Abstract
Acetaminophen (AAP) and N-acetylcysteine (NAC) have been found to have anti-inflammatory effects via the TLR–NF-κB pathway in LPS-challenged piglets. However, the action mechanisms employed by AAP and NAC have yet to be completely understood. This study investigated the anti-inflammatory effects and mechanisms of AAP and NAC on LPS-induced inflammatory responses via the NLRP3 inflammasome pathway in piglet mononuclear phagocytes. The results show that mRNA expression levels of IL-1β, IL-18 and NLRP3, as well as the protein level of cleaved caspase-1, are significantly increased after LPS challenge in vitro. LPS stimulation did not change ASC and caspase-1 mRNA levels, which were components of NLRP3 inflammasome complex. AAP (0.5–1.0 mM) and NAC (0.5–1.0 mM) used individually or in combination could down-regulate protein expression of cleaved caspase-1 and mRNA expression of IL-1β, IL-18 and NLRP3. NAC could significantly enhance the above inhibition actions of AAP. The combined use of AAP plus NAC had better inhibition action on the NLRP3 inflammasome pathway. These results indicate that the anti-inflammatory effects of AAP and NAC occur via the regulation on mRNA expression of NLRP3 and activation of caspase-1. The anti-inflammatory activity of AAP and NAC could be related to the suppression of NLRP3 inflammasome pathway under LPS stimulation.
Introduction
Acetaminophen (AAP) is widely used as an analgesic and antipyretic agent in the treatment of pain and fever. AAP is considered to have a weak anti-inflammatory ability owing to its weak inhibition action on cyclooxygenase (COX)-1 and COX-2. 1 AAP can affect the activity of NF-κB, and inhibit LPS/IFN-γ-induced iNOS protein and mRNA expression in macrophages, 2 as well as IL-1β-induced I-κB degradation and NF-κB nuclear translocation. 3 This compound also significantly suppresses the release of pro-inflammatory cytokines and NF-κB activation in LPS-challenged piglets. 4 The main side effect of AAP is hepatotoxicity when used in high doses.
N-Acetylcysteine (NAC) is an important cellular antioxidant and has been proven to have an anti-inflammatory effect.5–8 The effectiveness of NAC is primarily attributed to its ability to reduce extracellular cystine to cysteine, or act intracellularly as a source of sulfhydryl groups. NAC may suppress NF-κB activation and the production of pro-inflammatory cytokines.6,9–11 Furthermore, NAC reduces inflammation in the small intestine of piglets by regulating redox, epidermal growth factor and TLR4 signaling. 8 This compound is generally safe and well tolerated, even at high doses.
The most common reason for the combined use of NAC and AAP is that NAC has been used as an antidote for poisoning by high doses of AAP. Our previous work demonstrated that the combined use of AAP and NAC at their common dose had better inhibition of cytokine production and NF-κB activation in LPS-induced piglets. 4 In order to reduce the risk of hepatotoxicity, alleviate inflammation and protect the intestine, it is highly possible in the future that, in piglets, NAC will be used more commonly with the label dose of AAP. So far, anti-inflammatory effects of AAP and NAC have yet to be clearly elucidated, especially their action on inflammasome signaling pathway.
The generation of the IL-1 family of cytokines (IL-1β and IL-18) is controlled by inflammasomes. In mononuclear phagocytes, two temporally separate signals are required to yield the active pro-inflammatory cytokine. The first signal involves the activation of PRRs, such as TLRs or NOD-like receptors, by PAMPS and DAMPs, which triggers the expression of pro-IL-1β and pro-IL-18 via the NF-κB pathway. 12 Then, microbial products or endogenous signals as the potential second signals activate the inflammasome complex.13–16 To date, several proteins have been described that can initiate the formation of inflammasome complexes: the NLR proteins NLRP1, NLRP3 and NLRC4; and the pyrin and HIN200 domain-containing protein AIM2. 17
The NLRP3 inflammasome is tightly controlled by a priming step that is dependent on NF-κB. 17 Priming for NLRP3 inflammasome activation is highly inducible in response to pro-inflammatory stimuli such as LPS. It has been reported that LPS priming enhances neither the expression of NLRP1 nor that of NLRC4 or AIM2. 17 LPS stimulation led to both NLRP3 and pro-IL-1β protein induction in a dose- and time-dependent manner, which indicated that NLRP3 expression was tightly controlled by signals culminating in the activation of NF-κB. 18 The NLRP3 inflammasome complex mediate the maturation of inactive procaspase-1 into active caspase-1 (cleaved caspase-1), where casapse-1 is the central stimulator promoting the maturation of pro-inflammatory cytokines IL-1β and IL-18 from pro-IL-1β and pro-IL-18, respectively, and causing inflammation.19,20 We had found that AAP and NAC alone or in combination caused inhibition of IL-1β production and NF-κB activation in LPS-induced piglets. 4 Thus, it was hypothesized that AAP and NAC would be able to relieve the inflammation from immunological stress, at least in part, through the NLRP3 inflammasome signaling pathway in piglets.
In an effort to elucidate the effect and mechanism responsible for the anti-inflammatory action of AAP and NAC, the effects of mRNA expression and protein secretion of IL-1β and IL-18, and the protein level of cleaved caspase-1 by AAP and NAC were observed in LPS-induced piglet mononuclear phagocytes. Additionally, mRNA expression of the NLRP3 inflammasome complex [NLRP3, adaptor protein apoptosis-associated speck like protein (ASC) and caspase-1] was also evaluated. Our aim was to explore the combination effect of AAP and NAC on the NLRP3 inflammasome signaling pathway, and to evaluate its value in practical application.
Materials and methods
Isolation and culture of peripheral blood monocytes
Three crossbred healthy piglets (Duroc × Landrace × Yorkshire) obtained from Wuhan Xinnongyuan Livestock Co., Ltd (Wuhan, China) weighing 8–10 kg and aged 5 wk were used for in vitro experiments. The piglets had not been administered any drugs. The animal use protocol for this research was approved by the Animal Care and Use Committee of Wuhan Polytechnic University.
The blood used for the isolation of monocytes was collected by venipuncture from the piglets. Heparinized blood was layered carefully onto an equal volume of D-Han’s solution (pH 7.2) in a conical centrifuge tube, and then an equal volume of lymphocyte separation medium was added. The suspension was centrifuged at 1100 g for 5 min at room temperature (25℃). The mononuclear cell layer was collected and washed three times with D-Han’s solution (by centrifugation 1100 g for 2 min at 4℃). Cells were re-suspended in a known volume of RPMI 1640. Mononuclear cells were counted and their viability was determined by Trypan blue exclusion. The cells were seeded in a six-well plate, 1 ml of suspension was added in each well and were pre-incubated in a humidified incubator at 37℃ with 5% CO2 for 2 h in RPMI 1640 containing 10% FCS. Cells were then washed three times with RPMI medium 1640 pre-warmed to 37℃ to discard the unattached cells. Attached cells (monocytes) were detached using a cell scraper and suspended in RPMI medium 1640.
Experimental design
Piglet monocytes were treated in five different ways: (1) control treatment (normal cells with normal medium without adding LPS and drugs), (2) LPS (from Escherichia coli, serotype type O55:B5; Sigma, St. Louis, MO, USA) stimulation treatment (normal cells with normal medium adding LPS without drugs), (3) LPS stimulation + AAP (0.25, 0.5 and 1.0 mM) treatment, (4) LPS stimulation + NAC (0.25, 0.5 and 1.0 mM) treatment, and (5) LPS stimulation + AAP (0.25, 0.5 and 1.0 mM) plus NAC (0.25, 0.5 and 1.0 mM) treatment. Each treatment was repeated three times with monocytes from three piglets. After incubation with drugs for 1 h, LPS was added (the final concentration was 1 µg/ml), and incubation was continued for 3 h [reactive oxygen species (ROS), mRNA analysis of IL-1β, IL-18, TNF-α, NLRP3, ASC and caspase-1] or 10, 16 and 20 h (IL-1β, IL-18 and TNF-α), or 10 h (active caspase-1 protein analysis). Cytokines were measured by ELISA assay (in a 96-well plate at 3.0 × 105 cells/ml). ROS were observed by fluorescence microscope (in a 24-well plate at 3.0 × 106 cells/ml). IL-1β, IL-18, NLRP3, ASC and caspase-1 mRNA was measured by RT-PCR (in a 24-well plate at 3.0 × 106 cells/ml). Cleaved caspase-1 (20 kDa) was measured by Western blot analysis (in a six-well plate at 5.0 × 106 cells/ml).
Analysis of cytokines concentrations
Concentrations of IL-1β, IL-18 and TNF-α obtained from monocyte culture supernatants (in a 96-well plate at 3 × 105 cells/ml) were measured using ELISA kits according to manufacturer’s instructions (Blue Gene, Shanghai, China). IL-1β (10–320 pg/ml), IL-18 (10–500 pg/ml) and TNF-α (10–500 pg/ml) were used as standards for calibration, and the sensitivity of all assays was 3 pg/ml.
ROS detection
After 3 h of LPS stimulation, supernatants were discard, the un-stimulated and stimulated cells (1 ml, 3.0 × 106 cells/ml) were rinsed twice with ice-cold PBS; then, 1.5 ml 2,7-dichlorofurescin diacetate (10 µmol/l) was added and incubation continued at 37℃ with 5% CO2 for 45 min. ROS were observed by fluorescence microscopy and analysis by Image-Pro Plus (Media Cybernetics Inc., Rockville, MD, USA), which was performed using sum integrated optical density method in the same area.
RNA extraction and RT-PCR
Sequence of primers used in current investigation by RT-PCR.
Western blot analysis
For analysis of the level of cleaved (active) caspase-1 in cell lysate, the unstimulated and stimulated cells (1 ml, 5 × 106 cells/ml) were rinsed twice in ice-cold PBS and lysed in ice-cold buffer. Protein concentration was determined by the bicinchoninic acid assay method. The proteins were separated by SDS-PAGE using Tris-HCl Precast Gels (Amresco, OH, USA) and then transferred onto a PVDF membrane (60 mA, 2 h). The resulting membrane was blocked with Tris buffer saline solution containing 0.05% Tween-20 (TBS-T) supplemented with 15% skimmed milk at room temperature for 2 h on a rotary shaker, and followed by TBS-T washing. The membrane was incubated with cleaved caspases-1 Ab [cleaved caspases-1 (Asp297) (D57A2) Rabbit mAb; Cell Signaling Technology, Danvers, MA, USA] and β-actin Ab (Cell Signaling Technology), diluted in TBS-T solution (1:1000), at 4℃ overnight (12 h). Subsequently, the membrane was washed with TBS-T followed by incubation with the HRP-linked Ab (containing 15% skimmed milk TBS-T solution, 1:1000 dilution) at room temperature for 2 h. The level of active caspase-1 was detected using an enhanced chemiluminescence detection system.
Statistical analysis
SPSS Statistics version 17.0 (IBM, Armonk, NY, USA) was used for statistical analysis. The data were shown as the mean ± SD. The differences between the data sets were assessed by one-way ANOVA and multiple comparisons between the groups were performed using least significant difference (LSD) method. A probability value of P < 0.05 was considered significant.
Results
Effects of AAP and NAC on IL-1β, IL-18 and TNF-α production in LPS-stimulated piglet monocytes
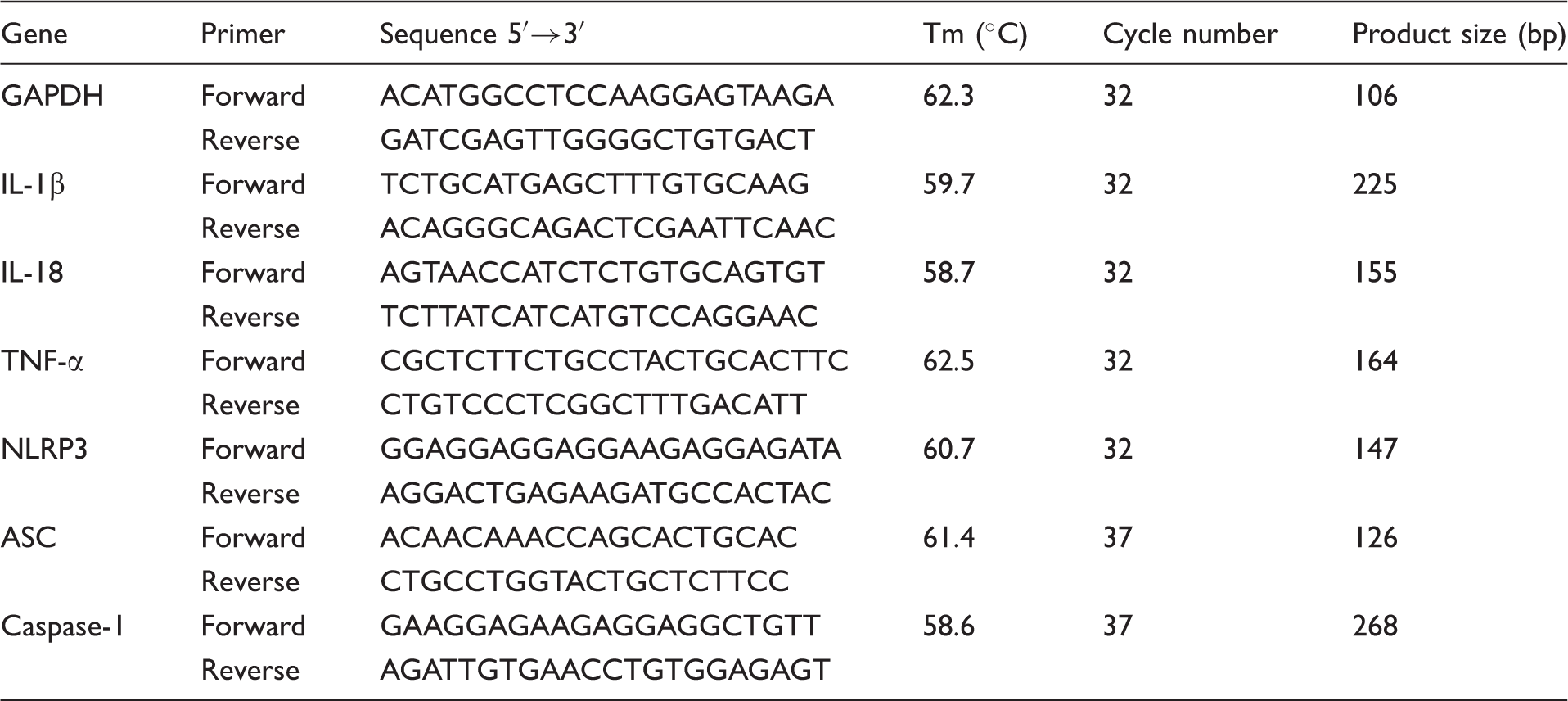
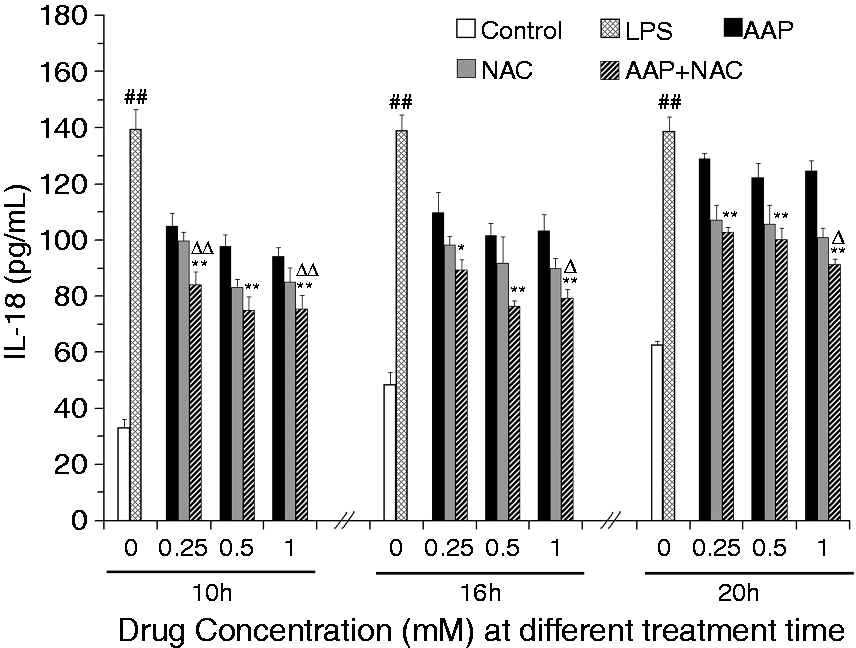
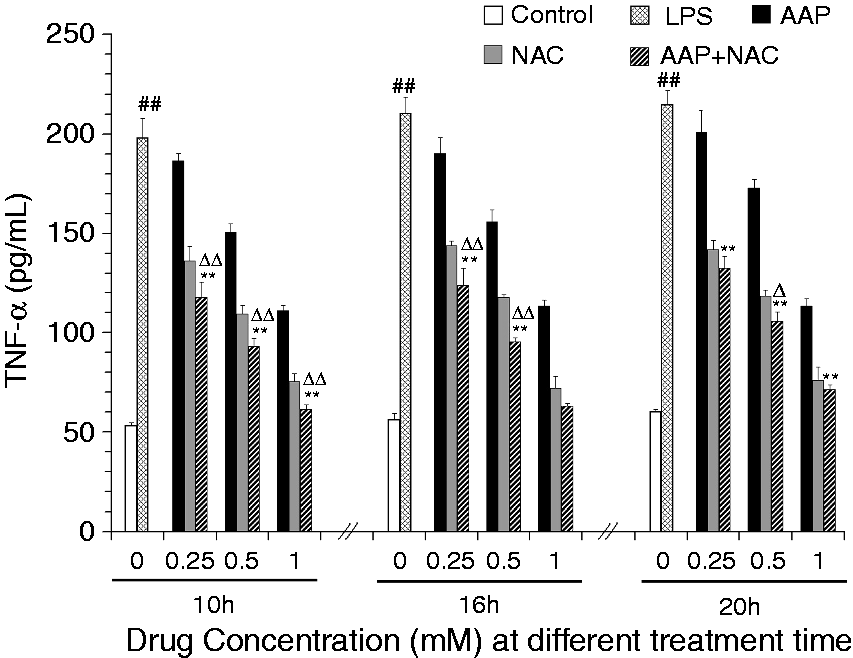
The effects of AAP and NAC on the production of IL-1β, IL-18 and TNF-α from LPS-simulated piglet monocytes were evaluated. LPS stimulation significantly enhanced the IL-1β and IL-18 level relative to that observed in the unstimulated cells. The production of IL-1β and IL-18 in response to LPS was significantly inhibited by AAP, NAC and AAP plus NAC (Figures 1 and 2). The addition of 0.25–1.0 mM NAC to the incubation mixture containing 0.25–1.0 mM AAP significantly (P < 0.05 or P < 0.01) enhanced the inhibitory effect on the secretion of IL-1β after 10 h, 16 h and 20 h of LPS exposure, with the exception of 0.25 mM NAC on 0.25 mM AAP after 20 h of LPS exposure. In the AAP plus NAC group the production of IL-1β was also significantly reduced compared with the NAC group. The addition of NAC to the incubation mixture containing AAP significantly (P < 0.05 or P < 0.01) enhanced the inhibitory effect on the secretion of IL-18 after LPS exposure. In the AAP plus NAC group, IL-18 production was also reduced compared with the NAC group. LPS stimulation significantly increased TNF-α production by blood monocytes relative to the control. The addition of AAP, NAC and AAP plus NAC to the cell were found to reduce the LPS-induced production of TNF-α in a dose-dependent manner (Figure 3). The addition of NAC to the incubation mixture containing AAP significantly enhanced the inhibitory effect on the secretion of TNF-α. In the AAP + NAC group, TNF-α production was also significantly reduced compared with the NAC group.
Effects of AAP and NAC on LPS-induced IL-1β production by blood monocytes of piglets in vitro. Cells (3 × 105 cells/ml) were pretreated for 1 h with drugs, and then stimulated for 10, 16 and 20 h with LPS (1 µg/ml). The levels of IL-1β in the supernatant were measured by ELISA. The values presented are the means ± SD of three independent experiments, and differences between mean values were assessed by LSD method. ##P < 0.01 vs. control; *P < 0.05 vs. AAP alone; **P < 0.01 vs. AAP alone; ΔP < 0.05 vs. NAC alone; ΔΔP < 0.01 vs. NAC alone. Effects of AAP and NAC on LPS-induced IL-18 production by blood monocytes of piglets in vitro. Cells (3 × 105 cells/ml) were pretreated for 1 h with drugs, and then stimulated for 10, 16 and 20 h with LPS (1 µg/ml). The levels of IL-18 in the supernatant were measured by ELISA. The values presented are the means ± SD of three independent experiments, and differences between mean values were assessed by LSD method. ##P < 0.01 vs. control; *P < 0.05 vs. AAP alone, **P < 0.01 vs. AAP alone; ΔP < 0.05 vs. NAC alone; ΔΔP < 0.01 vs. NAC alone. Effects of AAP and NAC on LPS-induced TNF-α production by blood monocytes of piglets in vitro. Cells (3 × 105 cells/ml) were pretreated for 1 h with drugs, and then stimulated for 10, 16 and 20 h with LPS (1 µg/ml). The levels of TNF-α in the supernatant were measured by ELISA. The values presented are the means ± SD of three independent experiments, and differences between mean values were assessed by the LSD method. ##P < 0.01 vs. control; **P < 0.01 vs. AAP alone; ΔP < 0.05 vs. NAC alone; ΔΔP < 0.01 vs. NAC alone.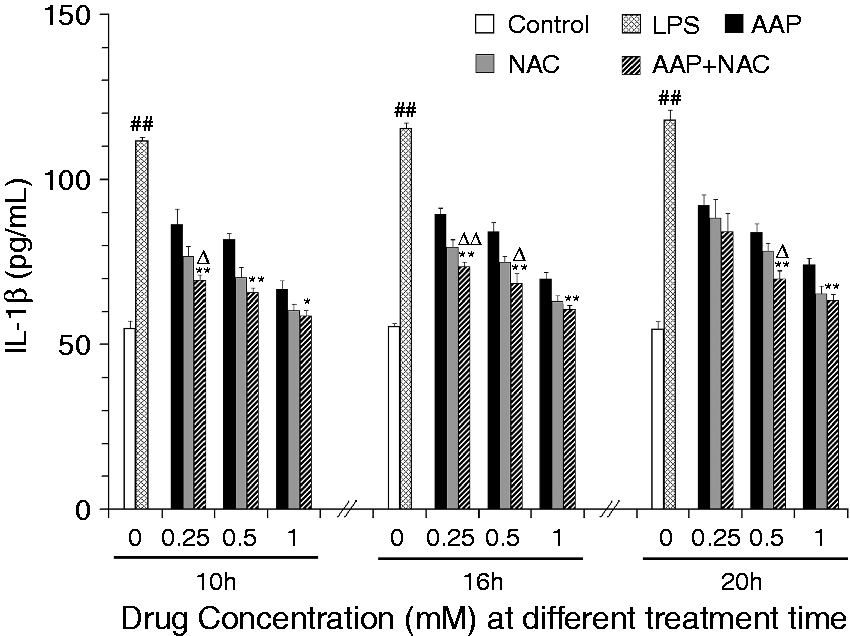
Effects of AAP and NAC on ROS production in LPS-stimulated piglet monocytes
The production of ROS in monocytes was significantly increased 3 h after LPS stimulation (Figure 4). The addition of AAP, NAC and AAP plus NAC to the cells reduced the production of ROS. NAC (0.5 mM) significantly (P < 0.01) enhanced the inhibitory effect of AAP (0.5 mM) on ROS production. In the AAP plus NAC group, the production level of ROS was also significantly reduced (P < 0.05) compared with the NAC group (0.5 mM).
Effects of AAP and NAC on ROS production by blood monocytes of piglets in vitro. Cells (3 × 106 cells/ml) were pretreated for 1 h with drugs, and then stimulated for 3 h with LPS (1 µg/ml). The levels of ROS in the cells were observed by fluorescence microscopy. The values presented are the means ± SD of three independent experiments, and differences between mean values were assessed by the LSD method. ##P < 0.01 vs. control; **P < 0.01 vs. AAP alone; ΔP < 0.05 vs. NAC alone.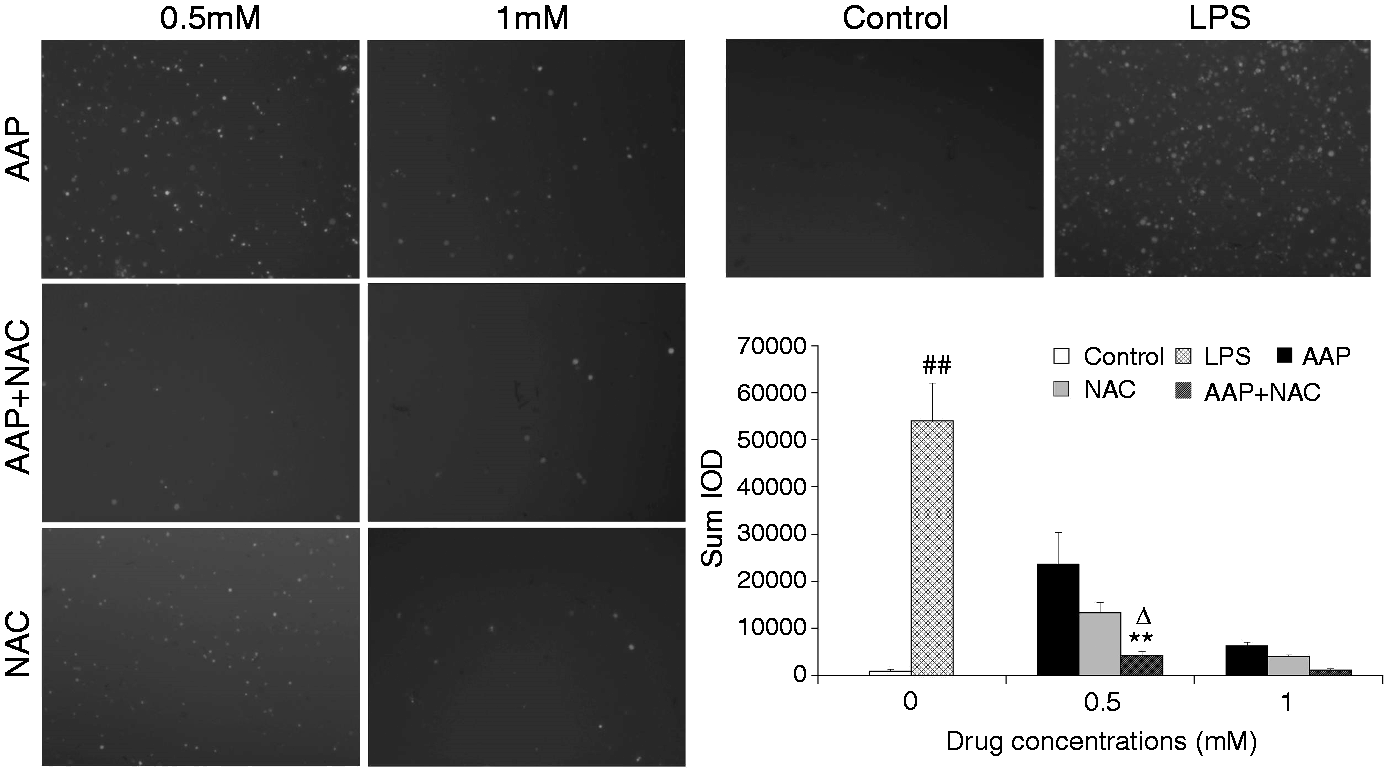
Effects of AAP and NAC on the mRNA expression of IL-1β, IL-18 and TNF-α in LPS-stimulated piglet monocytes
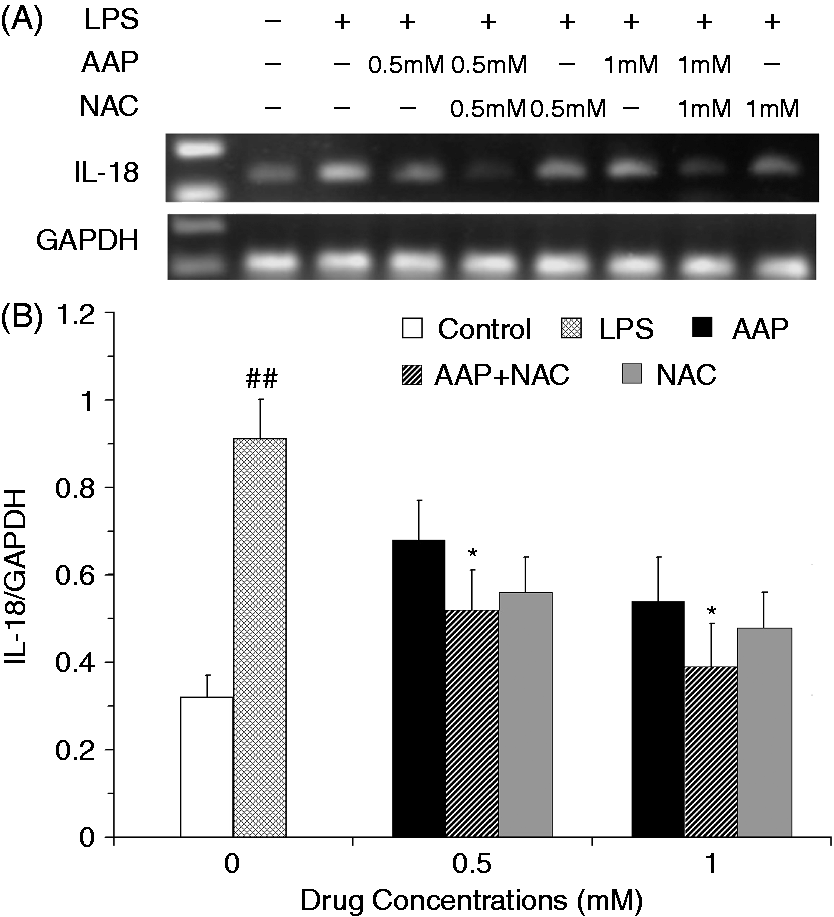
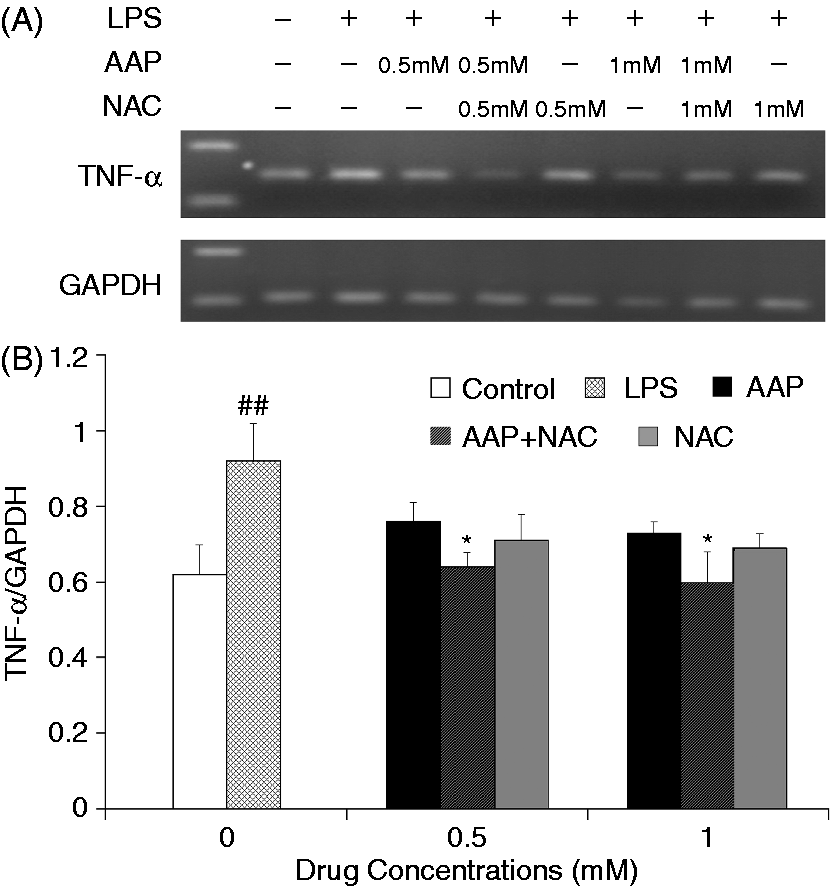
The mRNA expression levels of IL-1β and IL-18 in the monocytes were significantly increased 3 h after LPS stimulation (Figures 5 and 6). The addition of AAP, NAC and AAP plus NAC to the cell reduced the mRNA expression of IL-1β and IL-18. NAC (0.5 mM and 1.0 mM) significantly (P < 0.05) enhanced the inhibitory effect of AAP (0.5 mM and 1.0 mM) on IL-1β and IL-18 mRNA expression. The mRNA expression of TNF-α in the monocytes was significantly increased 3 h after LPS stimulation (Figure 7). The additions of AAP, NAC and AAP plus NAC to the cell reduced the LPS-induced mRNA expression of TNF-α. NAC (0.5 mM and 1.0 mM) significantly (P < 0.05) enhanced the inhibitory effect of AAP on TNF-α mRNA expression. In the AAP plus NAC group, the mRNA expression levels of IL-1β, IL-18 and TNF-α were reduced, but not significantly, compared with the NAC group.
Effects of AAP and NAC on LPS-induced mRNA expression of IL-1β by blood monocytes of piglets in vitro. (A) Cells (3 × 106 cells/ml) were pretreated for 1 h with drugs, and then stimulated for 3 h with LPS (1 µg/ml). The levels of mRNA expression of IL-1β in the cells were measured by RT-PCR. (B) The relative level of IL-1β was measured using an image analyzer. The values presented are the means ± SD of three independent experiments, and differences between mean values were assessed by the LSD method. ##P < 0.01 vs. control; *P < 0.05 vs. AAP alone. Effects of AAP and NAC on LPS-induced mRNA expression of IL-18 by blood monocytes of piglets in vitro. (A) Cells (3 × 106 cells/ml) were pretreated for 1 h with drugs, and then stimulated for 3 h with LPS (1 µg/ml). The levels of mRNA expression of IL-18 in the cells were measured by RT-PCR. (B) The relative level of IL-18 was measured using an image analyzer. The values presented are the means ± SD of three independent experiments, and differences between mean values were assessed by the LSD method. ##P < 0.01 vs. control; *P < 0.05 vs. AAP alone. Effects of AAP and NAC on LPS-induced mRNA expression of TNF-α by blood monocytes of piglets in vitro. (A) Cells (3 × 106 cells/ml) were pretreated for 1 h with drugs, and then stimulated for 3 h with LPS (1 µg/ml). The levels of mRNA expression of TNF-α in the cells were measured by RT-PCR. (B) The relative level of TNF-α was measured using an image analyzer. The values presented are the means ± SD of three independent experiments, and differences between mean values were assessed by the LSD method. ##P < 0.01 vs. control; *P < 0.05 vs. AAP alone.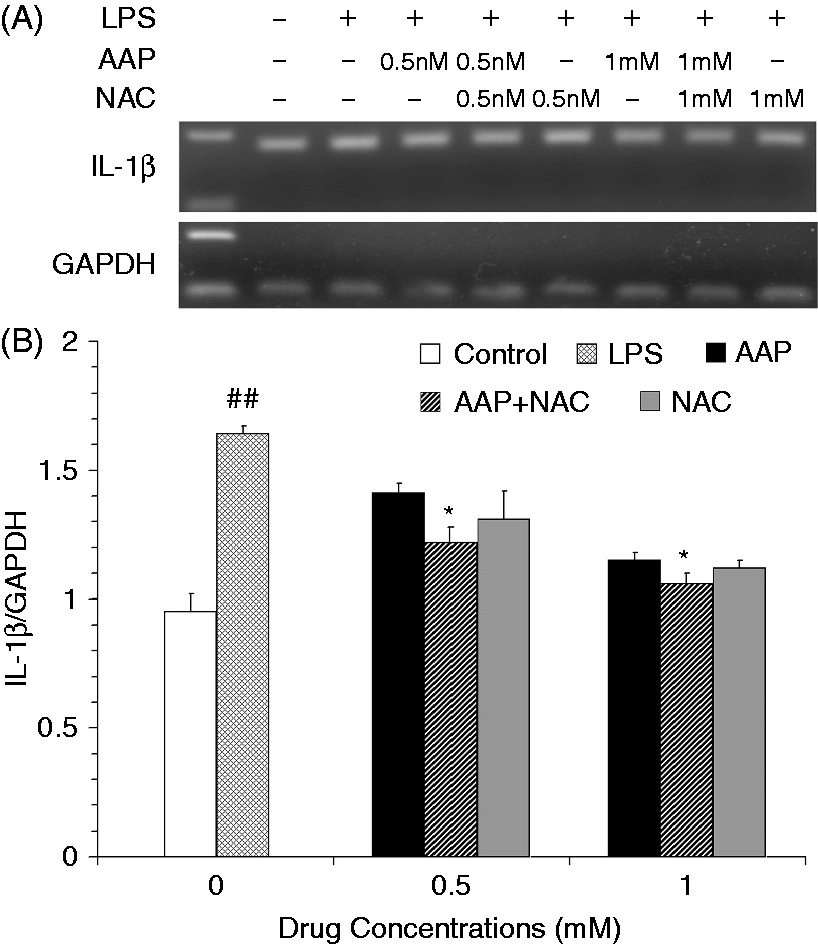
Effects of AAP and NAC on the mRNA expression of NLRP3 inflammasome components in LPS-stimulated piglet monocytes
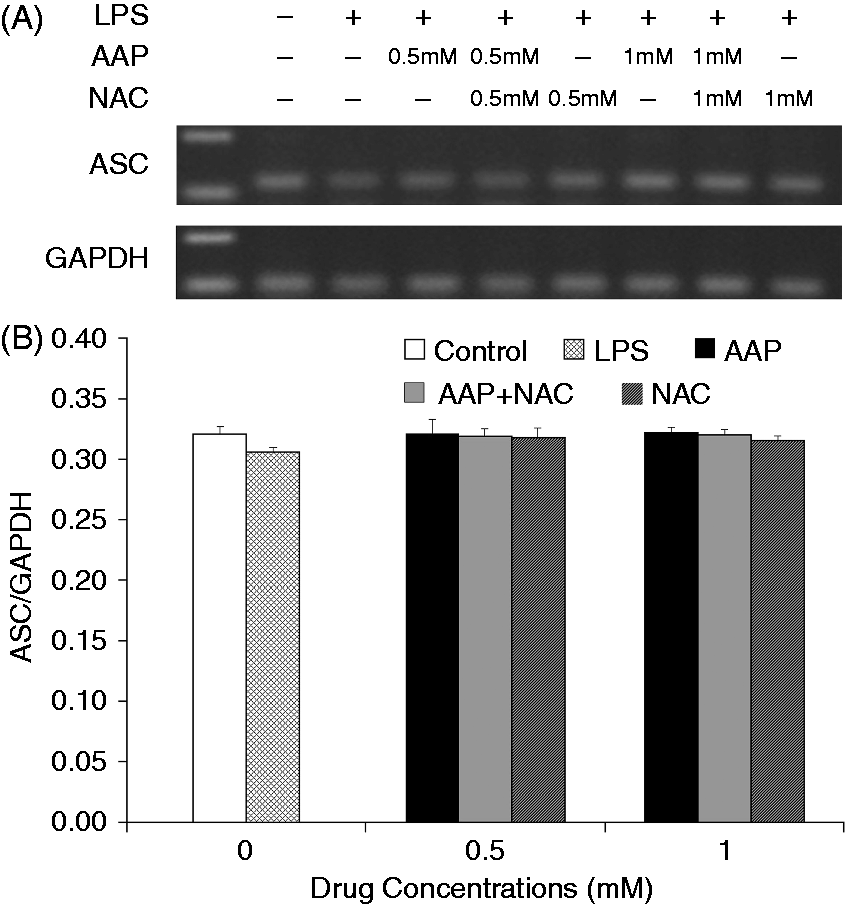
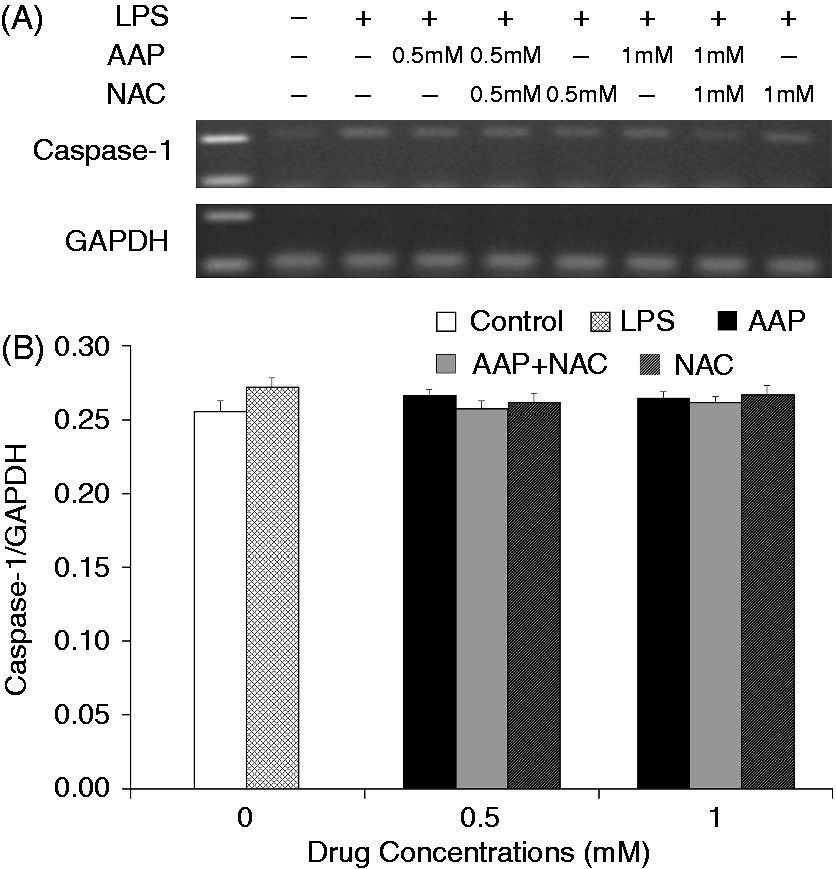
The mRNA expression level of NLRP3 in the monocytes was significantly increased 3 h after LPS stimulation (Figure 8). The additions of AAP, NAC and AAP plus NAC to the cell reduced the mRNA expression of NLRP3. NAC (0.5 mM and 1.0 mM) significantly (P < 0.05 or P < 0.01) enhanced the inhibitory effect of AAP on NLRP3 mRNA expression. In the AAP plus NAC group mRNA expression of NLRP3 was also significantly (P < 0.05) reduced compared with the NAC group. Of interest, LPS stimulation enhanced neither the mRNA expression of ASC nor that of caspase-1. The addition of AAP, NAC and AAP plus NAC to the treated cell had no significant effects on the mRNA expression of ASC and caspase-1 (Figures 9 and 10).
Effects of AAP and NAC on LPS-induced mRNA expression of NLRP3 by blood monocytes of piglets in vitro. (A) Cells (3 × 106 cells/ml) were pretreated for 1 h with drugs, and then stimulated for 3 h with LPS (1 µg/ml). The levels of mRNA expression of NLRP3 in the cells were measured by RT-PCR. (B) The relative level of NLRP3 was measured using an image analyzer. The values presented are the means ± SD of three independent experiments, and differences between mean values were assessed by the LSD method. ##P < 0.01 vs. control; *P < 0.05 vs. AAP alone; **P < 0.01 vs. AAP alone; ΔP < 0.05 vs. NAC alone. Effects of AAP and NAC on LPS-induced mRNA expression of ASC by blood monocytes of piglets in vitro. (A) Cells (3 × 106 cells/ml) were pretreated for 1 h with drugs, and then stimulated for 3 h with LPS (1 µg/ml). The levels of mRNA expression of ASC in the cells were measured by RT-PCR. (B) The relative level of ASC was measured using an image analyzer. The values presented are the means ± SD of three independent experiments, and differences between mean values were assessed by the LSD method. Effects of AAP and NAC on LPS-induced mRNA expression of caspase-1 by blood monocytes of piglets in vitro. (A) Cells (3 × 106 cells/ml) were pretreated for 1 h with drugs, and then stimulated for 3 h with LPS (1 µg/ml). The levels of mRNA expression of caspase-1 in the supernatant were measured by RT-PCR. (B) The relative level of caspase-1 was measured using an image analyzer. The values presented are the means ± SD of three independent experiments, and differences between mean values were assessed by the LSD method.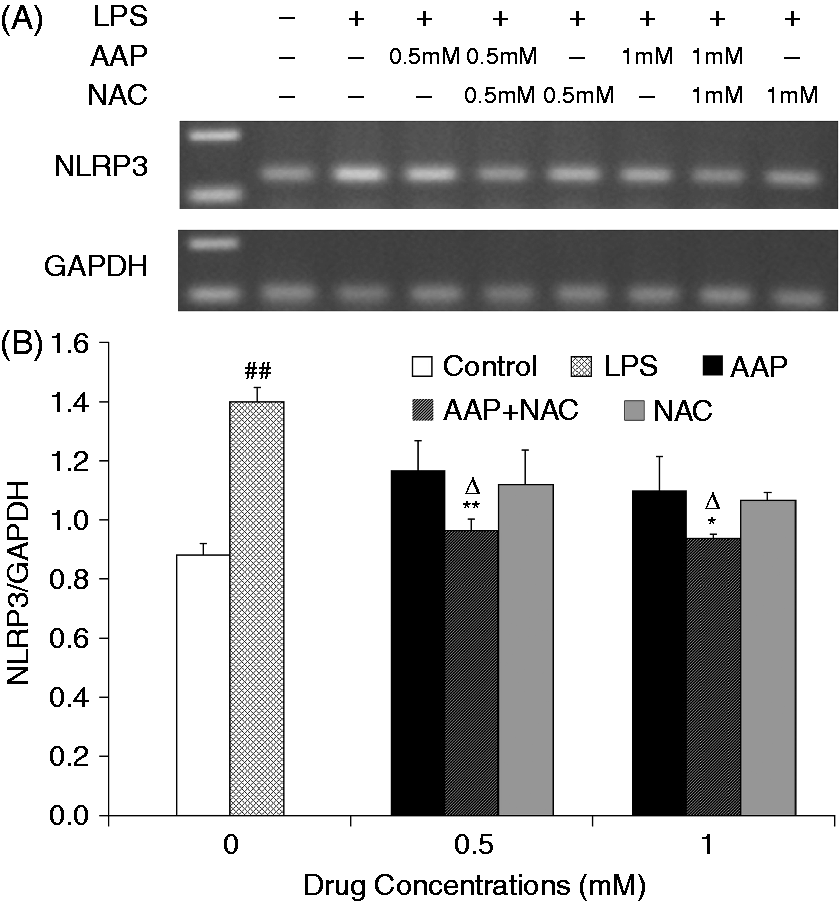
Effect of AAP and NAC on cleaved caspase-1 production in LPS-stimulated piglet monocytes
LPS stimulation significantly increased the cleaved (active) caspase-1 level relative to that observed in the unstimulated cells. The additions of AAP, NAC and AAP plus NAC to the stimulated cells were found to reduce active caspase-1 production in a dose-dependent manner (Figure 11). The maximal inhibition rate of cleaved caspase-1 production by AAP (1 mM), NAC (1 mM) and AAP plus NAC (1 mM in combination) were 57.42% ± 2.3%, 66.33% ± 8.6% and 67.96% ± 3.0%, respectively. The addition of NAC to the incubation mixture containing AAP significantly enhanced the inhibitory effect on cleaved caspase-1. In the AAP plus NAC group (only 0.5 mM in combination) the cleaved caspase-1 protein was significantly (P < 0.05) reduced compared with the NAC (0.5 mM) group.
Effects of AAP and NAC on LPS-induced active casapse-1 production by blood monocytes of piglets in vitro. (A–C) Cells (5 × 106 cells/ml) were pretreated for 1 h with different level of drugs, and then stimulated for 10 h with LPS (1 µg/ml). The levels of active caspase-1 in the supernatant were measured by Western blot analysis. (D) The relative level of active caspase-1 was measured using an image analyzer. The values presented are the means ± SD of three independent experiments, and differences between mean values were assessed by the LSD method. ##P < 0.01 vs. control; *P < 0.05 vs. AAP alone; **P < 0.01 vs. AAP alone; ΔP < 0.05 vs. NAC alone.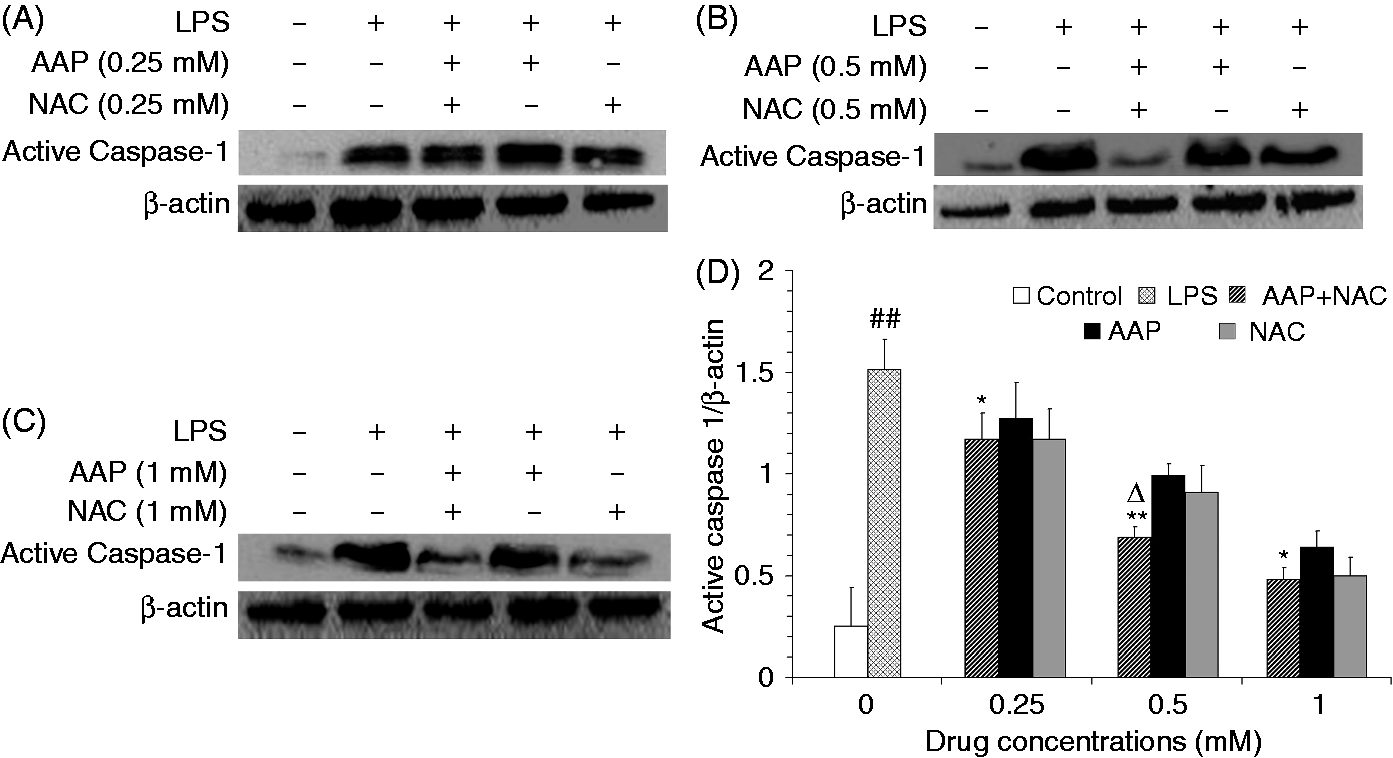
Discussion
The model of immunological stress provided by LPS-challenged (in vitro or in vivo) is generally used to increase the synthesis and release of excessive inflammatory mediators and pro-inflammatory cytokines,4,6,21 and also causes liver injury and intestinal function damage.7,22,24–26 It is clear that LPS stimulation activates the signaling pathway in monocytes/macrophages by acting on TLR-4 to activate NF-κB, and consequently activates the inflammatory gene expression and releases of mediators/cytokines.4,27 It had been reported that LPS stimulation alone enhanced the mRNA expression of NLRP3 in C57BL6/J macrophages and human PBMCs.17,18 The present study showed that LPS stimulation enhanced the mRNA expression of NLRP3 and the protein level of mature IL-1β, IL-18 and cleaved caspase-1 in a piglet monocyte model, which illustrated that NLRP3 inflammasome pathway may also play a role in immunological stress of piglet.
The IL-1 family of cytokines (IL-1β and IL-18) plays a vital role in promoting a variety of innate immune processes associated with infection, inflammation and autoimmunity.28,29 The generation of mature IL-1β and IL-18 is tightly controlled by inflammasomes, of which the NLRP3 inflammasome has been the most intensively studied. NLRP3 activation is intimately linked to NF-κB activation. 30 Upon sensing danger signals, such as LPS, NLRP3 proteins oligomerize and recruit caspase-1 through ASC. Subsequently, pro-caspase-1 undergoes an autocatalytic activation. Finally, mature caspase-1 cleaves pro-IL-1β and pro-IL-18 to produce mature IL-1β and IL-18.12,31
Inflammasome complexes control the regulated cleavage of pro-IL-1β and pro-IL-18 by assembling a multicomponent protein platform. So far, several proteins have been described that can initiate the formation of inflammasome complexes: the NLR proteins NLRP1, NLRP3 and LRC4, and the pyrin and HIN200 domain-containing protein AIM2. The NLRP3 inflammasome is critically dependent on priming because NLRP3 is expressed at limiting levels in unprimed macrophages. It had been reported that LPS stimulation alone (200 ng/ml) highly enhanced the mRNA expression of NLRP3 in C57BL6/J macrophages, but enhanced neither the expression of NLRP1 nor that of NLRC4, AIM2, ASC or caspase-1. 17 LPS stimulation alone (1 µg/ml) highly enhanced the protein expression of NLRP3 in human PBMCs, but did not activate the expression of ASC. 18 As expected, our present results showed that LPS stimulation (1 µg/ml) led to up-regulation of mRNA expression of NLRP3 in piglet blood monocytes in vitro, but had no effects on the mRNA expression of ASC and caspase-1. These results were in line with the report of Bauernfeind et al. 17
The NLRP3 inflammasome is tightly controlled by a priming step that is dependent on the transcription factor NF-κB. 32 Activation of NF-κB leads to cytokine secretion and activation of the NLRP3 inflammasome. TNF-α can activate downstream of the TLR–NF-κB signaling pathway, which plays an important role in rapid response to inflammatory immune stress. 30 ROS are also key signaling factors for the regulation of NLRP3 inflammsome activation. ROS inhibitors block NLRP3 inflammasome activation by interfering with the priming step, which is required to induce NLRP3 expression, whereas direct NLRP3 activation is not affected. 17 The present study showed that LPS stimulation significantly enhanced the mRNA expression and protein production of TNF-α, and suggested that TNF-α promotes activation of the NLRP3 inflammasome, at least in part, through activation of NF-κB. At the same time, AAP and NAC used alone or in combination could significantly reduce mRNA expression and the protein production of TNF-α.
In veterinary medicine, one AAP premix (Pracetam; Sogeval, Laval, France) is used to control the symptoms of fever associated with respiratory diseases at different growing stages of pigs. The present results show that addition of AAP to LPS-induced monocytes in vitro could significantly reduce the production of TNF-α, IL-1β and IL-18, and also inhibit the mRNA expression of these cytokines, which indicates that AAP had a certain anti-inflammatory effect. The present study showed, for the first time, that the addition of AAP (0.5–1.0 mM) could significantly reduce the mRNA expression of NLRP3 and the level of cleaved caspase-1, which differs from the previously published result that up-regulation of NLRP3 inflammasome occurred when AAP was used at a toxicity dose. 33 This result suggests that AAP inhibited activation of the NLRP3 inflammasome, at least in part, through reduce the production of TNF-α. We thus speculate that inhibition of NLRP3 inflammasome activation is one of the anti-inflammatory mechanisms employed by AAP, and AAP has different effect on NLRP3 inflammasome at different doses. This may be an important finding and may have value in practical applications combining the use of NAC and AAP with the label dose.
Recently, the production of ROS has been suggested to act as a common event upstream of the NLRP3 inflammasome machinery.
17
ROS inhibitors have been reported to potently inhibit the activation of NLRP3. NAC, as a scavenger of ROS regardless of the source of production, efficiently inhibits the expression of both NLRP3 and pro-IL-1β.
31
Previous studies in our laboratory have indicated that dietary NAC supplementation could inhibit the production of cytokines in vitro and in vivo,
4
and had protective effects on intestinal functions of piglets challenged with LPS;7,8 they also showed that NAC supplementation did not have a significant effect on the pharmacokinetics of AAP with the common dose in piglets.
34
In the present study, the results showed that the addition of NAC at 0.5–1.0 mM to LPS-induced monocytes could significantly reduce the production of IL-1β, and IL-18, and inhibit the mRNA expression of IL-1β, IL-18 and NLRP3, which also illustrated that NAC exhibited a systemic anti-inflammatory effect on the NLRP3 inflammasome pathway. These results might be helpful in suggesting the important molecular mechanism of AAP and NAC that reduces inflammation. A model of the anti-inflammatory mechanism of AAP and NAC based on the present work is provided (Figure 12).
Proposed anti-inflammatory mechanism of AAP and NAC in LPS-stimulated piglet mononuclear phagocytes.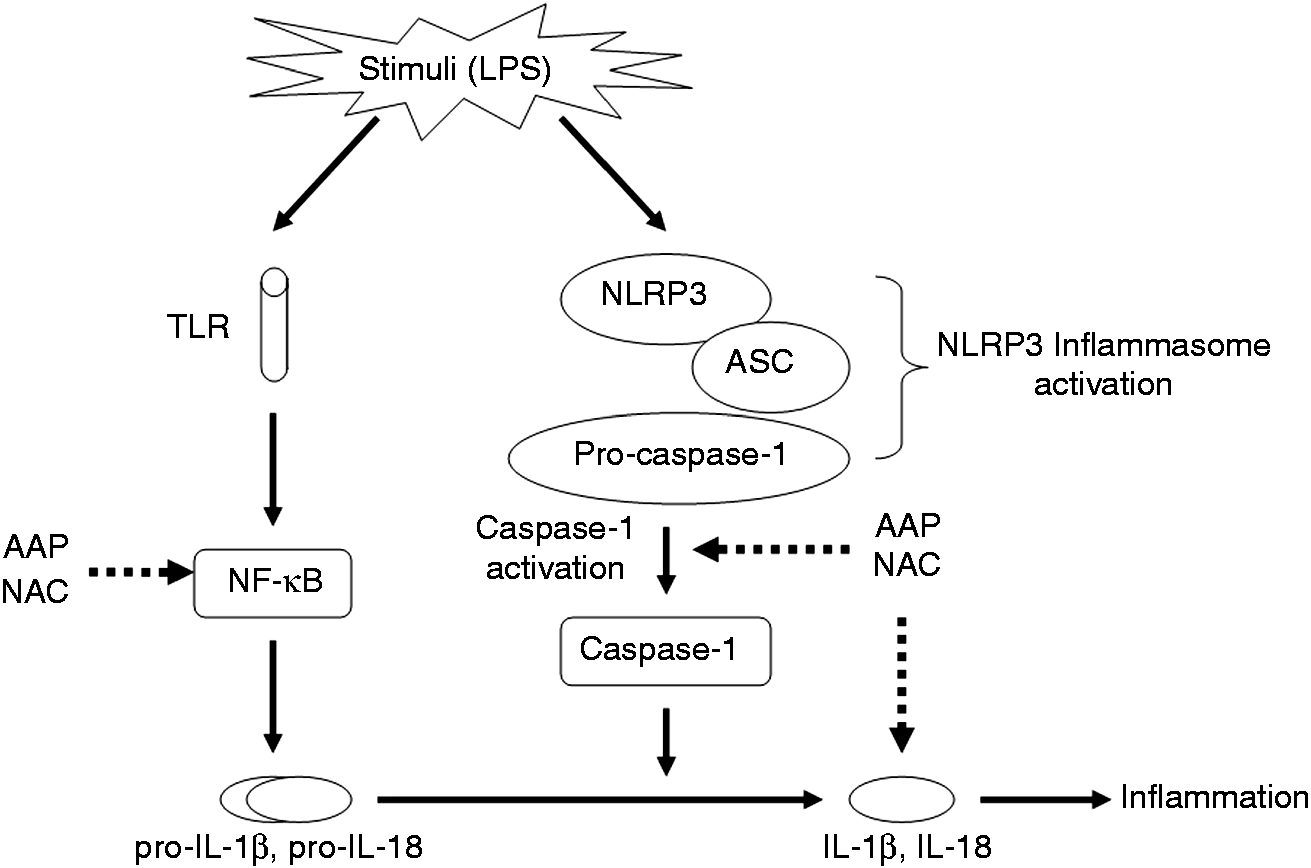
Caspase-1 is a member of a family of caspases with large prodomains, 26 and its activation induces inflammatory response via the production of pro-inflammatory cytokines and the recruitment of neutrophils.35,36 In the present study, the addition of AAP and NAC alone could reduce cleaved caspase-1 production in a dose-dependent manner, which showed that AAP and NAC could hinder the formation of active caspase-1. This result indicates that the regulatory effects of AAP and NAC on the inflammatory response might be partly attributable to the regulation of caspase-1 activation via NLRP3. We postulate that the anti-inflammatory effects of AAP and NAC are mediated by inhibition of the mRNA expression of TNF-α and NLRP3, and lead to suppression of the activation of caspase-1. It may be possible that the drugs act on one or all of the above process. Further studies will be carried out to determine the likely mechanism.
Our previous study showed that AAP plus NAC significantly suppresses the release of pro-inflammatory cytokines and NF-κB activation in LPS-challenged piglets. 4 The present study showed that AAP and NAC had better effects on NLRP3 inflammasome pathway in LPS-stimulated piglet mononuclear phagocytes. AAP and NAC could significantly inhibit the mRNA expression of IL-1β and IL-18, which indicated that the anti-inflammatory effect of AAP and NAC could be attributed to the suppression of the TLR–NF-κB pathway. Meanwhile, this study also confirms that AAP and NAC can suppress mRNA expression of NLRP3 and caspase-1 activation in LPS-stimulated piglet mononuclear phagocytes. This combination inhibition effect could be the result of the following possible mechanisms: in addition to the inhibition effect of AAP and NAC on TNF-α production, scavenging of ROS by NAC may also play a role in inhibition on mRNA expression of NLRP3. As a whole, the results of the inhibition effects of AAP and NAC on IL-1β, IL-18, caspase-1 and NLRP3 illustrated that AAP and NAC could alleviate the immune stress-induced inflammation by inhibiting both the TLR–NF-κB pathway and the NLRP3 inflammasome pathway.
In conclusion, AAP or NAC administrated individually had significant inhibitory effects on the production of cytokines, the mRNA expression of NLRP3 and the activation of caspase-1 in LPS-challenged piglet mononuclear phagocytes. The combined use of AAP and NAC had a better effect on the NLRP3 inflammasome pathway. These anti-inflammatory effects of AAP and NAC appear to be produced by the inhibition of LPS-induced NF-κB activation and mRNA expression of NLRP3, and subsequently inhibition of caspase-1 activation. The anti-inflammatory activities of AAP and NAC may be attributed to the regulation of the TLR–NF-κB and NLRP3 inflammasome pathways.
Footnotes
Funding
This study was supported by the National Natural Science Foundation of China (grant no. 30972213, 31372319); and National Basic Research Program of China (grant no. 2012CB126305).
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
