Abstract
Background. Curculigo orchioides Gaertn is an ancient medicinal plant (Family: Amaryllidaceae), well known for its immunomodulatory and rejuvenating effects. Cyclophosphamide (CPA) is an alkylating agent widely used for treating a variety of human malignancies, but associated with different toxicities too. Our previous reports regarding the hemoprotective and hepatoprotective effects of the plant against CPA toxicities provide the background for the present study, which is designed to analyze the ameliorative effect of the methanolic extract of C orchioides on the urotoxicity and nephrotoxicity induced by CPA. Methods. CPA was administered to male Swiss albino mice at a single dose of 1.5 mmol/kg body weight to induce urotoxicity after 5 days of prophylactic treatment with C orchioides extract (20 mg/kg body weight). Mesna (2-mercaptoethanesulfonate) was used as a control drug. Serum, tissue, and urine levels of kidney function markers and antioxidant levels were checked along with the serum cytokine levels. Results. The plant extract was found to be effective in ameliorating the urotoxic and nephrotoxic side effects of CPA. Upregulation of serum interferon-γ and interleukin-2 levels were observed with C orchioides treatment, which was decreased by CPA administration. Besides these, serum tumor necrosis factor-α level was also downregulated by C orchioides treatment. Conclusion. Curculigo orchioides was found to be effective against the CPA-induced bladder and renal toxicities by its antioxidant capability and also by regulating the pro-inflammatory cytokine levels.
Background
Cyclophosphamide (CPA), an alkylating agent, is a high efficiency anticancer drug belonging to the class of oxazaphosphorines. CPA is widely used in chemotherapeutic treatment and those clinical strategies following the maximum tolerable doses are often associated with therapy-related leukemia and bladder cancer.1-3 Besides, its immu-nosuppressive property 4 finds use in organ transplantation and in the treatment of some autoimmune diseases.5,6 It is a prodrug that requires enzymatic bioactivation by hepatic cytochrome P-450 (CYP) enzymes to form 4-hydroxycyclophosphamide (4-OH-CPA), which on spontaneous interconversion gives its tautomer aldophosphamide. This aldophosphamide undergoes an elimination reaction and fragments to acrolein and phosphoramide mustard (PM).7-9 Both acrolein and PM are reactive species toward DNA. Acrolein is responsible for the damage of host bladder epithelium leading to hematuria and hemorrhagic cystitis, which forms the major dose-limiting toxicity for the administration of CPA. 10 Besides these, N-dechloroethylation generates an inactive compound (dechloroethyl-CPA) and chloroacetaldehyde, which is both nephrotoxic and neurotoxic. 11 Supplementation of agents that can provide protection against the toxic side effects of antineoplastic drugs without affecting their chemotherapeutic efficacy will be of great promise as adjuvants in chemotherapeutic treatment regimens.
Mesna (2-mercaptoethanesulfonate) is an organosulfur compound used to lessen the prevalence of hemorrhagic cystitis and hematuria occurring as side effects of the administration of antineoplastic drugs like CPA. A sulfhydryl group present in mesna can act as an antioxidant, and detoxifies the urotoxic metabolite acrolein. 12 The free sulfhydryl (thiol) groups of mesna combine directly with a double bond of acrolein and with other urotoxic 4-hydroxyoxazaphosphorine metabolites (4-hydroxycyclophosphamide) to form stable nontoxic compounds. 13
A number of research studies suggest that antioxidant supplementation can influence the response to chemotherapy as well as the development of adverse side effects that result from treatment with antineoplastic agents. 14 In the present study ameliorative effects of the methanolic extract of Curculigo orchioides on the urotoxicity and nephrotoxicity induced by the administration of CPA were analyzed. C orchioides, or black musale, is a tiny herb with an edible tuber, seen in subtropical regions of Asia, including China, India, Malaysia and elsewhere, and is one of the important Rasayana drugs of the Ayurvedic system of medicine for vigor and vitality. The extracts of this plant contain a wide variety of glycosides, phenolic compounds, lignans, flavones, triterpenoids, saponins, and other phytochemicals. 15 Curculigosides are reported as the main bioactive components of C orchioides. 16 A number of reported medicinal properties of the plant, including immunomodulatory effects, 17 antioxidant and antitumor activities,18,19 mast cell stabilization, 20 and antiosteoporotic activities 21 emphasize the importance of the plant in combination therapies. Our previous studies showed that C orchioides extract can ameliorate the immune suppression along with the intestinal villi and hepatocellular damage brought about by CPA administration in experimental animals without affecting its chemotherapeutic efficacy. The immunostimulant and antioxidant activity of the plant may be the key players behind these biological effects; the most interesting finding was the synergistic effect of C orchioides and CPA on the tumor volume reduction. CPA at a dose of 20 mg/kg body weight when administered along with C orchioides extract (20 mg/kg body weight) reduced tumor volume significantly relatively to that of animals treated with CPA only at a dose of 25 mg/kg body weight. 22 The aforementioned properties of C orchioides were primarily considered as a precondition in the conception of protective activity against CPA-induced renal and bladder toxicities.
Methods
Animals
Swiss Albino mice (25-28 g, male) were taken from the small animal breeding section of Amala Cancer Research Centre. The animals were fed with normal mouse chow (Krish Scientific Stores, Bangalore, India) and water ad libitum. All the animal experiments were performed according to the rules and regulations of the Animal Ethics Committee, Government of India.
Chemicals and Reagents
Thiobarbituric acid (TBA) was purchased from Sigma Chemical Company Inc (St Louis, MO, USA). Hydrogen peroxide (H2O2), n-butanol, ascorbic acid, pyridine, and ethylenediamine tetraacetic acid (EDTA) were obtained from Merck India Ltd. Nitroblue tetrazolium (NBT), glutathione (GSH), and 5-5-dithiobis-2-nitrobenzoicacid (DTNB) (Ellman’s reagent), and riboflavin were obtained from SISCO Research Laboratory (Mumbai India). Sodium 2-mercaptoethane sulfoxide (mesna), total protein, creatinine and urea analysis kits were obtained from SPAN Diagnostic Ltd. All other chemicals used were of analytical reagent grade. Enzyme-linked immunosorbent assay kit: Highly specific quantitative sandwich enzyme-linked immunosorbent assay kits for mouse interleukin-2 (IL-2), interferon-γ (IFN-γ), and tumor necrosis factor-α (TNF-α) were purchased from Pierce Biotechnology (Rockford, IL, USA).
Extract Preparation
Authenticated C orchioides (Specimen No. 62653) obtained from Amala Ayurveda Pharmacy was shade-dried and powdered. 100 g whole plant powder was extracted with 70% methanol in a Soxhlet apparatus for 24 hours. The solvent was evaporated to dryness using a rotary evaporator at 42°C under reduced pressure. The yield of the extract was 10.2% (w/w). Preliminary phytochemical analysis showed the presence of phenolic glycosides. The extract was redissolved in a minimum amount of dimethyl sulfoxide (DMSO) for in vitro studies. For animal experiments the extract was dissolved in minimum quantity of methanol, and then resuspended in 1% gum acacia. A detailed toxicity analysis was conducted (data not shown) and a nontoxic concentration of 20 mg/kg body weight was given intraperitoneally. The dose of the plant extract was selected from the preliminary screening as the lowest concentration with good physiological activities. 22
Effect of Curculigo orchioides on CPA-Induced Nephrotoxicity
Swiss Albino mice (24 animals/group) were divided into four groups and group I was kept as normal, treated with normal saline intraperitoneally for 5 days. The other three groups of animals were treated with a single acute dose of CPA (1.5 mmol/kg body weight). Group II animals were kept as CPA-only control. Group III animals were treated with C orchioides extract (20 mg/kg body weight) for 5 days prior to CPA administration. Group IV animals were treated with a single dose of mesna (150 mg/kg body weight). Eight animals from each group were anesthetized using haloethane and then sacrificed by cervical dislocation, at 4th, 24th, and 48th hour time points after CPA administration. Body weights of the animals were recorded prior to the drug administration and at the time of killing, the weights of bladder and kidney were also recorded. Urine was collected in a water-jacketed, chilled chamber (4°C) before killing the animals and centrifuged (2000 × g, 5 minutes). 23 Blood was collected by heart puncture, and serum separated. Serum and urine were used for estimating total protein contents (Biuret and Dumas method), 24 serum creatinine (alkaline picrate method), serum and urea nitrogen by diacetylmonoxime (DAM) reagents. 25
Morphological Examination
The urinary bladder and kidney were excised from each animal immediately after killing, washed thoroughly in phosphate buffer (pH 7.4) and the entire surrounding connective tissues were removed. Three different persons performed morphological analysis of urinary bladder and kidney by observing the inflammation, coloration, and size of the bladder.
Biochemical Analysis
Kidney and bladder homogenate was prepared using ice-cold phosphate buffered saline (PBS; 50 mmol/L, pH 7), after thorough washing with cold isotonic saline. This homogenate was used for the following biochemical investigations.
Lipid Peroxidation and GSH levels
Lipid peroxidation in kidney homogenate was estimated by the method of Ohkawa et al., 1979. 26 MDA-thiobarbituric acid (TBA) adduct formed during the reaction in acidic medium was extracted to the pyridine-butanol organic layer and measured in a spectrophotometer at 532 nm. Measurement of GSH in kidney and bladder homogenate was done according to the method of Moron et al. 27 GSH was measured by its reaction with DTNB (Ellman’s reaction) to give a compound that absorbs at 412 nm. A portion of tissue homogenate (0.5 mL) was precipitated by adding 25% TCA (trichloroacetic acid) and the tubes were cooled on ice for 5 minutes. The mixture was further diluted with 0.6 ml of 5% TCA, centrifuged at 1000 rpm for 10 minutes and supernatant (0.1 mL) was made up to 1 mL with 0.2 M sodium phosphate buffer (pH 8.0). Freshly prepared DTNB solution (2 mL) in 0.2 M sodium phosphate buffer was added to the tubes and a yellow color that formed after 10 minutes was measured using a spectrophotometer.
Antioxidant Profile
The kidney homogenate was centrifuged at 5000 × g for 10 minutes at 4°C; after removal of the cell debris, the supernatant was used for different assays. Superoxide dismutase (SOD) was performed by NBT reduction method of McCord and Fridovich, 28 and glutathione peroxidase (GPx) by the method of Hafemann et al. 29 based on the degra-dation of H2O2 in the presence of GSH, whereas catalase (CAT) activity was determined from the rate of decomposition of H2O2 at 240 nm following the addition of tissue homogenate. 29
Histopathological Analysis
Urinary bladder as well as kidney was fixed using 10% formaldehyde, and tissue dehydration was performed by several treatments in different concentrations of alcohol. The dehydrated tissue was embedded in paraffin wax. Sections were cut and stained with hematoxylin and eosin and histopathological analysis was performed.
Effect of Curculigo orchioides on IL-2, IFN-γ, and TNF-α Level After CPA Administration
Serum IL-2, IFN-γ, and TNF-α level was checked using a sandwich ELISA Kit specific for the murine cytokines according to the manufacturer’s protocol (Pierce Bio-technology). Serum from the above experiment was used this.
Statistical Analysis
Data were expressed as mean ± standard deviation. Significance levels for comparison of differences were determined using one-way analysis of variance (ANOVA) followed by Tukey-Kramer multiple comparison test using Graphpad Instat version 3.00 for Windows 98; P value <.05 was considered to be significant.
Results
Effect of Curculigo orchioides on Urinary Bladder and Kidney Morphology
A noticeable difference between the control and treated groups was observed on the morphological analysis of the urinary bladder. After 4 hours of CPA treatment, the bladder of CPA-only control animals was inflamed with evident red coloration due to hemorrhage, whereas the CPA + C orchioides–treated group showed only a slight inflammation, but normal coloration. Severe bladder hemorrhage and coloration was observed in the CPA-only control group at 24 hours after treatment whereas the C orchioides–treated group showed normal bladder morphology. Even after 48 hours the urinary bladders of the CPA only–treated group were severely inflamed whereas those of the CPA plus C orchioides–treated group were similar to those of normal animals (Table 1). No considerable changes were observed in kidney morphology.
Morphological Examination of Urinary Bladder After Cyclophosphamide (CPA) Administration. a

All the animals were treated with CPA single dose (1.5 mmol/kg body weight). One group was treated with 5 doses of C orchioides (20 mg/kg body weight) and other group with mesna (150 mg/kg body weight) along with CPA.
Effect of Curculigo orchioides on Body Weight Loss After CPA Administration
Body weights of CPA only–administered animals were found to be decreasing due to its toxic effects at all the 3 time points and maximum decrease was observed at 48 hours (−2.00 g), whereas in the CPA + C orchioides–treated group, there was a gradual increase in body weight at the 3 time points compared with control animals (Table 2).
Effect of Curculigo orchioides Treatment on Change in Body Weight (in Grams). a

All data are represented as mean ± SD. Data were statistically analyzed using one-way analysis of variance followed by Tukey-Kramer multiple comparison test.
P < .05 significantly different from normal.
P < .05 significantly different from that of CPA alone.
Effect of Curculigo orchioides on Relative Organ Weight
At all time points, relative bladder weight was found to be increased in the case of the CPA only–treated group, indicating inflammation of the bladder by CPA; the maximum weight was observed at 4 hours (0.26 ± 0.08 g/100 g body weight). The values are very high when compared with normal value of 0.081 ± 0.016 g/100 g body weight. Administration of mesna along with CPA maintained the relative bladder weight almost similar to that of normal controls (0.078 ± 0.009 g/100 g body weight) at 4 hours. In CPA + C orchioides–treated animals, the relative weight of bladder was decreased at 4 hours (0.13 ± 0.013 g/100 g body weight), 24 hours (0.098 ± 0.006 g/100 g body weight), and 48 hours (0.083 ± 0.002 g/100 g body weight) (Table 3), showing its uroprotective role. The relative weight of the kidney did not show much variation among groups (data not shown).
Effect of Curculigo orchioides Treatment on Relative Bladder Weight (g/100 g Body Weight). a

All data are represented as mean ± SD. Data were statistically analyzed using one-way analysis of variance followed by Tukey-Kramer multiple comparison test.
P < .01 significantly different from normal.
P < .01 significantly different from that of CPA alone.
Effect of Curculigo orchioides on Serum Creatinine Level
Increased serum creatinine level was shown by the CPA only–treated group at all the 3 time points, 4 hours (1.16 ± 0.062 mg/dL), 24 hours (0.78 ± 0.031 mg/dL), and 48 hours (0.52 ± 0.02 mg/dL). A significant decrease in the creatinine levels resulted from the administration of the plant extract at 4 hours (0.45 ± 0.04 mg/dL), 24 hours (0.42 ± 0.02 mg/dL), and reached a near normal level (0.37 ± 0.014 mg/dL) at 48 hours (0.38 ± 0.03 mg/dL), revealing the nephroprotective role of C orchioides (Figure 1) on CPA-induced damage.

Effect of Curculigo orchioides on serum creatinine level: All the animals except those from the normal group were treated with cyclophosphamide (CPA; 1.5 mmol/kg body weight) and the animals were sacrificed at different time points, blood was collected and serum was separated to estimate the creatinine level. The values are expressed as means ± SD. Data were statistically analyzed using one-way analysis of variance followed by Tukey-Kramer multiple comparison test.
Effect of Curculigo orchioides on Blood Urea Nitrogen and Urine Nitrogen
Figures 2A and 2B represent urea nitrogen levels in blood and urine, respectively. CPA only–treated animals showed a significant elevation in the levels of urea and creatinine, which are the kidney function markers, when compared with that of normal animals, indicating renal damage. This increased level was found to be decreased significantly by C orchioides extract treatment. Increase in blood urea nitrogen (BUN) level (126.8 ± 8.65 mg/dL) (4 hours) has been observed in the CPA only–treated group compared with the normal control (32.2 ± 4.0 mg/dL), Analysis of the urine of the CPA + C orchioides–treated groups of animals after 4, 24, and 48 hours showed that C orchioides pretreatment attenuated the level to 62.6 ± 4.3, 46.6 ± 5.6, and 36.2 ± 2.5 mg/dL at 4, 24, and 48 hours, respectively. The mesna-treated group retained a normal level of BUN at these time points. Urea nitrogen in urine was also enhanced drastically by CPA administration (Figure 3). Urine urea nitrogen levels were also enhanced significantly by CPA administration to 27.33 ± 0.69 mg/dL at 4 hours, which was slightly decreased to 24.98 ± 1.58 at 48 hours, whereas on treatment with mesna, the levels (16.78 ± 1.68 at 48 hours) were brought toward normal (16.52 ± 1.68). C orchioides treatment also reduced the urea nitrogen levels significantly to 17.53 ± 1.32 at 48 hours, showing its renal protective role.

Effect of Curculigo orchioides on blood urea nitrogen (BUN) and urine urea nitrogen levels: All the animals except those from the normal group were treated with cyclophosphamide (CPA; 1.5 mmol/kg body weight). (A) Animals were sacrificed at different time points to collect the blood and BUN was estimated by diacetylmonoxime method. (B) Urine was collected in cooled chamber before killing the animals and used to estimate urine urea nitrogen levels. All the values are mean ± SD, data were statistically analyzed using one-way analysis of variance followed by Tukey-Kramer multiple comparison test.

Effect of Curculigo orchioides on kidney lipid peroxidation: Cyclophosphamide (CPA; 1.5 mmol/kg body weight) was administered to the animals and they were sacrificed to collect the kidney to prepare homogenate and kidney lipid peroxidation (LPO) was measured by the method of Ohkawa et al. 26 All the values are mean ± SD, data were statistically analyzed using one-way analysis of variance followed by Tukey-Kramer multiple comparison test.
Effect of Curculigo orchioides on Bladder and Kidney GSH Levels
The effect of C orchioides on bladder GSH levels after CPA administration is represented in Table 4. CPA treatment reduced the bladder GSH level to 1.12 ± 0.08 nmol/mg protein at 4 hours from the normal level of 3.01 ± 0.46 nmol/mg protein, which was slightly increased to 1.75 ± 0.05 at 48 hours. But there was an increase in bladder GSH level in the CPA + mesna–treated group at 4 hours (3.28 ± 0.51 nmol/mg protein) and 3.66 ± 0.71 nmol/mg protein at 48 hours. Pretreatment with C orchioides extract to the CPA-administered animals could also increase bladder GSH level, which was at 4 hours (2.13 ± 0.031 nmol/mg protein), 24 hours (2.50 ± 0.36 nmol/mg protein), and the level was normalized by 48 hours (3.15 ± 0.40 nmol/mg protein). Kidney GSH levels were also reduced drastically with CPA treatment (1.18 ± 0.09 nmol/mg protein, at 4 hours) when compared with normal level (4.21 ± 0.67 nmol/mg protein). A significant reestablishment in GSH level was observed in CPA + C orchioides–treated animals (2.89 ± 0.33 nmol/mg protein) when compared with CPA only–treated group. Even after 48 hours, only an insignificant increase was shown by the CPA-only experimental animals (1.92 ± 0.25 nmol/mg protein) whereas the CPA + C orchioides group reached close to the normal GSH levels by 48 hours (3.97 ± 0.42 nmol/mg protein), showing the renal protective role of C orchioides (Table 5).
Effect of Curculigo orchioides Treatment on Bladder Glutathione (GSH) Levels (nmol/mg Protein). a
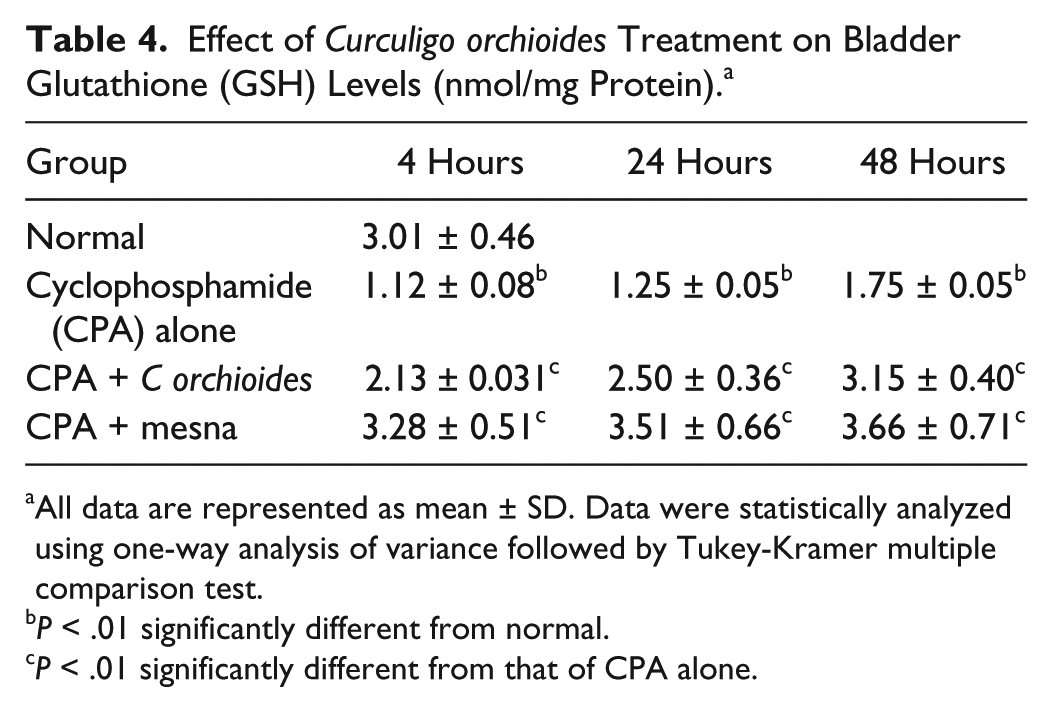
All data are represented as mean ± SD. Data were statistically analyzed using one-way analysis of variance followed by Tukey-Kramer multiple comparison test.
P < .01 significantly different from normal.
P < .01 significantly different from that of CPA alone.
Effect of Curculigo orchioides Treatment on Kidney Glutathione (GSH) Levels (nmol/mg Protein). a

All data are represented as mean ± SD. Data were statistically analyzed using one-way analysis of variance followed by Tukey-Kramermultiple comparison test. NS, nonsignificantly different from that of CPA alone.
P < .01 significantly different from normal.
P < .01 significantly different from that of CPA alone.
P < .001 significantly different from that of CPA alone.
Effect of Curculigo orchioides on Lipid Peroxidation
Lipid peroxidation in CPA-treated animals was significantly (P < .01) increased compared with normal animals at 4 hours (2.60 ± 0.24 mol/mg protein), 24 hours (2.1 ± 0.17 mol/mg protein), and 48 hours (2.0 ± 0.18 mol/mg protein) (Figure 4), whereas treatment with C orchioides along with CPA significantly decreased lipid peroxidation to 1.62 ± 0.08, 1.38 ± 0.15, and 1.3 ± 0.11 mol/mg protein at 4, 24, and 48 hours, respectively, indicating the cellular-level protection of C orchioides from the toxic effect of CPA (Figure3).

Effect of Curculigo orchioides on kidney superoxide dismutase (SOD) and catalase: All the animals except those from the normal group were treated with cyclophosphamide (CPA; 1.5 mmol/kg body weight). Animals were sacrificed at different time points to excise out the kidney to check kidney SOD (A) and kidney catalase levels (B). All the values are mean ± SD, data were statistically analyzed using one-way analysis of variance followed by Tukey-Kramer multiple comparison test.
Effect of Curculigo orchioides on Antioxidant Status
The activities of renal SOD, CAT, and GPx at the 3 time points are represented in Figure 4 and 5. The renal antioxidant status, comprising SOD, CAT, and GPx activities, is significantly (P < .01) decreased in the CPA only–treated group of animals compared with the normal ones. Administration of C orchioides was found to significantly elevate the lowered activities of these antioxidant enzymes thereby scavenging the free radicals produced as a result of CPA insult.

Effect of Curculigo orchioides on kidney glutathione peroxidase (GPx) levels: Cyclophosphamide (CPA; 1.5 mmol/kg body weight) was administered to the animals and they were sacrificed to collect the kidney and GPx levels were measured by method of Hafemann et al. 29 All the values are mean ± SD, data were statistically analyzed using one-way analysis of variance followed by Tukey-Kramer multiple comparison test.
Effect of Curculigo orchioides on Serum Cytokine Levels
Table 6 illustrates serum cytokines levels, including IFN-γ, IL-2, and TNF-α, of CPA-treated as well as CPA + C orchioides-treated animals. When compared to the normal animals, the serum IFN-γ levels of CPA only–treated animals were decreased at all 3 time points and maximum decrease was obtained at 48 hours. But in CPA + C orchioides as well as CPA + mesna groups, an increase in IFN-γ was observed as time passed, which was normalized by 48 hours. A significant reduction was also observed in the serum IL-2 levels of CPA-treated control animals after 4 and 48 hours in comparison with normal levels. C orchioides treatment was found to elevate the reduced level of IL-2 by CPA treatment to normal level by 48 hours, as also observed in the mesna-treated group. A significant increase in both IFN- γ and IL-2 level in the C orchioides only–treated group was also noticed during the study, showing its modulating activity on the immune system. At the same time, there was a significant increase in the level of serum TNF-α in CPA-only control animals compared to the normal controls. The protective effect of C. orchioides was evidenced by the decrease in TNF-α level in the C orchioides and mesna-treated groups.
Effect of Curculigo orchioides Treatment on Serum Cytokine Levels (pg/mL). a

Abbreviations: CPA, cyclophosphamide; IFN-γ, interferon-γ; IL-2, inerleukin-2; TNF-α, tumor necrosis factor-α.
All data are represented as mean ± SD. Data were statistically analyzed using one-way analysis of variance followed by Tukey-Kramer multiple comparison test. NS, nonsignificantly different from that of CPA alone.
P < .01 significantly different from normal.
P < .01 significantly different from that of CPA alone.
Histopathological Analysis
Figure 6 shows the histopathological analysis of the urinary bladder. The bladder morphology of CPA only–administered animals showed denudation of bladder mucosa with inflamed bladder walls. Hemorrhage of mucosal and submucosal region was also observed with diffuse infiltrate of lymphocytes, plasma cells, and polymorphs. At 48 hours after CPA administration the bladder wall showed edema as well as hemorrhage and extensive necrosis of the mucosal layer. Whereas in the case of CPA + C orchioides–treated animals, the urinary bladder wall and epithelium showed minimal damage and mild edema in the submucosal region even after 48 hours and the morphology was almost similar to the normal urinary bladder.

Histopathological analysis of the urinary bladder: (A) Normal urinary bladder showing normal bladder wall (normal fibrous tissue: a; normal muscularis: b; normal urothelium: c; lumen to collect urine: d. (B) Bladder excised from cyclophosphamide (CPA) alone administered animals after 48 hours of treatment. (Hemorrhage to fibrous tissue: a; damaged muscle layer: b; denudation of bladder mucosa: c; lumen to collect urine: d). (C) Urinary bladder of CPA + mesna–treated animals. (D) Urinary bladder of CPA + Curculigo orchioides–treated animals taken out after 48 hours (200×).
Morphological analysis of kidney (Figure 7) revealed the damage of renal tubules with irregular Bowmans capsule in CPA only–administered animals. Renal tubules showed vacuolation of lining epithelial cells and the interstitial tissue exhibited hemorrhage, foci of necrosis, and diffuse infiltration by lymphocytes and plasma cells. But in the case of CPA + C orchioides–administered animals, the renal tissue appears with normal glomeruli and interstitial tissue with mild hemorrhage and a few inflammatory cells. Thus the histopathological examination of kidney and urinary bladder accentuate the protective effects of the plant extract over the damage caused by CPA administration.

Histopathological analysis of kidney: (A) Normal kidney (normal glomorulus: bent arrow; normal renal tubule: arrow]). (B) Kidney excised from cyclophosphamide (CPA) alone administered animals after 48 hours of treatment (irregular Bowmans capsule: bent arrow; renal tubule damage and foci of necrosis: arrow). (C) Kidney of CPA + Curculigo orchioides–treated animals (mild hemorrhage to renal tissues: arrow; normal Bowmans capsule: bent arrow). (D) Kidney of CPA + mesna–treated animals (normal Bowmans capsule: bent arrow and damaged renal tubule: arrow) taken out after 48 hours (400×).
Discussion
Cyclophosphamide is a major antineoplastic agent and immune suppressor with a main drawback of the side effects caused by its toxic metabolites. A large portion of these metabolites are excreted through the urine and during the excretion process they react with the urothelium of the bladder and reduce its urine retention capacity. The deleterious effects of CPA on the bladder include mucosal oedema, haemorrhage, ulceration, subendothelial telangiectasia and, in severe cases, fibrosis of the bladder.31-33 A strategy to diminish the side effects of anticancer drugs with the preservation of its chemotherapeutic efficacy is required. C orchioides extract was found to be effective in ameliorating CPA-induced side effects.
Administration of CPA and many other anticancer drugs leads to depression of the hepatic mixed function oxidase system due to bioreactive molecule formation during the hepatic activation process. 34 Acrolein, a by-product of CPA activation is responsible for its dose-limiting urotoxic and nephrotoxic side effects. Acrolein reacts with the free and tissue bound thiols 35 and leads to depression in the hepatic glutathione and microsomal enzyme activities, thus resulting in kidney and bladder toxicities. Our previous studies revealed that C orchioides is effective in ameliorating the hepatic and intestinal toxicities induced by the administration of CPA without interfering its chemotherapeutic efficacy. The results obtained from the present study point out a possibility of blocking acrolein-related nephro- and urotoxicities mediated by the antioxidant and anti-inflammatory properties of C orchioides.
Creatinine and GSH levels along with the urine volume are the specific markers to identify renal malfunction. Oxidative stress generated by the free radicals formed due to the administration of CPA and further by its toxic metabolic by-product, acrolein, are together responsible for the pathological mechanism of these changes in renal parameters. 36 During our studies, decreased urine volume and significantly elevated levels of serum creatinine were observed in CPA-administered animals, which is associated with renal dysfunction. Blood urea nitrogen (BUN) level was also found to be elevated in the case of CPA- administered animals. Elevated BUN levels and creatinine concentrations are markers for kidney damage and nephrotoxicity. 37 C orchioides whole plant extract, when administered along with CPA, significantly de-creased the serum creatinine and BUN levels to near normal level.
Increased release of pro-inflammatory cytokines forms a major reason for the inflamed kidney and urinary bladder. C orchioides extract, by its anti-inflammatory activity and downregulatory effects on pro-inflammatory cytokine levels, was found to effectively reduce the CPA-induced inflammatory responses in test animals. Histopathological studies of kidney also indicated glomerular necrosis and hemorrhage to renal tubules whereas the urinary bladder showed edema with hemorrhagic cystitis and necrosis. Back flow of urine from bladder to the renal tubules due to the decreased storage space for urine within the inflamed bladder may be the reason for these impairments. Treatment with the extract minimized the structural damage occurring to the bladder and kidney by CPA administration along with a reduction in inflammation, and the protective effects may be due to the downregulation of pro-inflammatory cytokines by C ochioides extract.
Glutathione peroxidase is the enzyme participating in the elimination of reactive intermediates by reducing hydroperoxides and thus is involved in the repair of radical-caused biological damage. C orchioides administration was found to enhance the GPx levels in kidney, which were significantly reduced by CPA administration. The present study discloses the efficacy of C orchioides extract in scavenging superoxide radicals generated inside the body during normal metabolism and most importantly in the presence of xenobiotics. Moreover administration of C. orchioides significantly increased the catalase and glutathione levels in kidney and urinary bladder. Lipid peroxidation refers to the reaction of oxidative deterioration of polyunsaturated lipids. The extract could inhibit the process of lipid peroxidation, and thus reduced the free radical toxicity, which strongly supports its chemoprotective activity.
Conclusion
Morphological, histopathological, biochemical, and enzyme analysis showed that C orchioides could alleviate the severe urotoxicity and nephrotoxicity induced by cyclophosphamide. The present study reveals the protective role of C orchioides extract against cyclophosphamide induced multiple organ toxicity. At the same time, the extract does not hinder the antitumor efficacy of cyclophosphamide, which was revealed from our previous studies. 22 Thus C orchioides, which is a nontoxic, immunostimulating and antioxidant plant, can be used as a natural adjuvant with cyclophosphamide to inhibit tumor development.
Footnotes
Acknowledgements
The authors are thankful to Dr Ramadasan Kuttan, Director of Amala Cancer Research Centre, for his valuable suggestions and support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by DST INSPIRE Fellowship, Government of India, New Delhi, India.
