Abstract
This study investigates the expression of RIG-I-like receptors (RLRs) – RIG-I, MDA5, and LGP2 – in Lagovirus europaeus/GI.2 infections causing Rabbit Hemorrhagic Disease. Despite over 30 years of study, the acute pathogenesis remains incompletely understood. Lack of in vitro culture systems hampers vaccine development. The experiment, conducted on European rabbits, reveals a significant increase in RLR expression during infection, particularly in organs associated with severe pathology. The findings suggest a pivotal role for RLRs, especially RIG-I, in the host's defense against Lagovirus europaeus. Further research is needed to unravel the complete molecular pathway and associated factors, offering potential insights for therapeutic interventions.
Introduction
Viral infections with acute characteristics pose significant challenges to public health, especially in regions where they can rapidly spread and cause high morbidity and mortality. 1 Understanding the intricate mechanisms underlying these infections is crucial for the development of effective treatment strategies. While several viruses have been studied extensively in this context, there is a need for research on emerging viral infections that display acute pathogenesis. 2
This study focuses on Lagovirus europaeus, specifically Lagovirus europaeus/GI.2, which cause severe and deadly Rabbit Hemorrhagic Disease (RHD) among rabbits (Oryctolagus cuniculus). RHD was first recorded over 30 years ago, and its pathogenesis remains incompletely understood.3,4 Lagovirus europaeus has a genome consisting of single-stranded RNA with positive polarity, which is approximately 7.4 kb in length. 5 This genome encodes two open reading frames (ORF1 and ORF2), of which ORF1 is responsible for the production of structural and nonstructural proteins, while ORF2 encodes a capsid protein (VP2), essential for virus virulence. 3 The structure of the genome and the presence of regions of high genetic variability are key to the virus’ adaptation and its ability to evade the host immune response. 6 Lagovirus europaeus infections exhibit acute, subacute, and chronic clinical forms, characterized by severe organ damage, intravascular coagulation, and widespread apoptosis of immune cells. 7
Despite the high virulence and rapid spread of Lagovirus europaeus, there is currently no in vitro culture system for these viruses. Consequently, vaccines are based on inactivated virus isolated from infected rabbits, offering limited cross-immunity between the two strains.7,8
This study aims to investigate the expression of RIG-I-like receptors (RLRs), specifically RIG-I, MDA5 and LGP2, in Lagovirus europaeus infections. RLRs are key intracellular sensors of viral RNA and play a crucial role in innate immune responses.9,10 Understanding the role of RLRs in Lagovirus europaeus infections can provide valuable insights into the host-virus interaction and the pathogenesis of these acute infections.
Acute viral infections are characterized by their sudden onset and rapid progression, often resulting in a high mortality rate. 11 Innate immunity, including physical barriers and pattern recognition receptors (PRRs), is the first line of defense against viral invasion. These PRRs, such as RIG-I, MDA5 and LGP2, sense viral components and trigger downstream signaling pathways that lead to the production of interferons and pro-inflammatory cytokines. 12 These molecules, in turn, activate antiviral genes, including interferon-stimulated genes (ISGs), which are crucial for limiting viral replication and spread. 13
Viruses, however, have evolved multiple strategies to evade innate immune surveillance, allowing them to establish successful infections. They can suppress PRRs and their downstream signaling cascades, subvert type I interferon responses, and circumvent host innate immune surveillance. 14 In Lagovirus europaeus infections, understanding the interplay between RLRs and the virus may shed light on the mechanisms of immune escape and pathogenesis in these acute viral infections.
While research on established viral infections has provided valuable insights into viral pathogenesis, studying emerging infections, like Lagovirus europaeus, can contribute to a deeper understanding of the complex relationship between the virus and the host's innate immune system. This study seeks to bridge this knowledge gap and provide a foundation for future research into the pathogenesis of acute viral infections using RHD as a model. Given the lack of in vitro models and limited vaccine efficacy, studying innate immune biomarkers such as RLRs can provide an important foundation for understanding host defense mechanisms against rapidly progressing viral diseases.
Materials and methods
Ethicality of the experiment
Animal experimentation was conducted at the Pomeranian Medical University in Szczecin, Poland with approval from the Local Ethical Committee for Experiments on Animals in Poznań, Poland (Approval No. 35/2022). The rabbits were cared for in compliance with both European Union and national regulations for animal research. The study was carried out in line with the ARRIVE guidelines.
Scheme of the experiment
The study utilized European rabbits (Oryctolagus cuniculus/Crl:KBL (NZW)/0052) that were not vaccinated against Lagovirus europaeus/GI.1 and GI.2. These rabbits were procured from a licensed breeder, with a total of twenty rabbits involved in the experiment. All rabbits were six months old and had body weights within the range of 4.5 kg (±10%). They were randomly divided into two groups: a control group consisting of ten rabbits and an experimental group infected with the Lagovirus europaeus/GI.2 strain, also comprising ten rabbits. In both groups, there was an equal distribution of male and female rabbits.
Upon arrival at the laboratory, the rabbits underwent a three-week adaptation period. Environmental enrichment was provided in the form of hay balls and wooden blocks in their cages. The rabbits were housed under standard laboratory conditions, including a 12-h light/12-h dark cycle, a temperature range of 19 ± 21 °C, humidity maintained at 50 ± 10%, and 15–20 air changes per hour.
The virus strain used in the experiment was sourced from a naturally deceased rabbit in Italy (RHDV2_Ri2017 strain; GenBank: OQ680671). In the experimental group, infection was induced by intramuscular injection of an inoculum containing 2 × 104 hemagglutination units of the virus, determined using a hemagglutination assay in line with the previous results for Lagovirus europaeus infection. The control group received an equal volume (1 mL) of a placebo consisting of phosphate-buffered saline (PBS) via intramuscular injection.
The experiment officially commenced at the time of inoculum administration (marked as 0 h). Subsequently, the health status of all rabbits, including rectal body temperature measurements, was monitored at least twice daily. The experiment concluded when severe symptoms of rabbit hemorrhagic disease appeared, at which point qualified animals were humanely euthanized. Euthanasia was carried out by administering intravenous sodium pentobarbital at 90 mg/kg, followed by sodium pentobarbital at 250 mg/kg to induce cardiac arrest. The blood samples were collected at appropriate time points: 0-h, 12-h, 24-h, 36-h, and 48-h. Postmortem tissue samples were collected from the rabbits and preserved in RNAlater RNA Stabilization Solution (Thermo Fisher Scientific, Waltham, Massachusetts, USA), stored at −80 °C for subsequent analysis.
Quantitative real-time PCR (qRT-PCR)
Total RNA was extracted from frozen livers, lungs, spleens, kidneys, and blood samples using the Total RNA Mini Kit (A&A Biotechnology, Gdańsk, Poland) following the manufacturer's instructions. The quantity and quality of the RNA isolates were assessed using a NanoDrop™ 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, Massachusetts, USA).
To prepare cDNA, reverse transcription of the RNA isolates was carried out using the RevertAid First Strand cDNA Synthesis Kit (Thermo Fisher Scientific, Waltham, Massachusetts, USA) in accordance with the manufacturer's recommendations.
For quantitative real-time PCR, the LightCycler® 480 Instrument II (Roche Diagnostics GmbH, Mannheim, Germany) and the PowerUp™ SYBR™ Green Master Mix (Thermo Fisher Scientific, Waltham, Massachusetts, USA) were employed, following the manufacturer's protocols. In the study, specific primers were used for the genes of interest, including RIG-I, MDA5, LGP2, and the housekeeping gene β-actin. These primers were as follows:
RIG-I: Forward (5′-GGATAATGGCAGGTGCAGAGA-3′) and Reverse (5′-TTTTGGGCCAGTTTTCCTTGT-3′)
15
MDA5: Forward (5′- CTTGGAGAGCAATGCAGAGGCT-3′) and Reverse (5′-AGCTGACACTTCCTTCTGCC-3′) LGP2: Forward (5′- CGGTGCTCAAAGTCCGAAGC-3′) and Reverse (5′- GGTCAGTCCAGCGAGAGGTC-3′) β-actin: Forward (5′- TGGCATCCTGACGCTCAA-3′) and Reverse (5′- TCGTCCCAGTTGGTCACGAT-3′)
In addition, quantitative PCR (qPCR) using specific primers was used to quantify the copy number of RHDV2 (Lagovirus europaeus GI.2) virus. The process followed the method described by Hrynkiewicz et al.
16
All PCR reactions were performed in triplicate (triplicates) to ensure reproducibility of results. A series of dilutions of full-length Lagovirus europaeus GI.2 transcript standards were prepared for the RHDV2 strain, with concentrations ranging from 1 × 108 to 1 × 101 A standard curve was prepared using the LightCycler 480 Instrument II (Roche Diagnostics GmbH). This curve was used for quantitative analysis, enabling the determination of the RHDV2 virus copy number in the test samples.
RHDV2: Forward (5′-TGGAACTTGGCTTGAGTGTTGA-3′) and Reverse (3′-ACAAGCGTGCTTGTGGACGG-5′)
The threshold cycle (Ct) values were normalized using the expression of the β-actin gene, and relative gene expression was calculated using the Pfaffl method. 17
Western blot
Four rabbits in each group were euthanised as described previously. Tissue samples were incubated with T-PER™ Tissue Protein Extraction Reagent (cat.no. 78510, Thermo Scientific™) and a mixture of protease and phosphatase inhibitors (Roche Applied Science, Penzberg, Germany; #05892791001 and #11873580001)), and homogenized for 30 s with maximum speed using Bead Ruptor Elite (cat. no. SKU 19-040E, Omni International) to obtain a homogenous mixture. The samples were then centrifuged for 10 min (12,000 rpm, 4 °C). Protein lysates were transferred to new Eppendorf tubes. Analysis of protein levels in lysates was conducted using Bradford reagent. Proteins were separated using the WES system (WES - Automated Western Blots with Simple Western; ProteinSimple, San Jose, California, USA), with a 12–230 kDa separation module (#SM-W003), and detected using an Anti-Mouse (#DM-002) detection module, according to the manufacturer's instructions. The Total protein module (#DM-TP01, ProteinSimple, San Jose, CA, USA) was used as a loading control. The RIG-I Antibody (D-12) primary antibodies (sc-376845, Santa Cruz Biotechnology) were used in the study 1:50.
Statistical analysis
Statistical analysis was conducted using Tibco Statistica 13.3 (StatSoft, Palo Alto, CA, USA). The parameters were reported as arithmetic means, standard deviations (SD), and standard errors (SE). The normality of the data distributions was assessed using the Shapiro-Wilk Test. Data with a normal distribution were subjected to analysis using Student's t-test, while data that did not exhibit a normal distribution were analyzed using the Mann-Whitney U test.
Results
Clinical results
Postmortem examination was performed to document the typical characteristics of RHD in all the animals. Clinical observations during the experiment revealed that some animals did not exhibit any signs of infection (n = 6). In these cases, death occurred suddenly between 12- and 36-h post-infection, resulting in a 40% survival rate at 36 h post-infection. Survival curve of animals during the experiment has been shown in Figure 1.
For the remaining animals (n = 4), clinical signs of infection became apparent starting at 24 h post-infection. These signs included apathy, dyspnea (difficulty breathing), and conjunctival hyperemia (redness of the conjunctiva). In these cases, humane endpoints were reached between 36- and 48-h post-infection, leading to euthanasis.
Number of virus copies in the examined tissues
To investigate whether the activity of RLR receptors in the examined organs was influenced by the RHDV2 virus, a real-time PCR reaction was performed using primers specific to the mentioned virus to measure the number of virus copies in each tissue. The results of this analysis are presented on a graph (Figure 2), as an average for each tissue. On the basis of the results, it can be assumed that the activity of the tested receptors proofs the infection with L. europaeus virus.

Survival curve of animals during the experiment.

Average values of the number of virus copies in the examined organs, i.e. lungs, liver, spleen, and kidneys.
Number of virus copies in blood samples
The results of number of virus copies in blood samples are presented on Figure 3, also allowed for the identification of the peak moment of virus replication in the body. This study was carried out using the same methods that were used to determine the number of virus copies in tissues - i.e. real time PCR. The results showed a dramatic increase of the virus copies after 12 p.i., being a proof of the active infection with Lagovirus europaeus virus.

Average values of the number of virus copies in collected blood samples.
Expression of RIG-I, MDA5 and LGP2 receptors in the examined tissues
To assess the expression of RLR receptors during Lagovirus europaeus/GI.2 infection, quantitative real-time PCR analysis was performed on the genes encoding these receptors using specific primers (Table 1). For clarity and completeness, both tabular and graphical representations of the data are provided. The tables allow precise comparison of mean values and statistical parameters, while the figures illustrate overall expression trends across tissues. The expression of all three genes was detected, which may suggest the theory that the genetic material of Lagovirus europaeus is recognized by RIG-I and MDA5. The highest expression level of LGP2 is recorded in liver tissues (p ≤ 0.01) (Figure 4), but a high level of activity of this gene is also found in spleen tissues (p ≤ 0.05) (Figure 5) and in the kidneys (p ≤ 0.01) (Figure 6) and lungs (p ≤ 0.03) (Figure 7). RIG-I shows the highest activity in the lungs (p ≤ 0.01) (Figure 7) and kidneys (p ≤ 0.01) (Figure 6). MDA5, as a receptor capable of detecting viral RNA, shows its activity in the spleen (p ≤ 0.03) (Figure 5), kidneys (p ≤ 0.03) (Figure 6), although the highest level was tested in the lungs (p ≤ 0.01) (Figure 7).

Expression levels of RLR receptors in liver in rabbits infected with Lagovirus europaeus/GI.2. Real-time PCR results showed that expression levels of all the genes were higher in the experimental group than in the control group. * p ≤ 0.05 **; p ≤ 0.03; *** p ≤ 0.01.

Expression levels of RLR receptors in spleen in rabbits infected with Lagovirus europaeus/GI.2. Real-time PCR results showed that expression levels of all the genes were higher in the experimental group than in the control group. * p ≤ 0.05 **; p ≤ 0.03; *** p ≤ 0.01.

Expression levels of RLR receptors in kidney in rabbits infected with Lagovirus europaeus/GI.2. Real-time PCR results showed that expression levels of all the genes were higher in the experimental group than in the control group. * p ≤ 0.05; ** p ≤ 0.03; *** p ≤ 0.01.

Expression levels of RLR receptors in lung in rabbits infected with Lagovirus europaeus/GI.2. Real-time PCR results showed that expression levels of all the genes were higher in the experimental group than in the control group. * p ≤ 0.05; ** p ≤ 0.03; *** p ≤ 0.01.
Results of RLR expression in the examined tissues.

CG: control group; EG: experimental group; SD: standard deviation; SE: standard error; * p ≤ 0.05; ** p ≤ 0.03; *** p ≤ 0.01.
Results of western blot analysis
Western blot analysis confirmed the presence of RIG-I protein in the tested tissues. Significantly higher levels of RIG-I protein were observed in samples from the experimental group infected with Lagovirus europaeus/GI.2 compared to the control group, as can be seen in Figure 8. The highest levels of RIG-I protein were observed in the liver and kidney, indicating intense activation of this receptor at the translational level in these organs. These differences were statistically significant (p ≤ 0.01) (Figure 8). The spleen, on the other hand, had lower levels of RIG-I protein compared to the liver and kidney, and at the same time slightly lower than the control group (Figure 8).

Relative levels of cleaved protein. (A) RIG-I activity in selected organs. Protein levels were assessed using Western blot method (the WES system). Panel (A) shows representative blots of cleaved protein. Panels (B) shows a bar chart of RIG-I activity in the liver, kidney and spleen. All values are expressed as mean value ± standard deviation (SD). *p ≤ 0.05; **p ≤ 0.03; ***p ≤ 0.01.
Expression of RLR's receptors in the tested blood
To assess the expression of RLR receptors during Lagovirus europaeus/GI.2 infection, quantitative real-time PCR analysis was performed on the genes encoding these receptors using specific primers (Table 2 and Figures 8–11).

Expression levels of RIG-I receptors in blood samples in rabbits infected with Lagovirus europaeus/GI.2. * p ≤ 0.05; ** p ≤ 0.03; *** p ≤ 0.01.
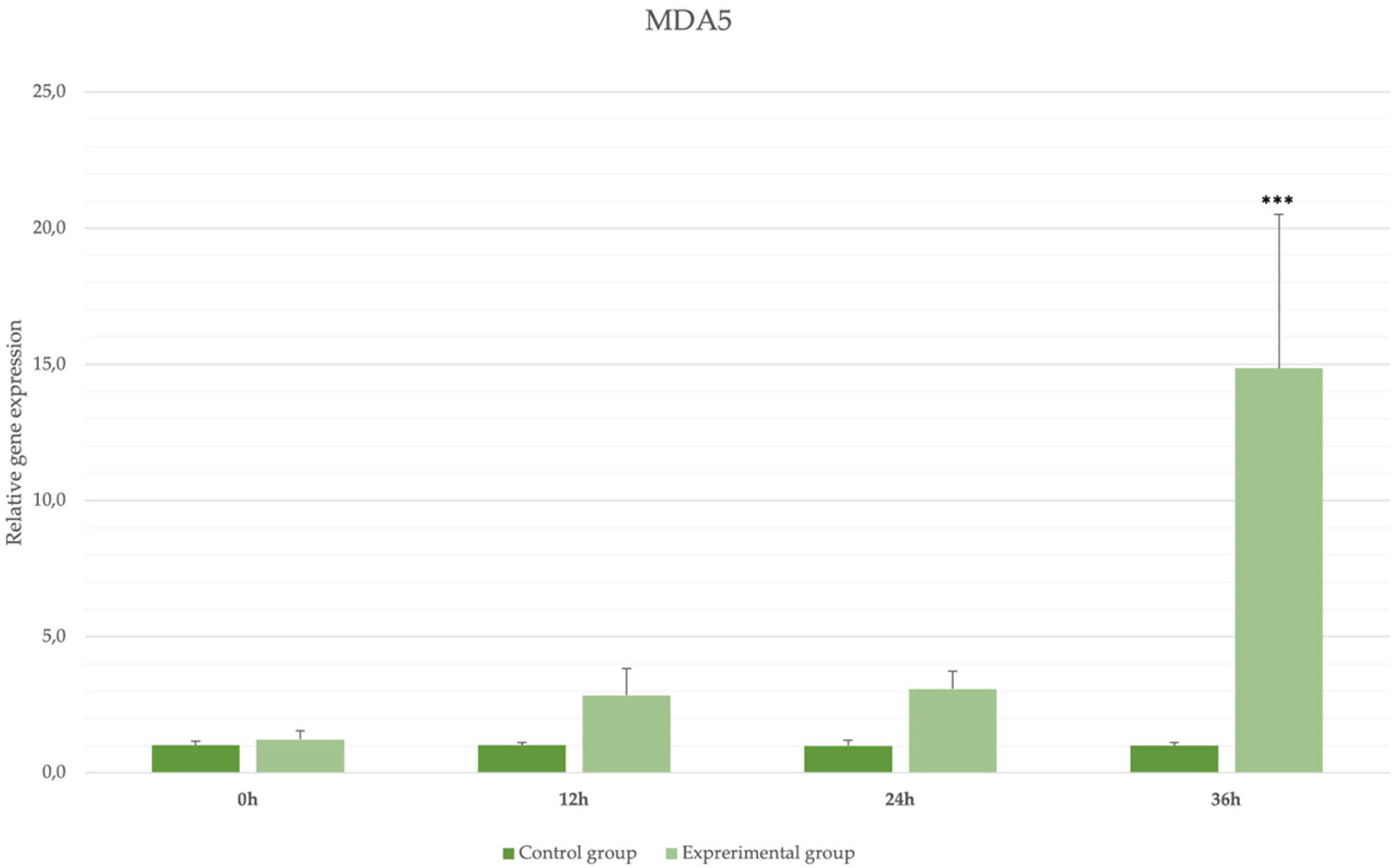
Expression levels of MDA5 receptors in blood samples in rabbits infected with Lagovirus europaeus/GI.2. * p ≤ 0.05; ** p ≤ 0.03; *** p ≤ 0.01.

Expression levels of LGP2 receptors in blood samples in rabbits infected with Lagovirus europaeus/GI.2. * p ≤ 0.05; ** p ≤ 0.03; *** p ≤ 0.01.
Results of RLR expression in blood samples.

CG: control group; EG: experimental group; SD: standard deviation; SE: standard error; * p ≤ 0.05; ** p ≤ 0.03; *** p ≤ 0.01.
Discussion
The study of RLRs, particularly RIG-I, MDA5, and LGP2, in the context of Lagovirus europaeus infection provides important insight into the immune response to acute viral infections. It is unquestionable to know that the expression of these receptors is detectable during infection, because due to their function, their activity must occur, which is not confirmed at this stage of research, however, having data on the number of virus copies both in the examined organs and in the tested samples blood, it is not wrong to hypothesize that this expression correlates with activity. Western blot analysis confirmed the presence of RIG-I protein in the tested tissues, with significantly higher levels observed in the liver and kidney of the experimental group infected with Lagovirus europaeus/GI.2 compared to the control group. These findings are consistent with the results of qPCR, which demonstrated elevated mRNA expression levels of RIG-I in these tissues. Interestingly, the spleen exhibited lower levels of RIG-I protein than the liver and kidney, and slightly lower than the control group, suggesting tissue-specific regulation of RIG-I during infection. The high levels of RIG-I protein in the liver and kidney indicate intense activation of this receptor at the translational level, which may reflect the critical role of these organs in the systemic response to viral replication. These observations align with the hypothesis that RIG-I plays a pivotal role in detecting viral RNA and initiating antiviral responses during Lagovirus europaeus infection. Furthermore, the lower protein levels in the spleen, despite detectable mRNA expression, may suggest post-transcriptional regulation or differential degradation of RIG-I in this tissue. The increase in the activity of RLR receptors in the blood must have occurred because these receptors are dedicated to the nucleic acids of the virus, however, with such basic results it is difficult to discuss what their mutual interaction is in the course of the tested infection, but all three receptors are expressed. Going further, their expression can also be correlated with the multiplication of the virus in the body, i.e. the increase in the number of virus copies correlates with the increase in the expression of receptors.
The mechanisms of action of the receptors and the factors influencing their activation have not been fully described. Jounai et al. states that the activation of RIG-I and MDA5 is influenced by many factors, including autophagy, which is crucial during RHDV2 infection. 18
The results of our study reveal a pronounced increase in the expression of RLRs, with RIG-I demonstrating the highest activity, particularly in liver, lung, and kidney tissues during Lagovirus europaeus/GI.2 infection. These observations are consistent with prior research by Pichlmair et al. 19 and Schlee et al.,20,21 which demonstrated the activation of RIG-I and MDA5 by RNA viruses. Furthermore, the binding of RLRs to the signaling adaptor MAVS and the downstream activation of NF-κB and type I interferon response align with the current findings. 21
It is essential to recognize the vital role played by the innate immune system in the early response to acute viral infections. RLRs are crucial pattern recognition receptors that sense viral components and initiate downstream signaling pathways, ultimately leading to the production of interferons and pro-inflammatory cytokines. The results of this study provide strong evidence that RLRs are pivotal players in the host's defense against Lagovirus europaeus, activating immune responses that limit viral replication and spread.
RIG-I and MDA5 are known to recognize various viral components, such as the 5′-triphosphate moiety and higher-order structures of dsRNA, respectively. As indicated by the study by Loo et al., 22 these receptors are not limited to dsRNA viruses but can also recognize ssRNA viruses that produce dsRNA during their life cycle. This diversity in viral recognition emphasizes the adaptability of the host's immune response. 23
Moreover, the recognition of ssRNA viruses by RIG-I and MDA5 underscores the complex nature of virus-host interactions. Different viruses can produce agonists for both RIG-I and MDA5, 24 while others may selectively activate one of these receptors. For instance, the study cites examples of viruses like Vesicular Stomatitis Virus (VSV), 25 Influenza, 26 Respiratory Syncytial Virus (RSV), 27 Hepatitis C Virus (HCV), 28 and Polio virus, 29 which trigger RIG-I or MDA5 responses. The specific viral triggers for these receptors play a crucial role in determining the nature of the host's immune response. 13
Continuation of our research may confirm the key role of the MAVS signaling adapter in RLR-mediated immune responses during Lagovirus europeaus infection. Interestingly, MAVS has been found to localize to both peroxisomes and mitochondria, leading to the rapid, type I interferon-independent expression of antiviral genes by the former pool of MAVS and the delayed, type I interferon-dependent expression of antiviral genes by the latter pool of MAVS.30,31
Limitations of the study
RLR receptors, as receptors that recognize viral patterns, are unconditionally associated with viral infections, and it is therefore extremely important to thoroughly understand their role and factors influencing their expression, as well as to know their expression level during viral infections. Perhaps the future of virology is to use this knowledge to use their activity to combat or biomarker viral diseases. Lagovirus europaeus infection as an acute infection, at the level of our preliminary research, provides only rudimentary information regarding the activity of these receptors, but to confirm all our hypotheses, further analyzes and more detailed studies are necessary, which will provide a picture of the entire molecular pathway of these receptors during infection and the factors involved. correlating with them. There are many unanswered questions at this pioneering stage of research, which we intend to continue in the future in the hope of obtaining answers. Although the present study provides a first insight into the molecular activation of RLRs during Lagovirus europaeus infection, it is limited by the sample size and the lack of protein quantification for MDA5 and LGP2. Future studies should include comprehensive signaling analyses to fully map the downstream pathways.
Conclusion
In conclusion, this study contributes significantly to our understanding of RLR expression and function in the context of Lagovirus europaeus infections. The findings align with existing literature on RLRs and their role in recognizing viral components, initiating immune responses, and ultimately combating viral infections. This knowledge can inform future research and therapeutic strategies aimed at mitigating the impact of acute viral infections, particularly those caused by Lagovirus europaeus. Overall, the study provides novel molecular evidence for RLR activation in an acute viral infection model in rabbits. Even though these results are preliminary, they lay the groundwork for further mechanistic studies and may aid in developing antiviral strategies or diagnostic markers based on RLR pathway modulation.
Footnotes
Acknowledgments
We would like to thank Patrizia Cavadini (Istituto Zooprofilattico della Lombardia ed Emilia Romagna, Brescia, Italy) for supplying the virus strain used in this study. The datasets used and analysed during the current study are available from the correspondent author upon reasonable request.
Author contributions
Conceptualization, F.L. and P.N.-R.; methodology, F.L.,R.H. and K.P.; software, F.L., R.H. and P.N.-R.; validation, F.L., K.W. and R.H.; formal analysis, F.L., R.H., K.W., K.P. and P.N.-R.; investigation, F.L.; resources, P.N.-R.; data curation, F.L.; writing—original draft preparation, F.L., R.H., K.W., K.P. and P.N.-R.; writing—review and editing, F.L., R.H. and P.N.-R.; visualization, F.L.; supervision, P.N.-R.; project administration, P.N.-R.; funding acquisition, P.N.-R. All authors have read and agreed to the published version of the manuscript.
Funding
Financed by the Minister of Science under the “Regional Excellence Initiative” Program. Agreement No. RID/SP/0045/2024/01.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Institutional review board statement
The animal study protocol was approved by the Local Ethical Committee for Experiments on Animals in Poznań, Poland (Approval No. 35/2022, 25.03.2022).
