Abstract
Exacerbated inflammation upon persistent barn organic dust exposure is a key contributor to the pathogenesis of lung inflammation and lung function decline. Barn dust constituents and the mechanisms contributing to the exacerbated inflammation are not clearly known. We set out to understand the inflammatory effects of Swine Barn Dust Extracts (SBDE) on human lung epithelial (BEAS2B) and macrophage (THP-1 monocyte derived) cell lines on a kinome array to determine phosphorylation events in the inflammatory signaling pathways. Upon identifying events unique to SBDE or those induced by innate immune ligands in each cell line, we validated the signaling pathway activation by transcriptional analyses of downstream inflammatory cytokines. Our findings indicate that SBDE-mediated pro-inflammatory effects are predominantly due to the induction of neutrophilic chemokine IL-8. Differentially phosphorylated peptides implicated in IL-8 induction in BEAS2B cell line include, TLR2, 4, 5, 7, 8, 9, PKC, MAP kinases (p38, JNK), inflammasomes (NLRP1, NLRP3), NF-κB and AP-1. In the THP-1 cell line, in addition to the aforementioned peptides, peptides corresponding to RIG-I-like receptors (RIG-I, MDA5) were found. This is the first report to demonstrate the application of a kinome array to delineate key inflammatory signaling pathways activated upon SBDE exposure in vitro.
Introduction
Agricultural workers, including swine production workers, are at an increased risk for developing respiratory disorders such as rhinosinusitis, chronic bronchitis, and chronic obstructive pulmonary disease (COPD) accompanied with lung function decline. 1 This is due to an increase in concentrated animal feeding operations (CAFOs) that predispose the farmers working 8 h shifts and are exposed to significant amounts of airborne dust generated in the barns.1,2 Repeated or chronic exposure of farmers to the barn dust enriched in microbial and non-microbial organic components is implicated in respiratory disease development; symptoms appear to be proportionate to the dust levels and dust reduction strategies have resulted in symptom reduction.3,4
Microbial components of the dust from swine farming facilities were found to comprise a large number (90%) of sequences pertaining to Gram-positive anaerobic bacteria and methanogenic Archaea upon shot gun pyrosequencing.5,6 The top four genera in the swine facility dust were assignable to the Clostridium, Lactobacillus, Ruminococcus, and Eubacterium species. 7 In addition, gas chromatography combined with MS analysis of settled swine barn dust revealed the presence of high amounts of muramic acid, a peptidoglycan (PGN) component present mainly in Gram-positive bacteria. 8 In addition, fungal β-glucans were also present and were implicated in the modulation of agricultural dust-induced inflammation. 8 Although endotoxin, a cell envelope component of Gram-negative bacteria, is present in significant amounts in barn dusts and is linked to barn dust-induced airway inflammation, a considerable amount of research attributes a very minor to no role to endotoxin in contributing to airway inflammation.9-11
Airway inflammation upon exposure to barn dust occurs in response to recognition of microbial components or PAMPs present in the barn dust by PRRs. These PRRs are present in the interior cell compartments or exterior cell surface. Three main families of PRRs present in the cell include; TLRs, retinoic acid inducible gene-I like receptors (RLRs), and NLRs. Among TLRs, TLR4 forms a complex with CD14 and MD2, and recognizes bacterial LPS presented via LPS binding protein (LBP), resulting in the downstream activation of adaptors (MyD88 and TRIF) and a cascade of kinases, ultimately leading to transcriptional induction of pro-inflammatory cytokines (IL-1β, IL-6, IL-8, TNF-α) and relatively less Type I IFNs (IFN-β, -α). Studies delineating the role of TLR4 in swine barn air-induced lung dysfunction in wild type (WT) and TLR4 mutant mice have implicated TLR4 independent mechanisms responsible for airway inflammation upon chronic exposure to swine barn dust air. 2 Studies using endotoxin-depleted barn dust samples have elicited inflammatory responses in a variety of cells to indicate inflammatory potential of components other than endotoxin in the dust samples.12-14 Following exposure to swine barn dust extract, airway epithelial cells up-regulated TLR2 mRNA and protein in a concentration-dependent manner and specific Ab-mediated blocking of TLR2 dampened the pro-inflammatory cytokine release in vitro.15,16 In a TLR2-deficient mouse model, single and repeated exposures to swine barn dust resulted in a significant reduction in neutrophil influx, cytokine production and loss of lung parenchyma was observed. However, airway hyper responsiveness (AHR) and NO release still persisted in these mice.17-19 Finally, cytoplasmic NLR, NOD2, which recognizes muramyl dipeptide from Gram-positive bacterial cell wall is implicated in airway inflammation. 20 Taken together, these evidences indicate the complexity of host inflammatory response upon swine barn dust exposure.
Airway epithelial cells in general are the primary cells that interface respiratory tract to the environment and are the first responders to the constituents of dust extracts. 21 BEAS-2B or primary human bronchial epithelial cells (HBEC) are the two frequently used cell types to model the responses to swine barn dust extracts (SBDE) in vitro.12,22 SBDE exposure of either BEAS-2B or primary HBEC leads to production of IL-6, IL-8 and TNF-α in a PKC-dependent manner. 22 SBDE exposure leads to sequential activation of PKC isoforms with an early activation of PKCα isoform within 1 h post exposure to promote TNF-α and IL-6 production. 23 Apart from epithelial cells lining the respiratory tract, lung alveolar macrophages play a critical role in maintaining lung homeostasis by limiting the inflammation, at the same time functioning to promote defense towards respiratory pathogens. Monocytes, when differentiated into macrophages in the presence of endotoxin depleted organic dust extracts (ODE), a significant reduction in surface marker expression (HLA-DR, CD80, CD86), phagocytic activity and cytokine responsiveness was observed. 24 This differentiation model indicated that repeated exposure to organic dusts significantly impairs the normal function of monocyte-derived macrophages. 24 This impaired macrophage function was also observed in swine alveolar macrophages upon treatment with SBDE. 25 Furthermore, in swine macrophages an increase in porcine reproductive and respiratory syndrome virus (PRRSV) receptor CD163 was observed.
In the current study, inflammatory responses to SBDE treatment were modeled in vitro in two well-established human cell lines namely, BEAS-2B, a bronchial epithelial cell line 26 and THP-1, a human acute leukemic monocytic cell line established from a leukemic patient. 27 We chose these cell lines to represent airway epithelial (BEAS-2B) and resident or recruited alveolar macrophages (THP-1), since inhaled swine barn dust has been shown to induce airway epithelial as well as alveolar inflammation. 28 With the extensive existing knowledge of signaling event modifications occurring prior to and independent of changes in cellular transcription or translation, we took advantage of characterizing post translational modifications; in particular, kinase-mediated phosphorylation events. Using kinome peptide arrays, we characterized phosphorylation-mediated signal transduction events in BEAS-2B and THP-1 cell lines treated with SBDE or innate immune ligands LPS or PGN. We then compared SBDE-induced signaling pathways with those induced upon treatment with LPS and PGN. Upon comparison, unique or shared signaling pathways among treatments were identified and further validated by transcriptional analyses of downstream target genes using quantitative real time PCR. Finally, comparisons were made between the cell lines BEAS-2B and THP-1 in terms of magnitude of responses to SBDE. This comparison was made in order to implicate critical cell types responsible for airway inflammation and lung pathology. Although the cell types employed in the current study are not primary cell types, they were frequently employed in past studies to investigate responses to barn dust, with no significant response variation observed when compared with primary cell types.14,18,20,22
Materials and methods
Cell culture
Prior to all experiments, live/dead cell count was determined by 4% trypan blue dye exclusion for viability. Cell populations with a minimum of 95% viability were used for the experiments. HBEC line (BEAS-2B, ATCC CRL-9609) was seeded (1 × 106) onto type I bovine collagen (StemCell Technologies, Vancouver, BC)-coated T-75 flasks. Cells were maintained in serum-free LHC-9 medium (Gibco) containing penicillin/streptomycin (100 U/ml; Gibco) and amphotericin B (2 µg/ml; Sigma) in a humidified chamber at 37˚C/5% CO2 until 80% confluent. Immortalized human monocytic cell line (THP-1, ATCC TIB-202) was maintained in suspension culture in Roswell Park Memorial Institute medium (RPMI-1640, Gibco) containing 10% of heat inactivated FBS (Atlanta Biologics) and supplemented with HEPES buffer (10 mM; Gibco), sodium pyruvate (1 mM; Sigma), D-Glc (4.5 g/l; Sigma), sodium bicarbonate (1.5 g/l; Sigma), penicillin/streptomycin (100 U/ml; Gibco) and amphotericin B (2 µg/ml; Sigma). Cells were maintained in a humidified chamber at 37˚C/5% CO2. 2-3 × 106 cells were stimulated with 10 nM phorbol 12-myristate 13-acetate (PMA, Sigma) diluted in RPMI-1640 with 1% FBS for 24 h to differentiate monocytes into macrophages and incubated for an additional 24 h.
SBDE preparation
Settled dust was collected from commercial swine production units. Dust was brushed into zip lock bags containing a desiccant, transported to the laboratory on ice and stored at −80℃ until processed. SBDE was prepared as previously described. 22 Briefly, dust collected was weighed and 1 g of the dust was placed in 10 ml of Hank’s Balanced Salt Solution (HBSS) without calcium (Gibco). The mixture was vortexed and allowed to stand at room temperature for 1 h. The mixture was centrifuged for 20 min (1365 g, 4℃). Supernatant was collected, centrifuged again at 1365 g and 4℃. The final supernatant was collected, and filter sterilized by passing through a 0.22 µm filters (EMD Millipore). Filtered SBDE was aliquoted and stored at −80℃.
Experimental treatments
SBDE, LPS (Escherichia coli O127:B8; Sigma), and PGN (Staphylococcus aureus; Sigma) treatments were performed on BEAS-2B and THP-1 cell lines. Ligand stimulants (SBDE, LPS, PGN) were prepared by dissolving stock concentrations in a serum-free culture medium to achieve a final concentration of 10 µg/ml (LPS and PGN) and 5% (SBDE), respectively. Five percent SBDE is known to induce maximal pro-inflammatory response with limited cytotoxicity. 13 Stimulant treatments were performed at the respective final concentrations and media alone treated cells served as no treatment controls. Stimulant and no treatment control cell pellets were prepared at 0 − and 24-h post treatment for kinome and transcriptional analyses.
Kinome analyses
Kinome analyses was performed as previously described by Arsenault et al.29,30 Briefly, cell lysates were prepared in 100 µl lysis buffer comprised of protease and phosphatase inhibitors followed by a 10 min incubation period on ice. After a brief centrifugation for 10 min, 70 µl supernatants were mixed with 10 µl of activation mix followed by incubation on the human peptide array slides at 37℃ overnight. Slides were washed with 1% Triton – PBS, air dried and immersed in phospho-specific fluorescent ProQ Diamond Phosphoprotein Stain (Invitrogen) to agitate for 1 h. Slides were then washed three times in destaining solution containing 20% acetonitrile (EMD Biosciences, VWR Distributor, Mississauga, ON, Canada) and 50 mM sodium acetate (Sigma) at pH 4.0 for 10 min. A final wash was done with distilled deionized water. Slides were then air dried and briefly centrifuged at 300 g for 2 min to remove any residual moisture.
Array slides were read using a Tecan PowerScanner (TECAN Männedorf, Switzerland) at 532 to 560 nm with a 580-nm filter to detect dye fluorescence. Images were collected using GenePix, version 6.0, software (MDS), and spot intensity signal was collected as a mean of pixel intensity using local feature background intensity calculation with the default scanner saturation level. Differentially phosphorylated peptides in treatments relative to media alone treated control cells were determined.
RNA extraction, cDNA synthesis and qRT-PCR analysis
DAVID GO-term enrichment of innate immune pathways in BEAS-2B cell line treatments.

DAVID: Database for Annotation, Visualization and Integrated Discovery.
Differentially phosphorylated substrate peptides in SBDE-, LPS- and PGN-treated BEAS-2B cell line.
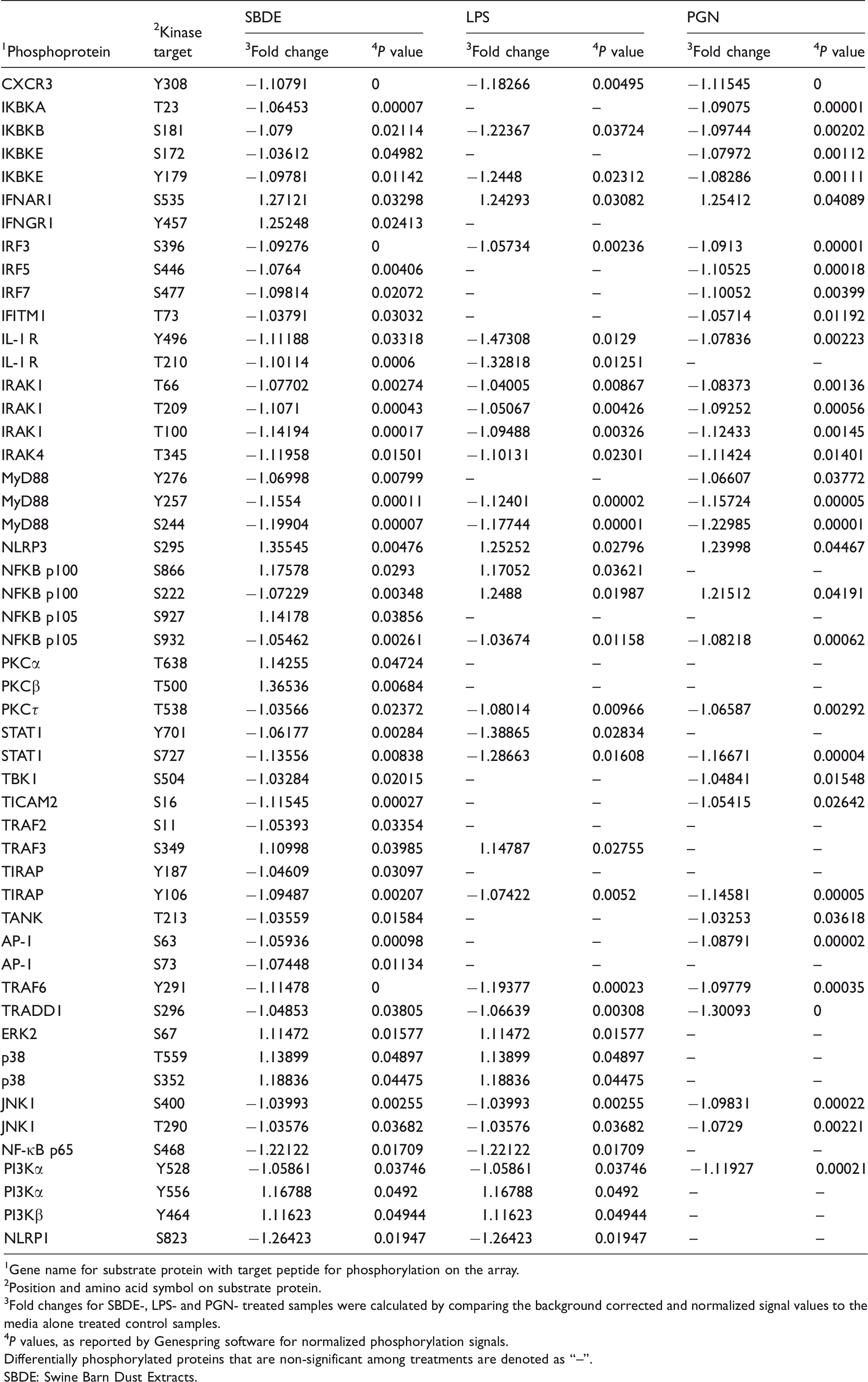
1 Gene name for substrate protein with target peptide for phosphorylation on the array.
2 Position and amino acid symbol on substrate protein.
3 Fold changes for SBDE-, LPS- and PGN- treated samples were calculated by comparing the background corrected and normalized signal values to the media alone treated control samples.
4 P values, as reported by Genespring software for normalized phosphorylation signals.
Differentially phosphorylated proteins that are non-significant among treatments are denoted as “–”.
SBDE: Swine Barn Dust Extracts.
Differentially phosphorylated substrate peptides in SBDE-, LPS- and PGN-treated THP-1 monocyte derived macrophage cell line.
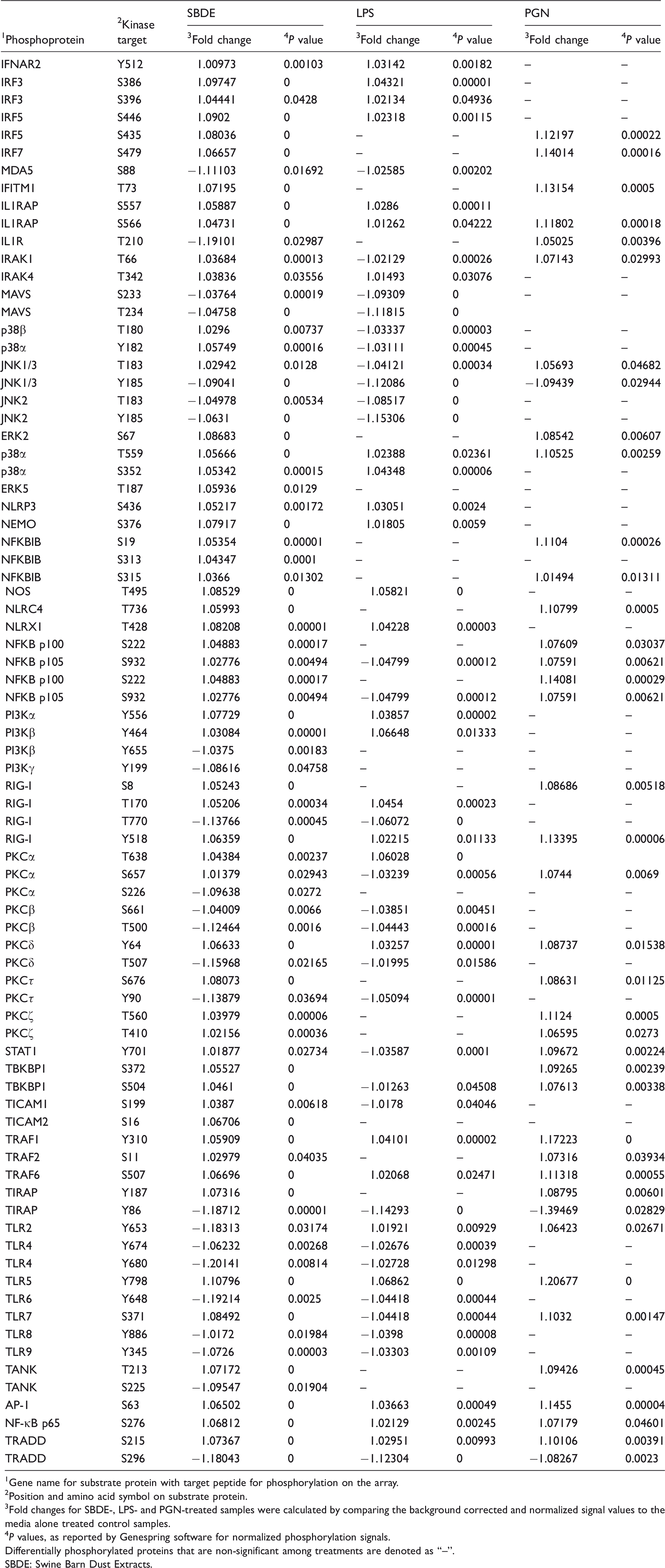
1 Gene name for substrate protein with target peptide for phosphorylation on the array.
2 Position and amino acid symbol on substrate protein.
3 Fold changes for SBDE-, LPS- and PGN-treated samples were calculated by comparing the background corrected and normalized signal values to the media alone treated control samples.
4 P values, as reported by Genespring software for normalized phosphorylation signals.
Differentially phosphorylated proteins that are non-significant among treatments are denoted as “–”.
SBDE: Swine Barn Dust Extracts.
Database for Annotation, Visualization and Integrated Discovery (DAVID) analysis
Differentially phosphorylated peptides and their corresponding UniprotKB entry ID lists (P ≤ 0.05) for each treatment were input and submitted to the gene list tab of DAVID with selected default background “Homo sapiens”. 33 This is due to the fact that a larger gene background tends to produce smaller P values than prebuilt backgrounds on the chips, in our case arrays. 33 Gene Ontology (GO) term enrichments under the section “Functional annotation chart” and annotation term relationships among the proteins in the input list under the section “Functional annotation clustering” were downloaded in XML format. Upon functional annotation, KEGG pathways under the DAVID pathway viewer were explored for significantly enriched pathways related to innate immunity and are described in the results section.
Ingenuity Pathway Analysis (IPA)
The kinome data set which included UniprotKB entry identifiers (IDs), fold changes and P values for each differentially phosphorylated peptide was input into IPA using the core analysis platform (Qiagen Inc.). The core analysis matched and retrieved proteins in our data set with those in the Ingenuity Knowledge Base, created molecular networks, divided the data set into biological functions that were significantly over represented and determined overrepresented canonical pathways. One hundred percent of input IDs were mapped to their corresponding proteins. Core analyses was performed with default settings: direct and indirect relationships between proteins supported by experimental treatments were considered, networks did not exceed 35 proteins, and all sources of data from human, mouse, and rat studies in the Ingenuity Knowledge Base were considered. Since the input IDs are selected based on their differential phosphorylation status and significance (P < 0.05), no cut-offs were applied with regards to fold changes and P values. IPA generated priority lists for enriched molecular networks, biological functions, canonical pathways, differentially abundant proteins, and predicted upstream regulators. Network scoring was based on the number of mapped proteins in the network, network size, and the number of proteins in the Ingenuity Knowledge Base that could be included in the network. Network scores were calculated based from hypergeometric distribution and right-tailed Fisher’s exact test.
Results
SBDE-induced kinome signaling in BEAS-2B cell line
In BEAS-2B cells treated with SBDE, significantly over-represented DAVID pathway clusters include chemokine signaling, pathways in cancer, TLR signaling pathways, TNF-α signaling pathway, and pathways associated with virus infections (Influenza A Virus (IAV), Hepatitis B Virus (HBV) and Hepatitis C Virus (HCV)) (Table 1). Among them, we intend to focus on relevant innate immune signaling pathways. To begin with, components in the SBDE are recognized by PRRs such as TLRs. Based on this, differential phosphorylation of TLR adaptor MyD88 was observed with subsequent downstream activation of kinases (IRAK1, IRAK4, IκBK-α, -β, -ɛ) that activate the NF-κB and AP-1 family of transcription factors (Table 2). MyD88 serves as an adaptor for TLR2, TLR4, TLR5, TLR7/8/9 whereas TLR4 can also signal via TRIF/TIRAP to activate downstream kinase complex TBK1/IKKɛ, responsible for activation of IFN regulatory factors (IRF3 and 7) leading to Type I IFN production. TLR4-TICAM-TRAF3-dependent IRF3/7 activation pathway also appears to be active, resulting in Type I IFN release and subsequent activation of IFN-αβ receptor and STAT1. Type I IFNs act in auto or paracrine manner to induce transcriptional activation of IFN-stimulated genes, among them include IFN-induced transmembrane protein 1 (IFITM1) which was found to be differentially phosphorylated (Table 2).
Based on the observed activation of IFN induction and downstream Type I IFN signaling pathways, we evaluated mRNA expression levels of Type I IFNs (IFN-β, -α) and one of the downstream effectors of IFN signaling (IFIT1). Interestingly, we found a significant induction of IFN-α but not IFN-β (Figure 1). However, no downstream IFN-induced signaling was observed as seen with a lack of induction of IFIT1 (IFIT1 threshold cycles remained > 35 among all the study treatments in both BEAS-2B and THP-1 cell lines). As expected, a lack of Type I IFNs and IFN signaling was also evident in controls, LPS-and PGN-treated cells, whose responses are primarily pro-inflammatory with no or little amounts of Type I IFNs.
Relative cytokine gene expression in BEAS-2B cells treated with SBDE (red bars), LPS (blue bars) and PGN (green bars). Error bars indicate standard deviation and asterisk denotes statistical significance (P < 0.05). Y-Axis indicates mRNA fold change compared with media treated control. Target gene expression was normalized to endogenous reference GAPDH.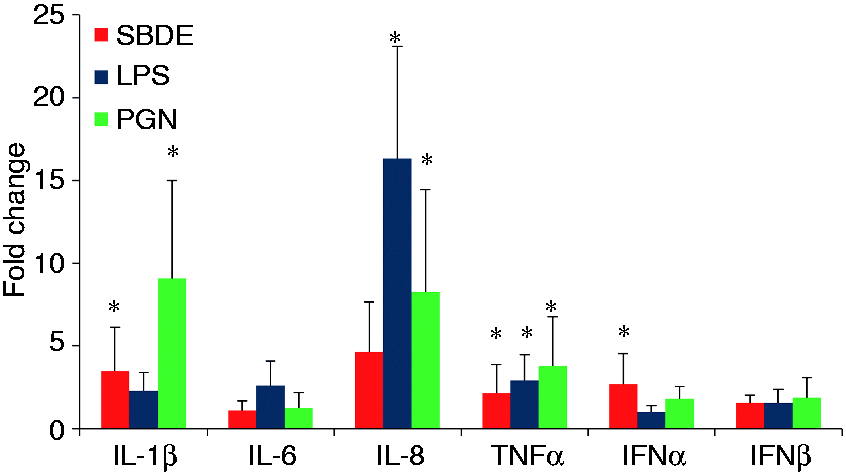
TLR2, upon stimulation with heat-killed S. aureus, is known to activate NF-κB in a Rac1-PI3K-dependent manner in THP-1 cells. 34 This pathway appears to be active in the SBDE-treated BEAS-2B cell line (Table 2). Finally, both TLR2 and TLR4 pathways appear to be activated in response to SBDE in the BEAS-2B cell line. Additionally, various TLR ligands are known to stimulate downstream activation of MAPKs such as ERK1/2, JNK and p38, with all three of them found to be differentially phosphorylated in SBDE-treated cells (Table 2). 35 LPS-treated BEAS-2B cells have shown proliferative response via thymidine [H3+] incorporation in a p38, JNK and PI3K dependent manner whereas this effect was ERK1/2-independent. 36
There was a significant enrichment of pathways related to cancer, among which protein kinase signaling mediated by PKC and PKA are evident. PKC and PKA signaling occur downstream of GPCR activation and play agonistic and antagonistic roles, respectively, in promoting IL-6 induction during SBDE exposure. 13 Finally, studies have shown that TLR-mediated Tpl2 activation activates ERK1/2 through MEK3/6 phosphorylation and this event regulates nucleocytoplasmic shuttling of TNF-α mRNA. 37 Both Tpl-ERK1/2 and TNF-α signaling were significantly enriched in our study (Table 3). Congruent with TNF-α signaling and PKC activation, TNF-α mRNA was > 2-fold up-regulated. IL-6 mRNA levels however, remained at basal levels (Figure 1). TNF-α mRNA levels in SBDE-treated cells were comparable with LPS- and PGN-treated cells.
As epithelial cells are the first cells that respond to organic dust, chemokine signaling is not unusual. Chemokines produced by these cells function in autocrine or paracrine manner to recruit other innate cells such as neutrophils (CXCL8) and monocytes (MCP1). By virtue of this, we decided to quantify key chemokines involved in neutrophil influx and airway pathology, IL-8 (CXCL8).1,18,22,23 Although not significant, we observed a 4.6-fold induction of IL-8 mRNA in SBDE-treated cells. A greater level of induction was seen in LPS-treated cells followed by PGN controls (Figure 1). The differentially phosphorylated receptor on the array was CXCR3. This is consistent with the constitutive expression of CXCR3 on HBEC. 38 HBEC also produce CXCR3 ligands in response to pro-inflammatory cytokines such as TNF-α. In line with this, TNF-α receptor signaling was found to be activated with subsequent downstream activation of the NF-κB/AP-1 family of transcription factors (Table 2). CXCR3 is a GPCR which upon ligand activation appears to stimulate cell proliferation under normal conditions. However, under pro-inflammatory milieu, it may inhibit epithelial cell proliferation leading to airway mucosal denudation and damage which appear to be the predominant mechanism in play, based on our results. 38 CXCR3 activation leads to downstream activation of PI3K and MAPK (ERK1/2 and p38 arms), both of which were found to be differentially phosphorylated (Table 2). 38
Lastly, we also found a significant enrichment of pathways related to inflammasome activation (Tables 1 and 2). NLRs, NLRP3 and NLRP1 were found differentially phosphorylated on our array. NLRP3 is activated via wide array of ligands including microbial (pore-forming toxins) host derived (ATP, urate, mitochondrial DNA (mtDNA), K+) and sterile substances (silica, asbestos, alum). Furthermore, brief LPS stimulation appears to activate NLRP3 independent of its transcriptional induction. This mode of activation is dependent on mitochondrial ROS and is further potentiated by ligand ATP.39,40 NLRP3 activation leads to ASC-mediated caspase 1 activation which processes pro-forms of IL-1β and IL-18 into mature secretory forms. In line with this, downstream activation of IL-1 R was seen. NLRP3 inflammasome activation and IL-1 family cytokine production are regulated at transcriptional and post transcriptional levels. A two signal model for NLRP3 activation indicates initial priming or the first signal being microbial or cytokine molecules that induce NLRP3 and pro-IL-1β mRNA via NF-κB activation followed by a second signal or NLRP3 activation triggered by NLRP3 ligands ATP, pore-forming toxins, viral RNA, or particulate matter.41,42 We investigated the priming signal by quantifying IL-1β mRNA levels and found a significant induction in SBDE-treated cells. In addition, a non-significant up-regulation and a significant up-regulation of IL-1β mRNA was found in LPS- and PGN-treated cells, respectively (Figure 1).
To identify protein interaction networks among the differentially phosphorylated proteins in SBDE-treated BEAS-2B cells and to connect them to the molecular networks deposited within the Ingenuity Knowledge Base, which contains biological interactions and functional annotations derived from literature or verified experimental models, we used IPA. IPA identified the top enriched molecular network to contain proteins with functions in cell signaling, inflammatory response, and antimicrobial response (score, 29; number of focus proteins in network, 17). Several protein hubs displayed high connectivity among others in the network (>8 direct or indirect connections), which included hubs involved in signal transduction by TLR (MyD88, IRAK4, IRAK1, TIRAP), IFN transcription (IRF-3, -5, -7), cytokine-mediated signaling (IL-1 R, IFNGR1, IFNAR) and chemokine signaling (CXCR3) (Figure 2a). In the second (score, 25; focus molecules, 15) and third (score, 25; focus molecules, 15) networks, proteins enriched in our kinome data set connected to major regulators of inflammation, such as inflammasome, NF-κB, STAT3, TNF-α and caspases (Figure 2b, 2c).
The top three enriched networks of interacting proteins in SBDE-treated BEAS2B cells, as determined by IPA. These networks were defined as: (a) cell signaling, inflammatory response, antimicrobial response (score, 29; 17 differentially phosphorylated proteins); (b) Cell signaling, cellular function and maintenance, cell death and survival (score, 25; 15 differentially phosphorylated proteins); (c) Cell death and survival, organismal injury and abnormalities, gastrointestinal disease (score, 25; 15 differentially phosphorylated proteins). Proteins represented in green (decreased) and red (increased) were differentially phosphorylated on the kinome array relative to control untreated cells. Proteins shown in white were not present in our data set but were added by IPA due to their connection to the enriched proteins. Scores were calculated from hypergeometric distribution and right-tailed Fisher’s exact test.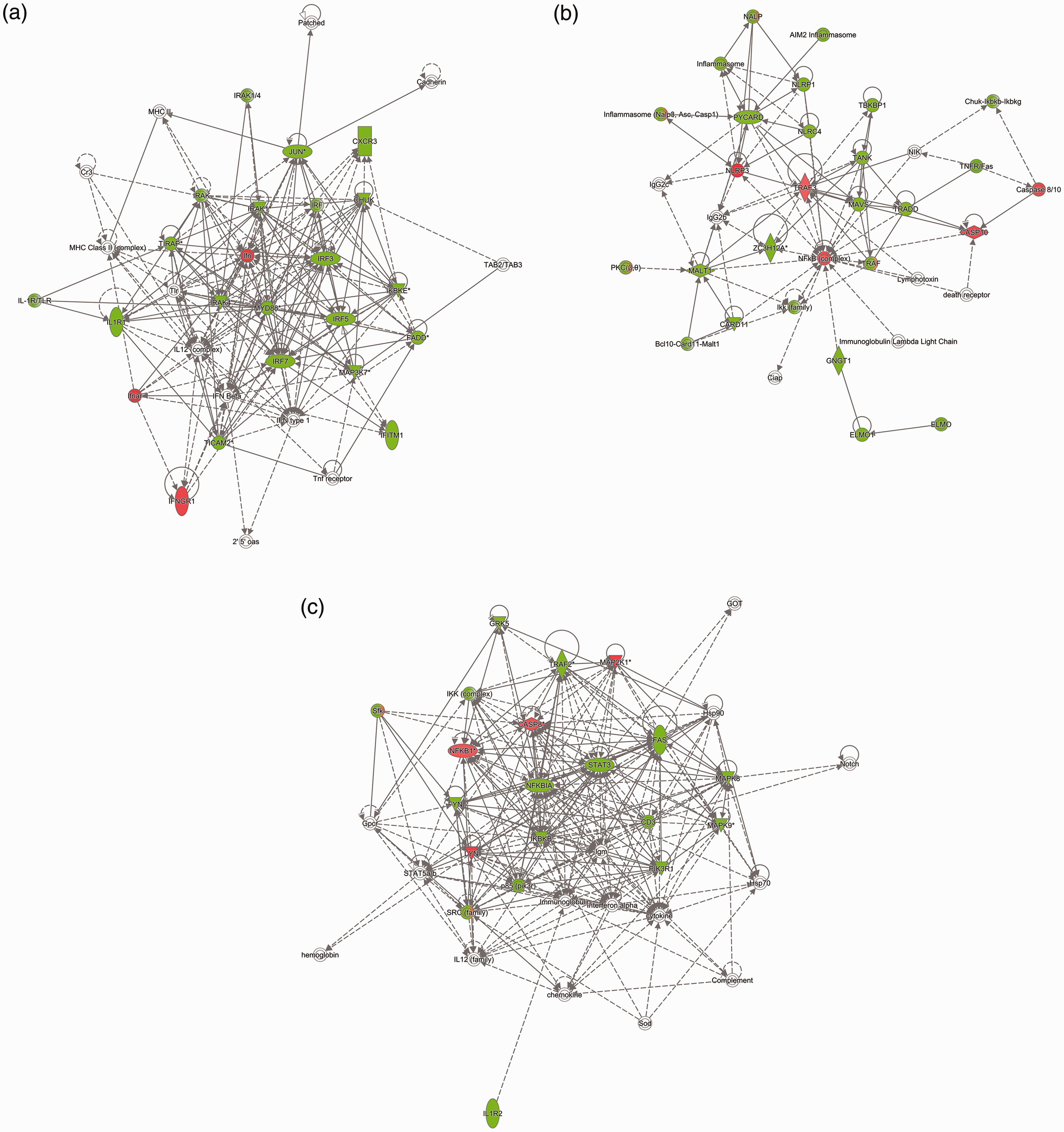
LPS-induced kinome signaling in BEAS-2B cell line
In BEAS-2B cells treated with LPS there was a stark enrichment of canonical TLR4 pathway. In addition, an activation of TLR2 signaling was also observed (Table 2). This could be due to cross talk among TLR signaling pathways or possible contamination of the LPS with other TLR ligands. In any case, the downstream consequence of signal integration was activation of NF-κB/AP-1 and IRF3 transcription factors. Lending further support, there was an activation of IFN-α/β receptor and downstream STAT1 indicating interferon responsiveness. Other pathways enriched included NF-κB (IL-1 R) and TNF-α receptor signaling, while the latter is interdependent on the former activation. Unsurprisingly, cancer pathways, similar to our observation in SBDE-treated cells, were found to be enriched indicating putative role played by LPS in stimulating cell proliferation.
PGN-induced kinome signaling in BEAS-2B cell line
In BEAS-2B cells treated with PGN, similar to the cross-talk patterns observed in the LPS-treated cells, there is an activation of TLR2 and TLR4 signaling. Consistent with the former, an enrichment of TNF-α signaling and chemokine signaling was observed (Table 2). Finally, cancer pathways commonly observed among the other two treatments (SBDE and LPS) were seen.
SBDE-induced kinome signaling in THP-1 cell line
In THP-1 cells treated with SBDE, interesting clusters that yield valuable information were observed. Although TLR signaling was one of the enriched pathways, we found novel signaling pathways that were not observed in BEAS-2B cells treated with SBDE. These include TLR5, TLR7/8 and TLR9 (Table 3). As a readout for TLR-mediated downstream activation of NF-κB, we investigated mRNA expression levels of pro-inflammatory cytokines (IL-1β, IL-6 and TNF-α) and chemokines (IL-8). A greater level of IL-1β and IL-8 was observed in SBDE-treated cells compared with LPS- and PGN-treated controls (Figure 3a, b and e). Furthermore, significant induction of both IL-6 and TNF-α was observed in SBDE-treated cells (Figure 3c and d). In addition, endosomal TLRs, TLR7/8 and TLR9 that recognize ssRNA and unmethylated bacterial CpG DNA sequences, respectively, were found to be differentially phosphorylated. Based on the enrichment of aforementioned pathways, the dust-associated dander may presumably encompass RNA and CpG DNA originating from viruses and bacteria, respectively.
Relative cytokine gene expression in THP-1 cells treated with SBDE, LPS and PGN. Panels a, b, c, d and e depict IL-1β, IL-6, TNF-α, IL-8 and Type I IFNs (-α, -β), respectively. Error bars indicate standard deviation and asterisk denotes statistical significance (P < 0.05). Y-axis indicates mRNA fold change compared with media treated controls. Target gene expression was normalized to endogenous reference GAPDH.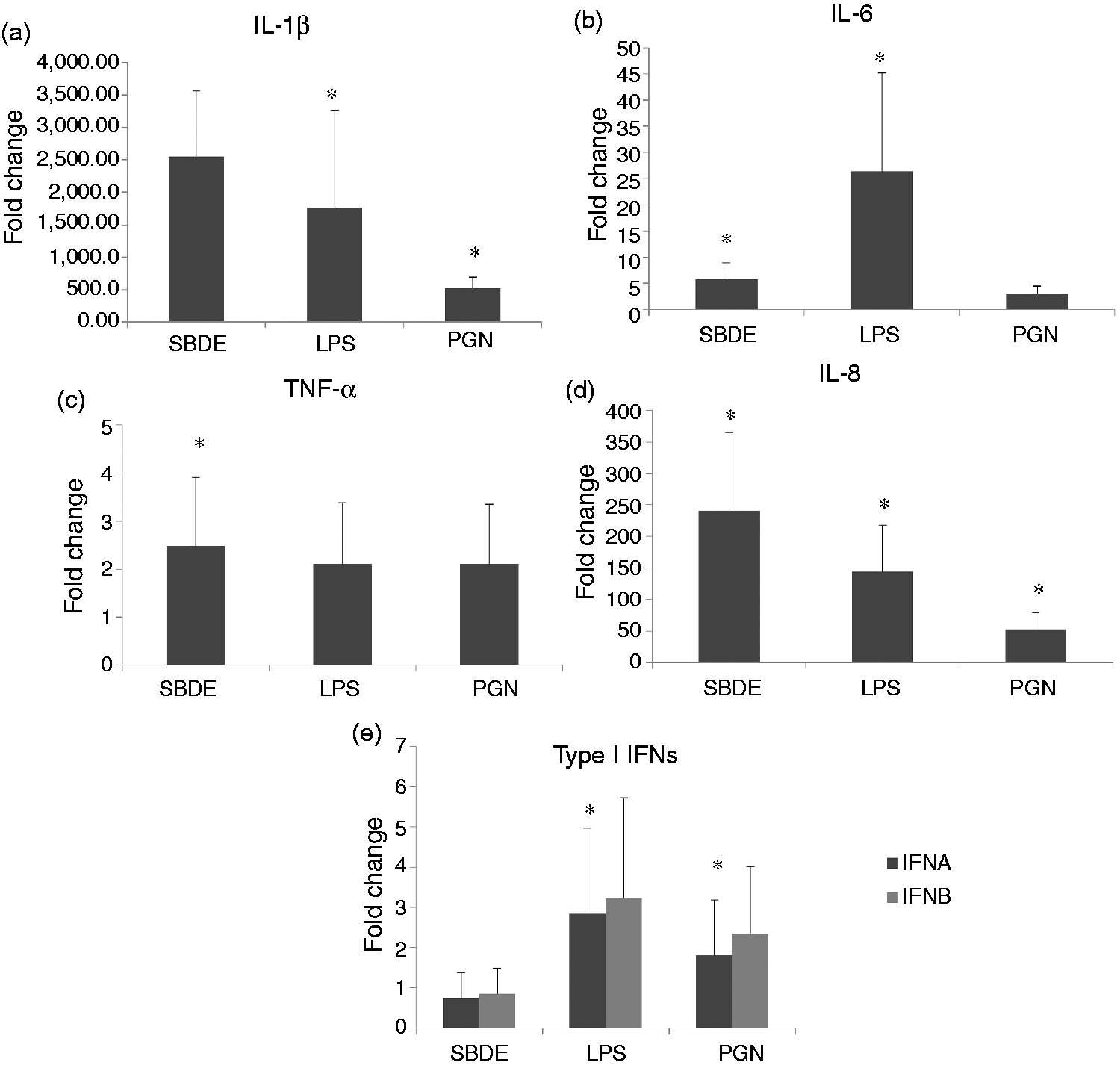
DAVID GO-term enrichment of innate immune pathways in THP-1 cell line treatments.

DAVID: Database for Annotation, Visualization and Integrated Discovery.
Using IPA, we identified protein interaction networks among the differentially phosphorylated proteins in SBDE-treated THP-1 cells and connected the proteins to molecular networks contained within the Ingenuity Knowledge Base. Upon molecular network analysis, IPA identified the top enriched molecular network to contain proteins involved in cell signaling, gene expression, cell death, survival (score, 294; focus proteins in network, 16). Protein hubs that displayed high connectivity in the network (>8 direct or indirect connections), include those involved in signal transduction by TNFR (TRAF, TRADD, TAB1), in line with the transcriptional induction of TNF-α (Figure 3d, Figure 4a). In the second (score, 24; focus molecules, 16) and third (score, 24; focus molecules, 16) networks, proteins enriched in our kinome data set connected to cellular reorganization and cell death, respectively (Figure 4b and c), implicating macrophage-mediated tissue reorganization due to SBDE-exposure-mediated airway pathology.
The top three enriched networks of interacting proteins in SBDE-treated THP-1 cells, as determined by IPA. These networks were defined as: (a) cell signaling, gene expression, cell death, survival (score, 24; 16 differentially phosphorylated proteins); (b) Cellular assembly, organization, cellular function and maintenance, tissue development (score, 24; 16 differentially phosphorylated proteins); (c) Cell death and survival, gastrointestinal disease, hepatic system disease (score, 24; 16 differentially phosphorylated proteins). Proteins represented in green (decreased) and red (increased) were differentially phosphorylated on the kinome array relative to control untreated cells. Proteins shown in white were not present in our data set but were added by IPA due to their connection to the enriched proteins. Scores were calculated from hypergeometric distribution and right-tailed Fisher’s exact test.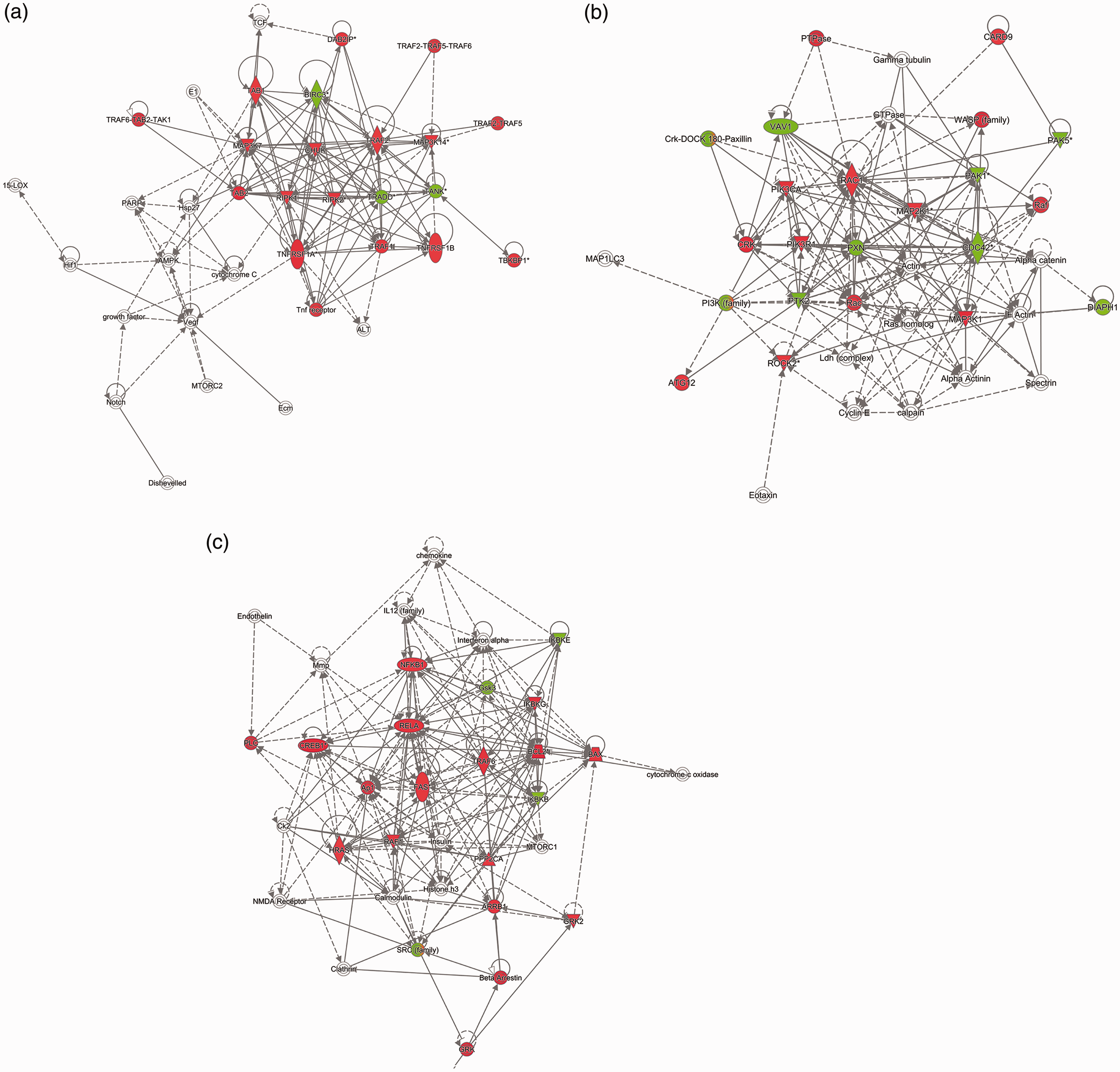
LPS-induced kinome signaling in THP-1 cell line
In THP-1 cells treated with LPS, canonical TLR4 signaling was seen. Additionally, due to cross talk among TLR receptors or possible contamination of the LPS with other TLR ligands, TLR2 and TLR5 activation was also observed (Table 3). TLR signaling leads to downstream activation of NF-κB and TNF-α production. In line with this, TNF-α receptor and the downstream targets were found to be differentially phosphorylated. Other enriched pathways include pathways related to Epstein Barr virus infection (EBV), and parasitic infections (Chagas disease caused by Trypanasoma cruzi and Toxoplasmosis caused by Toxoplasma gondii) due a significant overlap of these pathways with TLR and downstream NF-κB activation leading to their overrepresentation (Table 4).
PGN-induced kinome signaling in THP-1 cell line
In THP-1 cells treated with PGN, TLR2 signaling was seen (Table 3). Additionally, due to cross talk among TLR receptors, TLR5 activation was also observed. Other enriched pathways include pathways related to parasitic infections (Chagas disease and Toxoplasmosis) and TNF-α receptor signaling (Table 4).
Discussion
In this manuscript, we report on various cell-signaling pathways activated upon treatment of human airway epithelial or macrophage cells with SBDE or LPS or PGN. Our work highlights the complexity of SBDE constituents and SBDE-induced host response through activation of multiple PRRs-related pathways. Upon comparison of differential phosphorylation profiles of SBDE, LPS or PGN treatment, common trends observed include TLR2 and TLR4 activation, NF-κB signaling, TNF-α receptor signaling and chemokine signaling. These trends indicate significant amounts of TLR2 and 4 ligands in SBDE. Furthermore, although the contributory role played by endotoxin is thought to be minor, apart from TLR4 activation, LPS also contributes to the priming and activation of NLRP3 inflammasome. In addition, muramyl dipeptide, a constituent of Gram-positive cell walls, is known to activate NLRP1 in the presence of NOD2, leading to IL-1β secretion. The presence of other NLR ligands in the SBDE extracts is unknown. Finally, inflammasomes play an important role in the pathogenesis of many respiratory diseases such as ARDS, COPD, fibrotic lung disease and lung cancer. Based on the pathway responses seen in our study, it appears that chronic activation of TLRs and inflammasomes by SBDE appears to provide primary and secondary signals necessary for pro-inflammatory IL-1β production. This is corroborated by the observed induction of IL-1β mRNA in both BEAS-2B and THP-1 cell lines. On the other hand, TLR-mediated activation of NF-κB leads to IL-6, IL-8 (CXCL8) and TNF-α production. IL-8 is a chemotactic cytokine responsible for neutrophil influx and subsequent immune-related pathology seen in airways upon chronic exposure to barn dust.
In THP-1 cells, additional TLR and RLR pathways were enriched, indicating the presence of respective ligands. These include TLR5, TLR7/8, TLR9, RIG-I, MDA5 and MAVS. Besides TLR2 and TLR4, TLR5 is activated by flagellin proteins found in Gram-negative bacteria. In fact, highly purified recombinant flagellin (rFLA) was found to sensitize mice to allergic inflammation when administered with OVA. 44 In the same study, TLR5 was shown to be required for priming strong allergic responses to natural indoor allergens present in house dust extracts (HDEs). The role of TLR5 in barn dust-induced airway inflammation was not investigated. However, the presence of flagellin proteins or fragments in the barn dust cannot be excluded and necessitates detailed investigation. Henceforth, the putative role played by their respective ligands and downstream pathway activation during airway inflammation begs further investigation. This result is an important future avenue of research on the effects of swine barn dust and may lead to additional targets of intervention by reducing TLR5 responses in exposed individuals.
In our study, however, we observed a lack of RIG-I- and MDA5-mediated Type I IFN induction or downstream IFN signaling in THP-1 cells. On the other hand, in BEAS-2B cells a significant induction of Type I IFNs with a lack of downstream IFN signaling was observed. Nevertheless, a lack of downstream interferon signaling indicated by an absence of IFIT1 induction suggests no significant role played by Type I IFNs in SBDE-mediated inflammation. Furthermore, our efforts to amplify viral (IAV) RNA (RIG-I ligand) in SBDE inoculum remained futile. Henceforth, airway pathology seen upon chronic exposure to barn dust is predominantly pro-inflammatory cytokine mediated, including IL-8-mediated neutrophil influx. Based on the responses seen in BEAS-2B and THP-1 cells, both bronchial epithelial cells and resident alveolar macrophages appear to play a critical role in IL-8 production and neutrophil influx. We observed a significantly stronger immune activation both at the gene expression and kinome levels in THP-1 cells compared with BEAS-2B cells. This may be due to the greater activation potential of macrophage compared with epithelial cells, it also may indicate that while activated macrophages are highly inflammatory, epithelial cells require a more balanced and tolerant response to the constant exposure to immune ligands.
To dissect the specific pathways enriched by the SBDE other than those mediated by TLR4 and TLR2, we performed pathway enrichment analyses on the basis of mutual exclusion. Significant phosphorylation events that are either up- or down-regulated among all treatments were excluded whereas those events that exhibit temporal changes in at least one of the three treatments were retained for analyses. In this scenario, a higher number of events was detected in SBDE-treated THP-1 cells when compared with SBDE-treated BEAS-2B cells (453 vs 31). Enriched pathways upon exclusion in BEAS-2B cell line include cancer pathways, chemokine, VEGF, NF-κB, Ca2+, TNF-α and leucocyte trans-endothelial migration signaling pathways (data not shown). These pathways describe potent activation of downstream NF-κB and production of chemokines leading to further recruitment of other leukocytes such as neutrophils. In THP-1 cells, enriched pathways upon exclusion include cancer pathways, chemokine, TLR, RLR, NF-κB, TNFR apoptosis related, TNF-α and leucocyte trans-endothelial migration signaling pathways (data not shown). Additional activation of TLR5, TLR7/8, TLR9, RIG-I and MDA5 seen in THP-1 cells, is reiterated upon exclusion-based analyses.
Collectively, our kinome array data indicates that SBDE is a potent stimulator of pro-inflammatory cytokine production with greater levels of IL-8 secreted by BEAS-2B and THP-1 cells. In agreement with this, a recent study reported greater levels of IL-8 secretion by lung epithelial (BEAS-2B and A549) and THP-1 monocyte cell lines treated with poultry dust extract. 9 Furthermore, IL-8 induction was reported to be associated with activation of PKC and MAPK and binding of AP1 and NF-κB to IL-8 promoter, all of which were identified to be differentially phosphorylated on our arrays. Targeting the IL-8 response either by inhibiting up-stream pathway activation or inhibiting the IL-18 cytokine by anti-IL-8 therapies may prove beneficial in reducing the effects of swine barn dust exposure. Our kinome and gene expression analyses data in BEAS-2B and THP-1 were quite similar to the effects in dust-exposed primary normal human bronchial cells and human peripheral blood monocytes, respectively, attesting that pro-inflammatory effects of SBDE are independent of the nature of cell type employed. 22 The multiple immune ligands found in SBDE and the activation of multiple receptors makes the immune response to SBDE complex. In this study we have described the multiple and overlapping signal transduction pathways, with the goal of better characterizing the immune response to SBDE. Swine confinement unit workers exposed to persistent barn air are at occupational risk of developing a range of respiratory illnesses and there is a critical need to understand the mechanisms behind those illnesses.
Supplemental Material
Supplemental material for Kinome analyses of inflammatory responses to swine barn dust extract in human bronchial epithelial and monocyte cell lines
Supplemental material for Kinome analyses of inflammatory responses to swine barn dust extract in human bronchial epithelial and monocyte cell lines by Sabari Nath Neerukonda, Sanjana Mahadev-Bhat, Bridget Aylward, Casey Johnson, Chandrashekhar Charavaryamath and Ryan J Arsenault in Innate Immunity
Footnotes
Acknowledgements
The authors thank Dr Locke A Karriker (VDPAM, Iowa State University) for help with the collection of swine barn dust samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: funding from Iowa State University to C Charavaryamath and from the University of Delaware to R J Arsenault.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
