Abstract
Crohn's disease activates the inflammatory reactions to induce intestinal disorders. Enteral nutrition (EN) could exert general immunomodulatory effects. Cecal ligation and perforation (CLP) surgery was utilized to establish Crohn's disease mice models. Survival analysis, hematoxylin-eosin staining, flow cytometry, ELISA, Western blot and liquid chromatography-tandem MS were applied. Baicalein was added to inhibit lipoxygenases. The survival rate was restored and inflammatory injury, exudate neutrophils in peritoneal lavage and serum levels of IL-6 and TNF-α were ameliorated by EN treatment as compared with CLP treatment. EN also increased ILC-3 content, 5/15-LOX level and RvD1-RvD5 in peritoneal lavage. Baicalein reversed all the detected effects of EN except ILC-3 content. EN could activate special pro-resolving mediators (SPMs) through ILCs to mitigate injuries of Crohn's disease.
Keywords
Introduction
Crohn's disease is a chronic inflammatory disease in the gastrointestinal tract which is originated from diverse pathogenesis such as inherent, environmental factors, transposition of microbiota or unbalanced immune response with inflammatory infection. 1 Earlier therapies using aminosalicylates, steroids or thiopurine were just applied for controlling the progression of Crohn's disease while failing to avoid surgery interference. 2 So more efforts should be made to resolve the bowel damage and the complications to alleviate persistent histological lesion and recurrence. 2
Resolution is considered as a novel progression to terminate the inflammation and restore the tissue homeostasis, instead of only promoting acute inflammatory response which could lead to the persistent tissue injury. 3 Special pro-resolving mediators (SPMs) are produced among the progression of acute inflammatory response and they are underlined to be essential self-limiting factors that could be benefit to the clearance of inflammatory cells and tissue debris to reconstruct the homeostasis of internal environment.4–7 Polyunsaturated fatty acids (PUFA) are released from membrane phospholipids to arrive at the inflammatory sites and then are transformed into SPMs via exudate immunocytes. 8 SPMs are generated from PUFA eicosapentaenoic acid (EPA) or docosahexaenoic acid (DHA) in a lipoxygenase(LOX)-dependent manner through leukocyte-derived 5-LOX or platelet-derived 12-LOX in vasculature and leukocyte-derived 5-LOX or epithelial, eosinophil or monocyte derived 15-LOX.9,10 New bioactive docosanoids including 17-(R)-hydro(peroxy)-docosahexaenoic acid can be generated in mice after the combination treatment of acetylsalicylic acid (ASA) and DHA, and then can be transformed into resolvins D1-D4 under the modulation of cyclooxygenase-2.11,12 The synthesis of resolvins E series is similar to resolvins D series that the intermediate productions including 18-(R)-hydro(peroxy)-eicosapentaenoic acid could be generated under the combination treatment of ASA and EPA..11,13,14 Then the intermediate productions could be transformed into resolvins R series owing to the catalyzation of ASA-cyclooxygenase-2..11,13,14 Collectively, SPMs and 5/12/15-LOX shed light on acting as agonists in inflammatory response at a very low dosage to limit the duration and magnitude of inflammation,15,16 while they also overlap the function of antagonists that would ameliorate the disease.15,16
Resolvins, protectins and maresins are essential components of SPMs, which are instrumental to help the limitation of permeation of polymorphonuclear leucocytes (PMN) into tissue and the efferocytosis of PMN or macrophage as well as the removal of cellular debris. 7 The eliminated immune cells would alter the production of cascade reactions and prohibit the transformation of acute inflammation into chronic inflammation. 7 The biological effects of D-series resolvins which were derived from DHA were of multivariate functions. 7 For example, resolvin D1 (RvD1) had pro-protective function on the LPS-induced acute pulmonary impairments as well as the inflammation of central nervous system. RvD1 can partly block the NF-κB activation along with the following signaling targets. 17 Additionally, resolvin D2 (RvD2) could interfere the interaction of leucocyte with endothelial cells and promote the intra-phagosomal vacuolar production of reactive oxygen species to improve the survival rate of septic mice induced by cecal ligation and perforation (CLP). 18 Resolvins D3 (RvD3) could prohibit the infiltration of PMN into tissue and incite macrophages to clear the apoptotic cells as well as debris as mentioned above. Thereby, SPMs have novel self-limiting immunosuppressive behaviors which are optimized for organ defense and self-recovery. 19
Hence, we determined to explore the effects of enteral nutrition (EN) on the resolution of inflammation and the following mitigation of the inflammatory injuries in Crohn's disease. That may help release the patients’ pain and maintain the homeostasis.
Materials and methods
Animals, drugs, and groups
Seventy-five male C57BL/6J mice (specific pathogen-free grade, 7-wk-old, weighing 15–18 g, strain code: 219) were purchased from Vital River Laboratory Animal Technology Co. Ltd (Beijing, China). The mice were housed at 25°C (room temperature) under a 12 h light/12 h dark cycle and allowed for free food or water for 7 d (d) before the operation. The procedures were approved by Shanghai Tenth People's Hospital, Tongji University School (No. GMEN201706075). For minimizing animal suffering, all surgical resections were performed under anaesthesia induction with 50 mg/kg sodium pentobarbital (P5178, sigma-Aldrich, St Louis, Missouri, USA) and all animals were sacrificed by cervical dislocation under deep anaesthesia using 70 mg/kg sodium pentobarbital.
Baicalin (BAI; S2269) is an inhibitor of prolyl endopeptidase, which is soluble in water and could be stored at −80°C for 2 yr. The BAI adopted in our experiments was obtained from Selleck Chemical (https://www.selleck.cn/products/Baicalin.html, Huston, TX, USA), with a dosage of 10 mg/kg/d. 20
The mice were randomly separated into five average groups as follows: Sham group, Model group, EN group, another EN group and EN + BAI group (the controlled trial was conducted in the first three groups and the last two groups, each group contained 15 mice). Sham group or Model group mice were allowed for free diet, while EN group or EN + BAI group mice were fed with EN liquor (TPF, Nutricia pharmaceutical Co. Ltd, Shanghai Branch, China). No mice were dead during the process of establishing EN model. The number of subsequent deaths during the whole experimental process was counted for the calculation of survival rate of mice in each group.
Surgical procedure and collection of peritoneal lavage liquid
Mice were fasted 8 h before the surgery but were given free access to water. Under sterile conditions, mice were fixed and intraperitoneally injected with 50 mg/kg sodium pentobarbital. Concretely, the skin of mice was first disinfected with 75% ethanol (A500737, Sangon Biotech, Shanghai, China) and then an incision was created in the mid abdomen of mice. After that, mice in Model group, two EN groups and EN + BAI group underwent CLP to induce abdominal abscesses to mimic complication of Crohn's disease. The procedures of CLP were as follows: the content in cecum was pushed into distal end and the middle site of proximal and distal cecum was ligated by 5-0 silk purse string suture (Boda medical products Co. Ltd, Shandong, China). The cecal wall at distal part was perforated twice using affiliated sterile no. 5 needle. The content was pressed to exudate the cecal wall to make sure the open of perforation. Cecum was returned back into abdomen and the abdominal wall was sutured subsequently. 0.8 ml normal saline solution (ST341, Beyotime, Shanghai, China) was injected into each mouse soon after the antagonize shock. The corresponding surgical operations in mice of Sham group include opening abdominal wall and finding cecum without the ligation and perforation of cecum.
The EN group mice were inserted with silastic catheter provided in a professional gastrostomy tube package (Nutricia pharmaceutical Co. Ltd, Shanghai Branch, China). Mice were fasted 8 h before surgery. Then under anesthetic conditions, the abdominal skin of mice was disinfected, and the abdominal wall was opened. The stomach was opened to create an artificial fistula. The sterile catheter was inserted into the fistula via the neck and back subcutaneous tunnel and put at 0.5 cm under the pylorus. The catheter was fixed with medical adhesive (Beijing FuAiLe Science and Technology Development Co. Ltd, Beijing, China) at stomach wall, abdominal wall as well as neck and back skin. The EN liquid was provided using microinfusion pump (BF100-02, Qili constant current pump Co. Ltd, Baoding, Hebei, China) and the dose was 8–10 ml/d. Half amount of EN liquid was provided for EN group mice in the first d and afterwards whole amount of EN liquid was offered.
The mice in EN + BAI group were intraperitoneally injected with BAI at 10 mg/kg/d after the supply of EN liquor. The peritoneal lavage liquid of mice in each group on d 0, 2, 4, 6, and 8 was collected via the peritoneal injection of 4 ml normal saline solution and the extraction of the fluid soon afterwards. The peritoneal lavage liquid was immediately stored at −80°C for the following investigations.
Histomorphology
Three mice were randomly selected from Sham group, Model group, EN group and another EN group as well as EN + BAI group and 15 mice in total. After the sacrifice of mice at the 8th d, the colon tissues at the same sites were cut and immediately fixed with 4% paraformaldehyde (P0099, Beyotime) for 24 h, followed by the hematoxylin-eosin staining (HE stain) using related staining kit (C0105S, Beyotime).
Subsequently, the tissue was made into sections with a thickness of 4 μm. Next, the sections were deparaffinized using xylene (A530011, Sangon) for 5 min and fresh xylene for another 5 min. Then the sections were treated with absolute ethanol, 90%, 80%, and 70% ethanol and distilled water for 2 min, respectively. Afterwards, sections were stained with hematoxylin staining solution for 5 min and washed. Then the sections were differentiated using acid ethanol (C0161, Beyotime) for 10 s, and washed once again. Following that, the sections were stained with eosin staining solution for 1 min. Finally, sections were dehydrated using increasing concentrations of ethanol, permeabilized by xylene and mounted in neutral gum (N116470, Aladdin, Shanghai, China) for detection. Pictures were taken by microscope at 40 × and 100 × magnification separately (Biozero BZ-8000, Keyence, Osaka, Japan).
ELISA
Three mice were randomly selected from Sham group, Model group, EN group and another EN group as well as EN + BAI group and 15 mice in total. The blood of mice in each group at d 4 was collected through withdrawing tail vein blood and then incubated at room temperature for blood coagulation. The supernatant was centrifuged at 2000 g for 10 min. The supernatant of centrifuged liquor was transferred into sterile centrifuge tube to obtain serum. The serum was immediately stored at −80 °C for the subsequent investigations.
The concentrations of IL-6 and TNF-α in collected mice serum were measured using ELISA kits (PI326, PT512, Beyotime). Generally, standard control reagents, wash buffer and substrate solution were pre-prepared. Then mother concentration standard liquor was diluted using referred calibrator diluent. The final concentration of IL-6 standard liquor was 1000 pg/ml, and it was diluted to serial concentrations of 500, 250, 125, 62.5 and 31.25 pg/ml subsequently. The final concentration of TNF-α standard liquor was 2000 pg/ml and it was diluted to serial concentrations of 1000, 500, 250, 125 and 62.5 pg/ml respectively. The pure calibrator diluent was used as 0 pg/ml standard control. Then pre-coated reaction well was taken out and fixed on plates.
Various concentrations of standard control liquor and pre-diluted samples were added with a volume of 100 μl. The plates were sealed with provided adhesive strips and incubated at room temperature for 2 h. The plates were washed with washing buffer for 5 times and then patted to dryness using sterile absorbent papers. Then 100 μl biotinylated anti-IL-6 or anti-TNF-α was added and the plates were sealed and incubated at room temperature in the dark for 20 min away from light. The plates were patted to dryness again and added with 100 μl HRP-conjugated streptavidin. The plates were sealed and incubated at room temperature in the dark for another 20 min and patted to dryness. The 100 μl TMB chromogenic reagents were added to each well and the plates were sealed and incubated at room temperature in the dark for 20 min. Finally, 50 μl stop solution was added to each well and the solution was mixed, followed by the measurement of absorbance at 450 nm using xMark Microplate absorbance spectrophotometer (#1681150, Bio-red, CA, USA). The serum level of IL-6 or TNF-α was calculated according to the established standard curve.
Western blot (WB) assay
Three mice were randomly selected from Sham group, Model group, EN group and another EN group as well as EN + BAI group and 15 mice in total. Total protein in sorted ILC-3 cells (the procedure is mentioned below) was extracted through radio immunoprecipitation assay (RIPA) lysis buffer (P0013B, Beyotime) added with protease and phosphatase inhibitor (P1051, Beyotime) along with 0.1 mM phenylmethylsulfonyl fluoride (PMSF, ST506, Beyotime). Bicinchoninic acid (BCA) protein assay kit (P0010, Beyotime) was served to detect protein concentration. All protein samples to be used in one experiment were diluted to the same concentration. Then equal amounts of proteins were subjected to SDS-PAGE (P0012A, Beyotime), and transferred onto polyvinylidene fluoride (PVDF) membranes (IPSN07852, Millipore, Bedford, MA, USA). The PVDF membranes were blocked with 5% nonfat milk for 1 h and then cut into strips containing different protein bands according to protein marker. Strips were incubated with primary Abs against 5-LOX (ab169755, 78 kDa, dilution 1:1000, Abcam, Cambridge, MA, USA), 15-LOX (ab23691, 76 kDa, dilution 1:200, Abcam) and GAPDH (ab181602, 36 kDa, dilution 1:10000, Abcam) at 4°C overnight, and then washed by 0.1 M TBS (containing 0.1% Tween-20, ST673, Beyotime) for three times. Finally, strips were incubated with HRP-conjugated secondary Abs (ab205718, dilution 1:5000 Abcam) at room temperature for 2 h. Specific protein signals were tested using UltraSignal ECL Western Blotting Substrate (4AW011-200, 4A Biotech, Beijing, China) and quantified using Image J software (Wayne Rasband, NIH, USA).
Flow cytometry
Three mice were randomly selected from Sham group, Model group, two EN groups as well as EN + BAI group and 15 mice in total. Cells in peritoneal lavage liquid were enriched through low-speed centrifugation and re-suspension. Abs used in the subsequent experiments were purchased from BioLegend (San Diego, CA, USA) except specialized instructions. To test the neutrophils in peritoneal lavage liquid, enriched cells were incubated with PE-anti-Ly6G (no: 127607, clone: 1A8) and PE-anti-CD11b (no: 101207, clone: M1/70). The exudate neutrophils were detected using FACS Aria II (BD Biosciences, San Jose, CA, USA) and FlowJo software (Stanford University, San Francisco, CA, USA).
To sort ILC3 cells in the enriched cell solutions, the solutions were incubated with LIVE/DEAD fixable yellow dead cell stain (L34959, Invitrogen, Carlsbad, CA, USA). Then cells were incubated with lineage specific markers in staining solution at 4°C for 30 min following Ab combination: APC-anti-NK1.1 (no: 108709, clone: PK136), anti-RORγt (no:562197, clone: O28-835, BD pharmingen, San Diego, CA, USA), Brilliant Violet (BV) 650-anti-CD127 (no: 135025, clone: A7R34), PE-Cy7-anti-CD117 (no: 105813, clone: 2B8), PerCP-anti-IL23R (no: FAB16861C, clone: #753317, R&D System, distributer, Shanghai, China) and BV421-anti-CD335 (no:137611, clone: 29A1.4) Abs. ILC3 cells were sorted using FACS Aria II and FlowJo software as well. The gate strategies demonstrated as follows were used to validate the ILC-3 population with percentage values denoting the proportion of cells within the gate from the parent cell population: after gating for live, lineage negative and NK1.1 low-expressed cells, the cells were gated for RORγt positive coupled with CD127 positive, CD117 positive coupled with IL-23R positive, and CD335 positive coupled with IL23R positive. Finally, the expression of CD335 was determined. 21 IL-23R is the receptor of IL-23, which is the plastic feature of ILC-3 and is associated with the ILC-3 common marker IL-17.
Liquid chromatography-tandem MS (LC-MS-MS)
Three mice were randomly selected from Sham group, Model group, EN group and another EN group as well as EN + BAI group and 15 mice in total. The concentrations of resolvin serial molecular RvD1-RvD5 in concentrated peritoneal lavage liquid were detected using LC-MS-MS determination. Concentrated peritoneal lavage liquid was placed in 2 volumes of pre-cooling methanol (A601617, Sangon) and centrifuged at 1200 g and −20°C for 10 min and the supernatants were collected. The supernatants were brought to a gentle stream of nitrogen gas with less than 1 ml methanol content onto an automated evaporation system (TurboVap LV, Biotage, Uppsala, Sweden). Products for LC-MS-MS-based metabololipidomics in sample were subsequently extracted using automated extraction system (RapidTrace, Biotage) with C18 solid-phase extraction columns (500 mg, Biotage, Uppsala, Sweden). The C18 columns were equilibrated with 3 ml methanol and 6 ml double distilled water. Columns were then loaded with sample solutions coupled with 9 ml HCl water solution (pH 3.5) and the acid was neutralized with 4 ml water. After that, 5 ml hexane (A530007, Sangon) was supplemented and the samples were then eluted with 9 ml methyl formate (M103448, Aladdin) and evaporated to dryness through automated system followed by re-suspension in 50:50 (v:v) methanol water solution for LC-MS-MS automated injections. 22
Eluted methyl formate fractions were analyzed using Qtrap 5500 LC-MS-MS system (AB Sciex, Framingham, MA, USA) supplemented with Shimadzu SIL-20AC autoinjector (Shimadzu, Kyoto, Japan) and LC-20AD binary pump (Shimadzu) and the LC column applied in the experiment was Agilent Eclipse C18 column (100 mm × 4.6 mm × 1.8 μm, Agilent, Santa Clara, CA, USA). The C18 column was placed at column oven (ThermaSphere TS-130, Phenomenex, Torrance, CA, USA) to maintain 50°C column temperature. The deuterated internal standards (RvD1-5) representing regions of interest in chromatographic analysis (500 pg each sample) which were beneficial to conduct quantification were added. The procedures in LC were set as follows: initially, a gradient of methanol/water/acetic acid (acetate, A298827, Aladdin) was set at 55:45:0.01 (v:v:v), and then it was ramped to 85:15:0.01 (v:v:v) for over 10 min followed by being changed to 98:2:0.01 (v:v:v) for 8 min. The procedure was ultimately maintained at 98:2:0.01 (v:v:v) for 2 min and the flow rate was 0.4 ml/min in the whole progression.21,22
Statistical analysis
All groups of data were analyzed by SPSS20.0 (IBM Corporation, NY, USA). The entire results of multiple independent experiments were exhibited as the form of mean ± SD. The significance of differences between two groups was analyzed by independent-samples t-test, while that among multiple groups was analyzed using one-way analysis of variance (ANOVA). Tukey's multiple comparisons test were carried for post hoc test P < 0.05 was considered as statistical significance.
Results
EN treatment restored the survival impairment and colon inflammation in CLP treatment and inhibited the exudate neutrophils and inflammatory factors
CLP treatment was used to mimic Crohn's disease and inhibited the survival rate of C57BL/6J mice while EN treatment restored the survival rate in the period of experiment (Figure 1A). The inflammatory injury was induced by CLP treatment while it was ameliorated by EN treatment (Figure 1B). EN treatment significantly inhibited the exudate neutrophils in peritoneal lavage liquid (Figure 1C, P < 0.001). CLP treatment increased the expressions of inflammatory factors including IL-6 and TNF-α, while EN treatment significantly mitigated the overexpressed IL-6 and TNF-α (Figure 1D, P < 0.001).
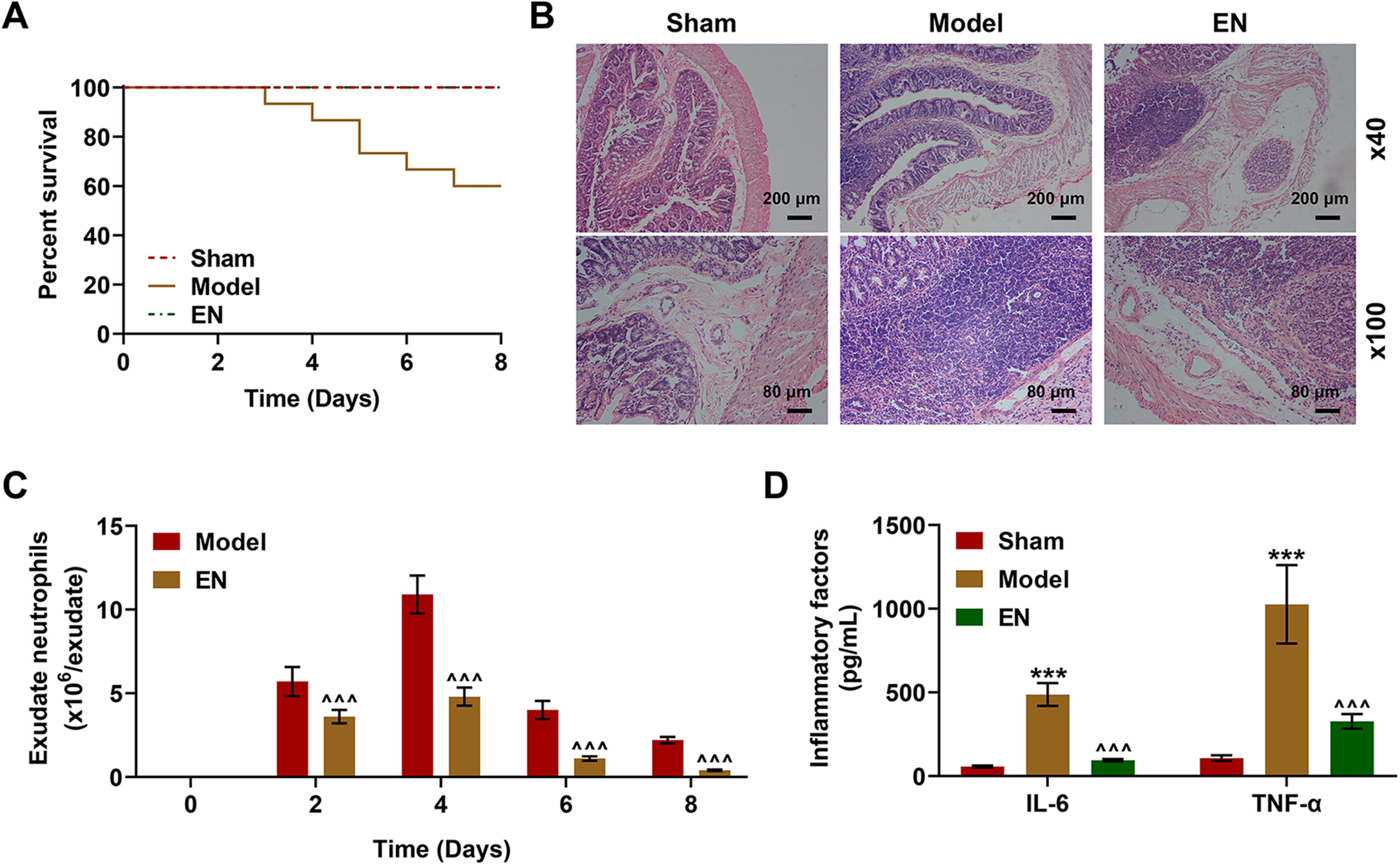
EN decreased the expressions of inflammatory cytokines and ameliorated the inflammatory injury of colon. (A) CLP decreased the survival rates of C57BL/6J mice, while EN treatment rescued the survival rate. (B) HE staining indicated that CLP surgery induced severe inflammatory injury of colon, while EN treatment mitigated the inflammatory injury (3 mice were randomly selected from each group). (C) Flow cytometry indicated that EN treatment remarkably decreased the exudate neutrophils in the peritoneal lavage fluid of mice (3 mice were randomly selected from each group). ^^^P < 0.001 vs. Model. Results of triplicate determinations were presented as means ± SD. (D) ELISA indicated that CLP treatment significantly increased the serum level of IL-6 and TNF-α while EN treatment reversed the effects (3 mice were randomly selected from each group). ***P < 0.001 vs. Sham, ^^^P < 0.001 vs. Model. Results of triplicate determinations were expressed as means ± SD.
EN treatment increased the D-series resolvins expressions via up-regulated 5/15-lipoxygenase
CLP treatment increased the expressions of 5-LOX and 15-LOX and the increasing trends as such were further promoted by EN treatment (Figure 2A, P < 0.001). In groups with EN treatment, expressions of the synthesis of SPMs including docosahexaenoic acid-derived resolvins D1-D5 were remarkably increased compared with those in model group (Figure 2B, P < 0.001). CLP treatment improved the enrichment of CD335 positive ILC-3 cells in peritoneal lavage liquid and the enrichment as such was further enhanced by EN treatment (Figure 2C–D, P < 0.001)
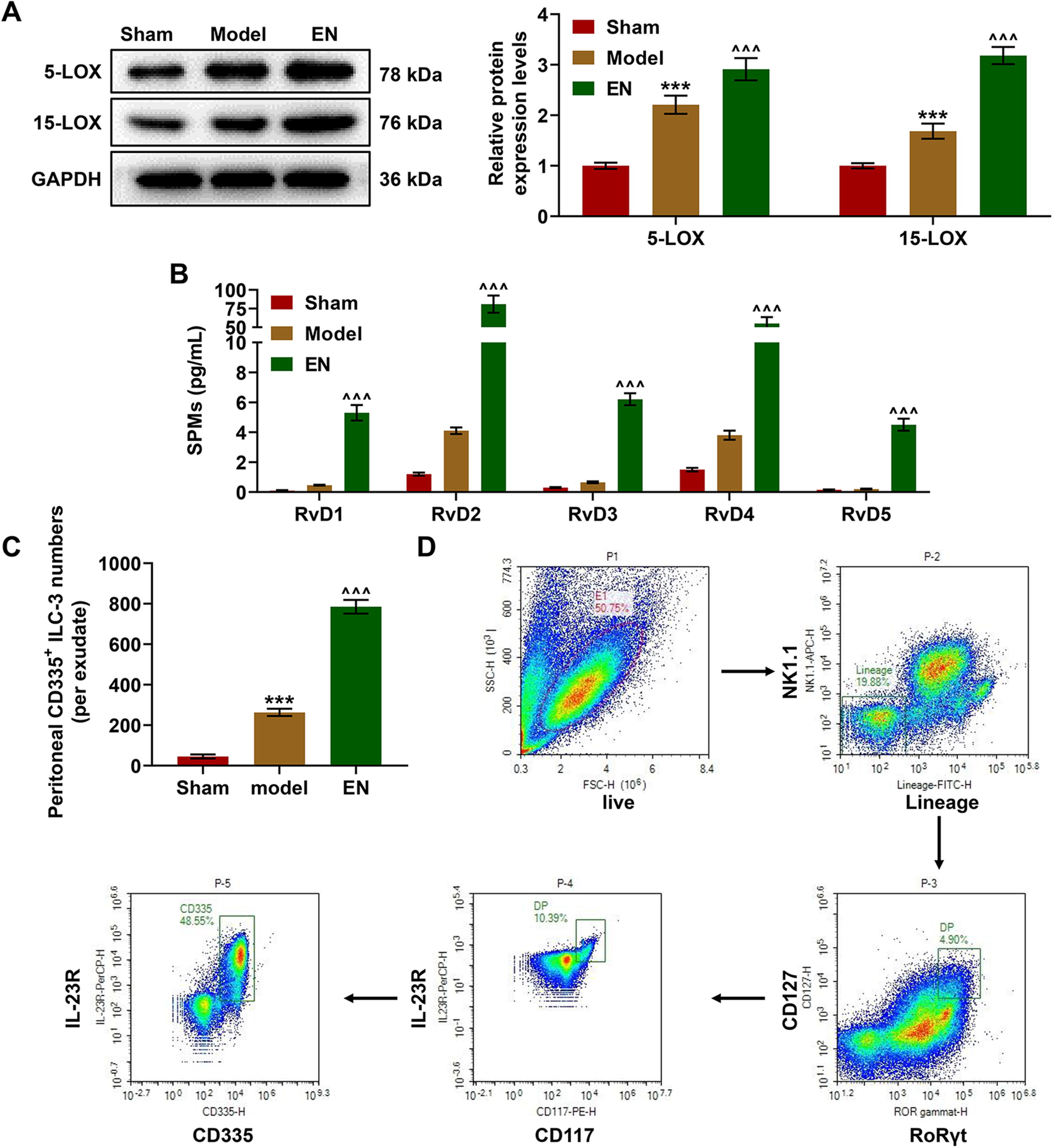
EN increased the expressions of lipoxygenases and special pro-resolving mediators (SPMs). (A) WB assay showed that CLP treatment significantly increased the protein expressions of 5-LOX and 15-LOX in peritoneal lavage fluid and EN treatment continued to promote the expressions of these two lipoxygenases (Random selected 3 mice in each group). ***P < 0.001 vs. Sham, ^^^P < 0.001 vs. Model. Results of triplicate determinations were presented as means ± SD. (B) LC-MS-MS showed that the contents of RvD1-RvD5 in peritoneal lavage fluid were significantly increased under the treatment of EN (3 mice were randomly selected from each group). ^^^P < 0.001 vs. Model. Results of triplicate determinations were expressed as means ± SD. (C) The FACS flow cytometry sorted the ILC-3 cells and the numbers of peritoneal CD335 positive ILC-3 cells were increased in CLP models and the results were further enhanced by EN treatment (3 mice were randomly selected from each group). ***P < 0.001 vs. Sham, ^^^P < 0.001 vs. Model. Results of triplicate determinations were described as means ± SD.
BAI reversed the effects of EN treatment on promoting survival rate, and decreasing inflammatory injury, exudate neutrophils and inflammatory factors
BAI was adopted as an inhibitor of LOX in the following experiments. BAI reversed the survival rate of EN group mice during experiment (Figure 3A). The inflammatory injury of mice in BAI group was more serious than that of mice in EN group (Figure 3B). BAI promoted the neutrophils exudation in peritoneal lavage liquid in all experimental period was promoted in mice of BAI group as compared with that in mice of EN group (Figure 3C, P < 0.001). BAI induced higher IL-6 and TNF-
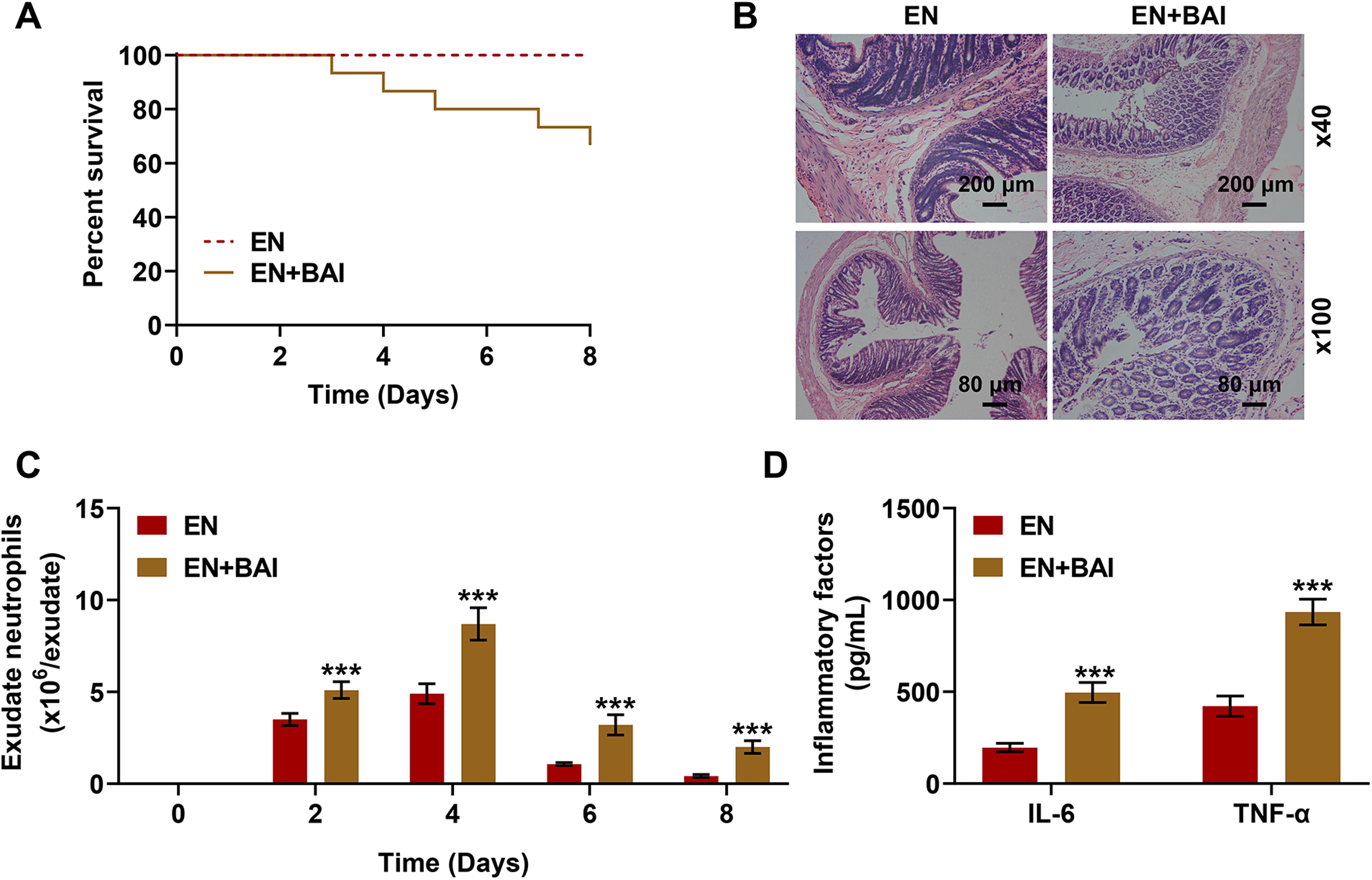
Lipoxygenase inhibitor BAI reversed the effects of EN treatment on inflammatory cytokines expressions and colon inflammatory injury. (A) BAI in combination with EN treatment decreased the survival rate of mice compared with EN treatment alone. (B) HE staining indicated that the use of BAI reversed the amelioration of EN treatment on the colon inflammatory injury (3 mice were randomly selected from each group). (C) Flow cytometry indicated that the use of BAI reversed the decrease of exudate neutrophils in peritoneal lavage fluid via EN treatment (3 mice were randomly selected from each group). ***P < 0.001 vs. EN. Results of triplicate determinations were presented as means ± SD. (D) ELISA indicated that BAI usage significantly reversed the decreased serum levels of IL-6 and TNF-α through EN treatment (3 mice were randomly selected from each group). ***P < 0.001 vs. EN. Results of triplicate determinations were described as means ± SD.
BAI reversed the effects of EN treatment on increasing 5/15-lipoxygenase and D-series resolvins expressions
BAI reversed the increasing expressions of 5-LOX and 15-LOX in EN treatment (Figure 4A, P < 0.05). Correspondingly, the produce of SPMs including resolvins D1-D5 was inhibited by combined treatment of BAI and EN compared with the treatment of EN alone (Figure 4B, P < 0.001). BAI had no significant influence on the enrichment of CD335 positive ILC-3 cells in peritoneal lavage liquid compared with EN treatment (Figure 4C).
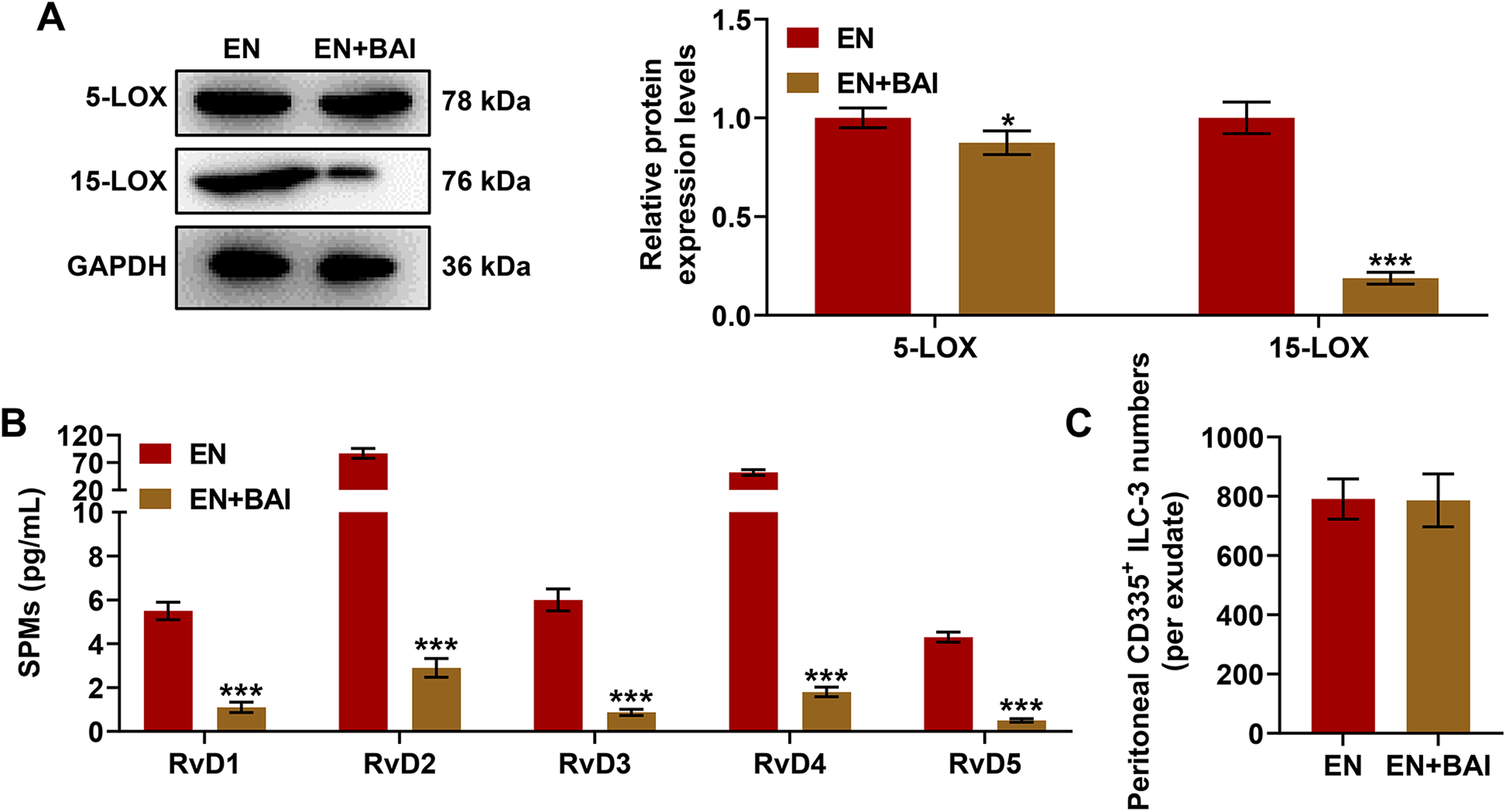
BAI treatment reversed the increased expressions of lipoxygenases and SPMs under EN treatment. (A) WB assay showed that BAI significantly reversed the increased protein expressions of 5-LOX and 15-LOX in peritoneal lavage fluid under EN treatment (3 mice were randomly selected from each group). *P < 0.05 vs. EN, ***P < 0.001 vs. EN. Results of triplicate determinations were presented as means ± SD. (B) LC-MS-MS assay showed that the increased contents of RvD1-RvD5 in peritoneal lavage fluid under EN treatment were significantly reversed via the use of BAI (3 mice were randomly selected from each group). ***P < 0.001 vs. EN. Results of triplicate determinations were presented as means ± SD. (C) The FACS flow cytometry indicated that BAI had no influence on the survival of peritoneal CD335 + ILC-3 cells (3 mice were randomly selected from in each group). Results of triplicate determinations were described as means ± SD.
Discussion
All results above indicated that EN is beneficial to the amelioration of inflammatory injury caused by intestinal barrier dysfunction. We took the survival rate, the production, saturation, and exudation of inflammatory factors into consideration and revealed the superior therapeutic effects of EN treatment in mitigating the inflammatory effects. We further elucidated the underlying mechanism of EN treatment may be involved the resolution of inflammatory reaction by D-series resolvins via the activation of ILC3 s, and the following increased 5-LOX and 15-LOX expressions all mediating in the transformation of precursor DHA into D-series resolvins. The results were ultimately validated in the interruption of the inflammatory resolution process through the inhibitor of LOX.
The EN treatment was directly used for some individuals suffering from eating disorder whose intestinal functions are normal to achieve normal diet. 23 The nasogastric tube feeding is frequently employed for short term enteral feeding while the percutaneous endoscopic gastrostomy tube is provided for long term EN. 24 Many mouse models demonstrated the tight associated relationships between diet and disease, however, this may be not evidenced in human. 25 Specific carbohydrate diet and exclusive EN are two main clinical feeding approaches,the two of which could alleviate the inflammatory symptoms but exert opposite impacts on the biodiversity of microbiome. 26 Meta-analysis has revealed that EN is an independent manner for curing Crohn's disease with well compatible with other therapies while the activity index increased first and then decreased during the long term EN treatment. 27 Our results validated the good efficacy of EN in the Crohn's disease. Meanwhile, the underlying inflammatory mechanisms such as the reduced neutrophils exudation and increased enrichment of inmunomodulatory cells were further explored.
The ILC-3 s enrichment was positively correlated with the expressions of LOXs and downstream immuno-resolvents. In the existence of RORγt and aryl hydrocarbon receptor, the stimulation of IL-23 with or without IL-1β could help ILC-3 produce more Th17 or Th22-like cytokines IL-22, IL-17 and granulocyte M-CSF. 28 They were confirmed as crucial regulator for maintaining tissue homeostasis and remodeling tissue. 29 Nevertheless, the improper activation of ILC-3 could lead to the unlimited production of IL-22, thereby recruiting numerous neutrophils to destroy the normal tissues. 30 So ILC-3 was concluded to be a double-edged sword in inflammatory bowel diseases including Crohn's disease and ulcerative colitis. 31 The ILC3 s were proposed to be regulated by vagal nerves and the disruption of vagal system via vagotomy of right vagal nerve prohibited the amount of ILC3 s and broke the host protective capacity against the Escherichia coli infection in mice. 21 The impairment of vancomycin pre-treatment on the dextran sodium sulfate-induced colitis was shown to be associated with the inhibition of ILC-3 cell population and the inactivation of downstream IL-22/p-STAT3 signaling axis in colon tissues. 32 We found that EN treatment remarkably stimulated the basal enrichment of ILC-3 cells during the inflammatory period caused by CLP treatment and enhanced the enrichment. Therefore, we should determine whether the ILC-3 cell enrichment was benefit to the resolution of inflammation.
We then figured out the expressions of rate-limiting enzymes 5-LOX and 15-LOX and their substrates were both at a basal level after the injection of microbiota from cecal content, whereas the expressions were significantly facilitated by EN treatment. When facing the stimulation of exo-pathogens, systemic M1 or M2 macrophage was stimulated to activate the downstream LOX or cycloxygenase (COX) signaling pathways to produce SPMs including RvD2, RvD5 and maresin 1. 33 The precursors of RvDs and RvEs including DHA and EPA exhibited protective effects in intestinal ischemia and reperfusion model through mediating TLR4 and NF-κB monomer p65. 34 At the meantime, the production of thrombus-promoted SPMs could not be inhibited by common COX inhibitors including celecoxib and indomethacin. 35 The SPMs in human blood mostly targeted leukocytes in single cell level. 35 An intriguing result indicated that a novel benzoxanthene lignan could induce the synthesis of SPMs through interfering 5-LOX rather than pro-inflammatory prostaglandins and leukotrienes and it exhibited potentials in the pharmacotherapy of arthritis. 36 Thereby the enrichment of ILC-3 cells promoted the synthesis of 5/15-LOX and accelerated the production of SPMs. All the results above suggested that EN treatment presented a proclivity towards inflammatory resolution.
Since the excellent performance of SPMs in inflammatory resolving, we speculated whether the supplementation of their origination n-3 PUFA such as DHA or EPA could be benefit for the relieving of inflammatory bowel disease (IBD) symptoms. A literature reported that n-3 supplementation could assist 5-ASA treatment to perform better in reducing the activation of NF-κB, thus reducing 5-ASA dosage in standard therapy of IBD. 37 Aside from ASA, standard treatment for IBD also contains corticosteroids, thiopurine and other immunomodulatory or biological drugs. 38 n-3 Supplementation was summarized to be benefit to the inhibition of inflammation, however, unbalanced ratio of n-6 to n-3 could stimulate the production of C-reactive protein and up-regulate the risk of IBD.39–41n-3 Supplementation was also concluded to be immunosuppressive, but it might induce oxidative stress through recruiting lipid peroxidation and these symptoms should be ameliorated through co-administration of vitamin E. 42 Thus, the clinical application of n-3 PUFA needs further exploration. There was still a common point that n-3 PUFA or SPMs were immunosuppressive.
To sum up, our experiments manifested that EN treatment as an unexpected favorable curing strategy could ameliorate the inflammation induced by Crohn’ disease in the intestinal infection model. In a nutshell, EN promoted the inflammatory resolution of the severe tissue injuries, and it could be applied in clinic to reduce the chronic inflammatory damages and assist other treatments.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number No. 81700480).
