Abstract
In Cerro de Pasco (CP), Peru (altitude 4338 m) 24% of men have migraine with aura. We studied 30 men. Twenty CP natives, examined in CP, were rated using a chronic mountain sickness (CMS) score to separate controls (10) from those with CMS (10), a maladaptation syndrome in natives to altitude which includes severe, recurring headache. We collected white cells in CP and, from the same men, within 1 h of arrival in Lima (150 m above sea level). Ten normal US men volunteered white cells for comparison. After RNA extraction we assessed gene expression by reverse transcription-polymerase chain reaction. Low ATP1A1 subunit of the ATPase gene mRNA expression in CP was correlated with headache (P = 0.002), acral paraesthesias (P = 0.004) and CMS score (P < 0.001). ATP1A1 subunit expression was increased in all Andeans in Lima (P < 0.001). There were no differences between Andean controls in Lima and US controls. Manipulation of Na+/K+ATPase could offer relief for migraineurs at sea level.
Keywords
Introduction
The World Health Organization ranks migraine 19th among the top disorders causing world-wide disability (1). Despite widespread interest for many years and intensive recent research, the pathogenesis of this common primary headache remains unknown.
Sea level dwellers venturing to the high mountains may have migraine (2). At altitude these headaches are classified as secondary migraine because of the prevailing hypoxia associated with these episodes (1). In Cerro de Pasco, Peru (CP) (altitude 4338 m), where this study was conducted, 47% of native inhabitants complain of headache. Twenty-four per cent of men have migraine with aura, three times the prevalence among men at sea level, suffering an average of 65 attacks a year. Most of the migraine sufferers have chronic mountain sickness (CMS), a maladaptation to altitude that leads to severe, especially nocturnal, hypoxia. The headaches increase in frequency and severity with advancing age, parallelling the clinical progression of this syndrome (3). Thus in Andeans the frequent migrainous headaches in their native environment are probably also classifiable as secondary migraine. Moreover, in sea level normoxia, all symptoms of CMS, including migraine, disappear.
CP natives have frequent acral paraesthesias. Low Na+, K+ adenosine triphosphatase (ATPase) content in sural nerves may be the cause (4), as low ATPase may generate abnormal impulses in peripheral nerves (5). ATPase has also been implicated in the pathogenesis of familial hemiplegic migraine type 2 (FHM2) at sea level, where impaired function of the enzyme makes the cortex susceptible to cortical spreading depression (CSD), one important antecedent thought to initiate the aura of migraine (6). The headache in a migrainous attack, however, has been attributed to impulses in perivascular nerves of the cerebral vessels, the trigeminal-vascular system (6). Since headache, irrespective of classification, is the result of activation of trigeminal nociceptors, the high Andes offer an opportunity to unravel the pathogenesis of migraine at sea level.
In FHM2, the loss of function of the ATPase has been traced to missense mutations in the ATP1A2 subunit. We report the expression of the ATP1A1 subunit of ATPase in white cells of Andean CMS patients and controls in CP and in the same subjects in Lima. We compared the results with the expression of ATP1A1 in normal US men living at an altitude of 1500 m. Our result points to a role for the ATP1A1 subunit in the genesis of secondary migraine in the Andes. This finding may have implications for the pathogenesis and management of primary migraine at sea level.
Methods
This study was approved by the IRB of the Universidad Peruana Cayetano Heredia, Lima, Peru. We studied 30 men; all gave informed consent. Twenty high-altitude Andeans were examined in CP, then transported to Lima (150 m above sea level) and examined within 1 h after arrival. They were rated using standard CMS scoring based on cardiovascular, neurological and haematological variables; a score of ≤12 is considered normal (Table 1) (7). After rating our 20 natives of CP we determined that 10 were normal and 10 had CMS. Standard procedures were used to obtain venous blood. The white cells were isolated in CP and in Lima, and placed in buffer. The cells were transported frozen on dry ice from CP to Lima. All cells were stored at − 70°C in Lima. RNA was subsequently extracted from the cells in Lima before transport to our laboratory. Ten normal US men who did not have migraine gave white cells for comparison of gene expression. These cells were similarly stored in our laboratory until RNA extraction.
Scoring chronic mountain sickness using symptoms and signs7

≤12 = normal; 12–18 = mild; 19–24 = moderate; > 25 = severe chronic mountain sickness.
RNA isolation
Total RNA was isolated using the QIAamp RNA Mini kit (Qiagen, Bothell, WA, USA). Leucocytes were homogenized using QIAshredder spin columns (Qiagen). RNA was extracted from the lysate by QIAmp spin columns, eluted into sterile RNase-free tubes and stored at 4°C.
Gene expression
Gene expression was assessed by reverse transcription-polymerase chain reaction (RT-PCR) (8). RT-PCR on all samples (from Andeans and US controls) was performed in our US laboratory by T.M. and Y.W. First-strand cDNA was synthesized using Ready Go You-Prime First-Strand Beads (Pharmacia, Piscastaway, NJ, USA) according to the manufacturer's instructions with 1 µg of total cellular RNA and 100 ng of random hexadeoxynucleotide primer (Pharmacia). Globin mRNA and two globin-specific primers supplied with the kit were used for evaluating the performance of the reverse transcription. After synthesis, the reaction mixture was immediately subjected to PCR.
PCR was carried out using the GenAmp PCR System 2400 (Perkin Elmer, Foster City, CA, USA) with the diluted first-strand reaction mixture, 1 U of Taq DNA Polymerase (Gibco BRL, Carlsbad, CA, USA), 0.5 µ
Statistical analysis
Data were analysed using single factor analysis of variance (
Results
The results are given in Tables 1 and 2 and Fig. 1.
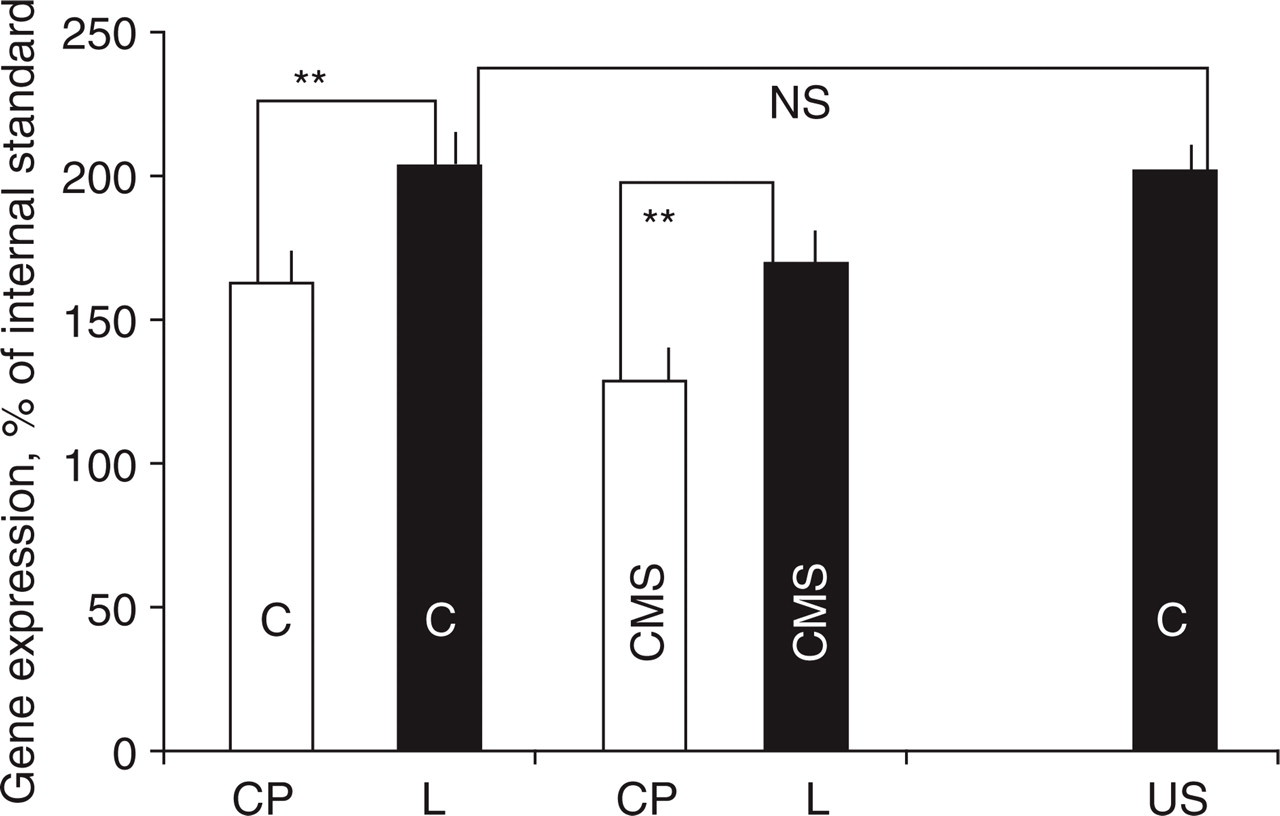
Expression of ATP1A1 subunit of the Na+/K+ATPase in Andeans in Cerro de Pasco (CP) and Lima (L) and in US controls (US). CMS, Chronic mountain sickness; C, Controls. Gene expression is shown as a percentage of internal standard (β2-microglobulin). The increase in expression in Lima in chronic mountain sickness patients (40.4%) vs. controls (41%) was not significantly different but CMS patients never reached the levels of controls at sea level. Mean ± SEM from 10 independent measurements are shown. ∗∗P< 0.001.
CMS patients had significantly lower ATP1A1 subunit of ATPase (ATP1A1sub) expression then Andean controls in CP (P= 0.008). Correlation, using symptoms and signs employed for scoring the severity of CMS with the expression of ATPase, showed low ATP1A1sub expression in CP was correlated with headache (P= 0.002), acral paraesthesias (P= 0.004) and CMS score (P< 0.001). A low ATP1A1sub expression in CP (<135% of internal standard) predicted low oxygen saturation or disturbed sleep (P= 0.003) and a high CMS score (mean of 21.3). Conversely, a higher expression of ATP1A1sub predicted a normal CMS score (mean of 11.4), higher oxygen saturation and no sleep disturbances in CP.
The ATP1A1 subunit of ATPase expression was increased in Lima in all Andeans within 1 h of arrival at sea level (P< 0.001). The absolute increase was similar in CMS and controls. However, patients with CMS had significantly lower levels when compared with CP controls (P= 0.004). There were no significant differences in ATP1A1sub expression between Andean controls in Lima and US controls (Table 2, Fig. 1).
Clinical characteristics of subjects and expression of the ATP1A1 subunit of the Na+/K+ATPase (mean ± SEM)

Gene expression is shown as the percentage ratio between the amount of RT-PCR product (mean optical density of band) of the gene mRNA to the internal standard β2-microglobulin (β2-M) mRNA.
P< 0.001 when compared with Lima.
CMS, Chronic mountain sickness; CP, Cerro de Pasco, Peru altitude 4338 m; L, Lima, Peru altitude 150 m; C, control Andeans; C-US, US controls altitude 1500 m; CMS-sc, chronic mountain sickness score; O2 Sat, oxygen saturation; HTC, haematocrit; –, not applicable.
Discussion
Natives of the high mountains are born, live and die hypoxic, and yet they usually function normally in ambient conditions that render visitors severely disabled. Thus, lessons learned among people living in the high Andes have relevance to medicine and especially to migraine at sea level.
Chronic mountain sickness, a syndrome of maladaptation to chronic hypoxia, is frequently seen in Andean natives who live at high altitudes. It was first described in 1925 by Monge in a miner from Cerro de Pasco, Peru (9), where the syndrome is especially common. All our CMS patients suffered from migraine defined according to International Classification of Headache Disorders (ICHD) criteria, although migraine is not a necessary feature for the diagnosis of the condition.
The oxygen transport laboratory of the Universidad Peruana Cayetano Heredia in Cerro de Pasco, Peru, maintains a roster of CMS patients and controls. We used this list to recruit our subjects. Because women are protected from CMS until after menopause (10), the list contains mainly men. Thus the result of this field study has unavoidable gender bias.
The overall migraine prevalence in western countries is 15–25% in women and 6–8% in men, but this pattern is apparently reversed in the Andes, mainly because of the association of migraine with CMS.
The symptoms of CMS are protean, suggesting dysfunction of the entire body, but when sufferers travel to sea level all symptoms remit within hours as the prevailing normoxia brings oxygen saturation back to normal. Lingering signs of the illness such as dilated veins and florid complexion vanish within about 2 months, when the laboratory hallmark of the disorder, polycythaemia, also disappears. We show here that gene expression in response to the ambient hypoxia of altitude also returns to sea level values within hours of arrival in Lima, parallelling the disappearance of all symptoms (3).
The present study implies that the characteristic high haematocrit of CMS patients plays, at most, a minor role. At sea level, headache is a prominent symptom of polycythaemia vera, a clonal stem cell disorder. The increased blood viscosity due to the increased red cell mass (polycythaemia) may cause headache by causing local hypoxia in the brain, and the headache and other symptoms respond to measures, such as blood letting, that reduce the hyperviscosity of the blood (11). CMS patients at altitude do respond to blood letting – the only treatment for the disorder. However, headache disappears at sea level in all altitude natives, even though haematocrit and presumably blood viscosity remain unchanged (Table 2). Thus, the prevailing hypoxia in Cerro de Pasco, not blood viscosity, is likely to be the major trigger of the migrainous pain.
Secondary headaches are important in unravelling the pathogenesis of primary headaches, such as migraine at sea level, since the immediate cause of head pain, the activation of trigeminal nociceptors, is the same irrespective of headache type. Numerous studies have demonstrated a vasodilator component to the migraineous pain, but the exact role of the cerebral vasculature in the genesis of attacks remains controversial. One idea now gaining proponents is that migraine is a neurovascular syndrome in which the trigeminal innervation of the cerebral vasculature dilates the cerebral vessels during attacks (12).
Both in sea level dwellers with migraine and high-altitude native migraineurs the cerebral vasculature reacts aberrantly to vasodilators. In migraineurs at sea level, cerebral vasodilation in response to increases in pCO2 is exaggerated between attacks (13). Dilator responses to NO donors, such as isosorbate dinitrate, are also exaggerated and longer lasting, especially in patients who develop delayed headache in response to the drug (14). In control and CMS Andeans vasodilation to these physiological vasodilators is diminished or, paradoxically, they induce vasoconstriction at altitude and during a short sea level sojourn (15). This abnormal vascular response may have an important genetic component. A multifactorial genetic ‘load’ is postulated to determine the individual's ‘migraine threshold’, which is modulated by many factors including so-called migraine triggers in the environment (6). In our men the most likely trigger was the ambient hypoxia.
Causative genes have been identified in a rare autosomal form of migraine with aura, FHM. In FHM2, two missense mutations have been found in the ATP1A2 subunit of the Na+/K+ATPase, with consequent loss of function (16). It has been postulated that this loss of enzyme function, especially in astrocytes, where expression of the α2-ATPase isoform is normally very high, could impair K+ clearance from these cells, making the cortex susceptible to CSD. CSD is thought to underlie the aura of migraine (17), with subsequent activation of the trigeminovascular system, which remains the prime suspect in causing the headache.
We made no attempt to identify causative genes responsible for migraine in the high Andes. Rather, we directed our attention to the expression of a normal ATPase subunit. There are at least four ATPase subunits, whose expression varies in different tissues and with age. We first directed our attention to the ATP1A2 subunit of the Na+/K+ATPase because of its relationship to FHM2 at sea level. We found, however, that this subunit was below the detection limit of our RT-PCR. We turned instead to the ATP1A1 subunit of the Na+/K+ATPase, which is abundant in normal white cells, and low levels of this enzyme are associated with the acral paraesthesias of diabetic neuropathy (18) at sea level; in CP, low ATPase content in sural nerves is linked to the prevailing paraesthesias (4). We postulated that low expression of the ATP1A1 subunit of Na+/K+ATPase in the high Andes might also trigger headache by causing trigeminal perivascular nerve depolarization, with consequent initiation of ectopic impulses and pain.
We found abundant evidence that levels of < 135% of the internal standard (β2-M) (8) of the ATP1A1 subunit were correlated with severe hypoxia and sleep disturbances (a symptom of profound oxygen desaturation during sleep). Moreover, such low expression correlated also with headache at altitude. Once CMS patients arrived in Lima, the prevailing normoxia increased ATP1A1 expression to levels above 135% of internal standard, mitigating all symptoms, even though the levels never reached those of Andean or US controls.
Migraine affects more then 10% of the population. Notwithstanding impressive recent progress, drug therapy remains unsatisfactory for many patients. We show here that unravelling the neurobiology of secondary migraine in the high Andes might offer a new therapeutic handle through manipulation of Na+/K+ATPase levels to prevent activation of the trigeminovascular system, which initiates the headache.
However, the analogy between the loss of function of the Na+/K+ATPase in the rare FHM2 at sea level (16), which inspired our study, and lowered expression of a normal protein in our Andean subjects is uncertain. Further studies are needed to determine whether low levels of the normal gene subunit at altitude are equivalent to the genetically disabled gene function, due to missense mutations in FHM2.
Footnotes
Acknowledgements
Funded by New Mexico Health Enhancement and Marathon Clinics Research Foundation with assistance of NIH grant no. IHSEP-02. O.A. was National Science Foundation Exchange Scholar and Fogarty International Exchange Scientist.
