Abstract
This study was aimed at investigating whether dietary copper/zinc-loaded montmorillonite (Cu/Zn-Mt) could alleviate Escherichia coli LPS-induced intestinal injury through pro- and anti-inflammatory signaling pathways (TLRs, NLRs and TGF-β1) in weaned piglets. Eighteen 21-d-old pigs were randomly divided into three groups (control, LPS and LPS + Cu/Zn-Mt). After 21 d of feeding, pigs in the LPS group and LPS + Cu/Zn-Mt group received i.p. administration of LPS, whereas pigs in the control group received saline. At 4 h post-injection, jejunum samples were collected for analysis. The results indicated that, compared with the LPS group, supplemental Cu/Zn-Mt increased transepithelial electrical resistance, the expressions of anti-inflammatory cytokines (TGF-β1) in mRNA and protein levels, and decreased FD4 and the mRNA expression of pro-inflammatory cytokines (TNF-α, IL-6, IL-8 and IL-1β). The pro-inflammatory signaling pathways results demonstrated that Cu/Zn-Mt supplementation decreased the mRNA levels of TLR4 and its downstream signals (MyD88, IRAK1, TRAF6) but had no effect on NOD1 and NOD2 signals. Cu/Zn-Mt supplementation did not affect NF-κB p65 mRNA abundance, but down-regulated its protein expression. The anti-inflammatory signaling pathways results showed supplemental Cu/Zn-Mt also increased TβRII, Smad4 and Smad7 mRNA expressions. These findings suggested dietary Cu/Zn-Mt attenuated LPS-induced intestinal injury by alleviating intestinal inflammation, influencing TLR4-MyD88 and TGF-β1 signaling pathways in weaned pig.
Keywords
Introduction
The intestinal tract plays a crucial role in preventing exogenous antigens, microorganisms and their toxins from entering the intestinal mucosa and systemic circulation.1,2 Early weaned piglets are commonly associated with intestinal barrier dysfunction, characterized by increased intestinal permeability. 3 Recently, weaning has been shown to trigger intestinal inflammation, which has a negative impact on gut integrity and epithelial functions. 4 Both in vitro and in vivo investigations show that the inflammatory response and subsequent overproduction of inflammatory cytokines induce an impaired intestinal epithelial barrier.4,5 Many researches have demonstrated that high dose of zinc (Zn) and copper (Cu) are involved in resistance to inflammation and barrier dysfunction in weaned piglets. 6 However, this feeding approach could cause large amounts of Zn and Cu to be excreted, leading to environmental problems. 7 Consequently, considering the environmental risk and sustainable development of the pig industry, it is necessary to seek an alternative to reduce Cu and Zn supplements in weaned piglets.
Montmorillonites (Mt), a naturally occurring hydrated aluminium silicate clay, is made up of layers formed by two silica tetrahedral sheets layered between an alumina octahedral sheet. Mt has high specific surface area, ion-exchange capacity, rheological properties, and low or null toxicity. These specific physical and chemical properties enable Mt as a drug delivery carrier. 8 When drugs are intercalated into Mt, the specific structure of Mt provides effective drug delivery systems with the ability to delay and/or prolong drug release, increase drug stability and modify drug delivery patterns. 9 Recently, special attention has been paid to Mt as a metal ion carrier, as metals exchanged in the Mt complex are superior in terms of safety, stability and long-term effectiveness when compared with conventional metal. 10 Researchers have found that Mt intercalated with two metals displays obvious synergistic effects in vitro. 11 We have synthesized Cu/Zn-loaded Mt (Cu/Zn-Mt) and found that this composite has a synergic effect in inhibiting the growth of pathogenic bacteria in vitro. 10 Our in vivo study showed that Cu/Zn-Mt had a beneficial effect in restoration of mucosal barrier integrity (unpublished data). However, whether the protective effect of Cu/Zn-Mt on intestinal integrity is related to alleviation of intestinal inflammation needs to be further investigated.
It is well known that cytokines, including pro- and anti-inflammatory molecules, play an important role in regulating the intestinal inflammation response. Pro-inflammatory cytokines (TNF-α, IL-1β and IL-6) can be induced by activating TLR- and NOD-mediated signaling pathways, which play a key role in recognizing PAMPs and initiating inflammation and immune response.12,13 TGF-β, a strong anti-inflammatory cytokine, regulates anti-inflammatory effects and its signaling pathway is mediated by the Smad family of proteins.14,15 Moreover, a previous study has demonstrated the role of TGF-β1 in the restoration of barrier integrity. 16
Accordingly, the purpose of this research was to investigate whether supplemental Cu/Zn-Mt could improve the intestinal epithelial barrier by regulating the expression of genes associated with inflammation in weaned pigs after Escherichia coli LPS challenge. In this study, E. coli LPS, a potent endotoxin, was administered to build the model of intestinal injury. The expressions of genes associated with inflammatory signaling pathways (TLR, NOD and TGF-β1) were analyzed to elucidate the underlying mechanisms.
Materials and methods
Animals and experiment design
Ingredient and composition of basal diets (as-fed basis).
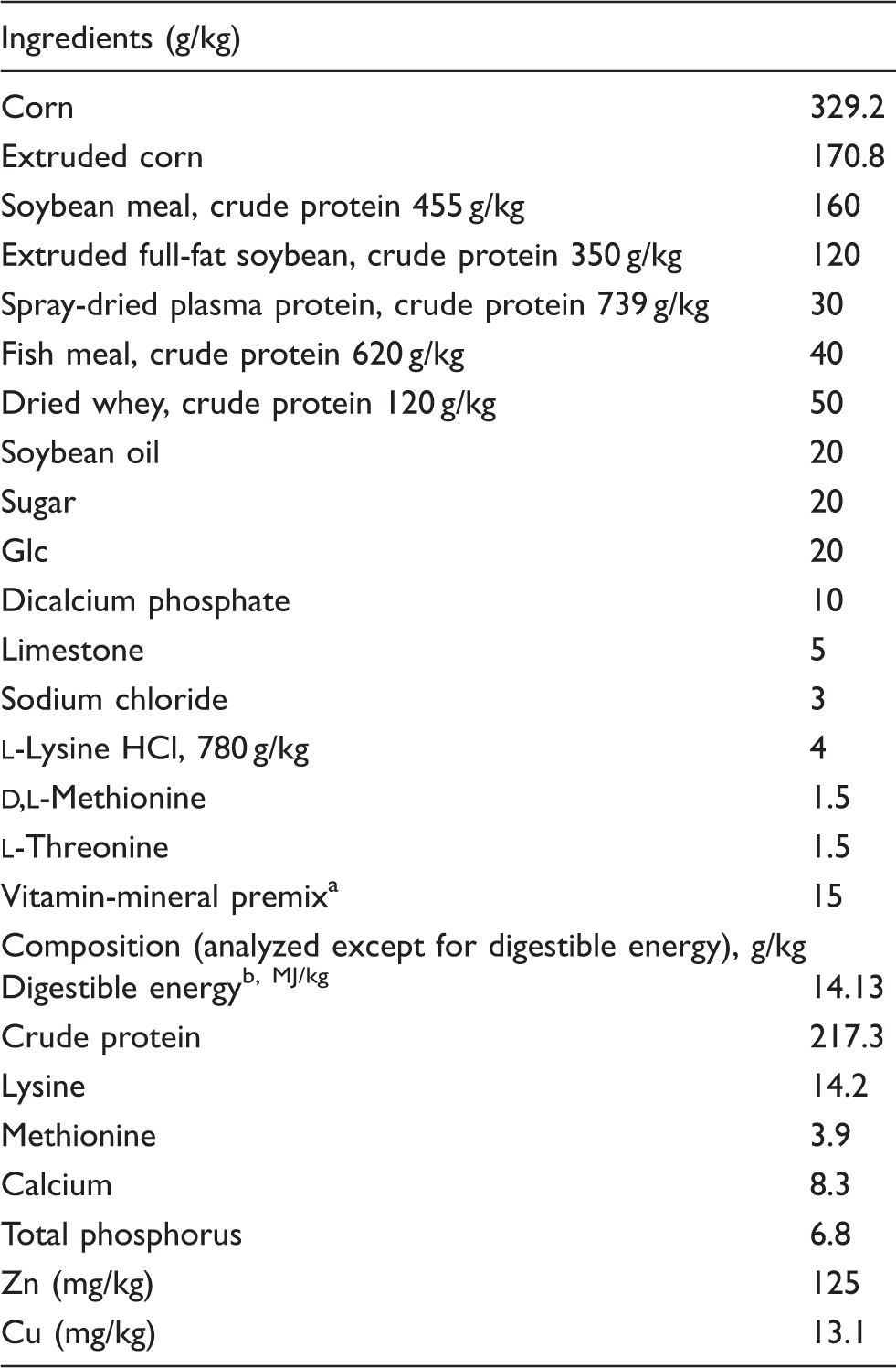
Supplied per kg of diet: vitamin A, 2200 IU; vitamin D3, 220 IU; vitamin E, 16 IU; vitamin K3, 0.5 mg; riboflavin, 4.0 mg; thiamine, 1.0 mg; niacin, 15 mg; pyridoxine, 1.5 mg; pantothenic acid, 10 mg; choline, 500 mg; folic acid, 0.3 mg; biotin, 0.05 mg; vitamin B12, 0.02 mg; Zn (ZnSO4), 100 mg; Cu (CuSO4·5H2O), 6 mg; I (KI), 0.14 mg; Fe (FeSO4), 100 mg; Mn (MnSO4·H2O), 4 mg; Se (Na2SeO3), 0.3 mg. bDigestible energy was calculated from data provide by Feed Database in China.
Sample collections
Four h after LPS or saline solution injection, all piglets were euthanized with an i.v. injection of sodium pentobarbital and the gastrointestinal tract was removed immediately. Segments of proximal jejunum were stripped from the seromuscular layer and put in oxygenated Ringer’s solution immediately for Ussing chamber studies. The mucosa samples from remaining jejunum were scraped using a glass slide, rapidly frozen in liquid nitrogen and stored at −80℃ for further analysis.
Intestinal barrier function analyses
Tissues were mounted in EasyMount Ussing chamber system (model VCC MC6; Physiologic Instruments, San Diego, CA, USA) as previously described. 17 Briefly, data were collected automatically using Acquire and Analyze software (Physiologic Instruments). Transepithelial electrical resistance (TER) was recorded at 15-min intervals over a 1-h period after a 15-min equilibration period. The flux of FITC dextran 4 kDa (FD4) was used to evaluate the epithelial barrier function as previously described. 17 A 400 μ/ml probe FD4 (FD4-100MG; Sigma-Aldrich, St. Louis, MO, USA) was added into the mucosal side. The concentration of FD4 in the serosal side was measured by fluorescence microplate reader (FLx800; BioTek Instruments, Winooski, VT, USA). The excitation and extinction wavelengths were 492 and 520 nm, respectively.
mRNA expression analysis by real-time PCR
Primer sequences used for real-time PCR.

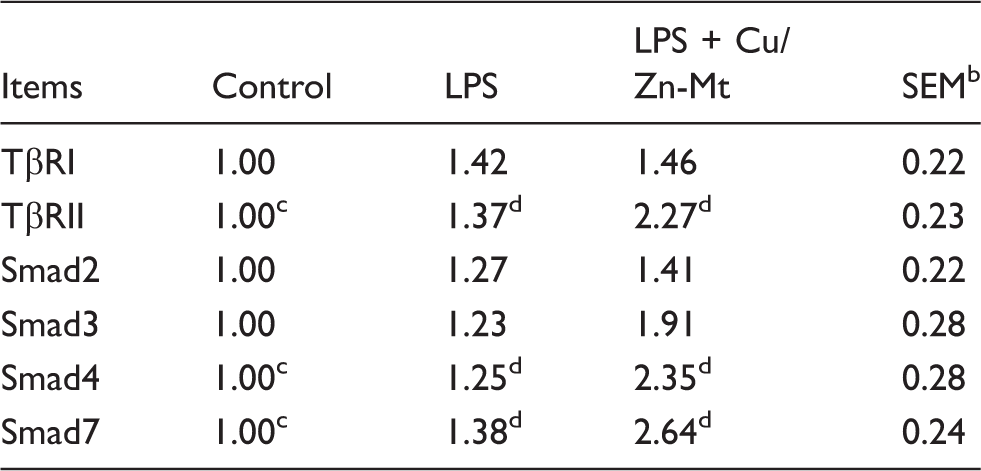
IRAK1: IL-1 receptor-associated kinase 1; TRAF6: TNF receptor-associated factor 6; RIPK2: receptor-interacting serine/threonine protein kinase-2; TβRI: TGF-β type I receptor; TβRII: TGFβ type II receptor; Smad2: mothers against decapentaplegic homolog 2; Smad3: mothers against decapentaplegic homolog 3; Smad4: mothers against decapentaplegic homolog 4; Smad7: mothers against decapentaplegic homolog 7.
Protein expression analysis by Western blot
The Western blot analysis was performed measured as previously described by Hu et al. 17 Briefly, the total protein was extracted by a commercial kit provided by NanJing KeyGen Biotech (Nanjing, China). Intestinal mucosa proteins were separated using a polyacrylamide gel and electrophoretically transferred onto a polyvinylidene difluoride membrane. The membranes were blocked with 5% non-fat dried milk proteins for 1 h and then incubated for 12 h at 4℃ with the primary Ab (TGF-β1 rabbit mAb, NF-κB p65 rabbit mAb, β-actin rabbit mAb), which were purchased from Cell Signaling Technology (Danvers, MA, USA). After washing with TBST, proteins were detected with HRP-conjugated secondary Abs (Beyotime, Haimen, China) for 1 h. After washing with TBST, specific bands were visualized with an ECL detection kit (Beyotime). Quantity One software was used to evaluate the Western blotting result (Bio-Rad Laboratories, Hercules, CA, USA). As an internal control, β-actin exhibited no difference within each group. The ratio of target protein and β-actin protein was used to evaluate the relative abundance of each target protein. The protein expression from two groups was calculated relative to the control group and expressed as fold changes.
Statistical analysis
Data were subjected to one-way ANOVA using SPSS 9.0 (IBM, Armonk, NY, USA). Duncan multiple range tests were used to define differences among treatments. Effects were considered significant at P < 0.05.
Results
Intestinal barrier function
Effects of Cu/Zn-Mt on barrier function of weaned piglets.
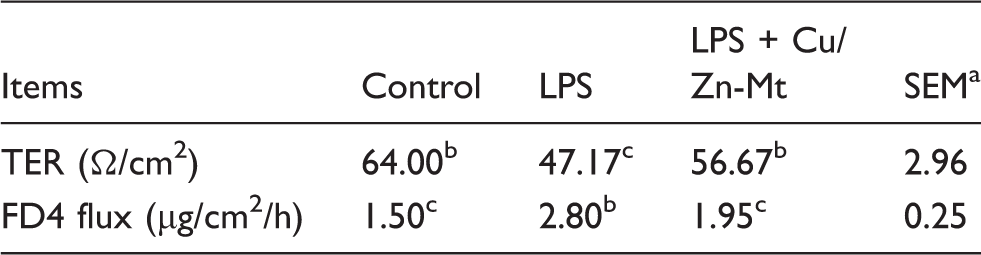
n = 6.
Means within a row with different letters differ significantly (P < 0.05).
Intestinal mRNA expressions of pro-inflammatory cytokines and anti-inflammatory cytokines
Effects of Cu/Zn-Mt on cytokine mRNA levels on the jejunal mucosa of piglets. a
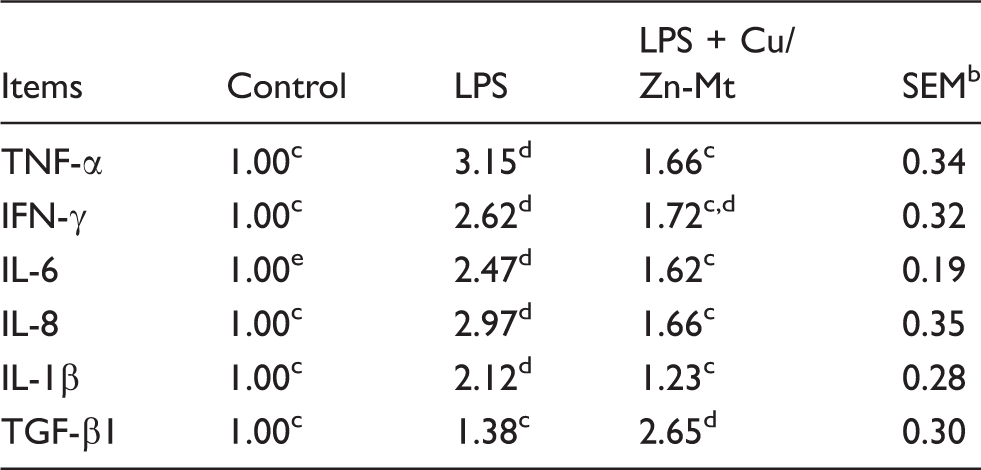
The 2-ΔΔCt method was used to analyze the relative expression (fold changes), calculated relative to the control group.
n = 6.
Means within a row with different letters differ significantly (P < 0.05).
mRNA expressions of TLR4 and NOD and their downstream signals
Effects of Cu/Zn-Mt on mRNA expressions of TLR4 and NOD and their downstream signals of weaned piglets. a
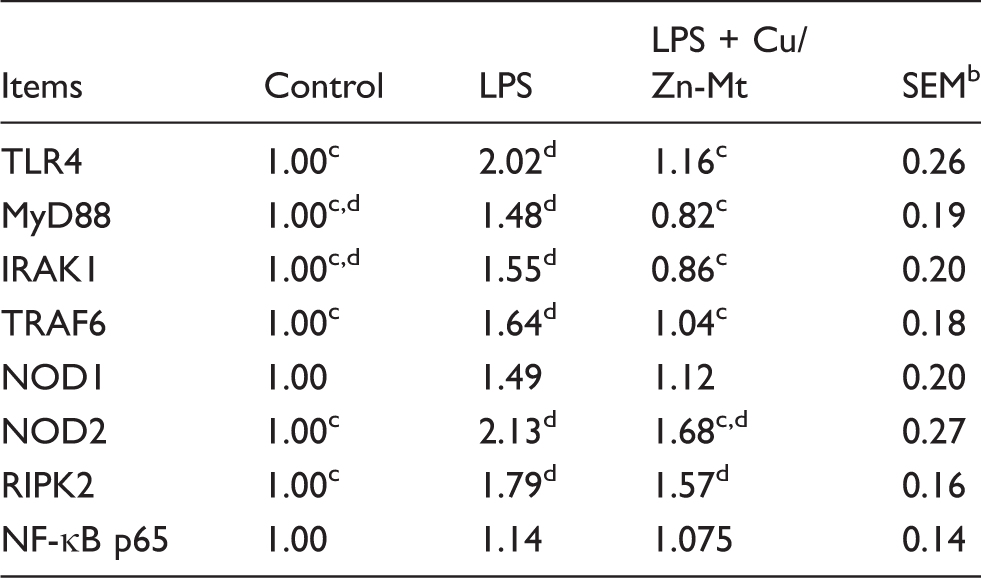
The 2-ΔΔCt method was used to analyze the relative expression (fold changes), alculated relative to the control group.
n = 6.
Means within a row with different letters differ significantly (P < 0.05).
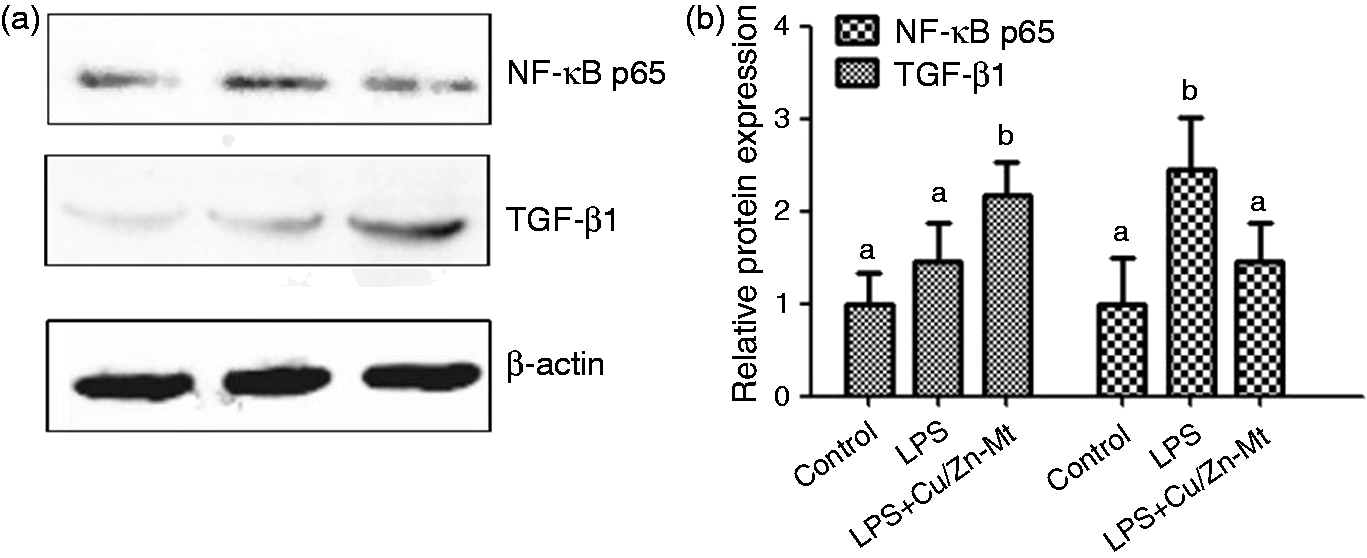
Effects of dietary Cu/Zn-Mt on the protein expression of TGF-β1 and NF-κB p65 in the jejunal mucosa of piglets. (a) Representative blots of TGF-β1, NF-κB p65 and β-actin. (b) Relative TGF-β1 and NF-κB p65 protein expression in jejunal mucosa of piglets. a,bMean values with different letters were significantly different (P < 0.05). The control sample was used as the reference sample. The protein expression of all samples was expressed as fold changes, calculated relative to the control group.
TGF-β1 protein expression and mRNA expressions of its canonical Smad signaling pathway
Effects of Cu/Zn-Mt on mRNA expressions of Smad signals on the jejunal mucosa of piglets. a
The 2-ΔΔCt method was used to analyze the relative expression (fold changes), calculated relative to the control group.
n = 6.
Means within a row with different letters differ significantly (P < 0.05).
Discussion
An intact intestinal barrier plays a crucial role in maintaining health and preventing tissue injury and several diseases. 19 Piglets often experience intestinal impairment of mucosal barrier function at weaning, which has a significant effect on subsequent growth performance and carcass characteristics. 20 In the current experiment, the results showed that LPS challenge resulted in a decreased TER and increased FD4 flux in jejunum of piglets, which indicated that LPS caused intestinal mucosa damage. This is consistent with the studies on the effect of LPS on intestinal barrier function in weaned pigs.2,21 Our study showed that dietary supplementation of Cu/Zn-Mt alleviated barrier function disorder caused by LPS. Our observations were supported by our previous study that dietary supplementation of Cu/Zn-Mt supported epithelial barrier integrity by decreasing plasma diamine oxidase (DAO) activities and increasing the protein expression of tight junctions (unpublished data). A large body of evidence has indicated that Zn has been involved in enhancing the restoration of epithelium after injury in weaned piglets.18,22,23 Cu is widely used in weaned pigs to prevent gastrointestinal ecosystem disorder during the post-weaning stress. 22 Previous studies have reported the utilization of Mt as a carrier for various active ingredients. 24 Moreover, a few studies have reported that a two-metal loaded Mt composite displayed obviously better synergistic effects in vitro than single-metal loaded Mt.10,11 Apart from being a carrier, Mt has been used as gastrointestinal protectors based on their high specific area and sorption capacity. 25 Mt was capable of adsorbing bacteria and bacterial enterotoxins, reinforcing the intestinal mucus barrier with the reduction of penetration of luminal antigens through the mucus layer, possibly directing modulatory action on pro-inflammatory cytokine production.24–26 It has been suggested that the protective barrier effect of Cu/Zn-Mt may be explained by interactions of Cu, Zn and Mt.
Cytokines, including pro-inflammatory and anti-inflammatory ones, are small peptide molecules that play a crucial role in the regulation of the immune and inflammatory responses in the gastrointestinal tract. 4 Accumulating evidence has demonstrated that they were also involved in regulating the intestinal barrier integrity. 27 It is increasingly recognized that overproduction of pro-inflammatory cytokines caused a disturbance in intestinal tight junction barrier and lead to epithelial cell apoptosis. 19 Anti-inflammatory cytokines protected against the disruption of intestinal tight junction barrier. 27 Therefore, the release control of pro-inflammatory cytokines in the intestine may alleviate intestinal disorders induced by LPS. In the present study, piglets challenged with LPS increased pro-inflammatory cytokine (TNF-α, IL-1β, IL-6, IFN-γ and IL-8) gene expression in the jejunal mucosa, which was in accordance with previous reports.21,28 The LPS-induced model of gut inflammation is a commonly used and well-established intestinal injury model. Previous studies have shown that LPS induces acute bacterial infection, followed by stimulating macrophages and monocytes to produce and secrete related pro-inflammatory cytokines, eventually leading to organism systemic inflammation. 29 Consistent with improved intestinal integrity, dietary Cu/Zn-Mt decreased pro-inflammatory cytokine expression (TNF-α, IL-1β, IL-6, and IL-8), which indicated that LPS-induced inflammation was minimized by Cu/Zn-Mt supplementation. Similar to our findings, Namkung et al. 28 reported that dietary addition of Zn and Cu could help maintain homeostasis and immune function in weaned pigs after LPS challenge. Recent reports suggested that the protective role of Zn in inflammatory responses was partially due to its inhibitory effects on the overproduction of pro-inflammatory mediators (TNF-α, IL-1β, and IL-8).6,18,30 It has been reported that increasing the content of Cu could inhibit the production of IFN-γ in vitro. 31 Gonzalez et al. 32 reported that Mt could be used for the treatment of chronic trinitrobenzene sulfonic acid colitis in rats, possibly having a direct role in regulating mucosal cell production of cytokines. Moreover, we found that dietary Cu/Zn-Mt increased anti-inflammatory cytokine expression (TGF-β). TGF-β, produced by activated monocyte macrophages and other inflammatory cells, plays a key role in regulating mucosal immune response and intestinal barrier restoration. 33 It has been reported that most of the cytokines promote epithelial restitution through a TGF-β-dependent mechanism. 21 Xiao et al. 16 found that the TGF-β1 was associated with the restoration of intestinal morphology and barrier function in weaning piglets. Shiou et al. 34 reported that oral administration of TGF-β1 reduces the incidence of necrotizing enterocolitis through an immune suppressive effect directly on intestinal epithelium in vivo and decreased systemic IL-6 and IFN-α levels. Similar to our results, Roselli et al. 35 reported that Zn up-regulated TGF-β mRNA in E. coli K 88-infected cells. Song et al. 18 found that Zn supplementation increased TGF-β1 expression in weaned piglets. Berthon et al. 36 demonstrated that the anti-inflammatory role of Cu is well known. Therefore, in this research, dietary addition of Cu/Zn-Mt may improve intestinal integrity, partially through its inhibitory effects on pro-inflammatory mediators and activation of anti-inflammatory mediator.
To explain the mechanisms by which Cu/Zn-Mt alleviates the intestinal pro-inflammatory response, we investigated the role of TLR and NLR signaling pathways. As the pathogen sensors, TLRs and NODs are triggered by markers of microbial non-self (PAMPs) to activate innate immune system in the gut. 12 TLRs are membrane-anchored proteins and have been demonstrated to cause systemic inflammatory response syndrome through directly identifying molecular patterns (LPS, RNA and DNA viruses, etc.) specific to microbes in the earliest stages of the host defense. 12 It is well known that TLR4 is the LPS receptor in the TLR family. Among the NOD family, both NOD1 and NOD2 can recognize specific structures from bacterial peptidoglycan and LPS, and then deliver a TLR-independent signal, which also cause NF-κB activation through RIPK2 and stimulates the expression of pro-inflammatory cytokines. 37 In the present study, we found that pigs challenged with LPS decreased the intestinal mRNA abundance of TLR4, TRAF6, NOD2 and RIPK2. Similar results were observed that TLR4 and NOD2 signaling pathways could be activated by LPS. 38 Dietary Cu/Zn-Mt decreased the mRNA expressions of TLR4, MyD88, IRAK1 and TRAF6 in jejunal mucosa. Although LPS group and LPS + Cu/Zn-Mt group had no effect on the NF-κB p65 mRNA expressions, LPS group up-regulate the NF-κB p65 protein expression and LPS + Cu/Zn-Mt group down-regulated the NF-κB p65 protein expression. Moreover, we observed that supplemental Cu/Zn-Mt did not influence the intestinal expressions of NOD1 and NOD2, and their adaptor molecule RIPK2. It has been suggested that Cu/Zn-Mt may inhibit the expression of inflammatory mediators through the TLR4–MyD88 signaling pathway.
To explain the mechanisms of Cu/Zn-Mt on the intestinal anti-inflammatory response, we investigated the role of TGF-β1 signaling pathways. The canonical TGF-β1 signaling pathway is mediated by intracellular substrates, known as SMAD family proteins. To explain whether the TGF-β1 signaling pathway is related to Cu/Zn-Mt’s beneficial effects on LPS-induced intestinal injury, we tested the canonical downstream substrates of the TGF-β1 signal (TβRI, TβRII, Smad2, Smad3, Smad4 and Smad7) and observed that Cu/Zn-Mt supplementation increased the gene expressions of TβRII, Smad4 and Smad7 after LPS challenge, indicating that the canonical Smad signaling pathway was activated. However, mRNA expression of Smad2 and Smad3 in the intestinal mucosa did not vary significantly compared with the LPS and control groups. In line with our study, we speculated that Cu/Zn-Mt-improved intestinal barrier restoration in LPS-induced intestine injury may be partially induced through the TGF-β1 Smad pathway, either directly or indirectly.
In conclusion, dietary supplementation with Cu/Zn-Mt has a positive effect in improving intestinal integrity. The protective effects of Cu/Zn-Mt on the intestine might be closely associated with regulation of the expression of intestinal inflammatory cytokines via TLR4 and TGF-β1 canonical Smad pathways.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by National Key R & D Program (2016YFD0501210), Zhejiang Province Key R & D Project (2015C02022), Zhejiang Province Major Science and Technology Project (2015C03006), Special Fund for Agro-scientific Research in the Public Interest (201403047), Dabeinong Funds for Discipline Development and Talent Training in Zhejiang University.
