Abstract
The recent Global Initiative for Chronic Obstructive Lung Disease (GOLD) chronic obstructive pulmonary disease (COPD) guidelines consider symptoms and exacerbation history in addition to the degree of airflow obstruction for classifying patients. The improvement of symptoms is principally provided by bronchodilators, using β2 agonists and antimuscarinic agents. Aclidinium bromide is a novel long-acting antimuscarinic agent licensed for use in patients with COPD. Novel fixed-dose combinations that are either licensed or in their late phase of development include vilanterol/umeclidinium, indacaterol/glycopyrronium, olodaterol/tiotropium and formoterol/aclidinium. Fixed-dose combinations of aclidinium/formoterol have been evaluated in COPD patients and evidence suggests that this is efficacious, safe, has a quick onset of action and is well tolerated. This review provides a clinico-pharmacological profile of this compound.
Introduction
Chronic obstructive pulmonary disease (COPD) is a preventable and treatable disease, characterized by nonreversible airflow limitation as a result of chronic inflammation in response to the inhalation of noxious particles and gases [Global Initiative for Chronic Obstructive Lung Disease, 2013]. At present, COPD is the fourth leading cause of mortality worldwide [World Health Organization, 2008], though it is projected to become the third leading cause of death by the end of the decade [Chapman et al. 2006]. In addition to its progressive nature, COPD also has a major economic impact as a consequence of both direct and indirect costs [National Institute for Health and Care Excellence, 2010; IMS Health LS, 2012].
The main objectives of pharmacotherapy in COPD are to alleviate and control symptoms, enhance health status and exercise capacity, and to minimize the frequency and severity of exacerbations [Global Initiative for Obstructive Lung Disease, 2013]. Guidelines promote the use of long-acting bronchodilators as a first-line maintenance therapy in COPD subjects. The logical progression from monotherapy in patients with persisting symptoms to the concept of dual bronchodilation with bronchodilators of differing modes of action has however been proposed [Cazzola et al. 2011, 2012a]. During 2011 a radical alteration to the guidelines for the Global Initiative for Chronic Obstructive Lung Disease (GOLD) saw a move away from the use of COPD staging by airflow limitation using spirometry, to the use of categorization using subjective symptoms and risk (spirometric airflow limitation as well as exacerbation history); thereby permitting a holistic therapeutic management approach to the patient [Global Initiative for Obstructive Lung Disease, 2013]. The new GOLD guidelines appear to predict COPD exacerbators better than the old system.
In this review we consider the rationale for the use of dual bronchodilators with a particular reference to the combination of aclidinium bromide and formoterol fumarate (aclidinium/formoterol) (Duaklir®, Almirall/Forest/Astra Zeneca, Spain). We discuss the evidence behind the monocomponents of aclidinium and formoterol, the delivery device, Genuair™ (Almirall SA/AstraZeneca, Spain) and, subsequently, preclinical, efficacy and safety data on the combination aclidinium/formoterol from various trials conducted to present day. Relevant information pertaining to the bronchodilators were made available by conducting a detailed literature search of PubMed, abstracts presented at international respiratory conferences (European Respiratory, British and American Thoracic Societies), clinical trials websites and relevant pharmaceutical industry websites.
Long-acting β2 agonists in COPD
Forced expiratory volume in 1 second (FEV1) has served as an important tool to categorize disease severity and to grade the effectiveness of treatment regimens. The control of symptoms using bronchodilators is the mainstay of treatment. Based on the assessment of the GOLD guidelines, the recommended choice of therapy for Group A patients are short-acting agents. Patients in or above Group B require a long-acting bronchodilator; this would involve either a long-acting β2 agonist (LABA) or a long-acting anticholinergic (LAMA).
Formoterol and salmeterol are LABAs that are effective in the treatment of COPD. Differences in their pharmacology exists with formoterol being a full β2 receptor agonist whilst salmeterol a partial agonist. Formoterol furthermore has a rapid onset of action compared with salmeterol. Seberova and Andersson showed that the onset of bronchodilatory effect, as measured by serial FEV1, for formoterol and salbutamol are similar in asthmatics [Seberova and Andersson, 2000]. This study proves that formoterol, though considered to be a LABA, has a faster onset of action similar to the short-acting bronchodilator salbutamol. This difference was thought to be due to differences in their physicochemical properties. Formoterol is water soluble with some lipophilicity that ensures rapid bronchodilation followed by a prolonged effect. On the other hand, salmeterol may diffuse slowly to the β2 receptors located on smooth muscle tissue thus explaining its slower onset of action [Rossi et al. 2008]. Several once-daily LABAs such as olodaterol, vilanterol and abideterol have either been recently approved or are in the late stages of clinical development. These agents are essentially full agonists at β2 receptors and produce dose-dependent rapid bronchodilation that is maintained for more than 24 h [Malerba et al. 2012; Babu and Morjaria, 2014].
Whilst the use of LABAs does not have any effect on the progression of COPD as measured by a rate of decline in FEV1, LABAs do improve lung function [Cope et al. 2013], reduce the rate of exacerbation, improve both health-related quality of life [Man et al. 2003; Sin et al. 2003] and small airway patency with a consequent reduction in forced residual capacity (FRC), and increase inspiratory capacity (IC) [Di Marco et al. 2003; O’Donnell et al. 2004].
LAMAs and aclidinium bromide
The parasympathetic network of the airway wall regulates bronchoconstriction and mucus secretion through muscarinic receptors. There are five different types of muscarinic receptors (M1–5), although only M1, M2 and M3 are expressed in human lungs. M1 receptors facilitate neurotransmission. M2 receptors are expressed on both presynaptic and parasympathetic nerve endings at the neuromuscular junction and on bronchial smooth muscles, thus are involved in either inhibiting or promoting bronchoconstriction depending on their site of stimulation. M3 receptors control bronchomotor tone and mucus secretion and are expressed on bronchial smooth muscle, submucosal glands and vascular endothelium [Sims and Panettieri, 2011], and are therefore the primary target for LAMAs.
Aclidinium bromide [(3R)-3-y-1-(3-phenoxypropyl)-1-azoniabicyclo (2,2,2) octane bromide] (Eklira™, Almirall SA/AstraZeneca, Spain) is a quaternary ammonium compound with a high binding affinity for the M3 receptor. Aclidinium, along with glycopyrronium and tiotropium, binds to the M1–5 receptors in a concentration-dependent manner with a greater selectivity for M3 receptors than for M2 [Prakash et al. 2013]. They also dissociate more slowly from the M3 than they do from the M2 receptors [Gavalda et al. 2009; Vogelmeier et al. 2011a]. The association rate of aclidinium with human M3 receptors is 2.6 times faster than that of tiotropium, whilst glycopyrronium associates 4–5 times faster with M3 receptors than tiotropium or aclidinium [Sykes et al. 2009; Ulrik, 2012]. The more rapid association of LAMA with the M3 receptor is believed to underlie its faster onset of action. The dissociation half-life of aclidinium from the M3 receptor is six times that of the M2 receptor, which will therefore provide bronchodilation via M3 receptors long after the less-desirable M2 effects such as tachycardia diminish. The quaternary nature of the compound also imparts a low oral bioavailability and a low blood–brain barrier permeability.
During early phase I studies, aclidinium exhibited linear and time-dependent pharmacokinetics with a steady state achieved within a 7-day treatment period compared with tiotropium which required 2–3 weeks to reach a steady state [Reid and Carlson, 2014]. Aclidinium is rapidly converted into inactive alcohol and carboxylic acid with a plasma half-life of 2.2 min compared with more than 60 min for both ipratropium and tiotropium. Furthermore, the metabolites of aclidinium do not have any affinity for muscarinic receptors. As aclidinium undergoes rapid hydrolysis to its inactive metabolites in the plasma, very little is excreted within the urine and renal dysfunction does not appear to have an impact on systemic exposure [Schmid et al. 2010].
Rationale for dual bronchodilation
The dose–response curve for LABA and LAMA demonstrates a linear response at lower concentrations followed by a flatter curve at higher concentrations [Ball et al. 1991; Casarosa et al. 2009]. This indicates only a marginal improvement in benefit with a greater side effect profile when increasing the dose of these medications. Interestingly, this attribute is also shared by other inhaled medications. Formoterol is a LABA with a faster onset of action and a relatively linear dose–response curve [Cazzola et al. 2012a]. Physicians have been using the concept of dual bronchodilation with short-acting β2 agonists and antimuscarinic agents in the form of a Combivent® inhaler and nebulizer solutions. Studies combining LABAs and LAMAs using separate devices have demonstrated an additive effect but the benefit achieved has varied greatly between studies [Van Noord et al. 2006; Tashkin et al. 2008; Hanania et al. 2009]. Despite this, these studies do support the rationale of adding bronchodilators with different mechanisms of action.
When one considers the primary outcome variable of symptoms and health-related quality of life, LABAs are more effective than LAMAs, while LAMAs with a favourable impact on the above endpoints appear to be more effective in reducing exacerbations [Vogelmeier et al. 2011b; Rodrigo and Neffen, 2012; Cope et al. 2013]. Combining these bronchodilators can thus have a favourable response effect. The symptoms of COPD vary throughout the day with symptoms being at their worst in the mornings. Hence, a morning dose of formoterol with its faster onset of action can help with improving these symptoms [Roche et al. 2013]. Adherence to medication is a concern if the medication does not relieve symptoms promptly. The addition of formoterol as a LABA would therefore provide reassurance to the patient that the combination is acting effectively. Currently three LABA/LAMA combinations have been licensed: glycopyrronium bromide/indacaterol malaete (Ultibro®, Novartis, Switzerland), umeclidinium bromide/vilantarol trifenatate (Anoro®, GlaxoSmithKline, UK) and more recently aclidinium bromide/formoterol fumarate (Duaklir®, Almirall SA/AstraZeneca, Spain). Another combination, tiotropium bromide/olodaterol (Boehringer Ingelheim, Germany) is currently undergoing phase III assessment.
Delivery devices
A number of inhalation devices are available for the delivery of medications to manage COPD. These include the pressurized metered dose inhalers (pMDIs) [Lavorini et al. 2011] and the increasingly popular breath-actuated dry powder inhalers (DPIs) [Nokhodchi and Larhrib, 2013]. Without a spacer device, pMDIs may pose a number of challenges for use compared with DPIs. These challenges include the lack of synchronized inhalation and actuation and attenuated oropharyngeal deposition in addition to drug delivery and potential resultant cost implications [Lavorini et al. 2008; Lavorini, 2013]. Despite numerous studies reporting excessive errors with the use of pMDIs, it has been suggested that this may also be the case with DPIs [Lavorini et al. 2008]. Critical errors in inhaler use may not only compromise treatment efficacy but may also compromise patient satisfaction, which is associated with improved inhaler concordance, resulting in improved clinical outcomes and reduced healthcare costs [Melani et al. 2011; Makela et al. 2013]. Thus, there is a need to develop easy-to-use inhalers that require minimal patient handling to ensure successful drug delivery which will ultimately enhance patient adherence and management of their underlying COPD.
Genuair® (Almirall SA/Astra Zeneca, Spain) is a novel breath-actuated multidose DPI delivery inhaler that has been developed to achieve a reliable and effective delivery of inhaled licensed aclidinium and more recently aclidinium and formoterol (Duaklir™, Almirall SA/Astra Zeneca, Spain) for the management of COPD. The Genuair device, which resembles the Novolizer® (MEDA Pharma GmbH, Germany), has a nonremovable powder cartridge and is a fully assembled ready to use inhaler that requires minimal patient handling and hence reduces the potential for error. The cyclone dispersion system and the Genuair mouthpiece deagglomerate the medication into small particles for a complete dose delivery within 2 l of inhalation as per recommendations [Chrystyn and Niederlaender, 2012]. There are two main steps for inhalation using the Genuair; pressing and releasing the blue button to activate the inhaler and inhaling strongly and deeply. The inhaler device has a number of feedback mechanisms for effective use including a control window that changes from red to green when the dose has been dispensed into the powder delivery channel and the dose is ready to be inhaled, an audible click sound when the patient inhales with the minimum inspiratory flow rate required, and following successful inhalation, the change of the window from green back to red [Magnussen et al. 2009; Newman et al. 2009; Chrystyn and Niederlaender, 2012]. For appropriate dosing, there is also a trigger threshold that prevents the release of a second dose into the powder delivery inhalation channel until complete inhalation has occurred in order to avoid double dosing. Importantly, on hearing the click the patient must continue to inspire, remove the inhaler from the mouth and hold their breath for as long as is comfortable before exhaling through their nose. The trigger flow rate is approximately 40 l/min; however, a constant fine particle fraction is achieved at flow rates above 35 l/min [Der Palen, 2014]. Moreover, the Genuair inhaler has a dose indicator with a warning line when only a few doses remain, in addition to having a lock-out mechanism which prevents use of the device after the final dose has been dispensed [Chrystyn and Niederlaender, 2012].
When choosing a new DPI for COPD patients, it is important to consider whether they can generate sufficient inspiratory flow in order to disaggregate the medication; the peak inspiratory flow (PIF) being inversely proportional to the intrinsic resistance of the device [Magnussen et al. 2009]. The PIF of the Genuair versus the Handihaler® (Boehringher Ingelheim, Germany) device has been investigated in an open-labelled randomized crossover study using 48 patients with moderate to severe COPD [Magnussen et al. 2009]. The report indicated the mean PIF achieved for the Genuair to be significantly higher than that with the Handihaler, and 97.2% of inhalation through the Genuair to be successful and optimal. More recently an open-labelled randomized crossover study of 130 patients with moderate to severe COPD was conducted to assess patient preference and willingness to continue with either the Genuair or Handihaler fitted with a placebo [Van Der Palen et al. 2013]. Not only was there a significant overall (79.1% versus 20.9%, respectively) and individual domain preference for the Genuair device, but also patient satisfaction and willingness to continue using the Genuair was significantly higher based on the fact that markedly fewer patients had critical errors with its use (2.9% versus 19%, respectively) compared with the Handihaler. In a similar double-blind, placebo-controlled study of 414 patients, a comparable preferential and willingness to continue with the Genuair compared with the Handihaler was reported [Beier et al. 2013]. More recently, an in vitro study comparing four different inhaler devices [Genuair; Repsimat®, Boehringher Ingelheim, Germany; Breezhaler, Novartis, Switzerland; and Handihaler], noted that the delivered dose was of the same order of magnitude for the former three devices (>89% of the label claim) but considerably lower for the Handihaler [Gjaltema et al. 2013].
Aclidinium bromide/formoterol fumarate (Duaklir®)
The individual characteristics of both aclidinium and formoterol represent appropriate components for a fixed-dose combination (FDC), twice-daily LABA/LAMA; although preclinical data of the interaction between these two bronchodilators is limited. In this section we discuss a preclinical and phase I study, phase II studies and subsequently phase III studies that have eventually led to the licensing of this FDC.
Preclinical and phase I studies
Cazzola and colleagues recently evaluated the synergistic effects between aclidinium and formoterol on the relaxation of human airway smooth muscle (ASM) [Cazzola et al. 2014]. Both aclidinium and formoterol relaxed precontracted segmental bronchi with acetylcholine, although only the latter eliminated acetylcholine-induced contraction. Importantly, at low concentrations of aclidinium and formoterol, a synergistic relaxant interaction was noted on prestimulated bronchial isolates therefore supporting the use of these therapies in combination. In an open-labelled, three-way crossover (phase I) study of 30 healthy subjects to assess the safety, tolerability and pharmacokinetics of FDC aclidinium/formoterol (400/12 µg), aclidinium (400 µg) and formoterol (12 µg) via the Genuair device, it was noted that there were no safety or tolerability issues and that systemic exposure was not significantly altered when administered together [Committee for Medicinal Products for Human Use, 2014].
Phase II studies
There are a number of phase II studies assessing the efficacy, safety, tolerability and appropriate dosing of the FDC of aclidinium/formoterol [Sliwinski, 2010; Magnussen, 2011; Cazzola et al. 2012b; Kerwin, 2013; Fogarty, 2014]. One of these was conducted to identify an appropriate dose using 566 moderate to severe COPD patients. During the study three doses of formoterol (6, 12 and 18 µg) combined with aclidinium (200 µg) was compared with aclidinium (200 µg) and formoterol (12 µg) monotherapy, and a placebo administered once daily using the Genuair device [Sliwinski, 2010]. All of the FDCs were significantly superior to the placebo and to both monotherapies (except for FDC aclidinium/formoterol 6 µg versus formoterol 12 µg). Importantly, the bronchodilation provided by the FDC using 18 µg of formoterol was comparable with that using 12 µg thus suggesting the optimal dose to be the latter. Another evaluator-blinded four-way crossover study using 24 moderate to severe COPD patients, compared a one-day treatment of formoterol (12 µg) delivered once daily via two different DPIs (Aerolizer® and Genuair) with FDC of aclidinium/formoterol (200/12 µg) via Genuair [Magnussen, 2011]. Not only were all of the treatments safe and well tolerated with improved lung function, but the absorption of formoterol was also comparable when delivered as a monotherapy or FDC. Fogarty and Ortiz have reported from an open-labelled pharmacokinetic study using aclidinium/formoterol (400/12 µg) via Genuair and formoterol (12 µg) via Aerolizer in 24 moderate to severe COPD patients. During the study both aclidinium and formoterol achieved a steady state after 5 days administration, irrespective of the inhaler type for the latter [Fogarty and Ortiz, 2014]. In a dose-ranging, double-blind, incomplete, crossover study conducted over 14-day blocks using FDC of aclidinium/formoterol (400/12 µg and 400/6 µg), aclidinium (400 µg), formoterol (12 µg) and a placebo, all active treatments demonstrated significant improvements on day 14 FEV1 12 h area under the curve (AUC0–12; the primary endpoint) and all were well tolerated with no safety concerns. Moreover, there were significant improvements in other FEV1 assessments in favour of the FDCs compared with the monotherapies, and the monotherapies compared with the placebo [Kerwin et al. 2013]. There is another exploratory phase II study for which results are not currently available [ClinicalTrials.gov identifier: NCT00706914; Cazzola et al. 2012b].
Phase III studies
To date there have been two randomized, double-blind, placebo-controlled studies in various centres worldwide, evaluating the efficacy, safety and tolerability of two FDCs of aclidinium/formoterol (400/12 µg and 400/6 µg twice daily) versus aclidinium (400 µg) twice daily, formoterol (12 µg) twice daily and a placebo administered via the Genuair device over a 24-week period in moderate to severe stable COPD patients (AUGMENT (ITT, n = 1668) and ACLIFORM-COPD (ITT, n = 1726)) [D’Urzo et al. 2014a; Singh et al. 2014]. Both studies had the same primary outcome measures of morning predose (trough) FEV1 and morning 1h postdose FEV1 at week 24. The predefined coprimary endpoints in both the AUGMENT and ACLIFORM-COPD studies, reflect the Food and Drug Administration (FDA) guidelines regarding FDC drugs with each component of the FDC making a contribution to the claimed effects; that is a rapid onset bronchodilation with formoterol (change from baseline in FEV1 at 1 h post morning dose) [Benhamou et al. 2001; Tashkin and Ferguson, 2013] and 24 h bronchodilation with twice daily aclidinium (change from baseline in trough FEV1) [Jones et al. 2012; Kerwin et al. 2012]. In the AUGMENT study, both the FDCs (400/12 µg and 400/6 µg) showed significantly greater 1 h post-dose FEV1 compared with aclidinium monotherapy (108 and 87 ml, respectively, p < 0.0001) as well as trough FEV1 in patients treated with 400/12 µg FDC versus formoterol monotherapy (45 ml; p = 0.0102) and numerical improvement of 26 ml in favour of 400/6 µg FDC (p = 0.133) at 24 weeks [D’urzo et al. 2014a]. Similar significant improvements were noted in the ACLIFORM–COPD study at 24 weeks in 1 h post-dose FEV1 (400/12 µg and 400/6 µg versus aclidinium, 125 and 69 ml, respectively, p < 0.001 both) and trough FEV1 (400/12 µg and 400/6 µg versus formoterol, 85 and 53 ml, respectively, p < 0.01) [Singh et al. 2014]. Other secondary endpoints in both studies included the transitional dyspnoea index (TDI) focal score, a change from baseline in St George’s Respiratory Questionnaire (SGRQ), peak FEV1, FEV1 during 3 h post-dose, TDI and SGRQ responders [percentage of patients achieving the minimum clinically important difference (MCID) in TDI (⩾1 unit increase) and SGRQ (⩾4 units)]; changes from baseline in COPD symptoms [total exacerbations of chronic obstructive pulmonary disease tool (EXACT)–respiratory symptoms (E-RS) score and night time and early morning symptom scores]; rate of COPD exacerbations [healthcare resource utilisation (HCRU) and EXACT (only in ACLIFORM-COPD study)]; and change from baseline in relief medication use. These have been summarized in Tables 1–3.
Changes in peak and 3 h FEV1 from baseline at day 1 and week 24 in the AUGMENT and ACLIFORM-COPD studies.

FDC, fixed-dose combination; FEV1, forced expiratory volume in 1 second; LSM, least squares means
Change compared with placebo
-¥Actual numerical data not available
p < 0.05 FDC versus aclidinium; ΨΨ p < 0.01FDC versus aclidinium; ΨΨΨ p ⩽ 0.001FDC versus aclidinium.
p < 0.05 FDC versus formoterol; ΩΩ p < 0.01 FDC versus formoterol; ΩΩΩ p < 0.001 FDC versus formoterol.
Changes in subjective parameters in the AUGMENT and ACLIFORM-COPD studies.

E-RS, Exacerbations of Chronic Obstructive Pulmonary Disease Tool (EXACT) Respiratory Symptoms (11 items of the 14-item EXACT); FDC, fixed-dose combination; LSM, least squares means; NS, not significant; SGRQ, St George’s Respiratory Questionnaire; TDI, transitional dyspnoea index.
Actual numerical not data available.
TDI responders defined as patients who had a minimal clinical important difference (MCID) ⩾1 unit improvement in TDI focal score at week 24 versus placebo.
SGRQ responders defined as patients who had a minimal clinical important difference (MCID) ⩾4 units reduction in the SGRQ score at week 24 versus placebo.
p < 0.05 FDC versus aclidinium; ΨΨ p < 0.01 FDC versus aclidinium.
p < 0.05 FDC versus formoterol; ΩΩ p < 0.01 FDC versus formoterol .
Exacerbation frequency over 24 weeks in the ACLIFORM-COPD study.
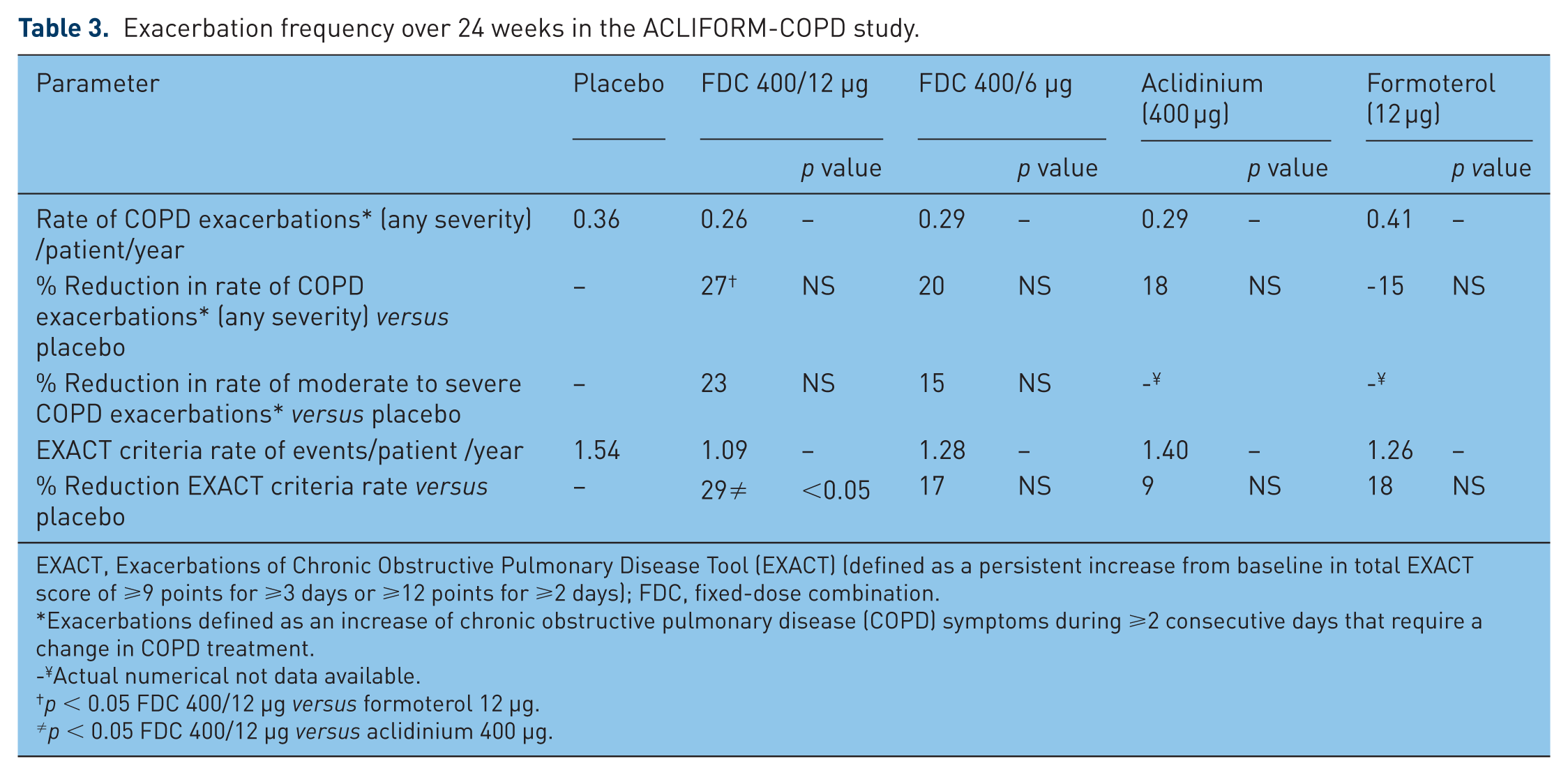
EXACT, Exacerbations of Chronic Obstructive Pulmonary Disease Tool (EXACT) (defined as a persistent increase from baseline in total EXACT score of ⩾9 points for ⩾3 days or ⩾12 points for ⩾2 days); FDC, fixed-dose combination.
Exacerbations defined as an increase of chronic obstructive pulmonary disease (COPD) symptoms during ⩾2 consecutive days that require a change in COPD treatment.
Actual numerical not data available.
p < 0.05 FDC 400/12 µg versus formoterol 12 µg.
p < 0.05 FDC 400/12 µg versus aclidinium 400 µg.
More recently at an international conference, a 28 week extension study [ClinicalTrials.gov identifier: NCT01572792] of the 24-week AUGMENT study compared the FDC aclidinium/formoterol (400/12 µg), FDC aclidinium/formoterol (400/6 µg), aclidinium (400 µg), formoterol (12 µg) and a placebo administered twice daily in 1240 moderate to severe stable COPD patients [D’urzo et al. 2014b]. As with the primary study, the 28-week extension reported that treatment with FDC at 52 week resulted in significant and sustained improvements in lung function and dyspnoea compared with the placebo; with numerical improvements in favour of FDC aclidinium/formoterol 400/12 µg versus 400/6 µg. In a pooled analyses of the ACLIFORM-COPD and AUGMENT studies on the efficacy of FDC aclidinium/formoterol (400/12 µg) on exacerbations in moderate to severe COPD patients it was reported that the FDC attenuated the rate of moderate to severe COPD (29%) and EXACT (22%) exacerbations [Bateman et al. 2014].
Safety and tolerability
In both the AUGMENT and ACLIFORM-COPD studies, there were more premature withdrawals in the placebo group (30% and 17.5%, respectively) [D’Urzo et al. 2014a] compared with the active treatment arms (18–21% and 8.8–13%, respectively) [Singh et al. 2014]. Insufficient therapeutic responses and patient requests to withdraw from the study in the placebo group were more common compared with the active treatment arms. The frequency of treatment-emergent adverse events (TEAEs) and withdrawals from the two phase III studies was comparable across all treatment groups; of which the former were mainly of mild or moderate intensity and unrelated to the treatment, and for the latter dyspnoea [D’Urzo et al. 2014a; Singh et al. 2014]. In the AUGMENT study, irrespective of treatment administered, headaches, nasopharyngitis and urinary tract infections were the most common TEAEs [D’Urzo et al. 2014b] while that in the ACLIFORM-COPD study were COPD exacerbations, headaches and nasopharyngitis [Singh et al. 2014].
Serious TEAEs in both the studies, overall ranged between 3.1% and 5.7% with the most common serious adverse event (SAE) being pneumonia in the AUGMENT study and COPD exacerbations in the ACLIFORM-COPD study [D’Urzo et al. 2014a; Singh et al. 2014]. Similarly there were five and four fatalities, respectively, which were considered unrelated to the study medication. The number of major adverse cardiac events (MACEs), whether TEAE or SAE, were low and similar across all study arms and were thought to be unrelated to the treatment [D’Urzo et al. 2014a; Singh et al. 2014]. Moreover, there was no evidence of the dose-related increase in TEAEs, serious TEAEs, SAEs or MACEs with FDC treatment and the overall safety profiles of both doses were similar to those of the individual monotherapies.
Overall, in both phase III studies there were no major clinically relevant changes from baseline in clinical laboratory parameters, vital signs or electrocardiograms. Holter monitoring did not show any ECG effects in any active treatment arms [D’urzo et al. 2014b; Singh et al. 2014].
Of late, another phase III 52-week study [ClinicalTrials.gov identifier: NCT01437540] evaluating the long-term safety and efficacy of FDC aclidinium/formoterol (400/12 µg twice daily) compared with formoterol (12 µg twice daily) in 2:1 randomization in 590 moderate to severe stable COPD patients [Make et al. 2014]. As with the ACLIFORM-COPD [Singh et al. 2014b], AUGMENT [D’Urzo et al. 2014a] and the 28-week extension of the AUGMENT [D’urzo et al. 2014b] studies, significant improvements in lung function with FDC aclidinium/formoterol (400/12 µg) versus formoterol (12 µg) were maintained over 1 year [Make et al. 2014]. Moreover, FDC aclidinium/formoterol (400/12 µg) was well tolerated and had a safety profile similar to formoterol (12 µg) alone. The safety, tolerability and efficacy of aclidinium/formoterol (400/12 µg) observations from the two 52-week studies, substantiate its use as a long-term therapeutic option for patients with moderate to severe COPD [D’Urzo et al. 2014b; Make et al. 2014].
In pooled analyses from three phase III studies [D’Urzo et al. 2014a, 2014b; Singh et al. 2014], both doses of FDC aclidinium/formoterol taken twice daily for up to 1 year had similar MACE, cardiovascular and cerebrovascular safety profiles to placebo and monotherapies [Donohue et al. 2014]. Moreover, there were no clinically relevant differences noted in ECG and Holter assessments.
Clinical perspectives
Current international recommendations propose the combined use of LABAs and LAMAs to maximize bronchodilation in COPD patients not controlled on a single bronchodilator [Global Initiative for Obstructive Lung Disease, 2013]. Prior to the invention of single-device dual bronchodilators, some patients were managed using single agents delivered in different inhalers, although this was uncommon, probably due to the inconvenience of handling two separate devices [Bourbeau and Bartlett, 2008; IMS Health LS, 2012]. The perspectives of physicians and payers regarding these newly licensed dual bronchodilators are awaited, although it would be a logical and appropriate choice for avoiding multiple drugs at high doses and hence attenuating adverse effects, improving concordance and maintaining simple treatment regimens for patients [National Institutes of Health, National Heart, Lung, and Blood Institute, 2012; Cazzola et al. 2013a]. Furthermore, it has been retrospectively reported that there may be pharmacoeconomic benefits of using a LAMA and LABA compared with monotherapy for managing stable COPD patients; in fact there was a suggestion that dual bronchodilator use was associated with lower healthcare-related costs compared with the use of triple therapy (LAMA, LABA and inhaled steroids) [Kozma et al. 2011]. These findings would advocate the rapid development of FDC dual bronchodilator therapy, especially on the basis of improved lung function and safety and tolerance in patients with moderate to severe COPD that has been observed with this class of therapy [Malerba et al. 2012; Prakash et al. 2015].
With the availability of once-daily and twice-daily LABAs and LAMAs, an important question is whether to choose a once-daily or a twice-daily administration of medication for patients with COPD. There is marked modulation of airway calibre over a 24-h period with short-acting bronchodilators. With extended duration of action, the 24-h profile of bronchial tone smoothens out leading to a net increase of AUC0-24 for FEV1 (and additional lung function parameters such as forced vital capacity), and second to an increase of morning FEV1 after the last respective inhalation (trough FEV1) versus twice-daily and short-acting drugs [Beeh and Beier, 2010]. However, Wu and colleagues used a pharmacokinetic–pharmacodynamic model of longitudinal FEV1 response to an inhaled LAMA in COPD patients to show that with the same twice-daily regimen one can achieve a better trough FEV1, maximal response and AUC0-24 [Wu et al. 2011]. There are no large-scale comparative studies that have looked at once-daily and twice-daily bronchodilators. However, a recent study showed that aclidinium provided improvements in early morning and night time symptoms that were greater than those observed with once-daily tiotropium [Beier et al. 2013]. Whilst the jury is out, twice-daily dosing should be considered a useful approach for COPD patients with nocturnal and early morning symptoms. The use of FDC circumvents an important problem of nonadherence to some medications. Though the adherence to a medication is multifactorial, tedious frequent dosing and the use of multiple inhalers do play an important role [Bourbeau and Bartlett, 2008]. Novel approaches such as bifunctional muscarinic β2 agonists (MABAs) combine muscarinic antagonism and β2 agonism in a single molecule [Cazzola et al. 2013b] which bypasses the potential problem of formulating different drugs in one inhaler and provides a simplified formulation and pharmacokinetics compared with individual combination therapy.
Conclusion
Bronchodilators are the mainstay of treatment for COPD and outcome data from FDC LABA/LAMA combinations have shown greater improvements in lung function parameters, dyspnoea, symptom scores, rescue medication use and quality of life than monocomponent long-acting treatments. This is also supported by the GOLD COPD guidelines for patients in group C (patients with few symptoms but with a high risk of exacerbations) and patients with severe breathlessness in Group B (patients with significant symptoms with a low risk of exacerbations) [Global Initiative for Obstructive Lung Disease, 2013].
Aclidinium bromide is a safe and well-tolerated anticholinergic with a rapid onset of action. It has a pharmacokinetic selectivity for M3 over M2 muscarinic receptors. Moreover, its rapid plasma hydrolysis into inactive metabolites should provide a good safety profile, particularly in patients with renal impairment. The combination of aclidinium with formoterol provides a simplified means of achieving bronchodilation for patients with COPD and the clinical studies show that this is efficacious with a good tolerability and side effect profile in the novel Genuair device. Patients with nocturnal symptoms and symptoms on awakening are better controlled with twice-daily dosing and the FDC of aclidinium/formoterol fits this bracket aptly. Clinical data do support that the addition of formoterol to aclidinium, results in greater bronchodilation and improvements in breathlessness and health related quality of life than formoterol or aclidinium monotherapies [Cazzola et al. 2013c]. With the availability of various LABA/LAMA FDC combinations in clinical practice further studies to address the superiority of one combination over another and the role of once or twice daily dosing will need addressing.
Footnotes
Acknowledgements
DHC Ltd assisted JBM and KSB to write a first draft of the manuscript and provided subsequent editorial support as directed by the authors. Almirall SA/AstraZeneca had no involvement in the writing or reviewing of the manuscript at any point.
Conflict of interest statement
KSB received honoraria and financial support from Wyeth, Chiesi, GSK, Teva, Novartis and AstraZeneca for speaking and to attend meetings. JBM received honoraria and financial support from Wyeth, Chiesi, Pfizer, MSD, Boehringer Ingelheim, Teva, GSK/Allen & Hanburys, Napp, Almirall, AstraZeneca and Novartis for speaking and to attend meetings/advisory boards.
Funding
This work was supported by a medical eduction grant from Almirall SA/AstraZeneca.
