Abstract
Hepcidin is a known key modulator of iron homeostasis and an innate immune molecule secreted by the liver. The transcriptional mechanism of hepcidin in hepatocytes during inflammation is mediated via the IL-6/STAT3 pathway. Recently, hepcidin demonstrated an anti-inflammatory function in endotoxic mice, and a TLR4-dependent inducible expression of hepcidin was detected in myeloid cells. In this study, we explored the expression and signaling mechanism regulating hepcidin mRNA expression in peripheral blood leukocytes. The mRNA levels of hepcidin in peripheral blood leukocytes from patients with severe sepsis (n = 14) was significantly higher than those in healthy controls (n = 16;0.286 ± 0.065 vs 0.068 ± 0.025;
Keywords
Introduction
Hepcidin is a small, cysteine-rich, cationic peptide mainly produced by the liver and is a principle iron regulatory hormone. Hepcidin binds to ferroportin, inducing its internalization and degradation, thereby decreasing the iron absorption of duodenal enterocytes and release of iron from the monocyte/macrophage system.1,2 This restricts the utilization of iron by microbes and enhances the body’s defense to infections. 3 A recent study showed that hepcidin could suppress inflammatory response in an endotoxic mice model. 4 Hepcidin may play an important role both in iron metabolism and in the host immune response.
The expression of hepcidin in hepatocytes can be upregulated by iron overload and inflammation while suppressed by hypoxia and anemia. 5 Previous studies have shown that IL-6 is the key mediator that induces hepcidin production in hepatocytes by LPS stimuli, or under the circumstances of chronic inflammatory disease. 6 – 8 The expression of hepcidin in liver was demonstrated to be mediated via the IL-6/STAT3 signaling pathway.9,10 Other cytokines, such as IL-1, but not TNF-α and IFN-γ, could also stimulate hepcidin expression in hepatocytes, which is consistent with the fact that hepcidin transcripts in the liver of IL-6 knockout (IL-6−/−) mice could still be upregulated in an endotoxic model.7,11
As well as hepatocytes, a number of immune cells, including murine abdominal-cavity neutrophils and macrophages in spleen, lung and bone marrow, display a baseline expression of hepcidin, as well as an increased expression under stimulation with LPS, group A
Therefore, we hypothesized that the mechanism of hepcidin production in myeloid cells may differ from that in hepatocytes. To address this issue, we investigated expression levels of hepcidin in peripheral blood leukocytes both
Materials and methods
Research subjects
The study was approved by the Ethics committee of Zhejiang University (Hangzhou, China) and written, informed consent was obtained from the patients or a next of kin. Fourteen patients with severe sepsis meeting the criteria recommended by the American College of Chest Physicians/Society of Critical Care Medicine (ACCP/SCCM) Consensus Conference were included in the study. 17 In addition, 16 age- and gender-matched healthy volunteers were included as controls.
Blood sampling and culture
Whole blood from both septic patients and controls was sampled into EDTA-containing tubes and serum isolation tubes. Blood samples from patients with severe sepsis were drawn within 24 h after diagnosis of severe sepsis. Whole blood from healthy controls was co-cultured with 100 ng/ml LPS (
Isolation of peripheral blood mononuclear cells and neutrophils
Isolation of PBMCs and neutrophils was carried out as previously described.18,19 Briefly, 20 ml EDTA-anticoagulation blood diluted 1:1 in Hank’s balanced salt solution (HBSS) as underlayed over Hypaque-Ficoll, centrifuged at 400
Treatment of anti-TNF-α antibody and inhibitors
One milliliter of the whole blood was incubated with 100 µl RPMI-1640 medium (Invitrogen) and was stimulated with 0 ng/ml LPS (as control), 100 ng/ml LPS alone, or 100 ng/ml LPS plus 10 µg/ml anti-TNF-α Ab (Catalog number: AB-210-NA; R&D systems) for 6 h at 37°C in an atmosphere of 5% CO2.
To investigate the possible transcriptional factor involved in hepcidin mRNA expression, two inhibitors, Ro106-9920 and rapamycin, were used in this study. Ro106-9920 selectively inhibits IκBα degradation and NF-κB activation. 20 Rapamycin suppresses phosphorylation of STAT3 on ser727. 21 Peripheral blood mononuclear cells or neutrophils were stimulated with 0 ng/ml LPS (as control), 100 ng/ml LPS, 100 ng/ml LPS plus 10 µmol/l Ro106-9920 (Sigma-Aldrich), or 100 ng/ml LPS plus 0.5 µmol/l rapamycin (Merck Biosciences, Darmstadt, Germany) for 6 h in a 5% CO2 incubator at 37°C.
Reverse transcription and quantitative PCR
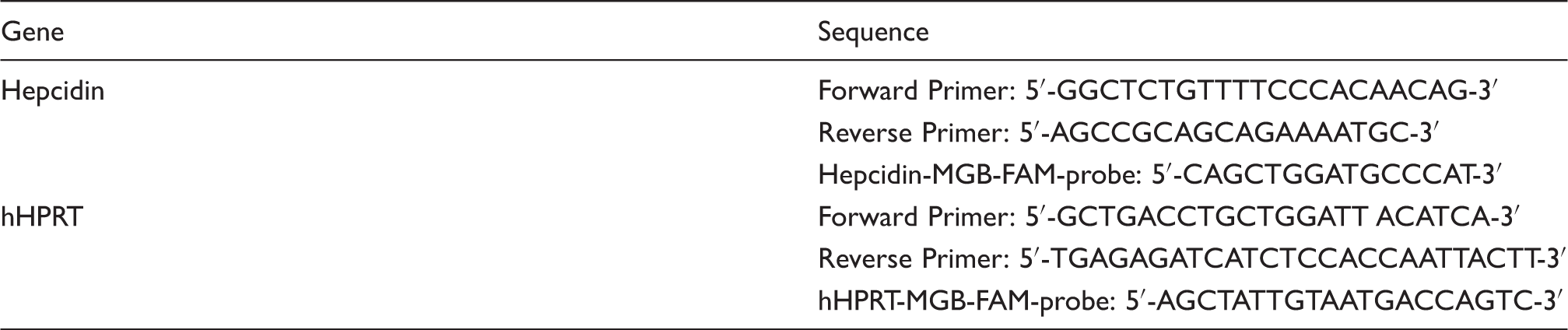
Primers and probes for quantitative PCR
Serum iron and ferritin assay
Total serum iron was measured by colorimetric assay using ascorbate/FerroZine reagents (Hitachi 747, Roche, Sydney, NSW, Australia). Serum ferritin was measured by a solid-phase, 2-site chemiluminescent immunometric assay (Immulite 2000; Diagnostic Products, Los Angeles, CA, USA).Statistical analysis
The expression levels of hepcidin among different groups were analyzed using
Results
Demographic data of subjects
The severe septic group consisted of nine males and five females, with a mean age of 59.6 ± 14.5 years. The healthy controls included 11 males and 5 females, with mean age of 57.5 ± 10.3 years. Gender and age between the two groups were comparable (
The mRNA levels of hepcidin in peripheral blood leukocytes are elevated in patients with severe sepsis
The mRNA levels of hepcidin and hHPRT in peripheral blood leukocytes were determined in patients suffering from severe sepsis and in healthy controls. The hHPRT mRNA levels in peripheral blood leukocytes are not affected by stimulation or donor health status and were therefore used to standardize hepcidin mRNA levels to compensate for potential variations of total RNA used in individual assays. The relative mRNA levels of hepcidin in the severe septic group were 0.286 ± 0.065, which were significantly higher than those in healthy controls 0.068 ± 0.025 ( The mRNA levels of hepcidin in peripheral blood leukocytes and correlation to serum levels of ferritin and iron in patients with severe sepsis. (A) The mRNA levels of hepcidin in peripheral blood leukocytes from 14 patients with severe sepsis and 16 healthy controls. The mRNA of hepcidin and hHPRT were analyzed by quantitative PCR; the relative mRNA levels of hepcidin were normalized to hHPRT. Data are presented as mean ± SD, *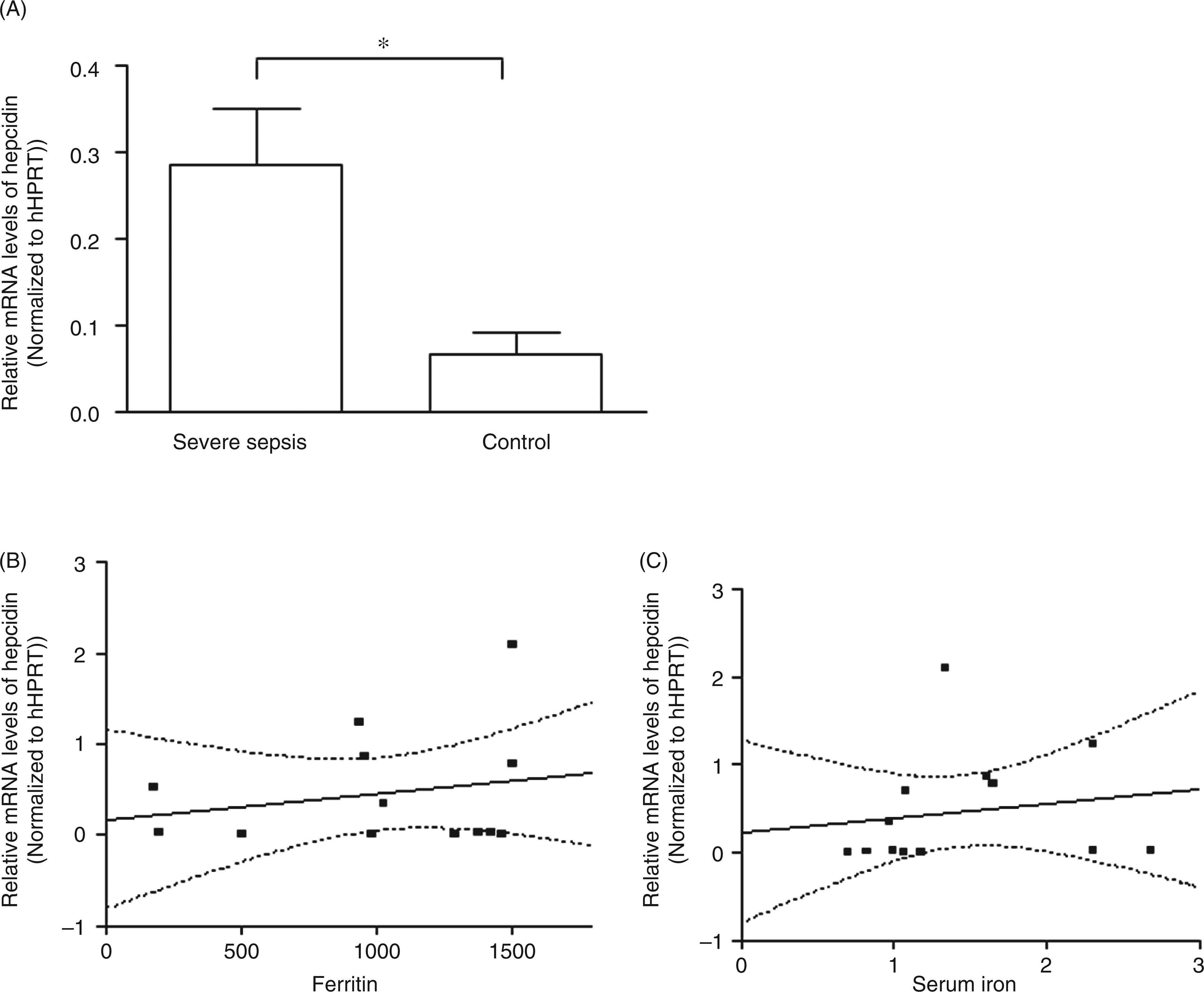
Furthermore, serum levels of ferritin and iron in septic patients were also determined. However, linear regression analysis showed that the levels of hepcidin transcripts in peripheral blood leukocytes from septic patients correlates with neither serum levels of ferritin (Figure 1B) nor those of iron (Figure 1C).
Tumor necrosis factor-α plays an important role in mRNA expression of hepcidin induced by LPS in peripheral blood leukocytes
Compared to unstimulated expression, time course experiments show significantly enhanced relative hepcidin mRNA expression after LPS stimulation for 3 to 12 h ( The role of TNF-α in the expression of hepcidin in peripheral blood leukocytes stimulated by LPS. The mRNA of hepcidin and hHPRT were analyzed by quantitative PCR, the relative mRNA levels of hepcidin were normalized to hHPRT. Data are presented as mean ± SD, ***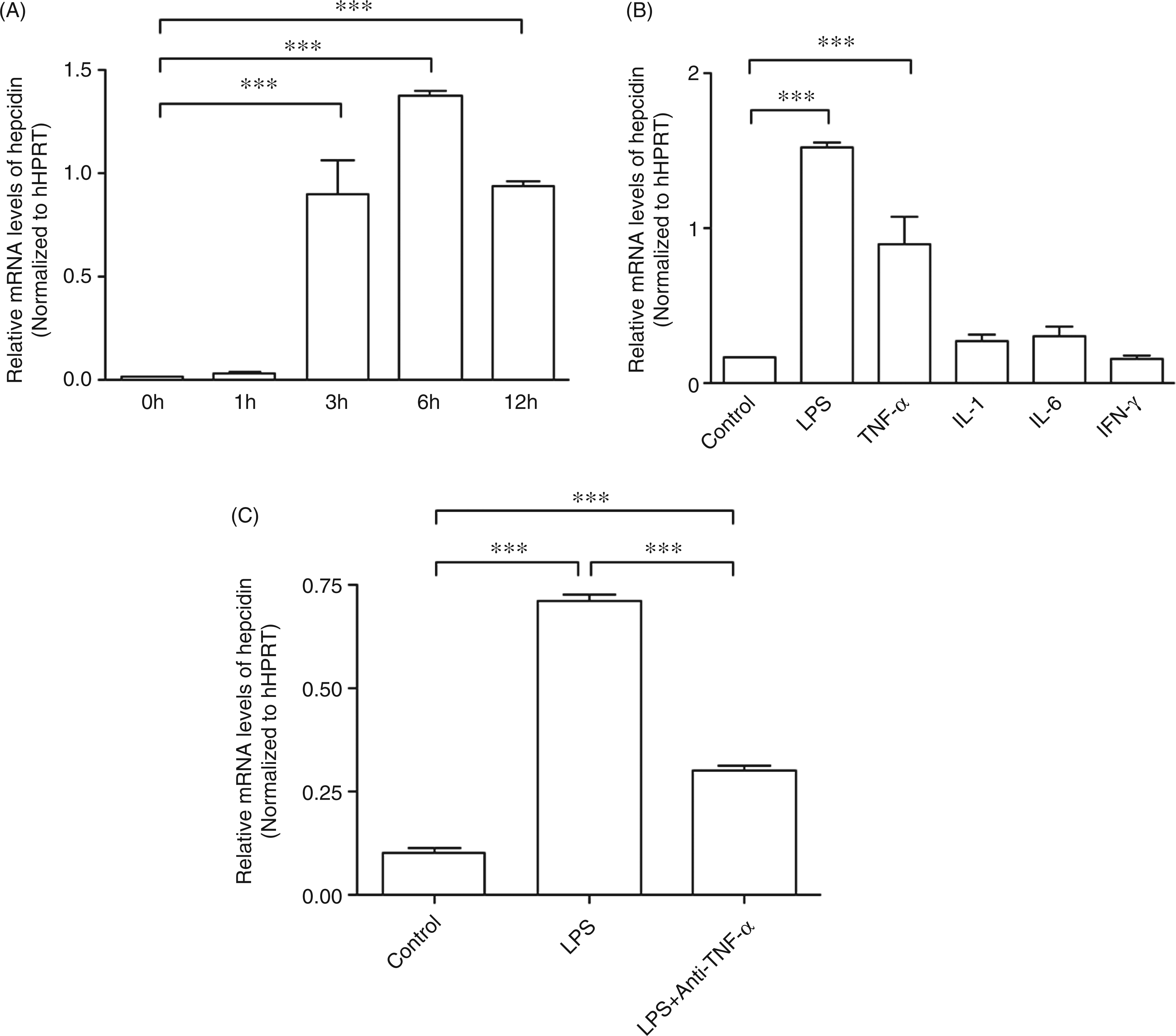
Nuclear factor-κB-mediated mRNA expression of hepcidin induced by LPS in peripheral blood leukocytes
To further explore the possible signal transduction pathway involved in the expression of hepcidin in peripheral blood leukocytes, the isolated PBMCs and neutrophils were treated with NF-κB inhibitor Ro106-9920 and STAT3 inhibitor rapamycin before LPS stimulation. In both types of cell, Ro106-9920 treatment completely depleted the inducibility of hepcidin mRNA by LPS ( Effect of Ro106-9920 and rapamycin on mRNA expression of hepcidin induced by LPS in PBMCs and neutrophils. The mRNA of hepcidin and hHPRT was analyzed by quantitative PCR; the relative mRNA levels of hepcidin were normalized to hHPRT. Data are presented as mean ± SD, ***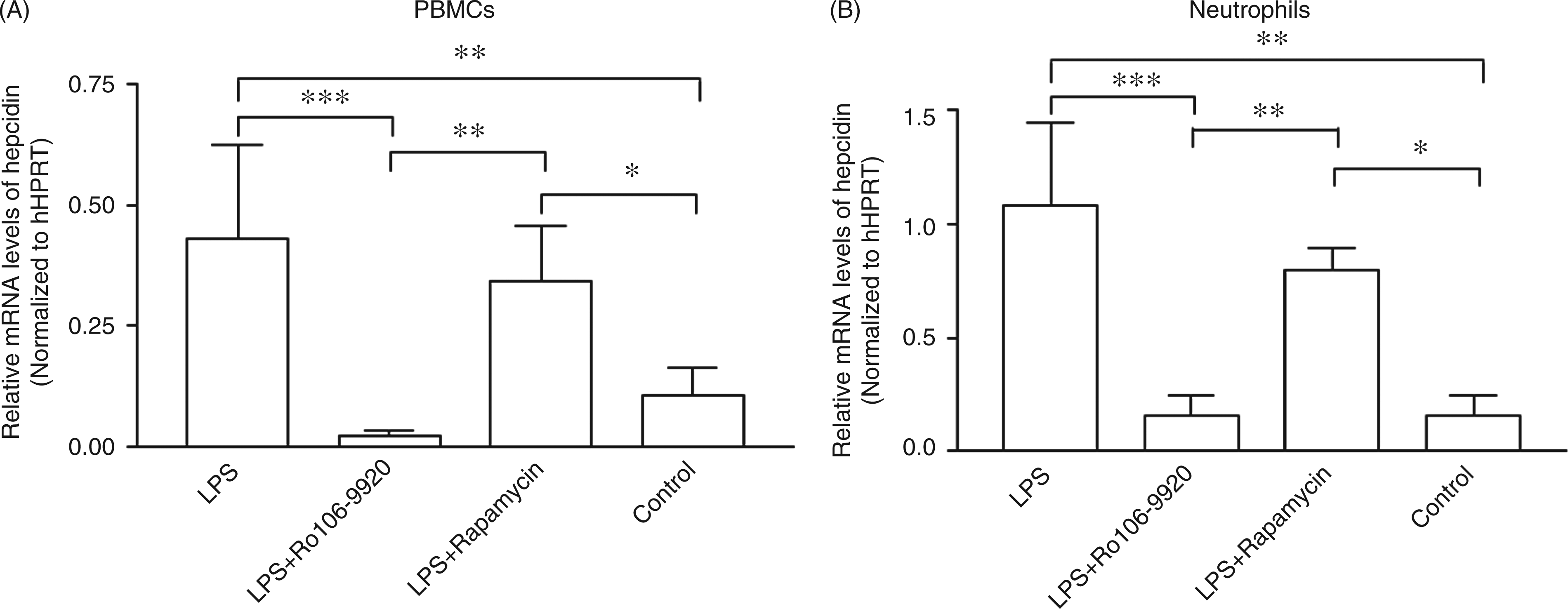
Discussion
In this study we found that the transcription levels of hepcidin in peripheral leukocytes were significantly elevated in patients with severe sepsis. Increased hepcidin mRNA levels were neither related to the concentration of ferritin nor that of total serum iron. LPS and TNF-α could induce the expression of hepcidin mRNA in peripheral blood cells and anti-TNF-α antibody significantly decreased the LPS-induced transcriptional level of the hepcidin gene. Furthermore, inhibition of NF-κB completely abolished the inducibility of hepcidin mRNA in neutrophils and PBMCs by LPS, while inhibiting STAT3 showed no detectable impacts on LPS-induced transcriptional levels of hepcidin in these cells.
In liver, the inducible regulation of hepcidin by inflammation has been well studied. 9 – 11 Interestingly, in contrast to studies in hepatocytes,7,11 the present study identified TNF-α as a strong inducer of the expression of hepcidin mRNA in peripheral leukocytes rather than IL-6, IL-1 or IFN-γ. This observation was consistent with recent studies which showed that TNF-α contributed to the up-regulation of hepcidin mRNA in human lymphocytes, 16 while IL-1 and IL-6 did not affect hepcidin expression in alveolar macrophages.13,14 In addition, the mRNA of hepcidin induced by LPS in peripheral blood cells is strongly dependent on TNF-α. This is supported by the finding that neutralization of TNF-α in LPS-treated peripheral blood abolished most of the mRNA induced by LPS. The time-course of TNF-α mRNA and protein expression in LPS-stimulated peripheral blood have shown that TNF-α mRNA peaked at 2 h and returned to baseline levels by 8 h, while the protein concentration reached a maximum at 4 h and remained at a high level until 24 h. 23 This expression pattern is consistent with the evolution of hepcidin mRNA in LPS-stimulated peripheral blood cells in our study. These data delineated that LPS stimulated and TNF-α mediated hepcidin expression in peripheral blood leukocytes. However, the expression level of LPS-induced hepcidin in the presence of blocking anti-TNF-α antibodies was still significantly higher than that in controls. It is a possibility that the TNF-α antibody may not completely block the effect of TNF-α which left some free TNF-α molecules to mediate hepcidin mRNA expression. Alternatively, other mediators, in addition to TNF-α, may regulate the transcription of hepcidin during inflammation. A very recent study has shown that IFN-α could induce hepcidin mRNA expression in human monocytes. 24 Therefore the role of IFN-α in LPS induced hepcidin expression should not be excluded.
Studies have shown that in liver, transcriptional factor STAT3 as a key effector was involved in the transcriptional regulation of hepcidin, in both baseline and inflammatory conditions.9,10 In the current study, inhibition of STAT3 did not significantly affect the mRNA level of hepcidin in either neutrophils or PBMCs, while inhibition of NF-κB did completely abrogate the inducibility of hepcidin mRNA by LPS. As the LPS/TLR4 pathway (which contributes to iron homeostasis) was independent of NF-κB, 12 this novel NF-κB mediated transcriptional regulation of hepcidin may indicate a different function of hepcidin in leukocytes. It is likely that NF-κB-mediated inducible expression of hepcidin in neutrophils and PBMCs regulates the function of these immune cells through a direct paracrine/autocrine pattern during local infection and inflammation. Further studies are required to demonstrate this notion in the future. Interestingly, the hepcidin mRNA levels in PBMCs pre-incubated with NF-κB inhibitor Ro106-9920 were found to be even lower than the background level of control cells in the absence of any stimuli. This indicates that the background hepcidin expression in PBMCs may also be NF-κB dependent. Also, the possibility that the 10% FBS within the RPMI-1640 medium used in the present study acted as a potential stimulus and regulated the background expression of hepcidin mRNA cannot be excluded.
The major limitation of the present study is that we did not measure the protein levels of hepcidin in the tested immune cells. Recent studies have shown that there are three known isoforms of mature hepcidin. The 25-amino acid peptide (hepcidin-25) is thought to be the major isoform and the only one known to be involved in iron metabolism. 25 While physiological roles of the other two isoforms (hepcidin-20 and -22) have been detected in human urine and hepatoma-derived cells, they are poorly understood.25,26 As hepcidin mRNA expression in peripheral leukocytes was found to be associated with neither serum ferritin nor iron in the present study, and its regulatory mechanism was different from that in hepatocytes, it is reasonable to speculate that hepcidin expressed in peripheral leukocytes in response to inflammation may exert immune modulating functions beyond iron metabolism. Further studies testing the protein levels of hepcidin via validated methodology, such as mass spectrometric assay, 26 are warranted to verify whether the hepcidin proteins expressed in peripheral immune cells are the same as those from hepatoma-derived cells.
In summary, the present study demonstrated that the mRNA expression of hepcidin in peripheral blood leukocytes during acute inflammation response is NF-κB-dependent. It is different from that in hepatocytes, which is in dependent on STAT3. Further studies to elucidate the role of hepcidin in neutrophils and PMBCs will highlight the importance of hepcidin in host innate immunity.
Footnotes
Acknowledgements
This study was financially supported by National Science Fund for Distinguished Young Scholars (30825037), Program for Changjiang Scholars, and Zhejiang Provincial Program for the Cultivation of high-level Innovative Health Talence.
The authors thank Professor Tomas Ganz, M.D., from Department of Medicine, David Geffen School of Medicine, University of California at Los Angeles, Los Angeles, CA, USA, for his critical review.
