Abstract
The aim of this study was to explore the effects of ethanol extracts from Portulaca oleracea L. (ePO) on joint inflammation and to explain the underlying mechanisms. A joint inflammation mouse model was constructed by injecting zymosan, and the Von Frey method was employed and the joint thickness measured. The numbers of leukocytes, neutrophils, and monocytes were counted in the joint cavity and the infiltration of inflammatory cells was assessed by joint histopathological analysis. The mRNA levels of inflammatory cytokines were determined by quantitative RT-PCR and their secretion levels were determined by specific ELISAs. Pre-treatment with ePO inhibited articular mechanical hyperalgesia and edema and ameliorated the recruitment of mononuclear neutrophils and leukocytes. In addition, pre-treatment with ePO improved pathological alternations in the joint tissues by reducing the number of inflammatory cells. Pre-treatment with ePO regulated the nuclear factor erythroid 2-related factor 2 (Nrf2)-related proteins and thereby inhibited oxidative stress. In addition, ePO inhibited NLR family pyrin domain containing 3 (NLRP3) inflammasome-related genes (NLRP3, ASC, pro-caspase-1 and pro-IL-1ß), modulated inflammatory cytokines and the activation of NF-κB. ePO attenuated zymosan-induced joint inflammation by regulating oxidative stress, NLRP3 inflammasome, and NF-κB.
Keywords
Introduction
Rheumatoid arthritis (RA) is an autoimmune condition that occurs when the immune system attacks the joint tissues.1,2 In addition, the development of RA damages other tissues including the heart, lung, vascular system, and skin. 3 The incidence and prevalence of RA are 0.03% and 1%, respectively, around the world. Every year, 41 out of 10,000 people in the United States are diagnosed with RA. 4 Although RA is widely known as an autoimmune disease, the detailed underlying molecular mechanism is still elusive.
RA is a chronic inflammation process which is associated with an immune response. Immune system cells (T lymphocytes, B lymphocytes, and macrophages) and other types of cells, including osteoclasts and fibroblasts, are involved in the development of RA.5,6 The production of metalloproteinases results in the migration of polymorphonuclear cells, which further cause the erosion of the bone and cartilage. Inflammatory cytokines (IL family cytokines including IL-1 and IL-6 and TNF-α) have important roles in the pathogenesis of RA.7,8 When TNF-α binds to TNF-receptors (TNFRs), the downstream PI3K/Akt/NF-κB pathways are activated, thereby triggering the release of inflammatory cytokines and mediators. The activation of TNFRs also stimulates the release of adhesion molecules, leading to the recruitment of leukocytes to the damaged tissues. 9 The IL-1–IL-1R signaling pathway is another well-known participant in the pathogenesis of RA. 10 Interestingly, numerous studies have revealed that blocking these cytokines is an effective treatment for RA.11,12 For instance, anti-TNF-α therapy is the standard care in RA treatments. However, anti-TNF-α drugs also suppress the immune system, thereby leading to a significantly increased risk of infections. 13 In addition, as the mainstream drugs widely prescribed for RA have many undesirable side effects after prolonged use, 14 many have turned to natural products by complementary and alternative therapies to relieve symptoms of RA and its related ailments.
Portulaca oleracea L. is also called purslane, belongs to the family Portulacaceae, and has a wide distribution. 15 As a fast-growing succulent plant, it has more than 2,000 yr of medicinal use for various diseases and is authorized by the World Health Organization. P. oleracea L. is an edible herb and is widely distributed in Asia, Central Europe, the United States, the Mediterranean, and the Middle East. 16 Purslane is enriched with minerals, omega-3 fatty acids, vitamins and other active ingredients.17,18 Modern pharmacological studies have revealed that purslane has anti-oxidative stress properties, 19 cytotoxic effects on cancers,19,20 anti-bacterial properties, 21 neuroprotective effects, 22 and inflammatory modulation effects. 23 Pre-treatment with P. oleracea L. ethanol extract (ePO) inhibits reactive oxygen species and endothelial cell-derived cytokines (intercellular adhesion molecule-1, E-selectin) in endothelial cells induced by TNF-α. Interestingly, in this study, the modulatory effects of ePO are associated with NF-κB signaling proteins, including p65 and the inhibitor molecule (I-B) alpha. 24 In addition, ePO exerts anti-oxidative stress effects, which are supported by treatment with ePO and increase the antioxidant enzymes (glutathione (GSH), glutathione reductase, glutathione-S-transferase, glutathione peroxidase, catalase and superoxide dismutase) and suppress the secretion of malondialdehyde and NO in rat tissues (liver, kidney, and testis). 25 On the other hand, it is still unknown whether P. oleracea L. exhibits inflammatory modulation effects on joint inflammation.
The current study explored whether ePO exerts therapeutic effects on a joint inflammation mouse model and elucidated its underlying molecular mechanisms. The results demonstrated that pre-treatment with 100 mg/kg ePO suppressed joint inflammation and reduced inflammatory cytokines (IL-6, TNF-α and IL-1β) in the spinal cord. These inflammatory modulation effects were associated with the activation of NF-κB and the NLRP3 inflammasome. In addition to inflammatory modulation effects, ePO also demonstrated antioxidant properties by regulating Nrf2 in the recruited CD45+ cells. Taken together, ePO appears to be a promising agent to joint inflammation treatment.
Materials and methods
Drug preparation
The aerial parts of P. oleracea L. were harvested, dried, and ground into powder. Next, the P. oleracea L. powder (2 kg) was weighted and extracted by 80% ethanol reflux extraction. The supernatant liquid was concentrated in a rotary evaporator at 40°C to produce the ePO.
Animals and drug administration
Male Swiss mice were purchased from the Beijing Protein Biotech Company and were kept under temperatures of 22–24°C with 60 ± 5% humidity. The animals were randomly divided into five groups after 1 wk of acclimation. Animal protocol was approved by the Ethics Committee of the University of South China and a schematic illustration is shown in Figure 1a.
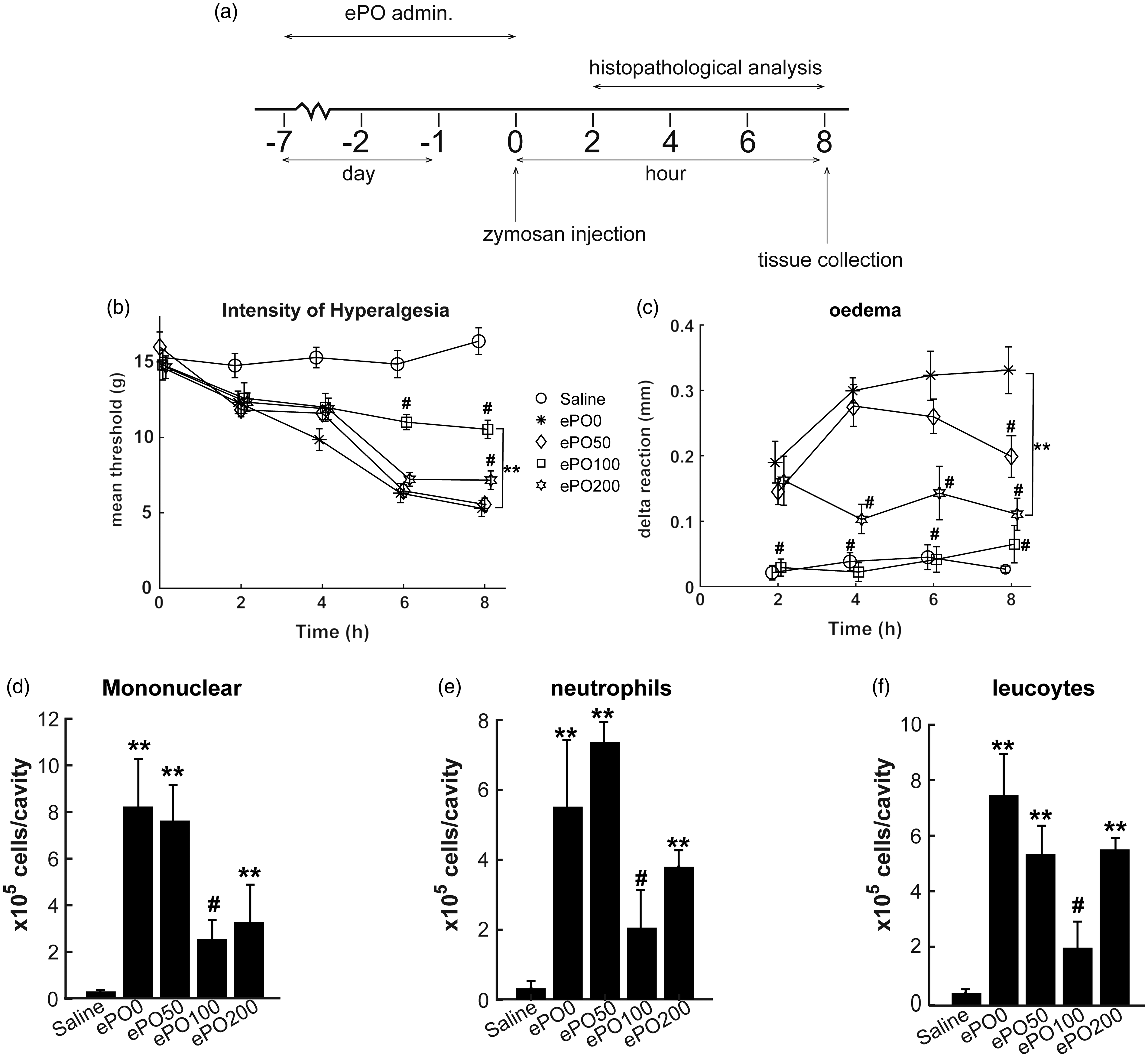
ePO inhibited zymosan-induced articular mechanical hyperalgesia, edema and reduced the numbers of leukocytes. (a) General procedures of model construction and drug administration. The mice were continuously administered with distilled water or P. oleracea L. ethanol extract (ePO) at doses of 50, 100, or 200 mg/kg. Next, the mice were injected with 10% zymosan solution and histopathological analysis was determined at 2, 4, 6, and 8 h. After zymosan injection was performed for 8 h, mice were sacrificed,, and tissue was collected for further assays. (b), (c) Joint mechanical pain allergy and edema were measured at different time points, including 2, 4, 6, and 8 h after zymosan injection. (d)–(f) After zymosan injection for 7 h, numbers of the total mononuclear, neutrophils, and leukocytes were counted in the knee membrane tissues. **P < 0.01, *P < 0.05 versus saline group; #P < 0.05 versus ePO0 group. n = 12 for each group.
The mice were orally gavaged with saline solutions in the saline group. The mice were injected with zymosan in the ePO0 group. The mice were orally gavaged with ePO (dissolved in 0.5 ml distilled water) at doses of 50, 100, or 200 mg/kg per d in the drug administration group in the ePO50, ePO100, and ePO200 groups, respectively. The doses were chosen based on the previous literature.26,27 The drug was continuously administered for 7 d, then a knee joint inflammation model was constructed by injecting 10% zymosan solution into the tibio-tarsal joint. The mice were utilized after 8 h of zymosan injections.
Finally, the tissues were collected and stored at −20°C for further analysis.
Evaluation of articular mechanical hyperalgesia and edema
To evaluate the effects of ePO on the articular mechanical hyperalgesia, the Von Frey method (electronic version) was performed after administration of ePO at 2, 4, 6, and 8 h, according to a previously reported method. In addition, a digital caliper was applied to measure the knee joint thickness. The delta reaction was calculated by the reduction in the diameter measured before the injection from the diameter measured after the injection.
Evaluation of leukocyte recruitment
After the mice were injected with 10% zymosan for 8 h, a sterile saline solution containing EDTA was used to wash the joint cavity. After that, the cell suspension was collected and the numbers of leukocytes, neutrophils, and monocytes were counted.
Histopathological analysis
After the mice were utilized, the joint tissues were harvested. The joint tissues were embedded in paraffin after fixing (10% formalin solution). Histopathological analysis was based on the hematoxylin and eosin (H&E) staining. The infiltration of inflammatory cells was evaluated in each group and histopathological scores were determined. Pixel dimensions of the image size are 279 × 195 in per field.
Immunofluorescent staining
The joint tissues were fixed with 4% paraformaldehyde. After Ag retrieval, the slides were then incubated with 0.25% Triton X-100. The tissues were stained with the PE-Cy5 conjugated primary Abs against CD45 or Nrf2. Next, the tissues were stained with the Alexa Fluor 488-conjugated secondary anti-rabbit Abs. After immunofluorescent staining, a confocal laser microscope was applied to evaluate the slides. ImageJ was applied to qualify the intensities of fluorescence.
Measurement of superoxide anion and GSH
A nitroblue tetrazolium (NBT) assay was used to measure the production of superoxide anions. A commercialized GSH assay kit (Abcam) was used to determine the levels of GSH according to its instructions.
RT-qPCR
RNA was isolated from the joint tissues by using a commercialized RNA extraction kit (Invitrogen). After the concentrations of RNA were determined, the cDNA library was synthesized by using reverse transcriptase. To analyze the accuracy, the melting curves were observed. The expressions of each gene were calculated using the 2–ΔΔCq method. The primers for target genes were:
NLRP3 forward: 5′-GTGGAGATCCTAGGTTTCTCTG -3′, and reverse: 5′–CAGGATCTCATTCTCTTGGATC-3′; ASC forward: 5′–CTCTGTATGGCAATGTGCTGAC-3′, and reverse: 5′–GAACAAGTTCTTGCAGGTCAG-3′; pro-caspase-1 forward: 5′- AATGAAGTTGCTGCTGGAGGA-3′, and reverse: 5′-CAGAAGTCTTGTCGTCTGGGC-3′; pro-IL1β forward: 5′-TGGACCTTCCAGGATGAGGACA -3′, and reverse: 5′- GTTCATCTCGGAGCCTGTAGT-3′. The mRNA expression values of each target gene were normalized to that of an internal control (GAPDH).
ELISA
Inflammatory cytokines (TNF-α, IL-1β, and IL-10) and p65 (total and phosphorylated p65) in the joint tissues were determined using specific ELISAs, according to the instructions of the manufacturers (R&D Biosystems).
Statistical analysis
GraphPad Prism 8 (GraphPad Software, Inc.) was applied to analyze the data. All experiments were repeated independently in triplicate. Data is presented as mean ± SD. One-way ANOVA for multiple comparisons followed by the Student–Newman–Keuls test were performed. P < 0.05 was considered to indicate statistically significant differences.
Results
ePO inhibited zymosan-induced articular mechanical hyperalgesia, edema and reduced the number of leukocytes
We first determined the effects of ePO on zymosan-induced articular mechanical hyperalgesia. The results showed a decrease of threshold in the ePO0 group compared with the saline group (Figure 1b). However, pre-treatment with ePO (100 or 200 mg/kg) significantly increased the threshold at 6 and 8 h compared with the ePO0 group, indicating that pre-treatment with ePO inhibited zymosan-induced articular mechanical hyperalgesia. In addition, we investigated delta reactions at several time points (2, 4, 6, and 8 h) after zymosan injection. The results demonstrated that pre-treatment with ePO (100 and 200 mg/kg) significantly reduced the delta reaction (Figure 1c), indicating that pre-treatment with ePO suppressed the zymosan-induced joint edema.
Furthermore, we evaluated the recruitment of leukocytes in the joint cavity. Zymosan injection significantly increased the numbers of mononuclear, neutrophils, and leukocytes, which could be significantly reduced by pre-treatment with ePO (100 and 200 mg/kg) (Figure 1d-f). Interestingly, we observed that pre-treatment with ePO (100 mg/kg) displayed significant inhibitory effects on the recruitment of mononuclear, neutrophils and leukocytes. These results demonstrated that ePO showed anti-inflammatory properties against zymosan-induced joint inflammation in mice.
ePO ameliorated zymosan-induced pathological alternations in the joint tissues
We performed a histopathological examination on the joint tissues. The results demonstrated that the leukocytes accumulated in the synovial tissue in the ePO0 group as compared with the saline group (Figure 2a).
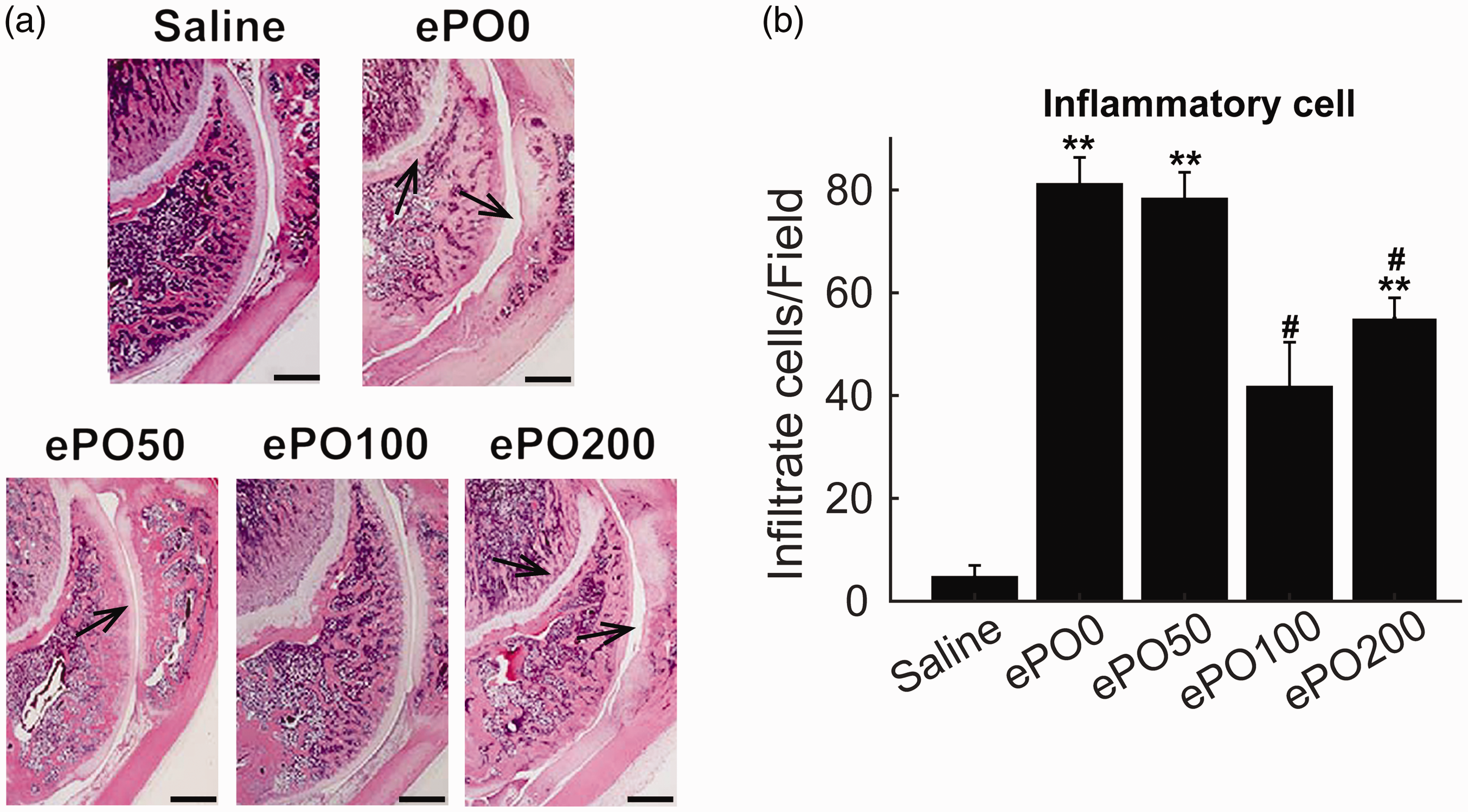
ePO ameliorated pathological alternations in articular cartilage in the patellofemoral joint tissue caused by the injection of zymosan. (a) Representative H&E staining pictures of joint histopathological examinations from different groups. (b) Infiltration of inflammatory cells was counted in slides of joint tissue from different groups. **P < 0.01 versus saline; #P < 0.05 versus ePO0 group. Scale bar = 1 mm. n = 12 for each group.
However, pre-treatment with ePO (100 and 200 mg/kg) inhibited the accumulation of leukocytes. We also observed a significantly increased number of infiltrated inflammatory cells in the ePO0 group, which was inhibited by pre-treatment with ePO (100 and 200 mg/kg) (Figure 2b). Notably, pre-treatment with ePO (100 mg/kg) demonstrated significant inhibitory effects on the infiltration of inflammatory cells. Taken together, these results supported that pre-treatment with ePO ameliorated the pathological alternations in the joint tissues by reducing the inflammatory cells.
ePO inhibited oxidative stress by regulating Nrf2-related signaling proteins
Oxidative stress-related biomarkers were then determined. The results demonstrated that mRNA levels of GP91 were significantly increased by the injection of zymosan, which was inhibited by the pre-treatment with ePO (100 and 200 mg/kg) (Figure 3a).
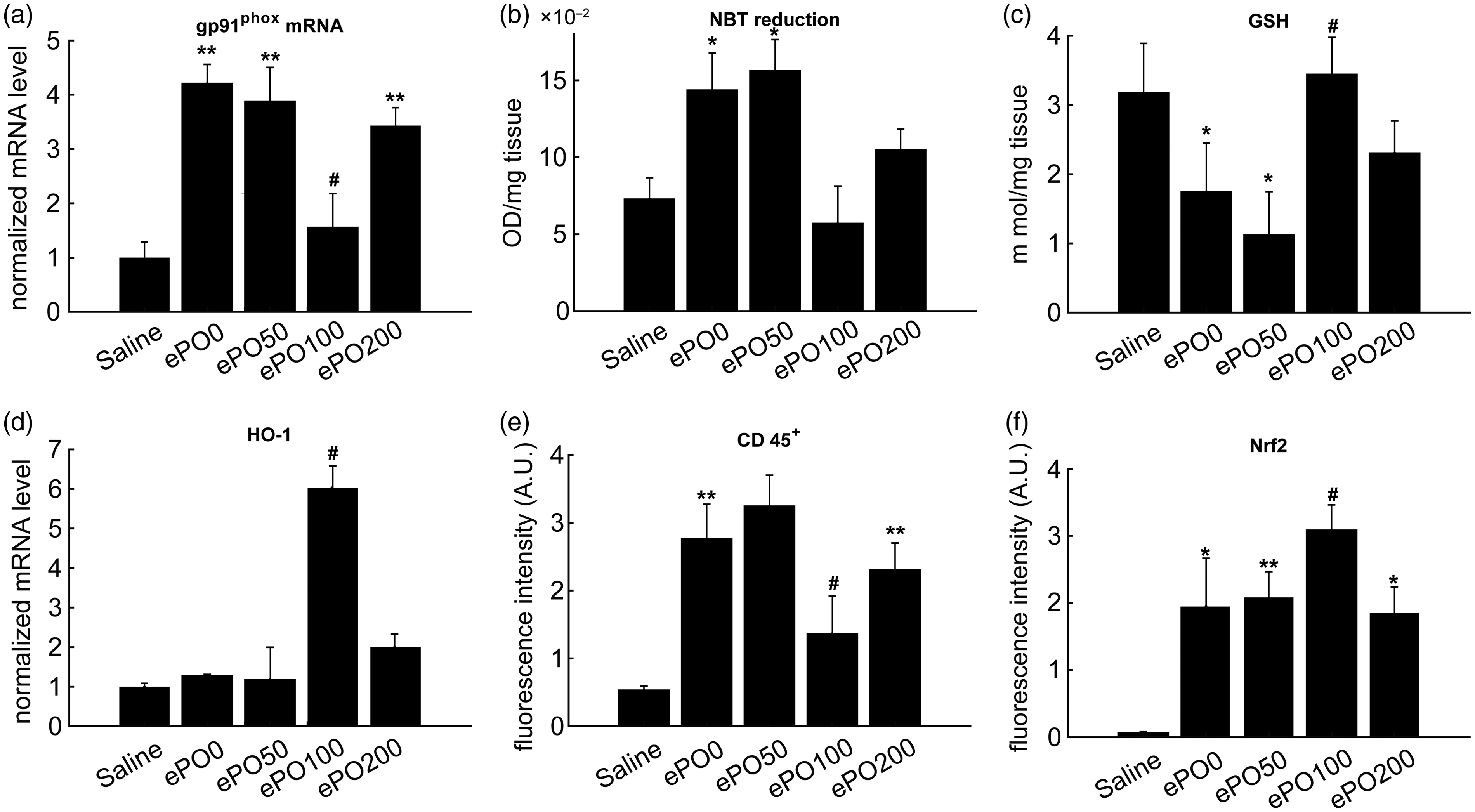
ePO inhibited oxidative stress by regulating the Nrf2-related signaling proteins. The joint tissue was collected after the mice were sacrificed. (a) qRT-PCR was used to determine mRNA levels of gp91phox. (b) NBT reduction assay was used to determine the production of superoxide anions. (c) GSH assay kit was used to measure levels of GSH. (d) qRT-PCR was used to determine mRNA levels of HO-1. (e), (f) The confocal immunofluorescent analysis was applied to determine the expressions of CD45 and Nrf2 in the synovial cells. **P < 0.01, *P < 0.05 versus saline; #P < 0.05 versus ePO0 group. n = 12 for each group.
Besides this, an elevation of superoxide anions was observed in the ePO0 group, which was inhibited by the pre-treatment with ePO (100 and 200 mg/kg) (Figure 3b). A reduction in GSH levels were also observed in the ePO0 group, which was enhanced by pre-treatment with ePO (100 mg/kg) (Figure 3c). In addition, pre-treatment with ePO (100 mg/kg) increased the mRNA levels of heme oxygenase 1 (HO-1) compared with the other groups (Figure 3d).
Immunofluorescent analysis of the synovial cells was performed by CD45 and Nrf2 staining. The results demonstrated significantly increased fluorescent intensities of CD45 and Nrf2 in the ePO0 group. Pre-treatment with ePO (100 mg/kg) could successfully decrease the CD45 expression, while enhancing Nrf2 expression (Figures S1, 3e, and 3f). These results suggested that pre-treatment with ePO inhibited oxidative stress by regulating the Nrf2-related signaling proteins.
ePO inhibited the mRNA levels of NLRP3 inflammasome components in the joint tissues
To explore the mechanism underlying the ePO effect on joint inflammation, next we analyzed the mRNA levels of the NLRP3 inflammasome components, including NLRP3, ASC, pro-caspase-1, and pro-IL-1ß. The mRNA levels of NLRP3, ASC, pro-caspase-1, and pro-IL-1ß were significantly increased compared with the saline group, which could be significantly decreased by pre-treatment with ePO (100 mg/kg) (Figure 4a–d). These results suggested that ePO ameliorated the joint inflammation, at least in part, by regulating the NLRP3 inflammasome.
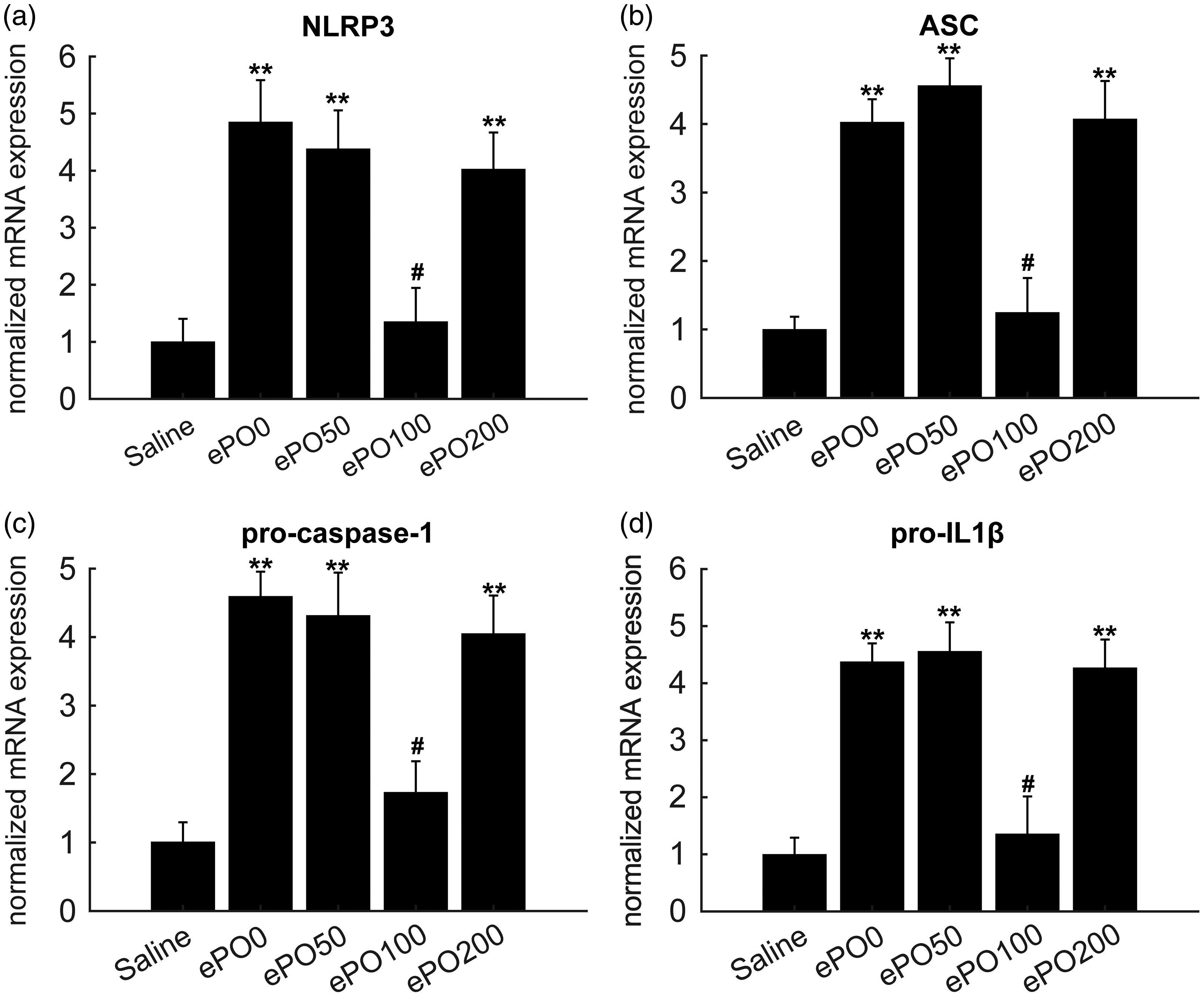
ePO inhibited NLRP3 inflammasome component mRNA levels in joint tissue. After zymosan injection was performed for 8 h, mice were sacrificed, and joint tissue was collected. (a)–(d) qPCR was performed to determine mRNA levels of Nlrp3, ASC, pro-caspase-1, and IL-1β. **P < 0.01, *P < 0.05 versus saline; #P < 0.05 versus ePO0 group. n = 12 for each group.
ePO Inhibited NF-κB and inflammatory cytokines
To confirm the anti-inflammatory effects of ePO, we evaluated the expressions of inflammatory cytokines and NF-κB activation in the joint tissues. The results demonstrated that inflammatory cytokines, including TNF-α and IL-1β, were significantly increased in the ePO0 group, which were inhibited by pre-treatment with ePO (100 mg/kg) (Figure 5a and b).
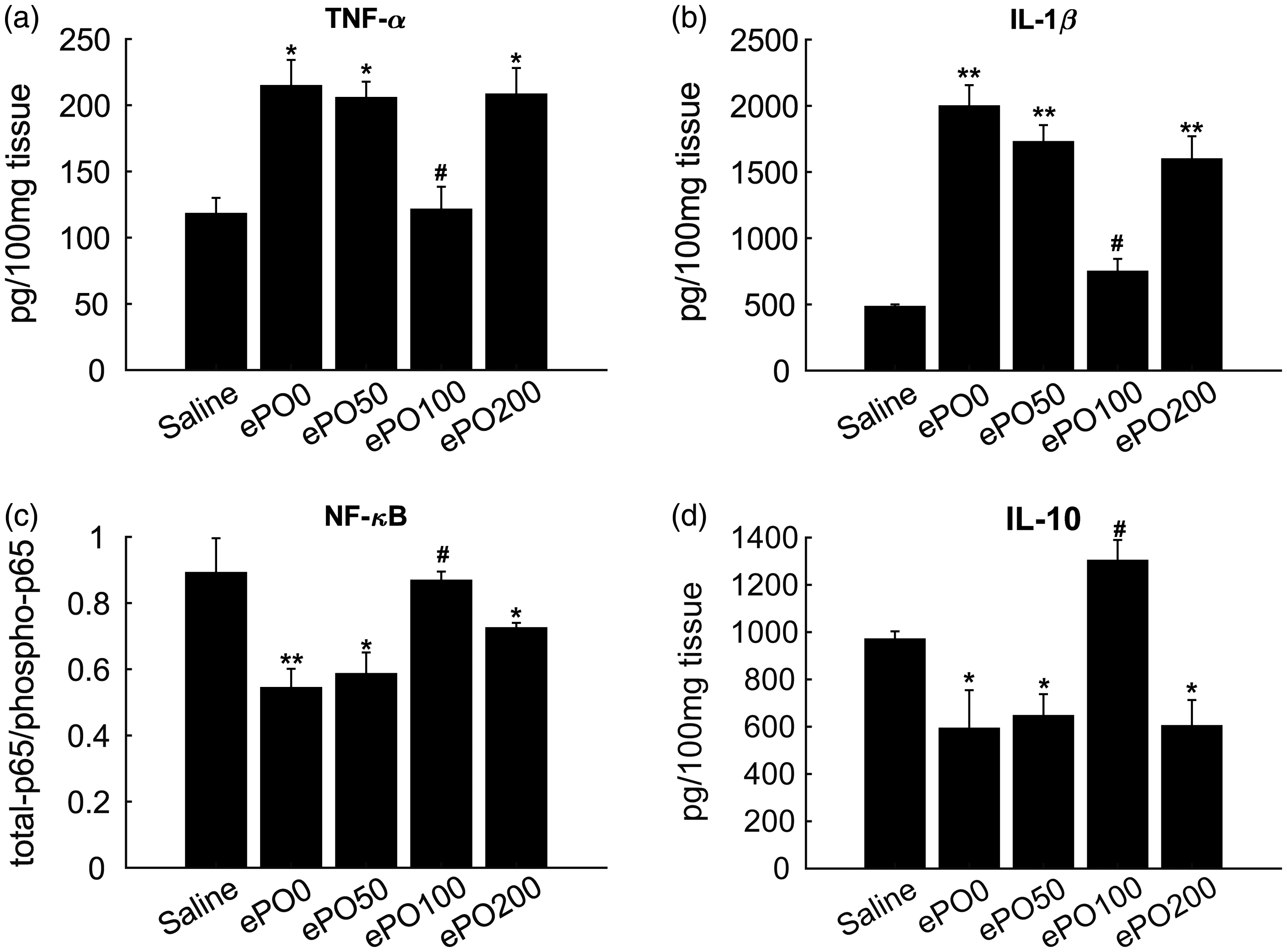
ePO inhibits NF-κB activation and inflammatory cytokines. After zymosan injection was performed for 8 h, mice were sacrificed, and joint tissue was collected. (a)–(d) Protein levels of TNF-α, IL-1β, and IL-10 were determined using specific ELISAs. In addition, levels of total p65 and phosphorylated p65 were measured using specific ELISAs and the ratio of p65 to phosphorylated p65 was calculated. **P < 0.01, *P < 0.05 versus saline; #P < 0.05 versus ePO0 group. n = 12 for each group.
In addition, an elevation of anti-inflammatory cytokine IL-10 was observed in the ePO0 group and was restored by pre-treatment with ePO (100 mg/kg) (Figure 5d). Moreover, NF-κB activation was evaluated by measuring the total p65 and phosphorylated p65. The results demonstrated that the decreased ratio of total p65 to phosphorylated p65 was restored in the ePO (100 mg/kg) group (Figure 5c). These results suggested that ePO ameliorated the joint inflammation by regulating NF-κB activation and inflammatory cytokines.
Discussion
The current study discovered that ePO exerted potential anti-nociceptive effects on a zymosan-induced joint inflammation mouse model. First, the results showed that the numbers of mononuclear neutrophils and leukocytes in the joint cavity were inhibited by the treatment of ePO (200 mg/kg and 100 mg/kg). Consistently, treatment of ePO ameliorated zymosan-induced joint pathological changes by inhibiting infiltrated inflammatory cells. In addition, ePO also showed anti-oxidative stress properties by regulating Nrf2. Moreover, treatment of 100 mg/kg ePO suppressed joint inflammation by regulating key components of NLRP3 inflammasome and NF-κB activation.
Zymosan-induced mouse joint inflammation has been widely employed in the evaluation of anti-nociceptive activities of candidate drugs.28–30 Zymosan is commonly used to induce hyperalgesia, edema, and leukocyte recruitment. In addition, some components, including arachidonic acid lipid products, complement system, and endothelin-1 (ET-1), are involved in the inflammation caused by zymosan. In addition, ET-1 and COX-2-derived prostaglandins are important mediators involved in the inflammation caused by zymosan.30,31 The current study found that immune cells, including mononuclear neutrophils and leukocytes, accumulated in the joint cavity, consistent with characteristics of joint inflammation. 32 We also observed decreased numbers of leukocytes in the ePO-treated group. Treatment of ePO also inhibited the number of CD45+ cells. In addition to the accumulation of inflammatory cells, other symptoms including swelling and joint pain were observed in the joint inflammation mouse model. Therefore, we evaluated the effects of ePO on the joint inflammation-induced pain and swelling symptoms. The results showed that pre-treatment of ePO inhibited zymosan-induced articular mechanical hyperalgesia and joint edema.
Oxidative stress is associated with various inflammatory diseases including arthritis, atherosclerosis, and chronic obstructive pulmonary diseases.33,34 In the pathogenesis of joint inflammation, oxidative stress triggers the inflammatory response, which in turn induces the release of additional oxidative stress products. 35 In the present study, we observed significant changes of GSH, superoxide anions, and HO-1, indicating that oxidative stress participates in joint inflammation. GSH is known as an important nutrient in the defense against joint inflammation, which neutralizes free radicals such as superoxide anions produced during joint inflammation. 36 We observed decreased GSH and increased superoxide anions in the zymosan-induced joint inflammation mice, which was inhibited by ePO (100 mg/kg). In addition, we further explored the mechanism responsible for the ePO effect on oxidative stress. Two key factors, Nrf2 and Gp91 phox, that are associated with oxidative stress, were evaluated in the present study. Nrf2 is known as a transcription factor that regulates anti-oxidative proteins. 37 Gp91 phox is the heme-binding unit of nicotinamide adenine dinucleotide phosphate. 38 Our results demonstrated that pre-treatment with ePO (100 mg/kg) increased Nrf2 while decreasing Gp91 phox, indicating that the regulatory effects of ePO on joint inflammation are associated with its modulatory effects on oxidative stress.
NLRP3 inflammasome is activated in the joint tissues when inflammation occurs. 39 The key components of the NLRP3 inflammasome are adaptor protein ASC and caspase-1 units. Once NLRP3 is activated, caspase-1 cleaves pro-IL-1β, the latter is converted into IL-1β and activates IL-1β signaling. As a result, these events promote the release of inflammatory mediators and adhesion molecules in the joint tissues. 39 In the present study, we observed that NLRP3, ASC, pro-caspase-1, and pro-IL-1ß were significantly increased, but were significantly decreased by pre-treatment with ePO (100 mg/kg). Previous studies have reported the anti-inflammation effects of ePO both in vitro and in vivo.40,41 Our results demonstrated that the anti-inflammatory effects of ePO are attributed to its regulatory effects on the NLRP3 inflammasome.
In addition to NLRP3 inflammasome, NF-κB activation also participates in the pathogenesis of joint inflammation. 39 IL-1β is a unique biomarker indicating the NLRP3 inflammasome activation. 42 However, NF-κB activation is associated with the production of a series of inflammatory cytokines (IL-1β, IL-6, and TNF-α).43,44 In the present study, in addition to IL-1β, we also observed increased IL-6 and TNF-α levels in the joint tissues, which could be inhibited by ePO. Moreover, pre-treatment with ePO also suppressed the NF-κB activation, indicating that ePO suppressed inflammatory cytokines by inhibiting NF-κB activation. Interestingly, we also observed an elevation of anti-inflammatory cytokine IL-10 in the ePO (100 mg/kg) group. However, further studies are needed to elucidate the regulatory mechanisms of ePO on IL-10. Another interesting phenomenon is that the anti-inflammatory effects of ePO100 and ePO200 were not consistent. Previous studies have demonstrated that high doses of ePO exhibit over-dosage effects or toxicity. For instance, it was reported that P. oleracea L. is moderately toxic at high doses. 45 Another study clarified that P. oleracea L. extracts contained toxic cardiac glycosides and oxalic acids, which may contribute to its toxicity.46 In the present study, the behavioral observations showed an anti-inflammation effect of ePO at the dose of 100 mg/kg, rather than 50 and 200 mg/kg, suggesting that the anti-inflammatory activity of ePO was restricted to a dose range. High doses of ePO may cause undesirable adverse effects, thereby negating its anti-inflammatory activity. However, to confirm this phenomenon, further toxicological studies should be performed.
One major challenge in the use of natural products for therapy is their poor bioavailability. In the present study, the pre-treatment of ePO showed potential therapeutic effects against RA. However, investigations into its distribution and bioavailability should be investigated in the future to further develop ePO into an anti-inflammatory drug for inflammatory disease treatment.
Taken together, the present study revealed that the ameliorating effects of ePO against joint inflammation are in part mediated through regulating NLRP3 inflammasome and NF-κB, both of which play important roles in the inflammatory conditions including Alzheimer’s, Parkinson’s and traumatic brain injury.47,48 Therefore, the data presented in the current study also encourages us to investigate the potential effects of ePO on neurological disorders in future studies.
The present study has identified ePO with anti-nociceptive effects on the zymosan-induced joint inflammation mouse model. Pre-treatment of 100 mg/kg ePO ameliorated the pathological alternations in the joint by attenuating the infiltration of inflammatory cells and oxidative stress. The mechanism responsible for ePO effects on joint inflammation is associated with its regulatory role on NLRP3 inflammasome and NF-κB activation.
Supplemental Material
sj-pdf-1-ini-10.1177_1753425921994190 - Supplemental material for Anti-nociceptive effect of Portulaca oleracea L. ethanol extracts attenuated zymosan-induced mouse joint inflammation via inhibition of Nrf2 expression
Supplemental material, sj-pdf-1-ini-10.1177_1753425921994190 for Anti-nociceptive effect of Portulaca oleracea L. ethanol extracts attenuated zymosan-induced mouse joint inflammation via inhibition of Nrf2 expression by Yunwu He, Hui Long, Cong Zou, Wuzhou Yang, Liping Jiang, Zhenping Xiao, Qing Li and Shiyin Long in Innate Immunity
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics statement
All animal experiments were approved by the Ethics Committee of the University of South China.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Nature Science Fund of Hunan Province (2018JJ2346 and 2018JJ2348).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
