Abstract
Exclusive enteral nutrition is established as an initial therapy to induce remission in active Crohn’s disease (CD), especially in children, but the mechanisms of action of this therapy are yet to be fully defined. CEACAM6 protein is an adhesion molecule that is up-regulated in active CD and implicated in the attachment of adherent-invasive Escherichia coli (AIEC) to the gut epithelium. Using the Caco-2 human adenocarcinoma cell line, this study showed that the incubation of human cells with a polymeric formula (PF) resulted in a dose-dependent increase in the expression of CEACAM6, and that this effect was most noticeable on the cell surface. Further investigation revealed that PF doubled the release of CEACAM6 protein by Caco-2 cells exposed to PF, and that an increase in release of soluble CEACAM6 inversely correlated with the ability of AIEC to associate with the intestinal epithelial cells. Our findings suggest that the secretion of cell surface-associated proteins acting as releasable decoys may be an aspect of the gut’s innate immune response to pathogenic bacteria that is strengthened by PF in the setting of CD.
Introduction
Crohn’s disease (CD) has become increasingly common over recent years, including in children.1,2 The role played by gut flora in the aetiology of CD is supported by studies that demonstrate diminished inflammation following antibiotic and probiotic treatment. 3 Likewise, dysregulation of immune responses (that may be fuelled by a genetically predisposed loss of tolerance by the immune system to the gut microbiota) is also implicated. 4 However, the precise pathogenesis of this chronic disease is unknown, and CD remains incurable. Accordingly, the various treatment options currently available serve only to decrease and/or modulate gut inflammation. 1
Over the last 20 yrs, nutritional therapy [or exclusive enteral nutrition (EEN)] has become established as an effective, non-medicinal treatment to induce remission in active CD, especially in children. 5 This therapy involves the provision of a liquid diet using elemental or polymeric formulae (PF), given exclusively over a prolonged period, usually for 6–8 wks. Meta-analysis of paediatric studies shows that EEN has equivalent efficacy to steroids in the induction of remission. 6 In addition to anti-inflammatory benefits, EEN leads to superior mucosal healing and nutritional improvements, has few side effects and permits avoidance of medication-related side effects. 5 However, although the clinical benefits of EEN have been well defined, 7 the mechanism(s) of these effects remains unclear.
The first line of resistance against gut flora is the epithelial cell barrier and bacteria that are able to bind to the apical surface of the cells forming this barrier are thought to trigger cell signalling pathways that result in weakened barrier function and/or a pro-inflammatory response. Our results show that PF, as used clinically, can completely rescue intestinal barrier function from degradation by a pro-inflammatory stimulus 8 and also has an anti-inflammatory effect. 9 Either (or both) of these effects may reflect an ability of PF to limit bacterial binding to the gut epithelium.
The aim of our study was to determine the effect of PF on carcinoembryonic antigen cell adhesion molecule 6 (CEACAM6) expression in a human colonic epithelial cell line. CEACAM6 is reportedly increased in the ileal mucosa of patients with CD 10 where it acts as a receptor for adherent invasive Escherichia coli (AIEC), bacteria that are implicated in the aetiology of CD. 11 Accordingly, we considered that one function of PF might be to down-regulate the expression of this glycosyl phosphatidyl inositol-anchored protein found on the surface of intestinal epithelial cells. However, we found decreased AIEC adherence to PF-treated cells that correlated with the release of extracellular CEACAM6, which leads us to speculate that up-regulated expression of this cell-surface associated receptor may, instead, serve to protect the gut by acting as a releasable decoy and that this mechanism may be strengthened by PF in the setting of CD.
Materials and methods
Reagents and Abs
Polymeric formula (Osmolite) was sourced from Abbott Nutrition (Christchurch, New Zealand).This nutritional product, comprising whole proteins, provides a complete nutritional intake and is widely used in clinical practice. 7 Human recombinant IFN-γ, Dulbecco’s PBS (D-PBS), BSA, trypsin–EDTA, Hoechst 33342 and Alexa 488 were all obtained from Life Technologies (Mulgrave, VIC, Australia), as were Trizol, DNase I, Superscript III and RNase H. Recombinant human CEACAM6 protein and the CEACAM6 Ab, CD66C (9A6) were bought from R&D Systems (Minneapolis, MN, USA) and Santa Cruz Biotechology (Dallas, TX, USA) respectively. The alkaline phosphatase-, FITC- and HRP-conjugated secondary Abs were from Sigma (St Louis, MO, USA). TMB was from DAKO (Carpinteria, CA, USA). CDP-Star substrate was purchased from New England Biolabs (Ipswich, MA USA). SsoFast EvaGreen Supermix and Coomassie Brilliant Blue R-250 were from BioRad Laboratories (Hercules, CA, USA). All other reagents were bought from Sigma (Auckland, New Zealand).
Cell culture
The human colonic Caco-2 epithelial cell line was cultured in Minimal Essential Medium (MEM) with Earle’s Salt and
Cell viability
Caco-2 cells (5 × 105 per well) were cultured in 24-well plates for 48 h before the addition of PF (in medium) for a further 24 h. Following exposure to PF, the medium was aspirated and saved to a sterile tube. The remaining adherent cells were rinsed three times with 37℃ D-PBS, with each wash added to the saved medium. After incubation with 0.25% trypsin–EDTA for 10 min at 37℃ the detached cells were transferred to the same tube. The well was then rinsed with 500 µl of cell culture medium to collect any remaining cells (also added to the tube). A 1.5-ml aliquot of the final cell suspension was centrifuged at 1300 g for 5 min and the supernatant discarded. The remaining pellet of cells was re-suspended in 150 µl of medium, an equal volume of trypan blue (in D-PBS, 0.025% final concentration) was added and the cells were loaded onto a haemocytometer (BLAUBRAND, Wertheim, Germany). Live (clear) and dead (blue) cells were counted under the 40×objective of a light microscope (Nikon, Tokyo, Japan) with cell number and viability calculated.
Bacteria
E. coli strain LF 82 (kindly provided by Dr Arlette Darfeuille-Michaud, Laboratoire de Bacteriologies, CBRV, Clermont-Ferrnand, France) was grown in 3 ml of Luria Bertani (LB) broth at 37℃ on a rotating platform (100 rpm) to log phase (approximately 2 h). Bacteria were harvested, washed and quantitated by density, based on a standard curve measured at an absorbance of 550 nm. Bacterial addition to cell cultures was at a multiplicity of infection (MOI) of 100 : 1, unless otherwise stated.
Preparation of whole cell lysates
Caco-2 cells (2 × 105 per well) were cultured for 48 h in 48-well plates and then exposed to (or not) PF and/or IFN-γ for a further 24 h before being lysed. Briefly, the culture medium was removed and the cells washed three times with D-PBS before addition of 200 µl per well of RIPA buffer [50 mM Tris-HCl pH 7.5, 100 mM NaCl, 5 mM EDTA pH 7.5, 1% (v/v) Nonidet P-40, 0.2% (w/v) SDS, 0.5% (w/v) sodium deoxycholate, 1 mM sodium vanadate and 1 mM phenylmethanesulfonyl fluoride (added immediately before use)]. The plate was incubated on ice for 20 min, during which time the RIPA buffer was triturated to ensure cell detachment and lysis. The lysates were centrifuged at 14,000 g for 30 min at 4℃ to remove remaining cellular debris before being assayed for protein content 12 and stored at −20℃.
SDS-PAGE and Coomassie staining
Caco-2 cell lysate (40 µg protein) was separated by discontinuous SDS-PAGE under reducing conditions on a 10% acrylamide gel and stained for 2 h at room temperature (18–23℃) with Coomassie Blue stain (0.25%, w/v, Coomassie Brilliant Blue R-250 in 10% glacial acetic acid, 45% methanol). The gel was destained overnight (18 h) in 10% glacial acetic acid and 45% methanol.
Immunoblot analysis of CEACAM6 expression profile in Caco-2 cells
SDS-PAGE-separated Caco-2 cell lysate was transferred to a polyvinylidene difluoride (PVDF) membrane (Bio-Rad, Hercules, CA, USA) by electroblotting and the membrane probed for 60 min at room temperature with an anti-human CEACAM6 Ab (diluted 1/200) as primary Ab, followed by alkaline phosphatase-conjugated goat anti-mouse IgG antiserum (Sigma) (diluted 1/10 000) as secondary Ab. The Abs were diluted in 2% (w/v) non-fat milk in 0.1 % (v/v) Tween-20 (TBS-T). The membrane was developed with a chemiluminescent substrate (CDP-Star) and the blot photographed using a Chemidoc XRS (Bio-Rad).
Dot blotting
Dot blotting was used to determine the level of CEACAM6 expression in treated and untreated Caco-2 cells. Whole cell lysates (50 µg protein) were transferred onto PVDF membrane and incubated for 1 h at room temperature in blocking buffer TBS (20 mM Tris-HCl pH 7.5, 140 mM NaCl) containing 5% (w/v) non-fat milk and 0.1% (v/v) Tween-20 (TBS-T). The membrane was probed overnight at 4℃ with an anti-CEACAM6 Ab [final concentration 10 µg/ml, diluted in 2% non-fat milk (w/v) in TBS-T], followed by an alkaline phosphatase-conjugated secondary Ab [1 : 10,000 dilution in 2% non-fat milk (w/v) in TBS-T] added for 90 min at room temperature. The membrane was developed with a chemiluminescent substrate (CDP-Star) and the blots photographed using a Chemidoc XRS (Bio-Rad). The relative amounts of CEACAM6 in each preparation were quantitated using Quantity-1 densitometry software.
RNA isolation and cDNA synthesis
Caco-2 cells (5 × 105 per well) were cultured (changing the medium every 2 d) in six-well plates until confluent, and then the total RNA was extracted using Trizol reagent following the manufacturer’s protocol. Contaminating DNA was removed by the addition of chloroform (200 µl) to the Trizol-extracted RNA. After gentle shaking to mix, the tubes were incubated for 3 min at room temperature before centrifugation at 12,000 g for 15 min at 4℃. The upper layer was transferred to a new tube, gently mixed with 500 µl of isopropanol and incubated for 10 min at room temperature. After centrifugation at 12,000 g for 10 min at 4℃ the supernatant was discarded. One ml of 75% ethanol was added to the remaining pellet and the tube vortexed for 10–15 s before centrifugation at 7500 g for 5 min at 4℃. The ethanol layer was then discarded and the pellet left to air-dry for 10 min before being re-suspended in 10 µl of nuclease free water. Total RNA content was measured using a NanoDrop spectrophotometer (Nanodrop Technologies, Montanin, DE, USA). Samples were treated with DNase I at 37℃ for 30 min, followed by 10 min at 75℃ and 5–10 min at 4℃. First strand cDNA was synthesised from total RNA (2 µg) using Superscript III reverse transcriptase followed by RNase H digestion.
Real-time quantitative PCR
PCR primers.

Fluorescence microscopy
Caco-2 cells (2 × 105 per well) were plated on ethanol-sterilised coverslips in tissue culture plates for 48 h before exposure to PF for a further 24 h. The supernatant was removed and the cells were fixed by adding 10% neutral buffered formalin for 30 min. Following four 5-min washes in PIPES buffer (60 mM PIPES pH 6.8), potential non-specific binding sites on the cells were blocked by incubating the coverslips in blocking buffer [5% BSA (w/v), in PIPES] for 45 min at room temperature before overnight incubation at 4℃ in primary Ab (anti-human CEACAM6 Ab, 1 µg/ml dilution in PIPES containing 2.5% BSA), followed by a FITC-conjugated sheep anti-mouse secondary Ab for 2 h at room temperature (1 : 1000 dilution, in PIPES buffer containing 2.5% BSA). Cell nuclei were stained with Hoechst 33342 (0.25 µg/ml, in PIPES) at room temperature for 20 min. After four 10-min washes [PIPES containing 0.05% (v/v) Tween-20], the cells were mounted with p-phenylenediamine dihydrochloride [0.1% (w/v) in 50 mM Tris-HCl pH 6.8] and examined using epifluorescence microscopy on an Axiolmager Z1 upright microscope with a 40×EC Plan Neofluar lens (Zeiss, Oberkochen, Germany). Images were taken at the same exposure for each replicate with an Axiocam MRm camera, coupled to Axiovision software. Images were adjusted for presentation with NIH ImageJ.
Flow cytometry
Flow cytometry was used to quantitate the increase in surface-associated CEACAM6 in treated cells. Caco-2 cells (2 × 105 per well) were plated (changing the medium every 2 d) in six-well plates until confluent before exposure to PF and/or IFN-γ for a further 24 h. Adherent Caco-2 cells were then incubated with trypsin (500 µl per well) until cells had lifted to give a single cell suspension. Residual trypsin activity was quenched with the addition of an equal volume of medium before centrifuging the cells (500 g for 5 min). After two washes (D-PBS) the cells were fixed in 10% buffered formalin (v/v, in PBS) for 30 min at room temperature before being re-suspended in blocking buffer (D-PBS containing 1% BSA) for 20 min at room temperature. After blocking, the cells were re-suspended in primary Ab (anti-CEACAM6, diluted 1 : 50 in blocking buffer) and incubated for 1 h at room temperature. After two washes in blocking buffer the cells were re-suspended in secondary Ab (goat anti-mouse IgG Alexa Fluor 488, diluted 1 : 500 in blocking buffer) and incubated for 30 min at room temperature. The cells were given a final wash in blocking buffer before being re-suspended in D-PBS. Fluorescence measurements were made using a flow cytometer (Beckman Coulter Cytomics FC 500 MPL) and CPX software (Beckman Coulter). Ten thousand events were collected for each sample, with results presented as mean fluorescence intensity (MFI) values of cells.
CEACAM6 release by Caco-2 cells
Caco-2 cells (2 × 105 per well) were cultured for 48 h in 48-well plates and then exposed to (or not) PF and/or IFN-γ for a further 24 h. The amount of extracellular CEACAM6 released by Caco-2 cells was determined by an ELISA of the culture supernatant using a commercial kit (Sino Biological, Beijing, China). The assay was performed according to the manufacturer’s instructions. Absorbance measurement was at 450 nm (SpectraMax 190 plate reader; Molecular Devices, Sunnyvale, CA, USA).
Semi-quantitative assay to measure secreted CEACAM6 binding to AIEC
Caco-2 cells (1 × 105 per well) were plated into six-well plates and grown for 7 d before the addition of PF for 24 h. E. coli (0.5 × 108) were grown overnight in LB broth, killed using 10% neutral buffered formalin and added to the cell cultures supernatants (200 µl) for 2 h at 37℃. Following three washes (in PBS), non-specific binding sites on the bacteria were blocked overnight at 4℃ with blocking buffer [PBS containing 5% (w/v) BSA] before incubation in anti-CEACAM6 Ab (1 : 50, diluted in blocking buffer) for 1 h at 37℃. After three washes (blocking buffer), the bacteria were re-suspended in secondary Ab (HRP-conjugated goat anti-mouse IgG, diluted 1 : 500 in blocking buffer) and incubated for 1 h at 37℃. The bacteria were given three more washes before incubation in TMB substrate for 30 min, followed by absorbance measurement at 450 nm (SpectraMax).
E. coli infection of Caco-2 cells
Caco-2 cells (1 × 105 per well) were plated into six-well plates and grown until confluent (approximately 7 d). The medium was removed and the cells were washed with PBS before the addition of antibiotic-free medium, with or without the addition of PF. The cells were cultured for a further 24 h in the presence of PF before the addition of E. coli (MOI 100 : 1) for 3 h. In a separate series of experiments, E. coli were pre-incubated with recombinant CEACAM6 (diluted in antibiotic-free medium) for 30 min at 37℃, before being added to confluent Caco-2 cells for 3 h (MOI 100 : 1).
In both experiments, the cells were then carefully washed three times with PBS before the recovery of cell-associated bacteria. Briefly, the infected cells were lysed using 0.1% Triton-X 100 (v/v) in PBS for 10 min at 37℃. The lysates were then serially diluted (in PBS) and cultured (in duplicate) on sheep blood agar plates (Fort Richard, Auckland, New Zealand) overnight for colony-forming unit (CFU) determinations.
Statistical analysis
Results are presented as the means ± SE of the means. Data were analysed by ANOVA followed by Tukey’s post hoc test, non-parametric t-test or Pearson’s correlation coefficient as appropriate. If P ≤ 0.05, the differences were considered to be statistically significant. Statistics were calculated with GraphPad Prism software (version 5.04), and IC50s were determined with GraFit v7 (Erithacus Software, Norwich, UK).
Results
PF has no effect on epithelial cell viability
We first assessed the effect of PF on intestinal epithelial cell viability. Caco-2 cells, a human colonic epithelial cell line, were exposed to concentrations of up to 30% (v/v) PF for 24 h and cell viability determined by Trypan blue exclusion. This determined that PF had no significant effect on the viability of Caco-2 cells at any concentration tested, with 97.1% ± 1.59 untreated cells still viable after 24 h compared to 97.4% ± 1.5 viability in those cells treated with 30% PF. These results were the mean ± SE of three independent experiments.
PF increases the expression of CEACAM6
We next investigated an association between PF and CEACAM6 by exposing Caco-2 cells to increasing concentrations of PF for 24 h. After determining the specificity of our anti-CEACAM6 Ab by immunoblotting a Caco-2 cell lysate (Figure 1), we used dot blotting to show that CEACAM6 expression was dose-dependent, increasing with concentrations of PF up to 20% (Figure 2A), and that 20% PF treatment significantly increased CEACAM6 protein expression by Caco-2 cells (175% ± 24, P < 0.05), compared with untreated control cells. We then compared the response of Caco-2 cells to IFN-γ, a cytokine known to increase CEACAM6 expression in colon epithelial cells.
14
Exposure to IFN-γ (100 ng/ml) for 24 h also significantly increased CEACAM6 expression (182% ± 18, P < 0.01) in Caco-2 cells (Figure 2B). However, when the cells were exposed to a combination of PF and IFN-γ at the same concentrations (20% and 100 ng/ml respectively) and for the same period of time, CEACAM6 expression did not increase significantly above the increased level observed when either were added alone (190% ± 13; Figure 2B).
Detection of CEACAM6 in a Caco-2 cell lysate. Coomassie stained (2) SDS-PAGE and immunoblot (3) of a Caco-2 cell lysate. Lane 1, protein molecular mass markers (kDA). Equal amounts of lysate were analysed, determined by the protein concentration (40 µg) of the original samples. Polymeric formula increases the expression of CEACAM6. (A) PF added to Caco-2 cells for 24 h had a dose-dependent effect on CEACAM6 expression that reached significance at 20% (total volume) (P < 0.05). Treating cells with PF (20%) and/or IFN-γ (100 ng/ml) for 24 h had a significant effect on CEACAM6 (B) protein expression and (C) mRNA levels when added separately or in combination. Results are ± SE for three independent experiments. *P < 0.05, **P < 0.01, ***P < 0.001.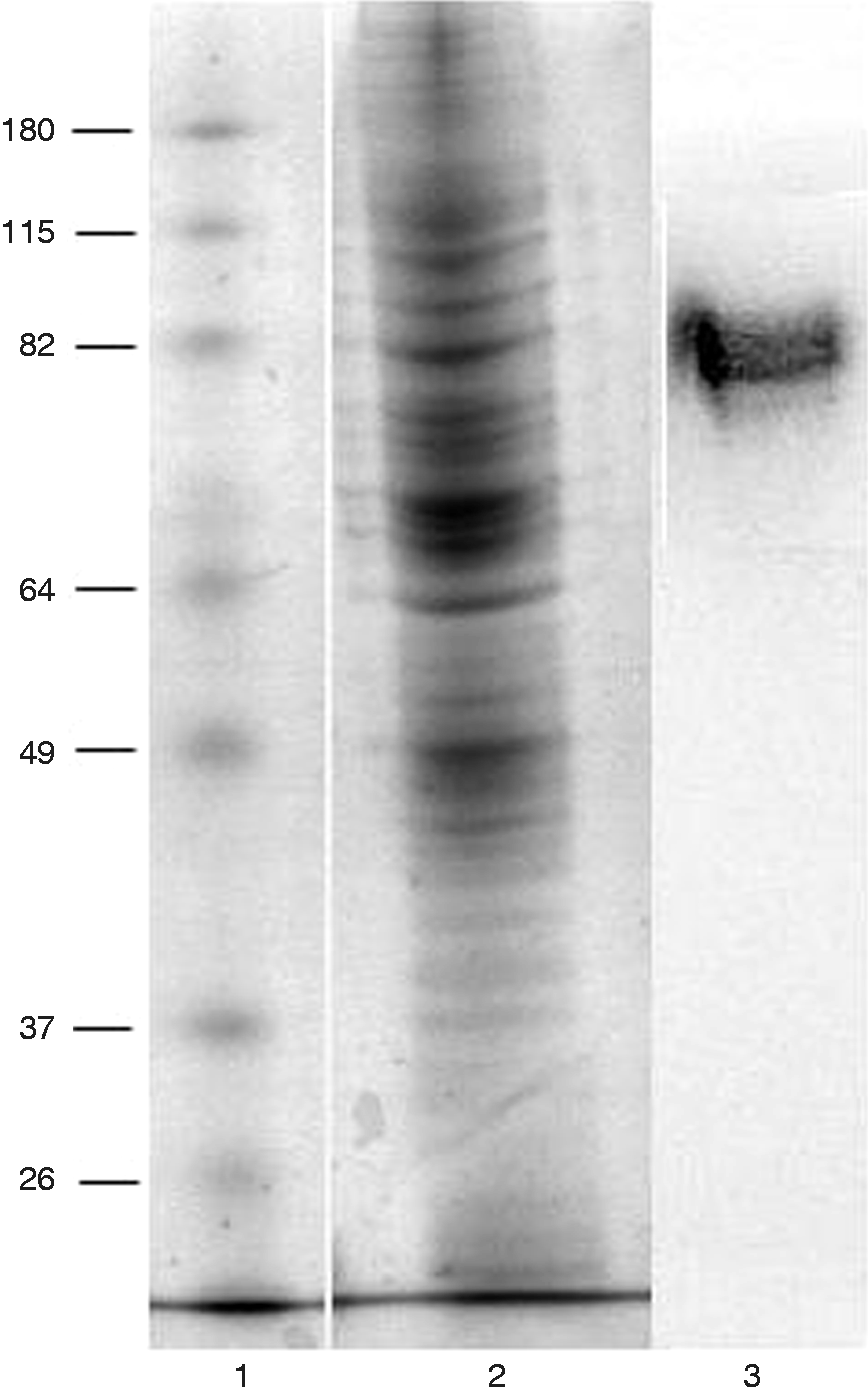
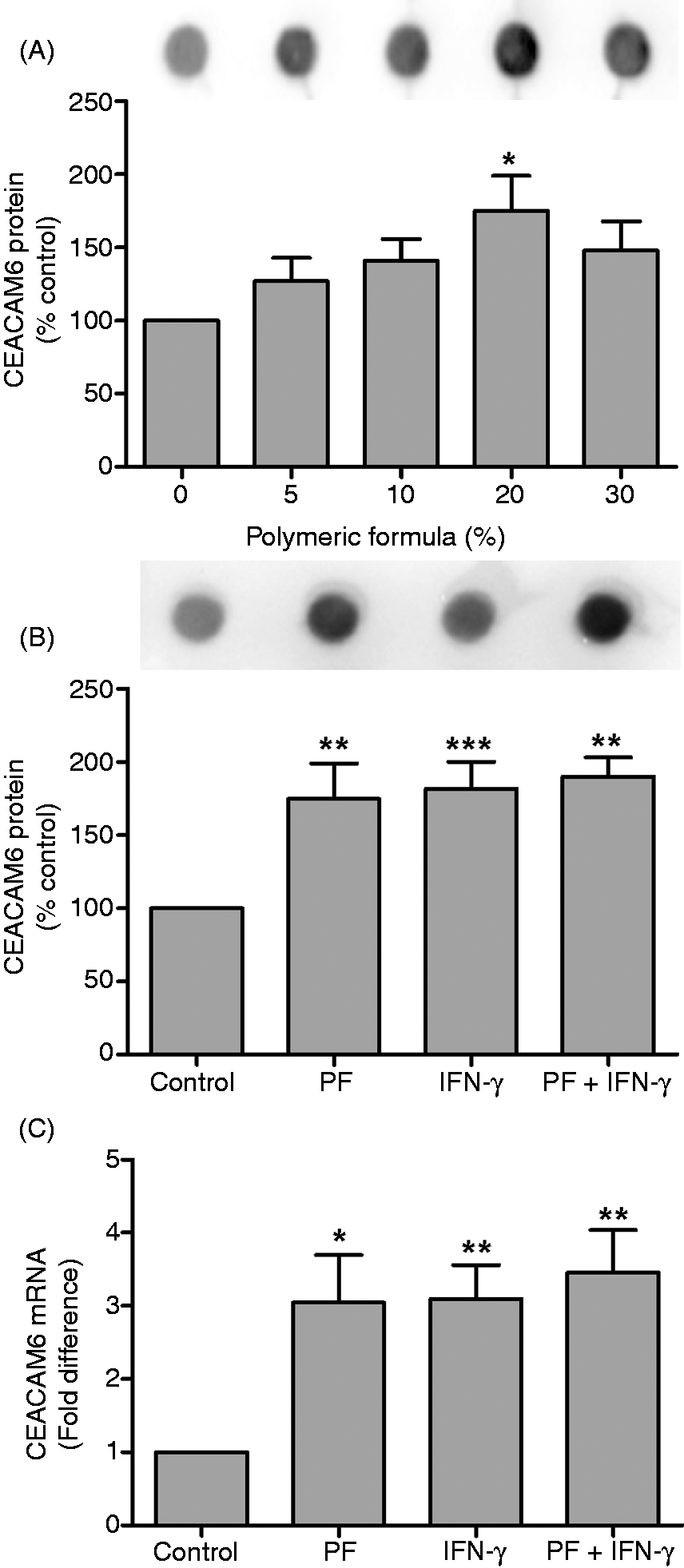
Next, we looked for changes in CEACAM6 gene expression following treatment of Caco-2 cells with PF and/or IFN-γ. We found a significant increase in the level of CEACAM6 mRNA in Caco-2 cells in response to both PF (3.05 ± 0.65, P < 0.05) and IFN-γ (3.01 ± 0.46, P < 0.01) compared with untreated control cells (Figure 2C). Collectively, these results suggest that PF and IFN-γ induce the expression of CEACAM6 by Caco-2 cells and that this expression reaches a threshold, an observation supported by a levelling off in the response of Caco-2 cells following 24 h exposure to 30% (v/v) PF (Figure 2A) and no evidence of any additive or synergistic effect on CEACAM6 protein (Figure 2B) or mRNA (Figure 2C) levels when cells were exposed to a combination of PF and IFN-γ.
PF increases CEACAM6 release by Caco-2 cells
CEACAM6 is normally localised to the apical surface of the normal colonic epithelium.
15
We used fluorescence microscopy to demonstrate that CEACAM6 is increased in Caco-2 cells after 24 h exposure to PF (Figure 3A, B). This observation was confirmed by flow cytometry of non-permeabilised Caco-2 cells following exposure to PF, which revealed a significant increase in the MFI values when compared to untreated control cells (36.8 ± 4.2 and 21.1 ± 4, respectively, P < 0.05; Figure 3C). Thus, CEACAM6 protein accumulates on the surface of Caco-2 cells following exposure to PF.
Polymeric formula increases CEACAM6 expression at the cell surface. Untreated (A) and PF-treated (B) Caco-2 cells exposed to PF for 24 h were fixed, stained and visualized for FITC-labelled CEACAM6 (green) and nuclei (blue). Scale bar =10 µm. (C) Flow cytometry of membrane-associated levels of CEACAM6 in PF and IFN-γ treated Caco-2 cells. *P < 0.05). Results are ± SE of three independent experiments.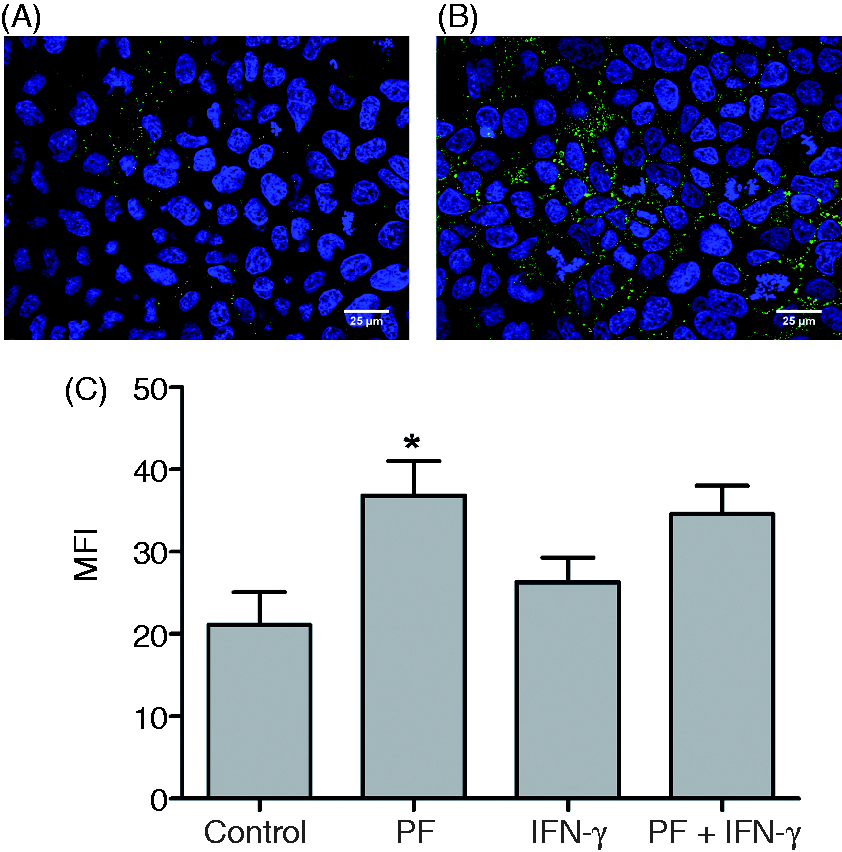
It has been suggested that colonic epithelial cells may release CEACAMs as part of the host nonspecific defence against microorganisms.
15
Thus, an accumulation of CEACAM6 at the surface of the PF-exposed cells suggested that these cells might be releasing this protein into the extracellular environment. To confirm this observation, we collected the culture medium from Caco-2 cells exposed to PF and/or IFN-γ for 24 h and assayed (by ELISA) for soluble CEACAM6. PF notably increased the level of extracellular CEACAM6 protein (284.9 pg/ml ± 62.3) when compared to untreated control cells (124.8 ± 34.2; Figure 4). This difference was statistically significant.
Polymeric formula increases the concentration of extracellular CEACAM6. The supernatant collected from Caco-2 cells after 24 h exposure to PF (20%) and/or IFN-γ (100 ng/ml) was assayed for extracellular CEACAM6 by ELISA. Results are the means ± SE of eight independent experiments. *P < 0.05 by unpaired t-test.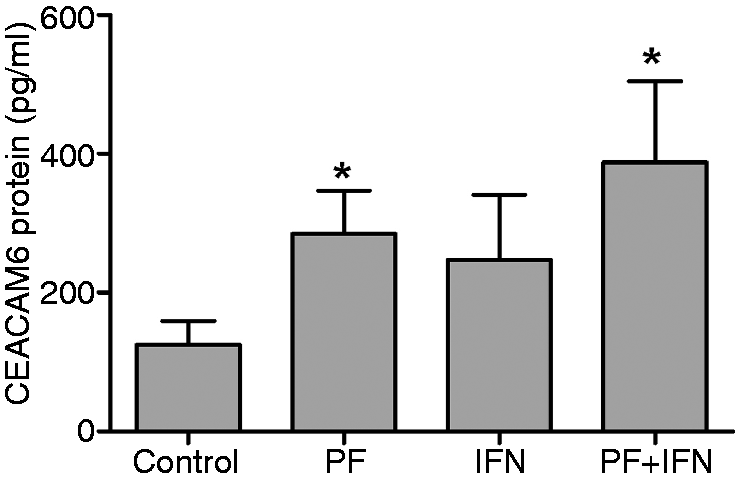
PF reduces the association of AIEC with Caco-2 cells
Our observation of a notable increase in the release of CEACAM6 from the surface of PF-treated Caco-2 cells (Figure 4) suggested that the release of this protein might act as a deterrent to AIEC invasion of gut epithelial cells.
10
To test this hypothesis the culture media from PF-exposed Caco-2 cells was collected and incubated with AIEC (MOI 100 : 1) for 4 h before being probed with an anti-CEACAM6 Ab. Using an HRP conjugate, we demonstrated that released CEACAM6 in the culture medium bound to the bacteria in a dose-dependent manner, with a Pearson’s correlation coefficient of 0.84 (P < 0.05; Figure 5).
Association of extracellular CEACAM6 with E. coli. The supernatant collected from Caco-2 cells after 24 h exposure to increasing concentrations of PF was incubated with AIEC (MOI 100:1) for 4 h. CEACAM6 binding to the bacteria was detected by labelling the pelleted and washed bacteria with anti-CEACAM6 Ab. Pearson’s correlation co-efficient was 0.84. Results are the means ± SE for three independent experiments.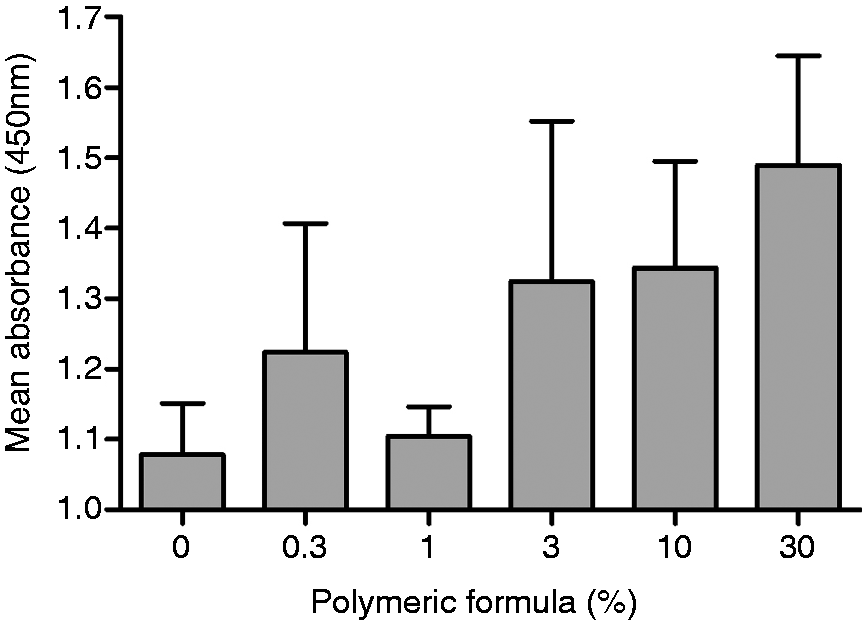
Next we determined whether PF could inhibit the association of AIEC with intestinal epithelial cells. Caco-2 cells were treated with increasing concentration of PF for 24 h before the addition of the bacteria at an MOI of 100 : 1 for 3 h. We observed that the presence of PF was associated with significantly reduced bacterial association with Caco-2 cells and that this effect was concentration-dependent, with a Pearson’s correlation coefficient of 0.898 (P < 0.05). Specifically, the addition of 10% and 30% PF reduced bacterial association to 52.75% ± 8.58 (P < 0.01) and 22.6% ± 4.24 (P < 0.001) respectively, when compared with untreated control cells (Figure 6A). The concentration of PF required to inhibit the association of 50% of the bacteria (IC50) was calculated at 10.19% ± 2.99. To demonstrate that this effect was due to soluble CEACAM6, AIEC bacteria were pre-incubated with recombinant CEACAM6 (Figure 6B). The concentrations of recombinant protein were based on the levels of soluble CEACAM6 released by Caco-2 cells in response to PF (Figure 4), and we observed the presence of recombinant protein was also associated with significantly reduced bacterial association with Caco-2 cells. Specifically, the addition of 500 pg/ml and 1000 pg/ml recombinant CEACAM6 reduced AIEC association to 51.67% ± 10.4 (P < 0.05) and 53.1% ± 7.2 (P < 0.05), respectively, when compared with untreated controls (Figure 6B). However, a Pearson’s correlation coefficient of 0.799 (P = 0.056) showed the effect was not significantly dose-dependent.
Polymeric formula has a dose-dependent effect on AIEC adherence to Caco-2 cells. AIEC (MOI 100:1) added to cells (A) pre-incubated with increasing concentrations of PF for 24 h or (B) following incubation in recombinant CEACAM6 for 30 min, were given 3 h to adhere to the cells before extensive washing to remove non adherent bacteria. Quantitative assessment of viable AIEC associated with Caco-2 cells 3 h post-infection was by serial dilution of cell lysates. Pearson’s correlation coefficients were (A) 0.898 and (B) 0.799 respectively. Results are the means ± SE for three independent experiments. *P < 0.05, **P < 0.01, ***P < 0.001.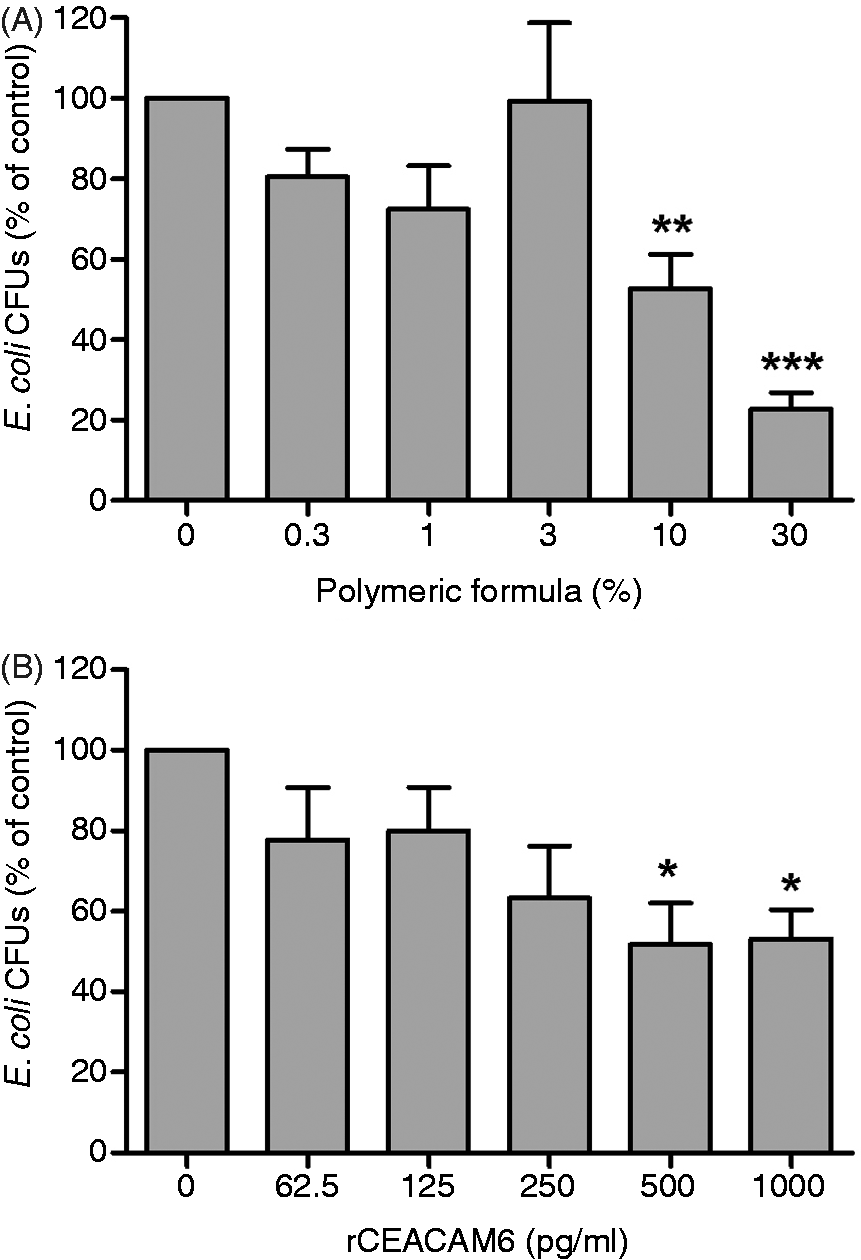
Discussion
Clinically, there is a growing awareness of the benefit of EEN as a non-pharmacological means to induce remission in active CD.5,6 However, whereas the clinical benefits have been well defined, 7 we are only now starting to understand the potential mechanisms underlying these effects. One effect of PF that we have reported is an ability to dampen the response of cultured gut epithelial cells to inflammatory stimuli. 9 We have also shown that PF can restore compromised gut barrier function, 8 an effect that gains significance when one considers that an intact intestinal epithelial monolayer is required to provide a physical barrier to luminal gut bacteria. Pathogenic bacteria that adhere to and/or invade the epithelial cells that form this barrier can weaken or impair barrier function, triggering the onset of gut inflammation. 16 However, to date, the effect (if any) that PF plays in abrogating the interaction of pathogenic bacteria with the intestinal epithelial cell barrier has not been considered.
The presence of a coat of highly glycosylated proteins serves to limit bacterial adherence to the luminal surface of the epithelial cells that form this barrier. 17 However, pathogenic strains of Salmonella and E. coli bacteria are able to bind to cell surface proteins that include CEACAMs. 18 We are particularly interested in AIEC as these bacteria are significantly over-represented in bacterial flora associated with the ileal mucosa in patients with CD.19,20 AIEC express a type 1 pilus variant on their surface. 21 Expression of this pilus enables these bacteria to bind to CEACAM6 10 thereby facilitating their invasion of the epithelium. 11 Invasion is associated with loss of barrier function in cultured epithelial cells 22 and in mice colonized with AIEC, 23 where it precedes the induction of gut inflammation. 24 AIEC appear to bind more readily to the brush border of enterocytes isolated from patients with CD than from healthy controls and this is thought to correlate with the finding of increased CEACAM6 expression in the ileum of patients with CD. 10 However, it is still not clear whether CEACAM6 over-expression is due to the presence of AIEC or AIEC colonisation is a consequence of CEACAM6 up-regulation. 10
Using Caco-2 cells, we showed that exposure to PF increased the expression of surface-associated CEACAM6 above the level found in untreated control cells. This effect was dose-dependent and reached a maximum following exposure to 20% PF. A corresponding increase in mRNA expression suggested that the PF-treated cells were continuously synthesising more protein, as evinced by increased expression of CEACAM6 at the cell surface with concomitant release into the extracellular environment. We also observed that the level of CEACAM6 in the 20% PF-treated cells was not significantly different to that observed in Caco-2 cells exposed to IFN-γ, a cytokine known to increase CEACAM6 expression in colon epithelial cells 14 and used in our study as a positive control. Intriguingly, there was no significant increase in CEACAM6 expression when IFN-γ was added to PF-exposed cells, suggesting that CEACAM6 levels may reach a maximum and plateau. This may reflect cell differentiation, as reported elsewhere, 25 and our results suggest that PF promotes this process. If so, this previously unrecognised effect of PF may prove to be an important mechanism for the mucosal healing observed in children with CD who are treated with PF. 26 In support of this hypothesis is our observation that PF restores barrier function in Caco-2 cells treated with agents that disrupt the integrity of the tight junctions between epithelial cells that are essential to maintain barrier function. 8
It may seem counter-intuitive that a formula able to induce remission in children with CD 7 acts by increasing the expression of an adhesion molecule that serves to facilitate the invasion of bacterial pathogens through the epithelial barrier. However, we suggest that the response of Caco-2 cells to PF instead signals a role for CEACAM6 in the host innate immune response, as hypothesised elsewhere. 25 There is precedence for this, with increased expression of intestinal alkaline phosphatase, an enzyme credited with detoxifying bacterial LPS and preventing bacterial invasion across the gut mucosal barrier, 27 also reportedly increased as Caco-2 cells differentiate. 28 Intriguingly, there is also evidence of a role for enteral nutrition in this process. 27 Fully differentiated gut epithelial cells are further characterised by their ability to constitutively shed small vesicles from the tips of their microvilli into the lumen of the gut. 15 These vesicles are enriched with both CEACAM6 15 and alkaline phosphatase, 28 thus enabling luminal gut epithelial cells to deploy decoy bacterial receptors, 25 as well as enzymes with the potential to detoxify and/or kill bacteria29,30 at the same time. Collectively, these findings suggest that epithelial cell differentiation is a key determinant in the innate immune response at the gut epithelial barrier.
Our findings support this hypothesis, with evidence that exposure to increasing concentrations of PF increased levels of extracellular CEACAM6 at a rate that was inversely proportional to limiting AIEC binding to Caco-2 cells. We did not determine whether the CEACAM6 shed from the surface of Caco-2 cells in response to PF was soluble or vesicle-associated. However, our results support the idea that constitutive shedding of cell surface-associated components as decoy receptors may serve to reduce pathogen binding to the intestinal barrier. If so, this may prove to have a downstream effect on barrier integrity and/or the release of inflammatory cytokines, as well as limiting the number of invading bacteria. Furthermore, our results suggest that non-pharmacological interventions could bolster this response.
In conclusion, we found that the addition of PF to an intestinal epithelial cell line increased the expression of CEACAM6 on the cell surface with concomitant release into the extracellular environment, and that the shedding of CEACAM6 correlated with decreased AIEC binding to the surface of Caco-2 cells. These findings suggest that the secretion of cell surface-associated proteins acting as releasable decoys may be an under-appreciated aspect of the gut’s innate immune response to pathogenic bacteria and our results suggest that this mechanism may be strengthened by PF in the setting of CD.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Acknowledgements
The authors wish to thank Dr RW Bentley for assistance in developing the RT-qPCR methodology.
