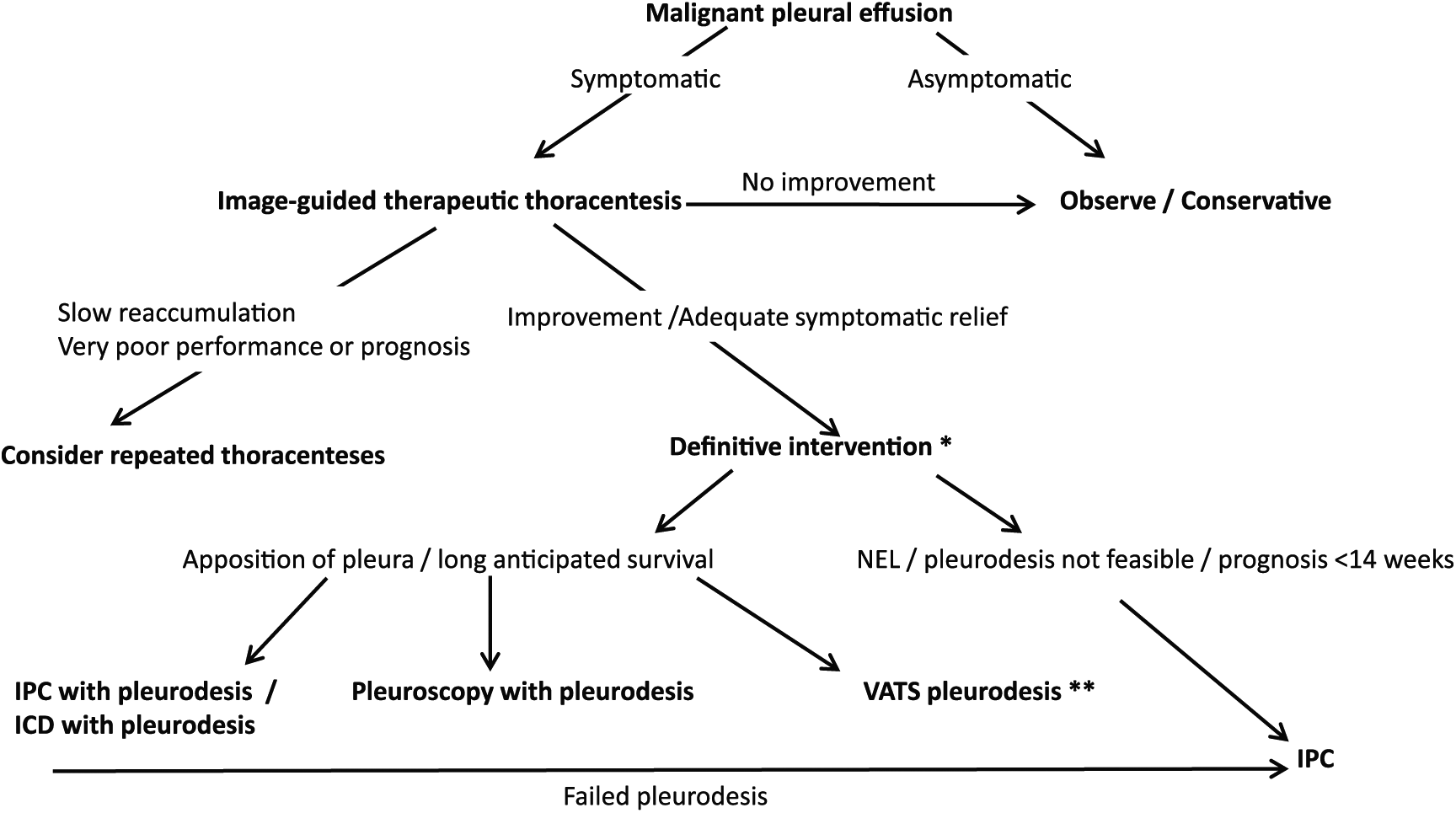
A suggested general approach to general management of MPEs.
Patients who do not have a NEL, and in whom apposition of the pleura can be achieved, are candidates for either chemical pleurodesis (via ICD) or an IPC (preferably with pleurodesis). Personal preferences, local expertise, and prognosis may dictate the choice in these cases, as both approaches have an equal impact on symptom relief and QoL, with the only real difference being less time spent in hospital with an IPC, and a lower rate of requiring further pleural interventions (22% versus 4%).
Many would advocate that an IPC, unless contraindicated, should be inserted in all patients with symptomatic MPE, as this approach would reduce the need for further pleural interventions and reduce hospital days in the remainder of the patients life. 20 Daily vacuum bottle drainage for at least 2 weeks is encouraged, based on higher pleurodesis rate. 37 If the lung fully expands, talc should be instilled via the IPC, and if pleurodesis is achieved, the IPC may be removed. 30 Patients with symptomatic MPE presenting with a NEL and those with a failed pleurodesis are candidates for symptom-guided drainage via IPC.
Chemical pleurodesis at the time of diagnostic pleuroscopy (when overt malignant changes are present) is a very reasonable intervention in patients where lung expansion is likely. Similarly, VATS pleurodesis may be offered to a subgroup of surgically fit candidates with a long-anticipated survival.
Conclusion
The presence of an MPE implies advanced disease, and as such clinicians should tailor their expectations of the outcomes of an intervention. Prolongation of life will not be possible, and palliation of symptoms is therefore the primary aim. A therapeutic thoracentesis provides immediate relief in most cases, provided other causes for dyspnea are not present. Therapeutic thoracentesis can be repeated, especially in patients with slow rate of recurrence or a very short anticipated survival. Definitive interventions, individualized according to the patient’s wishes, performance status, prognosis and other practical considerations (including the ability of the underlying lung to expand) should be offered to the remainder of patients with MPE. Chemical pleurodesis (achieved via ICD or pleuroscopy) and IPC have equal impact on patient-based outcomes, although patients treated with IPC spend less time in hospital and fewer pleural reinterventions. Talc slurry via IPC is a new and potentially attractive option for patients who do not have a NEL.
Expert commentary
People afflicted by MPE are a highly diverse group of patients. Yet, despite vast differences in the underlying malignancy and its chemosensitivity, involvement of the underlying lung (particularly its ability to expand), major comorbidities, performance status, anticipated survival and patient’s wishes, MPE is often still managed with a ‘one size fits all’ approach. While no intervention currently prolongs life or even directly decreases fluid formation, several interventions have been shown to impact on QoL, symptom control and the need for inpatient care.
One of the greatest challenges is to provide individualized care by phenotyping patients with MPE. 60 Clinicians have traditionally been poor at predicting prognosis. Even in large RCTs, where enrolment criteria included an expected survival of ⩾3 months, about a quarter of patients did not survive 3 months. 23 The LENT score has fortunately changed this landscape, allowing for more accurate prognostication and thus practical guidance in individual cases. 13 However, the estimation of prognosis may once again become challenging in the era of personalized chemotherapy based on cancer epigenetics. 1
The ideal prediction model for successful pleurodesis is still unknown, but female sex, a higher Karnofsky performance score, a normal fluid pH and a higher adenosine deaminase (ADA) level (>18 IU/l) have been statistically associated with successful pleurodesis, whereas a high pleural fluid C-reactive protein (CRP), a high lactate dehydrogenase (LDH) level and low pH have a been shown to predict failure of pleurodesis.61,62
Another challenge is predicting who will benefit from early pleurodesis or IPC placement, as symptomatic response after therapeutic pleural drainage remains variable. 60 A recent multivariate analysis found that patients with low pleural fluid pH, large effusions, and increasing age were more likely to require pleurodesis or IPC placement for fluid control. 60 The currently ongoing PLEASE study is the first large prospective study to characterize the physiological and symptomatic responses of fluid evacuation, which may represent the first step in the journey towards improved understanding of the mechanisms of dyspnea in MPE. 63
A significant advance in our approach to MPE was shifting the goalposts from ‘radiologically successful’ pleurodesis to patient-centered outcomes, particularly dyspnea. A novel breathlessness score for MPE, using a 100-mm visual analog scale, was recently validated, and subsequently incorporated into several prospective studies. 64 Despite these advances, it should be appreciated that even these endpoints have limitations. The advent of actigraphy has shown promise as an all-encompassing endpoint that incorporates control of breathlessness, pain and deconditioning as well as muscle wasting, mood/depression and comorbidity. A recent study of actigraphy found that MPE patients have very poor daily activity levels, and that measurements of activity (such as step counts and bouts of moderate/vigorous activities) correlated with conventional ECOG scores. 65
Despite the fact that VATS is still commonly performed in the management of MPE, its true efficacy compared with far less invasive procedures is largely unknown and its use chiefly supported by case series from large volume centers. 12 Clearly an RCT in which MPE patients with good prognosis are randomized to VATS or IPC (plus pleurodesis if appropriate) is long overdue.
The recent finding that talc instilled via an IPC in patients with no evidence of NEL at 10 days post-insertion has the potential to revolutionize the minimally invasive management of MPE. 30 Not only did it lead to a clinically and statistically significant improvement in pleurodesis rates without adverse events, but it also to improved QoL scores and symptom control. The implications of these findings are substantial: by facilitating early catheter removal this intervention reduces the inconvenience of regular drainage, risk of infection, mechanical failure and costs of the consumables.
A 5-year view
Pleural medicine, and particularly the management of MPE, will continue to come of age and move from an era of expert opinion (based on ‘collective experience’) and anecdotal reports to one based on high level evidence. The value and safety of existing and recent minimally invasive interventions such as chemical pleurodesis, IPC (with and without pleurodesis) have been rigorously tested by several RCTs over the past few years. Moreover, the long overdue shift from ‘successful pleurodesis’ as an endpoint in RCTs to patient-centered outcomes was finally made.
What still needs refinement is our ability to provide tailored care for patients with MPE, and to accurately guide clinicians and patients with regards to the choice of ICD with pleurodesis and IPC with or without pleurodesis. Greater understanding of the mechanisms of dyspnea and thus the optimal intervention to address symptoms will be investigated, and a shift away from invasive measures such as VATS is anticipated. Furthermore, the subgroup of relatively fit patients who may benefit from VATS (even with decortication or pleurectomy) will become better defined.
Novel sclerosants and drug-eluting IPCs will be investigated during the next 5 years, as well as the optimal manner of combining IPC and pleurodesis with regards to timing and practical considerations. 40 Ancillary therapy such as exercise and dietary interventions may also emerge as beneficial adjuncts to pleural interventions in the holistic approach to the patient with MPE. 1
Future studies may very well include more patient-based outcomes. In addition to visual analog scales for pain and dyspnea and QoL questionnaires, more objective assessments of daily activity by means of actigraphy via a triaxial accelerometer could provide better insight into the true influence on QoL of the various interventions. 65 Supportive therapy such as exercise training, nutritional interventions and psychological support have rarely been investigated, but could hold significant role in MPE care for patients. 66
Ultimately, large multicenter RCTs with various arms, stratified according to patient characteristics and comparing interventions ranging from surgical to minimally invasive methods of achieving pleurodesis or preventing fluid accumulation are needed to attain the ultimate goal of ‘personalized’ management.
Key issues
An intervention for the management of MPE will perforce be palliative in nature, as no pleural procedure has yet been shown to prolong life.
A therapeutic thoracentesis provides immediate relief in most cases, provided other causes for dyspnea are not present.
Therapeutic thoracentesis can be repeated in patients with slow rate of fluid reaccumulation and patients who have a very short anticipated survival or poor performance status.
Definitive interventions, individualized according the patient’s wishes, performance status, prognosis and other practical considerations (including the ability of the underlying lung to expand) should be offered to the remainder of patients with MPE.
Various surgical and nonsurgical options are available, but insufficient evidence exists to guide management in most clinical scenarios.
Chemical pleurodesis (achieved via ICD or pleuroscopy) and IPC have equal impact on patient-based outcomes, although patients treated with IPC do spend less time in hospital.
Talc slurry via IPC is a new and potentially attractive option for patients who do not have a NEL.
Footnotes
Funding
CFNK, JAS and EMI have nothing to declare. YCGL is a NHMRC/MRFF Practitioner Fellow of Australia and has received project grant funding from the NHMRC, iCARE Dust Diseases Authority, Sir Charles Gairdner Research Advisory Committee, Cancer Australia and Cancer Council of Western Australia.
Conflict of interest statement
CFNK, JAS and EMI have nothing to declare. YCGL has served on the advisory board of CareFusion/BD Ltd and has received an unrestricted educational grant from Rocket Ltd. YCGL led the AMPLE-2 study in which participants received drainage kits without charge from Rocket Ltd.
