Abstract
Streptococcus pneumoniae (Spn) is a common respiratory pathogen and a frequent cause of acute otitis media (AOM) in children. The first step in bacterial pathogenesis of AOM is the establishment of asymptomatic colonization in the nasopharynx. We studied Spn bacterial burden in conjunction with neutrophil recruitment and inflammatory gene transcription and cytokine secretion in samples of nasal wash collected from normal and otitis-prone children during health, viral upper respiratory infection without middle ear involvement (URI) and AOM. We found no significant associations between otitis-prone status and any of the measured parameters. However, Spn bacterial burden was significantly correlated with neutrophil recruitment, transcription of IL-8, TNF-α and SOD2, and secretion of TNF-α. We also found that transcription of IL-8 and TNF-α mRNA by neutrophils was significantly correlated with the secretion of these cytokines into the nasopharynx. We conclude that Spn bacterial burden in the NP is a major determinant of neutrophil recruitment to the NP and activity during URI and AOM, and that neutrophils are contributors to the secretion of IL-8 and TNF-α in the NP when the Spn burden is high.
Keywords
Introduction
In the first few years of life, infants and young children experience very frequent upper respiratory infections (URIs). Among these, acute otitis media (AOM) is one of the most prominent, responsible for widespread antibiotic prescription and pediatric surgery with a total economic burden estimated at US$3–5 billion annually in the USA.1–3 Approximately one-third of cases of AOM is caused by Streptococcus pneumoniae (Spn), while Haemophilus influenzae, and/or Moraxella catarrhalis are responsible for most of the remainder. 4
Spn and other otopathogens are frequently encountered as asymptomatic colonizers of the human nasopharynx (NP).5,6 These transient episodes of colonization are cleared in a matter of days to weeks by a mild inflammatory response spearheaded by neutrophils and supported by Th17 immunity.7,8 Asymptomatic colonization is generally an immunizing event, contributing to the development of Spn-specific adaptive immunity. 9 However, viral co-infection during a colonization episode induces remodeling of the NP, permitting Spn to reach a pathogenic inoculum and invade the middle ear through the Eustachian tubes. Virtually all episodes of AOM are precipitated by viral co-infection during bacterial colonization, most commonly respiratory syncytial virus and influenza.10,11 Approximately 10% of children are otitis prone, suffering from frequent, recurrent episodes of OM associated with both environmental and immunologic risk factors.12–14
The progression of inflammation in the NP during pathogenesis of AOM is an area of great interest. Infection by respiratory viruses has been shown to increase nasopharyngeal colonization by potential otopathogens in human children. 15 Murine studies found an important role for signaling through TLR2 and TLR4, where mice lacking these receptors experienced impaired bacterial clearance from the NP and delayed recovery associated with reduced neutrophil and macrophage recruitment. 16 A transcriptomic analysis of murine otitis media identified four distinct programs of inflammatory gene transcription activated at different time points during the 3 days following infection. 3 In a separate study, neutrophil recruitment and myeloperoxidase activity were found not only to promote Spn killing in the middle ear, but also increased inflammatory tissue damage, 17 indicating that inflammation must be carefully controlled during the course of an AOM episode. Although the nasopharyngeal bacterial burden of Spn and M. catarrhalis has been observed to correlate with AOM disease severity, 18 limited information is available on the dynamics of inflammation in the NP during AOM in humans.
The primary question of interest was the nature of the inflammatory host–pathogen relationship during Spn colonization and pathogenesis in the NP. To study this, Spn bacterial burden and host inflammatory factors were quantified during health and viral URI with or without concurrent asymptomatic Spn infection, and when AOM caused by Spn occurred concurrently with a viral URI. Direct measurements of inflammation in the human NP have been hampered by the lack of an effective means of quantifying neutrophil recruitment. We recently described a novel method for measuring neutrophil content in nasal secretions by PCR. 19 By applying this method to pediatric NP samples collected during health, viral URI without progression to AOM and viral URI with progression to AOM during colonization by Spn, we were able to measure and compare the influence of NP bacterial burden and physiological state on signals of local NP inflammation, including neutrophil recruitment and transcription and secretion of various innate immune cytokines.
Materials and methods
Sample collection
Subject samples analyzed in this study (n = 98).
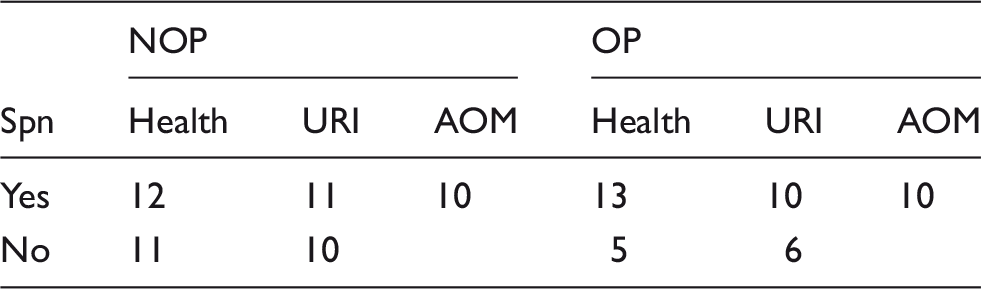
NOP: non-otitis prone; OP: otitis prone
RNA isolation and RT-qPCR
Sequences of primers used for RT-PCR.

Cytokine detection
Cytokines and total protein concentrations were measured in the supernatant of nasal wash samples after centrifugation. Total protein was measured with a BCA Protein Assay kit (Pierce, Rockford, IL, USA). Concentrations of IL-6, IL-8, MIP-1α, RANTES and TNF-α were measured using a multiplex ELISA kit (EMD Millipore, Billerica, MA, USA) according to the manufacturer's instructions. Reads were taken on a Bio-Plex 200 machine (BioRad, Hercules, CA, USA).
Statistics
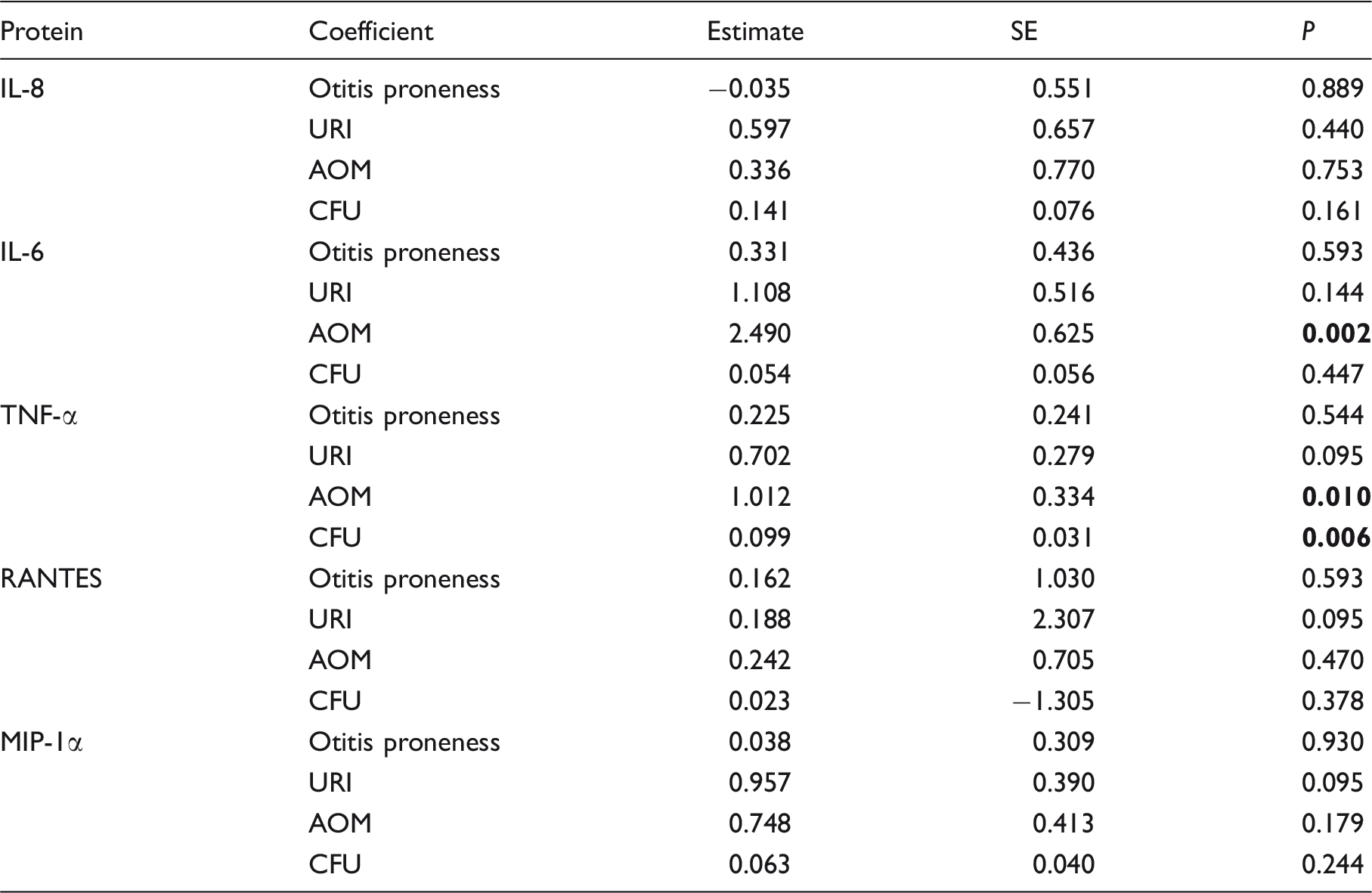
All statistics were performed using R version 3.1.1. 21 Linear models were constructed to describe dependent variables (neutrophil count, mRNA and cytokines) from all subjects as functions of independent variables (physiological state, Spn CFU bacterial burden and otitis proneness): total protein concentration was included as a covariate for protein analytes. Independent variables with P-values < 0.05 after Benjamini–Hochberg adjustment for multiple comparisons (false discovery rate = 5%) were judged to significantly affect the dependent variables. Measurements of bacterial burden were transformed by the inverse hyperbolic sine function (asinh) to include 0 values. Gene transcription and protein measurements were transformed by Box-Cox using the implementation in the MASS package of R. 22 Asymptomatic colonization rates in the whole population were compared by Pearson's χ2 test. Figures were prepared using the ggplot2 package in R. 23 All subjects were analyzed for every analyte. Tables of effect sizes from the linear models are shown in the results.
Results
Neutrophil recruitment and bacterial burden in the NP during health, URI and AOM
To study the effects of disease state (health, URI or AOM) on bacterial burden and inflammation, we measured Spn bacterial burden and neutrophil count in nasal wash samples. Spn was quantified using qPCR of the lytA gene,
20
and neutrophil counts were obtained by RT-qPCR of CD16, CD18 and CD62L.
19
All AOM samples were necessarily pathogen positive, but we further subdivided healthy and URI visits on the basis of Spn culture positivity in the NP. Spn-negative samples selected for study grew no otopathogenic bacteria (including Spn, H. influenzae and M. catarrhalis) when cultured and did not amplify lytA during qPCR. We observed no significant differences in Spn burden or neutrophil count between non-otitis-prone and otitis-prone samples (data not shown), so samples were combined for further analysis. In Spn positive samples, URI and AOM illness visits were associated with a significant increase in Spn burden in the NP relative to healthy visits. Spn burden did not significantly differ between URI and AOM samples (Figure 1).
Spn bacterial burden is elevated during URI and AOM relative to health (*P < 0.05, **P < 0.01).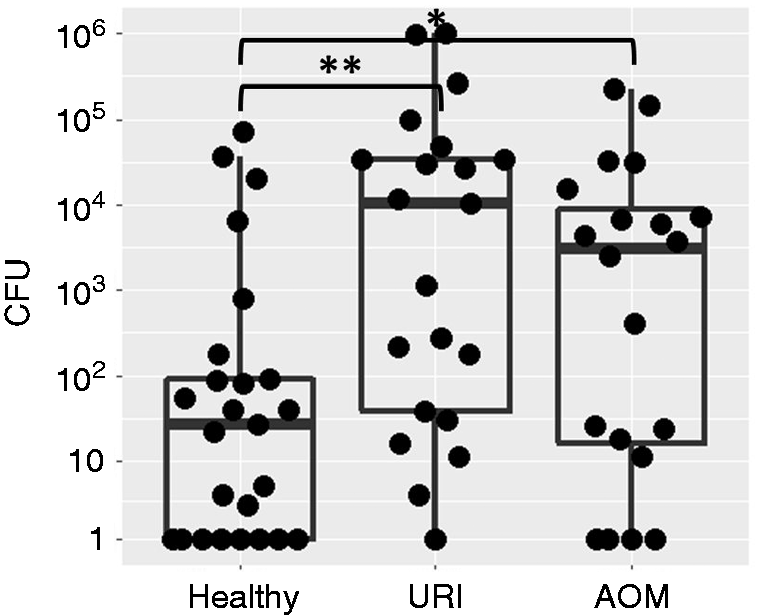
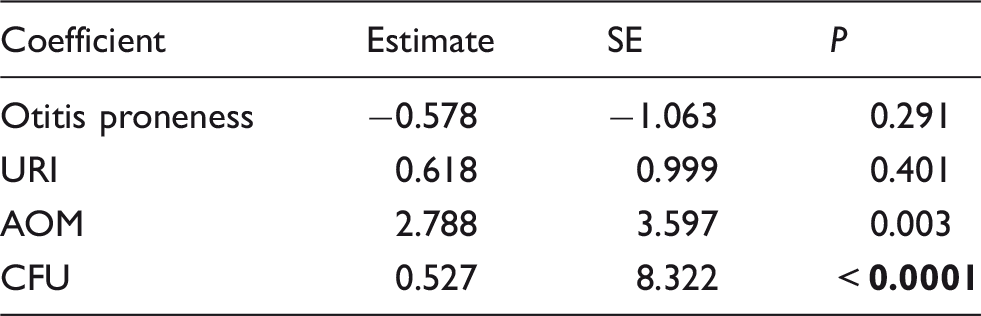
Correlations from a linear model expressing neutrophil count as a function of physiological group (health, URI or AOM) and Spn burden are shown in Figure 2. After correcting for the influence of bacterial burden, we found neutrophils to be significantly more abundant during AOM compared with health, but URI visits did not show an increase in neutrophil count independent of bacterial burden (Figure 2a) Neutrophil count was significantly correlated with NP Spn burden during health, URI and AOM (Figure 2b). Linear model analysis results are reported in Table 3. Significant positive coefficients are associated with increased neutrophil count. Since neutrophil count was log10 transformed for analysis, the coefficient for AOM signifies that neutrophil count is increased by 102.788 or roughly 600-fold during AOM. As described in the methods, CFU values were transformed by inverse hyperbolic sin (asinh). Thus, a unit increase in asinh(CFU) signifies an increase of 1.17 in bacterial colonies, and the attached coefficient means that every such increase in Spn burden increases neutrophil count by 100.527 or 3.4-fold.
Neutrophil count is elevated during AOM and correlates with Spn burden in the NP (effects summarized in Table 3). (a) Comparisons of raw neutrophil count between healthy, URI and AOM. (b) Comparisons of neutrophil count between health, URI and AOM after correcting for Spn burden. (c) Trend line representing correlation between neutrophil count and Spn burden after correcting for physiological state of healthy, URI and AOM (*P < 0.05, **P < 0.01, ***P < 0.001). Effect size estimates from linear model analysis of log10-transformed neutrophil count. Bold indicates significant result.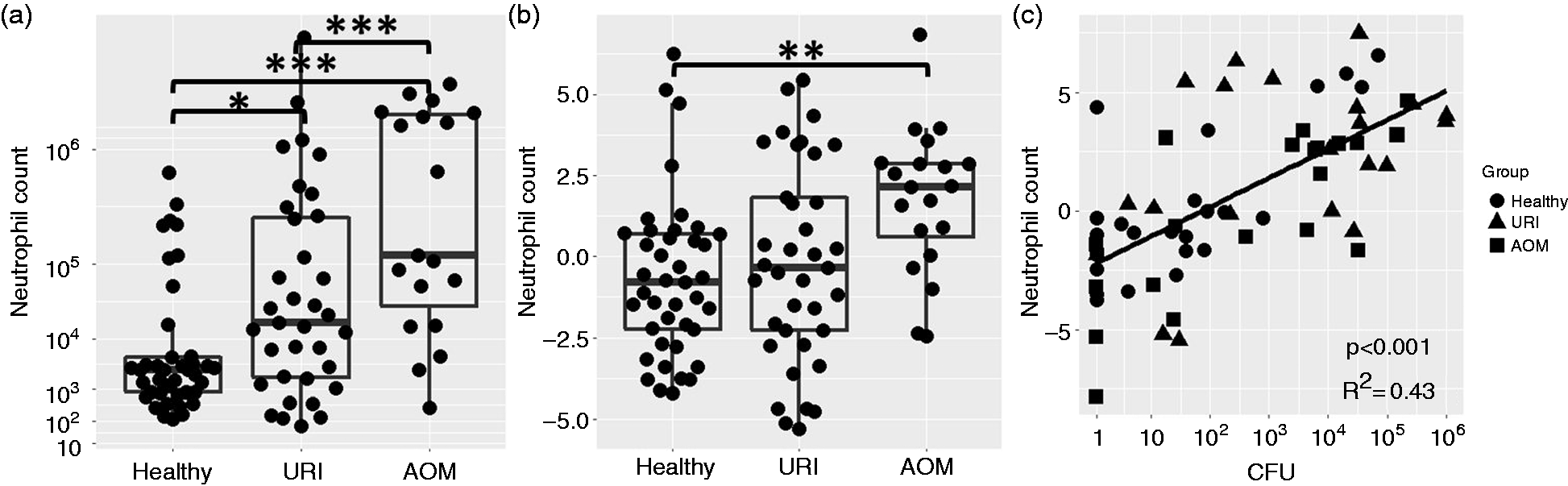
Transcription of inflammatory genes during health, URI and AOM
We next measured the transcription of a panel of inflammatory genes in nasal wash samples. We chose to study IL-8, a well-established neutrophil chemoattractant;24,25 arginase-1 (Arg-1), a marker of alternative inflammation involved in tissue repair and inflammatory resolution;26,27 superoxide dismutase 2 (SOD2), a critical component of the neutrophil respiratory burst pathway;
28
and the canonical pro-inflammatory cytokines IL-6 and TNF-α. Transcription was first analyzed by a linear model including Spn bacterial burden, physiological state (health, URI, and AOM) and otitis-prone status. Since otitis proneness was not significantly associated with variations in any of the tested genes, it was dropped from the model. Results of comparisons among raw measurements are shown in Figure 3: IL-8, Arg-1 and TNF-α mRNA were significantly more abundant during AOM, and IL-8, TNF-α and SOD2 were higher during URI.
Inflammatory gene transcription (IL-8, Arg-1, TNF-α, IL-6 and SOD2) in the NP during health, URI and AOM with or without Spn NP colonization (raw data; *P < 0.05, **P < 0.01).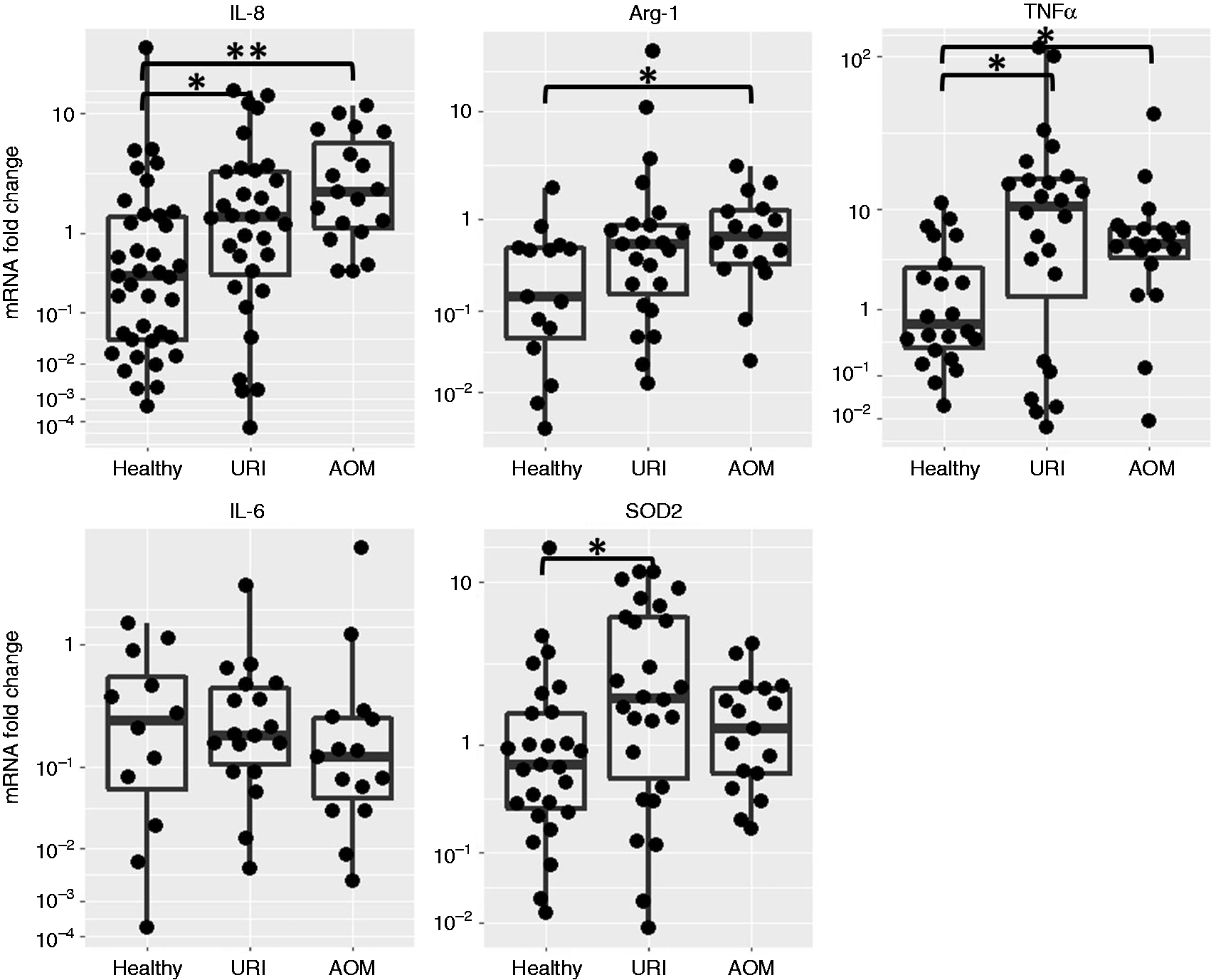
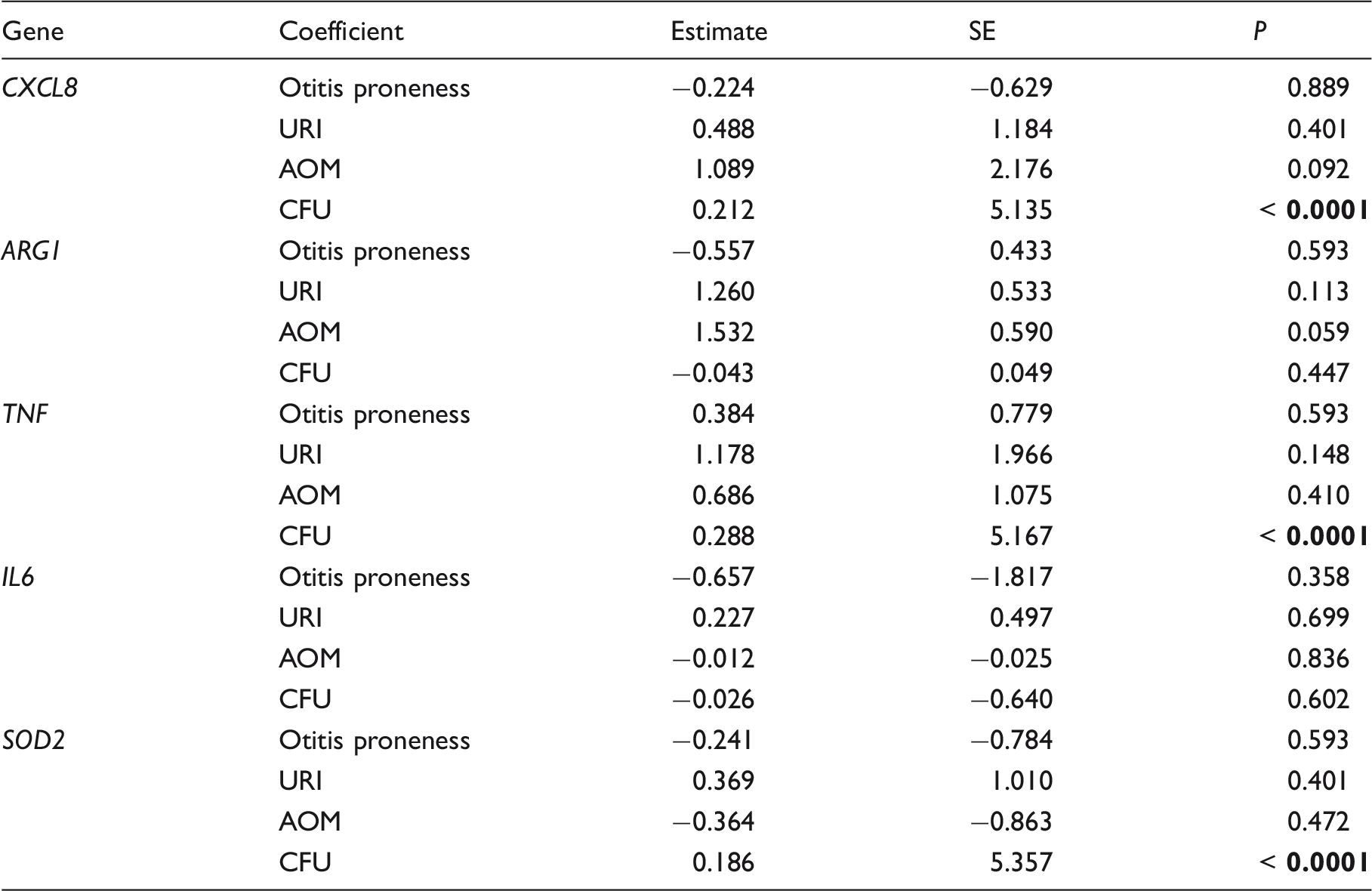
We applied the same approach described above to depict correlations of each analyte with Spn burden and physiological state. After correcting for Spn burden, we found a trend (P < 0.1) for elevated transcription of IL-8 and Arg-1 during AOM relative to health; no other significant differences emerged (Figure 4a). NP Spn bacterial burden was significantly correlated with transcription of IL-8, TNF-α and SOD2 (Figure 4b). Linear model analysis results are reported in Table 4.
Effects of physiological state (healthy, URI and AOM) and Spn burden on inflammatory gene transcription (IL-8, Arg-1, TNF-α, IL-6 and SOD2) in the NP (effects summarized in Table 4). Plots show residuals from linear models expressing each gene as a function of Spn bacterial burden or physiological state. (a) Physiological state effects on mRNA transcription in the NP after correcting for the influence of bacterial burden. (b) Spn burden correlations with mRNA transcription in the NP after correcting for the influence of physiological state. Effect size estimates from linear model analyses of log2-transformed inflammatory gene transcription. Bold indicates significant result.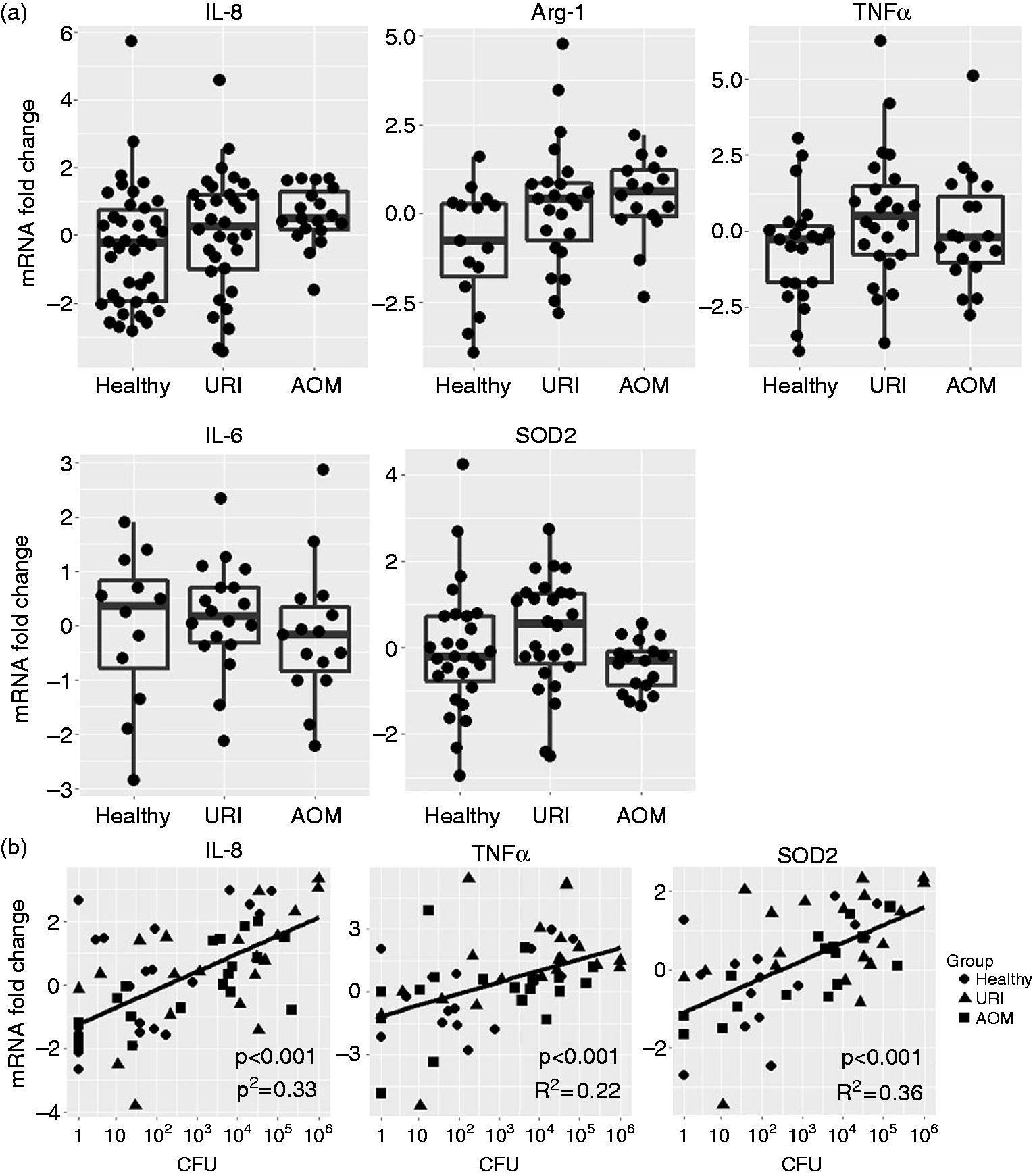
Secretion of inflammatory cytokines during health, URI and AOM
Measurements of RNA transcription in nasal wash samples were derived from the cellular pellets obtained after centrifugation, which is predominantly composed of neutrophils infiltrating the NP.
19
Inflammatory responses by the respiratory epithelium are necessary to begin the process of neutrophil recruitment and bacterial clearance.
7
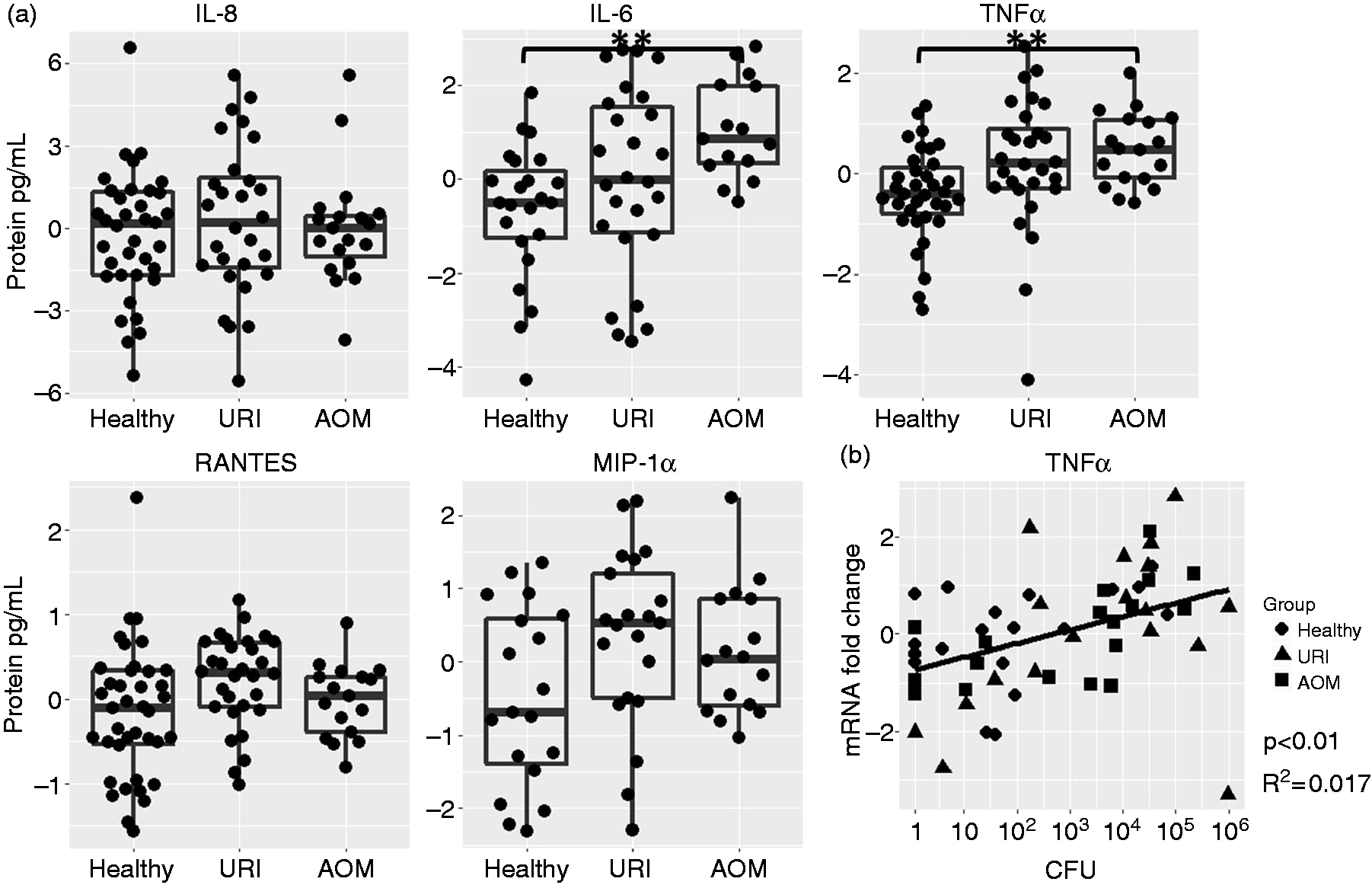
To understand these initial responses, it is necessary to measure the levels of inflammatory cytokines secreted into the NP lumen, in addition to the activation of recruited inflammatory cells. We measured the levels of inflammatory cytokines and chemokines: IL-6, IL-8, RANTES, TNF-α and MIP-1α in the supernatant of nasal wash samples. At the same time, we measured the total protein concentration in the supernatants by BCA, to correct for variations in the recovered volume of each sample. All protein analytes were significantly correlated with total protein concentration (data not shown). After correcting for this effect, we found no significant differences associated with otitis proneness. Linear models ignoring the influence of NP Spn burden found significant increases in IL-6, TNF-α and MIP-1α during URI and AOM relative to health (Figure 5). After including Spn burden, levels of IL-6 and TNF-α were significantly elevated during AOM compared with health, and we observed a trend (P < 0.1) for elevated secretion of RANTES, TNF-α and MIP-1α during URI compared with health (Figure 6a). Only TNF-α displayed a significant partial correlation with Spn burden (Figure 6b). Linear model analysis results are reported in Table 5.
Secretion of cytokines and chemokines in the NP during health, URI and AOM with or without Spn colonization (raw data; *P < 0.05, ***P < 0.001). Effects of physiological state and Spn burden on inflammatory cytokine and chemokine secretion in the NP. Plots show residuals from linear models expressing each protein as a function of Spn bacterial burden or physiological state (effect sizes reported in Table 5). (a) Influence of physiological state on protein secretion after correcting for Spn burden (**P < 0.01). (b) Influence of Spn burden on protein secretion after correcting for physiological state. Effect size estimates from linear model analyses of log2-transformed inflammatory cytokine and chemokine protein secretion. Bold indicates significant result.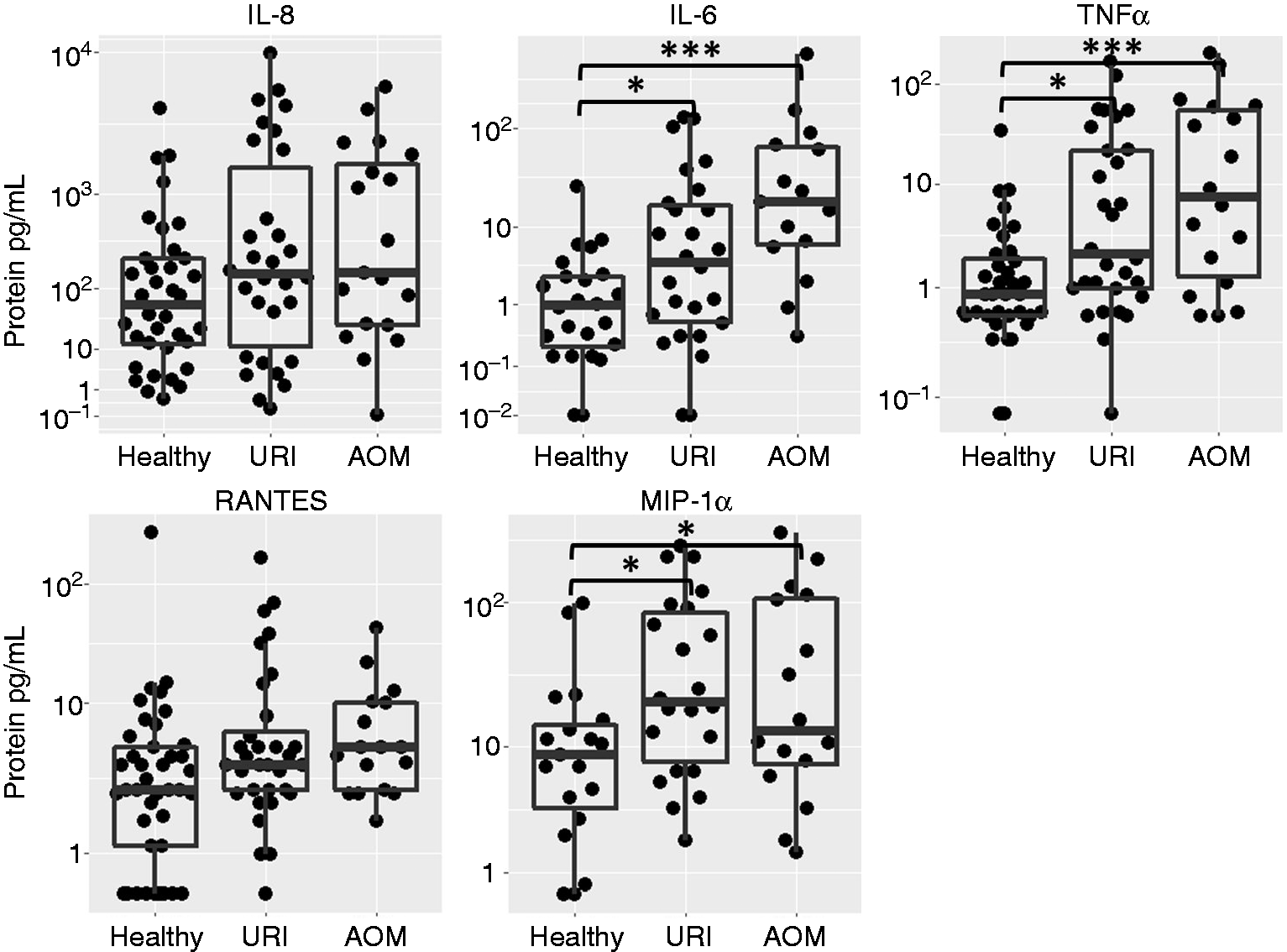
Neutrophil contribution to inflammatory signaling
Since neutrophils constitute the predominant cell type present in NP samples,
19
measurements of RNA transcription in the cell pellets can be attributed largely to neutrophils. In contrast, protein secretion in the supernatants of these samples could come from any cell present in the NP at the time of sample collection, especially epithelial cells. To determine whether neutrophils were meaningful contributors to the levels of inflammatory proteins, we assessed correlations between gene transcription and protein concentration of IL-6, IL-8 and TNF-α. In linear models expressing cytokine secretion as a function of Spn burden, total protein concentration and transcription level of the corresponding mRNA, we found significant correlations between cytokine secretion and mRNA transcription for IL-8 and TNF-α (Figure 7). This indicates a potential role for neutrophils in the secretion of inflammatory proteins during an antimicrobial response.
Relationships between mRNA and protein secretion for IL-6, IL-8 and TNF-α. Statistically significant correlations are denoted as trendlines (IL-6 correlation not significant).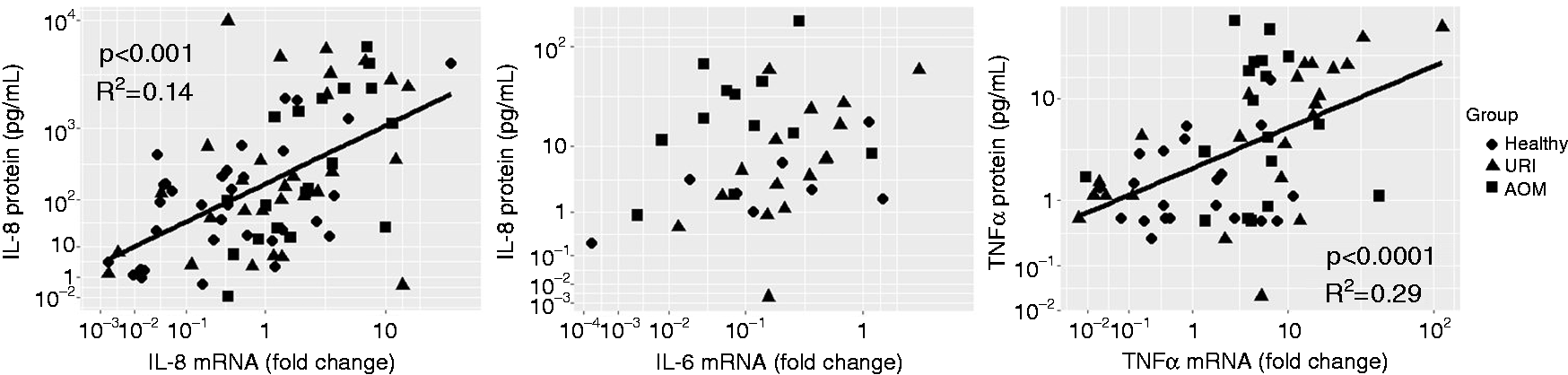
Asymptomatic colonization by otopathogens in non-otitis-prone and otitis-prone children
In our study population as a whole, asymptomatic colonization by one or more of the three main otopathogens (Spn, H. influenzae and M. catarrhalis) is frequently observed. Out of 3147 healthy visits (2800 non-otitis prone, 347 otitis prone), we observed significantly higher incidence of asymptomatic nasopharyngeal colonization by each of the three main otopathogens in otitis-prone children (Figure 8).
Incidence of asymptomatic colonization by the three main otopathogens in non-otitis-prone (NOP) and otitis prone (OP) children (bars depict mean ± 95% confidence interval; **P < 0.01, ***P < 0.001).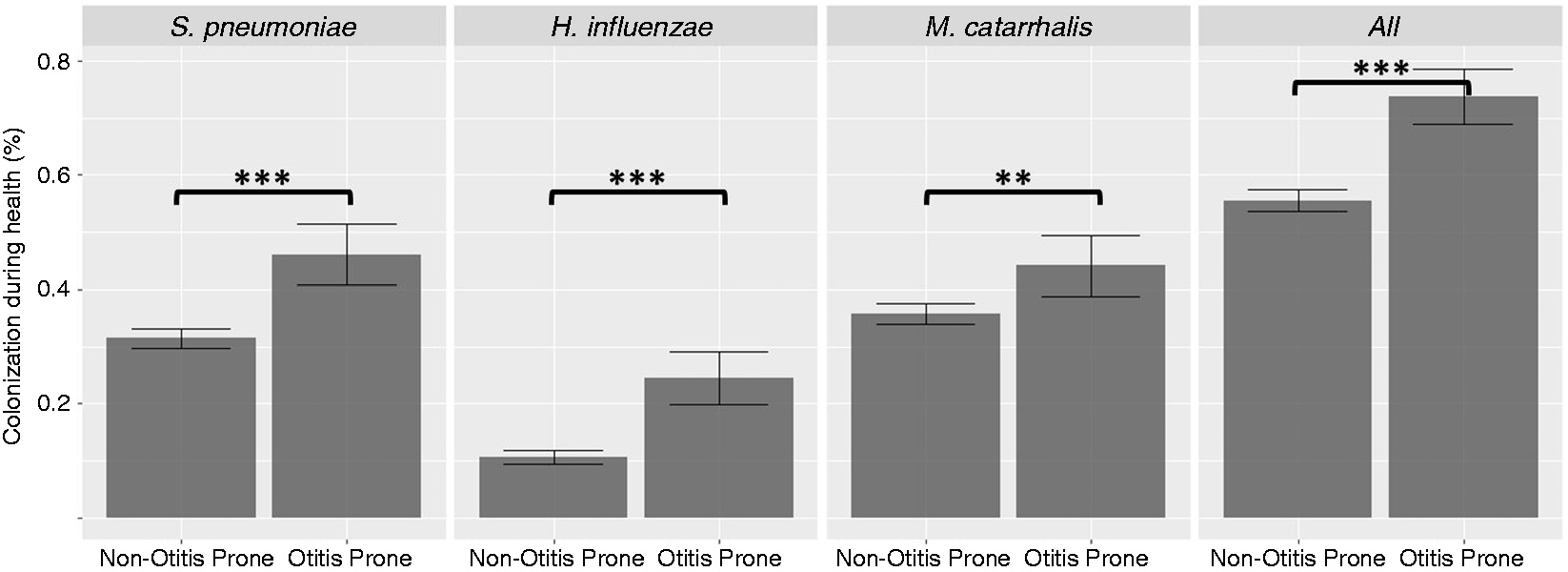
Discussion
We conducted studies of neutrophil recruitment and inflammation in children during health, viral URI and AOM in the presence or absence of NP colonization by Spn. We found no evidence of dysfunctional neutrophil recruitment or associated inflammatory responses by otitis-prone children, including inflammatory gene transcription, cytokine secretion and bactericidal ROS production by neutrophils. NP Spn bacterial burden appeared to be the predominating influence on the magnitude of neutrophil recruitment and inflammatory cytokine production, explaining much of the variation in inflammatory responses observed between health, URI and AOM.
Neutrophils are critical for the control and clearance of bacterial infections, especially Spn.17,29,30 However, their role during viral infection in the absence of bacteria is more complex. 31 Recent murine studies have implicated neutrophils in influenza pathology, 32 but neutrophil-mediated inflammation appears not to be involved in pathology caused by a pneumovirus related to respiratory syncytial virus (RSV; influenza and RSV commonly infect children in our study population). 33 We observed increases in neutrophil infiltration in the NP of children we studied correlating with the magnitude of Spn burden, regardless of whether patients were currently healthy or suffering from a viral URI. In other words, the apparent increase in neutrophil recruitment during URI (Figure 2) can be attributed to the increase in Spn bacterial burden during URI (Figure 1), and it is only during the robust inflammatory response associated with an active middle-ear infection that neutrophil recruitment is significantly elevated. The major determinant of neutrophil recruitment to the NP during a viral URI was Spn burden, not the presence of a viral infection as such. We found Spn burden to play a similar role with respect to transcription and protein secretion of various inflammatory genes: apparent differences between measurements taken during health, URI and AOM were, in fact, attributable to the Spn burden in the NP sample. While AOM independently increased neutrophil recruitment and secretion of IL-6 and IL-8, the presence of a viral URI did not appear to significantly increase the expression of inflammatory markers independently of bacterial burden.
The transcription of inflammatory genes followed similar patterns, with IL-8, TNF-α and SOD2 correlating positively with Spn bacterial burden, whereas transcription of CXCL1, a neutrophil chemoattractant, was significantly elevated during AOM. Since neutrophils are the predominant cell type present in nasal wash samples, 19 the bulk of gene transcription can be attributed to neutrophils. These measurements thus constitute a survey of the activation status, as well as the recruitment of neutrophils.
Secretion of inflammatory cytokines, however, can be attributed to any of the cell types present in the NP at the time of sample collection, including epithelial cells and T cells.34–36 However, human neutrophils are known to produce inflammatory cytokines in their own right; in particular, IL-8 and TNF-α.37,38 We observed that the transcription of IL-8 and TNF-α mRNA in nasal wash pellets (attributable mainly to neutrophils) correlated with the concentrations of these cytokines in the NP. This suggests that neutrophils may be meaningful contributors to the production of these and other cytokines during upper respiratory inflammatory responses.
Researchers from our laboratory have previously identified deficiencies in the immune responses of otitis-prone children. Specifically, otitis-prone children display reduced production of specific Abs against Spn, 39 associated with reduced generation of memory T and B cells.40,41 As a result, the NP of the otitis-prone child is a more permissive environment for the establishment of asymptomatic colonization by otopathogens (Figure 6), predisposing them to experience more frequent episodes of otitis media. Taken as a whole, our results suggest that neutrophil responses to Spn are normal in otitis-prone children, and that their heightened susceptibility to otitis media is instead the result of increased incidence of the transient colonization episodes that are a prerequisite for AOM. 11
This is the first study to analyze inflammatory gene transcription and cytokine production along with quantitative measurements of Spn bacterial burden and neutrophil recruitment in the same NP samples. The relationship between Spn and neutrophil-associated inflammation may also hold true for other major pediatric respiratory pathogens such as H. influenzae and M. catarrhalis. The increased incidence of otitis media in the otitis-prone population appears to arise not from defects in neutrophil recruitment or function, but rather from increased colonization by otopathogens as a result of diminished adaptive immune memory. We conclude that neutrophils are contributors to the secretion of inflammatory cytokines during Spn infection, and that Spn bacterial burden is the major driver of neutrophil recruitment and innate inflammatory responses in the NP during asymptomatic colonization, viral URI and AOM.
Footnotes
Acknowledgments
The authors thank Professor A. Almudevar (University of Rochester) for assistance with statistical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the National Institute on Deafness and Other Communication Disorders (R01 DC008671 to M.E. Pichichero, including a postdoctoral award to M.C. Morris).
