Abstract
This study explored whether zinc oxide (ZnO) supplementation could alleviate weanling-induced intestinal injury through TLR and NOD-like receptor signaling pathways. Twelve early-weanling piglets were allotted to two dietary treatments (control vs 2200 mg Zn/kg from ZnO) for 1 wk. The results showed that supplemental ZnO improved daily gain and feed intake, decreased post weaning scour scores, increased villus height and villus height:crypt depth ratio at the jejunal mucosa, and decreased diamine oxidase activity and endotoxin concentration in plasma. The intestinal mRNA levels of TLR4 and its downstream signals, including MyD88, IL-1 receptor-associated kinase 1 and TNF-α receptor-associated factor 6, were decreased, and the expressions of intestinal pro-inflammatory cytokines and chemokines were decreased simultaneously in the ZnO-supplemented piglets. Although NF-κB p65 mRNA abundance was not affected by ZnO supplementation, NF-κB p65 protein expression was down-regulated by ZnO. However, ZnO supplementation had no effect on intestinal expressions of NOD1 and NOD2, and their adaptor molecule receptor-interacting serine/threonine-protein kinase 2, as well as protein expressions of caspase-3 and heat shock protein 70. The results indicated that the protective effects of ZnO on intestinal integrity were closely related to decreasing the expressions of genes associated with inflammation through inhibiting the TLR4-MyD88 signaling pathways.
Keywords
Introduction
Weaning is the most significant event in the life of pigs as they are abruptly forced to adapt to nutritional, immunological and psychological disruptions. 1 The weaning transition of piglets is commonly associated with growth retardation and impaired intestinal barrier.1–4 One of the major problems in nursery pig management is post-weaning syndrome, which includes post-weaning diarrhea and endotoxin shock. 5 It is widely accepted that endotoxin plays an important role in the development of post-weaning diarrhea. 5 Endotoxin shock is a shock-like syndrome that is caused by the systemic effects of endotoxin, and usually occurs 1 wk after weaning. 5
Early-weaned piglets are susceptible to immunologic challenge as a result of their immature immune and digestive systems. 6 It has been well documented that weaning triggers intestinal inflammation, marked by an up-regulated expression of pro-inflammatory cytokines in the intestine, such as TNF-α, IL-6, IL-1β and INF-γ.4,7,8 The inflammatory response and subsequent overproduction of pro-inflammatory cytokines induce a disrupted intestinal barrier function.1,4,6 From the nutritional perspective, controlling early intestinal inflammation is certainly a challenge in managing post-weaning intestinal disorders in piglet.4,8
It is well known that zinc (Zn) is involved in resistance to inflammation, diarrhea and infection.9–11 The role of Zn in restoration of mucosal barrier integrity has been demonstrated both in vitro and in vivo.9,10,12 The duration and severity of diarrhea can be controlled by Zn supplementation.9–11 In particular, zinc oxide (ZnO) appears to have a strong effect in resistance to intestinal diseases. 12 Supplementing pharmacologic concentrations of ZnO (between 2000 and 4000 mg/kg of Zn) in weanling piglets alleviates post-weaning diarrhea and improves growth performance.9,10,13 Although there is evidence suggesting that the beneficial role of ZnO in the intestine is closely related to the down-regulation of pro-inflammatory cytokines,9,10,14 the underlying mechanisms remain largely unknown.
In the gut, two classes of PRRs, the TLRs and the NLRs, play a crucial role in the recognition of PAMPs by the mucosal immune system. 15 TLRs are membrane-anchored proteins that are expressed either at the cell surface or associated with intracellular organelles, whereas NLRs are localized in cell cytosol. 15 A key hallmark of inflammation is the induction of the NF-κB pathway, which is responsible for turning on pro-inflammatory cytokines and chemokines. 15 The activation of NF-κB is triggered both by TLRs and some NLRs.
We hypothesized that dietary ZnO supplementation would improve the intestinal barrier by regulating the production of pro-inflammatory cytokines through modulation of TLR and NLR signaling pathways. In the present study, we employed an early-weanling porcine model, to explore whether ZnO supplementation could alleviate weanling-induced intestinal injury through the TLR4 and NLR signaling pathways.
Materials and methods
Experimental design and pig care
Ingredient and composition of the basal diet (as-fed basis).
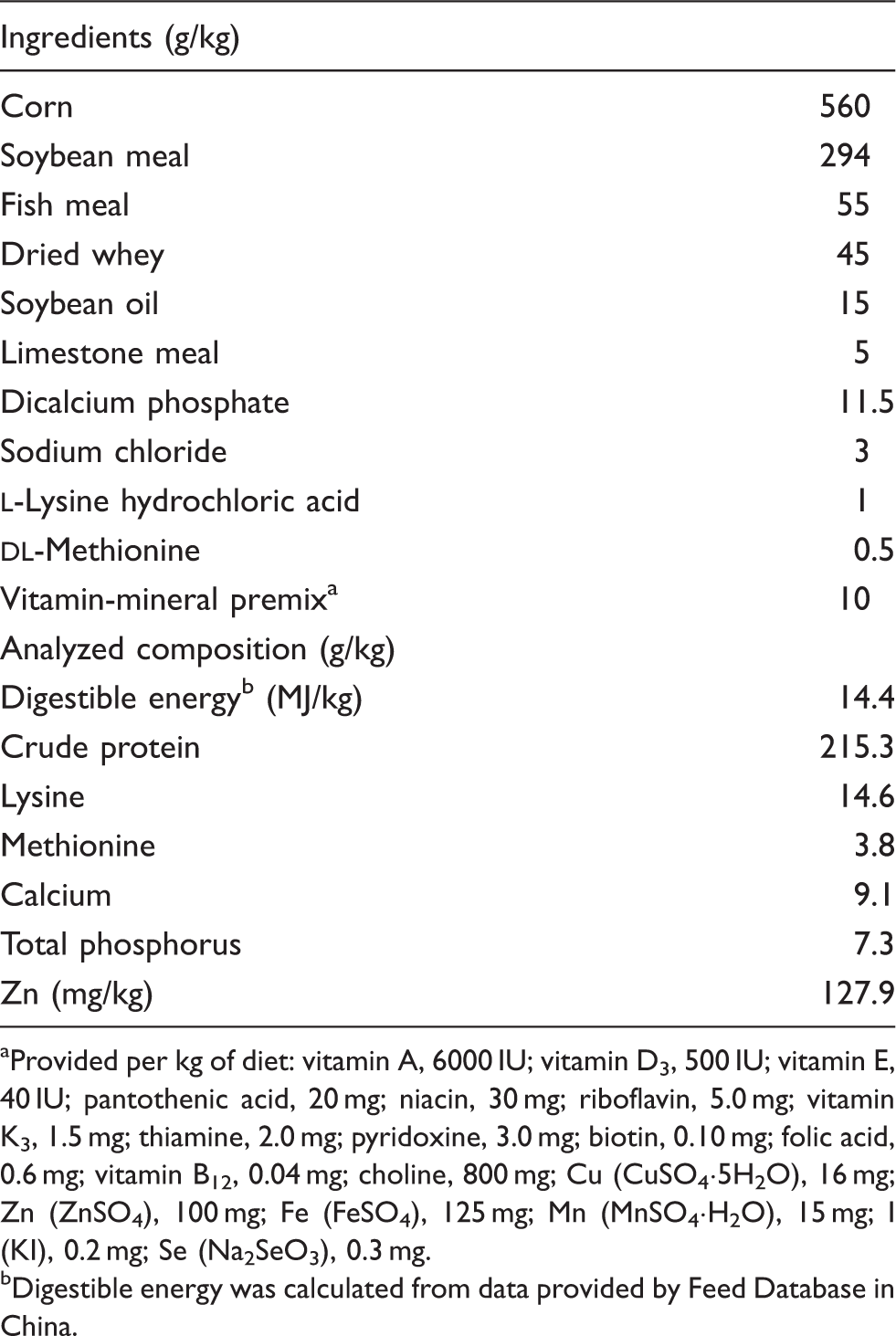
Provided per kg of diet: vitamin A, 6000 IU; vitamin D3, 500 IU; vitamin E, 40 IU; pantothenic acid, 20 mg; niacin, 30 mg; riboflavin, 5.0 mg; vitamin K3, 1.5 mg; thiamine, 2.0 mg; pyridoxine, 3.0 mg; biotin, 0.10 mg; folic acid, 0.6 mg; vitamin B12, 0.04 mg; choline, 800 mg; Cu (CuSO4·5H2O), 16 mg; Zn (ZnSO4), 100 mg; Fe (FeSO4), 125 mg; Mn (MnSO4·H2O), 15 mg; I (KI), 0.2 mg; Se (Na2SeO3), 0.3 mg.
Digestible energy was calculated from data provided by Feed Database in China.
Sample collections
After the feeding trial, blood samples for diamine oxidase (DAO; EC 1.4.3.6) analysis were taken from the anterior vena cava into tubes containing sodium heparin and mixed immediately to avoid coagulation. Plasma was obtained after centrifugation at 3000 g for 15 min at 4℃ and then stored at −80℃ for DAO analysis. Endotoxin-free needles and tubes containing sodium heparin were used to collect the blood samples for endotoxin analysis according to the procedure of Hagman et al. 17 After sampling, the tubes were immediately placed on ice and centrifuged. The plasma was then transferred by autoclaved pipettes to endotoxin-free glass tubes, frozen and stored at −20℃. Only sterile, pyrogen-free equipment was used. All samples were transported in a Styrofoam box with ice clamps, and arrived within 4 h at the laboratory, where the endotoxin analyses were performed. 17 After blood collection was complete, all the piglets were slaughtered under anesthesia. The gastrointestinal tract was removed immediately. The specimens from the mid-jejunum were excised, flushed with physiological saline and fixed in 10% formalin. The mucosa samples from the middle part of jejunum were harvested by scraping with a sterile glass microscope slide, rapidly frozen in liquid nitrogen and stored at −80℃ for further analysis.
Intestinal morphological analyses
The jejunal segments were dehydrated, embedded and stained with hematoxylin and eosin according to the procedures of Hu et al.4,13 Villus height and crypt depth were determined using an image processing and analysis system (Version 1; Leica Imaging Systems, Cambridge, UK).
DAO activity in plasma and intestinal mucosa
DAO activity in plasma and intestinal mucosa was measured using an enzymatic spectrophotometric assay, as described by Hu et al. 13 Cadaverine dihydrochloride, o-dianisidine dihydrochloride, HRP and DAO standard were purchased from Sigma Chemical (St. Louis, MO, USA). DAO activity in plasma and intestinal mucosa was expressed as U/ml and U/mg protein, respectively.
Concentrations of endotoxin in plasma
The plasma endotoxin was analyzed using a standard limulus amebocyte lysate assay. 17 The endotoxin testing kit was purchased from Zhanjiang Bokang Marine Biological (Zhanjiang, China). The procedures were carried out following the manufacturer’s protocol.
mRNA expression analysis by real-time PCR
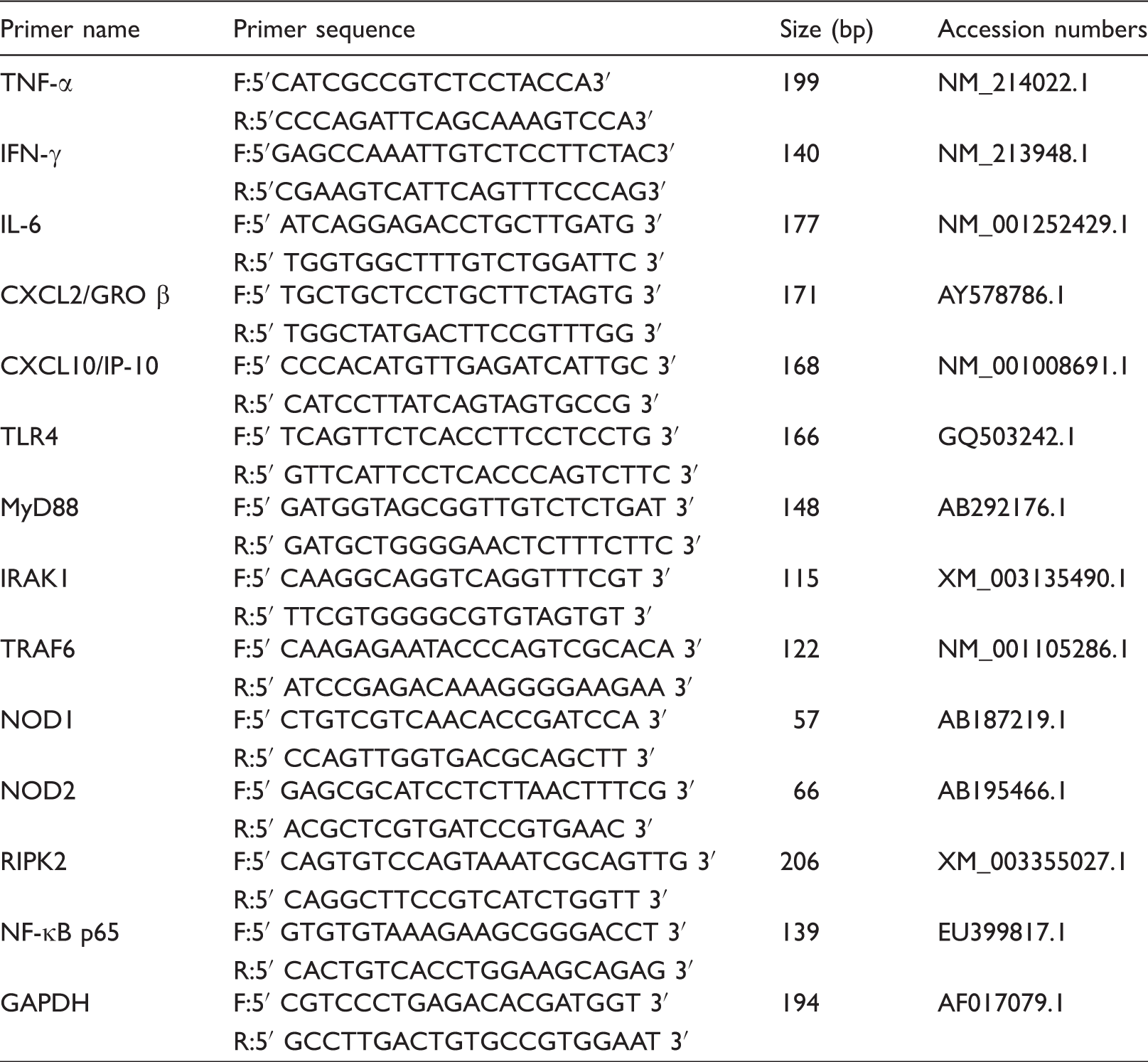
Primer sequences used for real-time PCR.a
Protein expression analysis by Western blot
Western blot analysis was performed as described by Liu et al. 18 Briefly, the intestinal mucosa samples were homogenized in lysis buffer and centrifuged to collect the supernatants. Intestinal mucosa proteins were separated on a polyacrylamide gel and transferred onto polyvinylidene difluoride membranes. The membranes were incubated overnight (12–15 h) at 4℃ with primary Abs and then with the secondary Ab for 120 min at room temperature (22℃). The primary Abs [Caspase-3 rabbit mAb, heat shock protein (HSP)70 rabbit mAb, NF-κB p65 rabbit mAb, β-actin rabbit mAb] were purchased from Cell Signaling Technology Inc. (Danvers, MA, USA). The secondary Ab was HRP-conjugated anti-rabbit Ab. Western blot was detected with an Enhanced Chemiluminescence detection kit (Amersham, Arlington Heights, IL, USA), photographed by the ChemiScope 3400 machine (Clinx Science Instruments, Shanghai, China), and analyzed using Quantity One software. The β-actin was used as an internal control, which exhibited no difference between the groups. The relative abundance of each target protein was expressed as target protein/β-actin protein ratio.
Statistical analysis
Data were analyzed using the SAS statistical package (SAS Institute, Cary, NC, USA), 21 with each animal as an experimental unit. Results are expressed as mean ± SD. Differences between means were tested using Student’s t-test. Differences were considered significant at P < 0.05.
Results
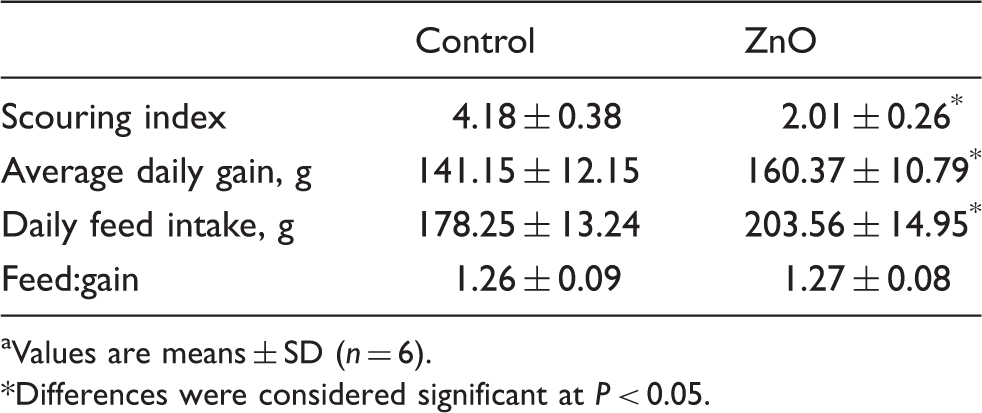
Scouring index and growth performance
Effects of ZnO on scouring index and performance of weanling pigs. a
Values are means ± SD (n = 6).
Differences were considered significant at P < 0.05.
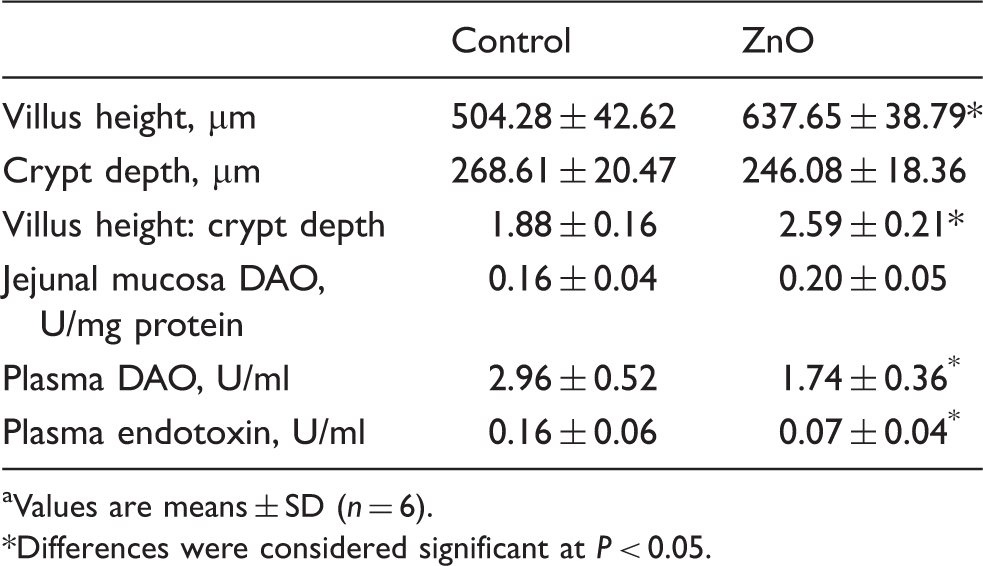
Intestinal morphology and barrier function
Effect of ZnO on jejunal morphology and barrier function of weanling pigs. a
Values are means ± SD (n = 6).
Differences were considered significant at P < 0.05.
Intestinal mRNA expressions of cytokines and chemokines
Effect of ZnO on cytokine and chemokine mRNA relative expressions in jejunal mucosa of piglets. a
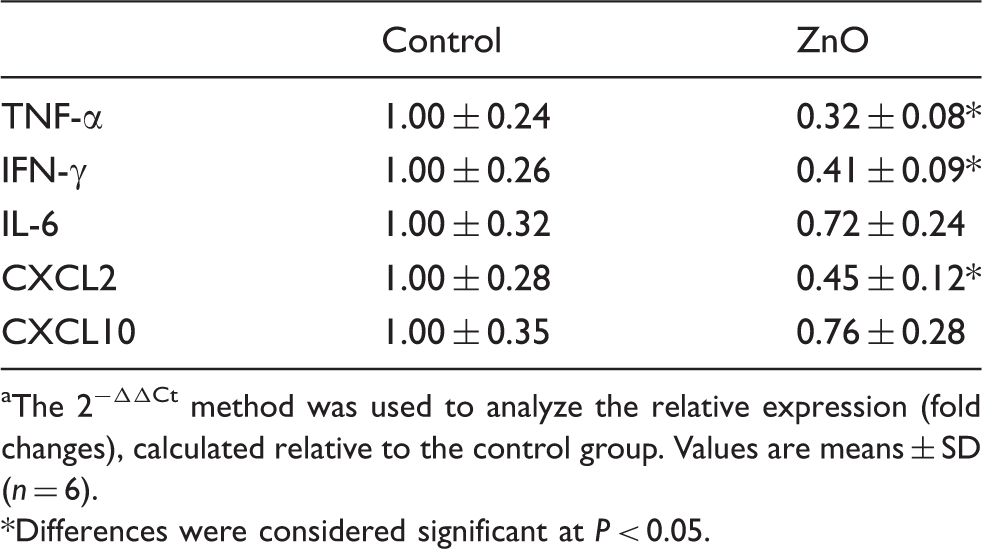
The 2−ΔΔCt method was used to analyze the relative expression (fold changes), calculated relative to the control group. Values are means ± SD (n = 6).
Differences were considered significant at P < 0.05.
Intestinal mRNA expressions of TLR4 and NOD and their downstream signals
Effects of ZnO on mRNA expressions of TLR4 and NOD and their downstream signals in weanling pigs. a
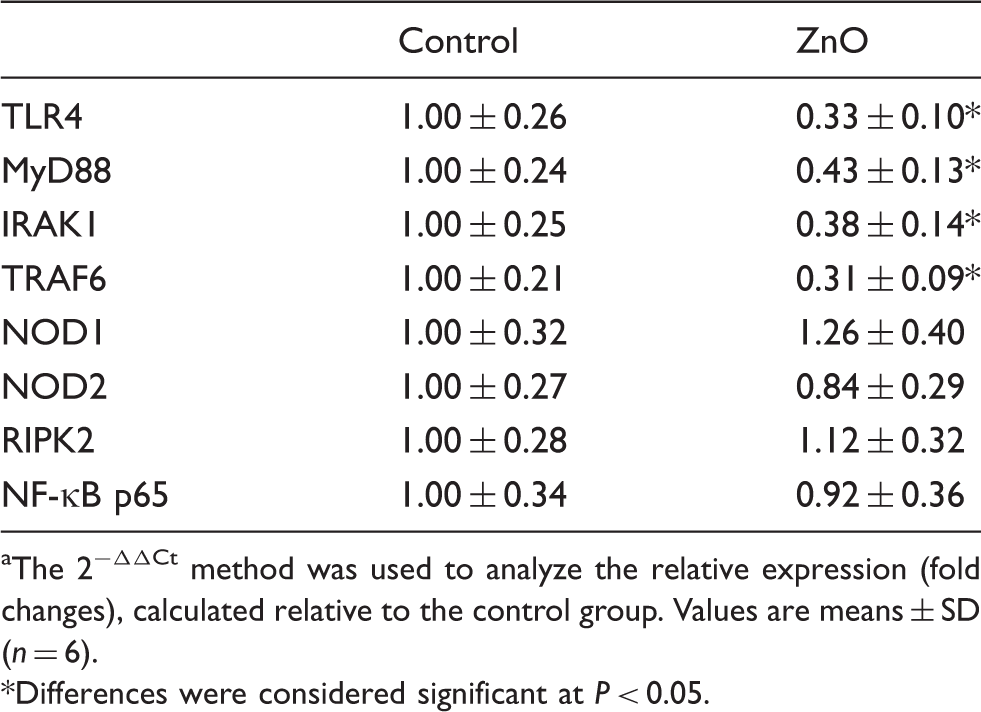
The 2−ΔΔCt method was used to analyze the relative expression (fold changes), calculated relative to the control group. Values are means ± SD (n = 6).
Differences were considered significant at P < 0.05.
Intestinal protein expressions of HSP 70, caspase-3 and NF-κB p65
The effects of ZnO on HSP 70, caspase-3 and NF-κB p65 protein expressions in intestinal mucosa of piglets are shown in Figure 1. Compared with the control, dietary addition of 2200 mg Zn/kg from ZnO decreased (P < 0.05) NF-κB p65 protein expression in jejunal mucosa. However, there was no difference (P > 0.05) for jejunal caspase-3 and HSP 70 protein expressions between the two groups.
Effects of ZnO on caspase-3, HSP 70 and NF-κB p65 protein expressions in jejunal mucosa of weanling piglets. The bands are representative blots from one of six pigs. The relative protein expression was expressed as the ratio of the target protein and β-actin. Data are means ± SD (n = 6). *Differences were considered significant at P < 0.05.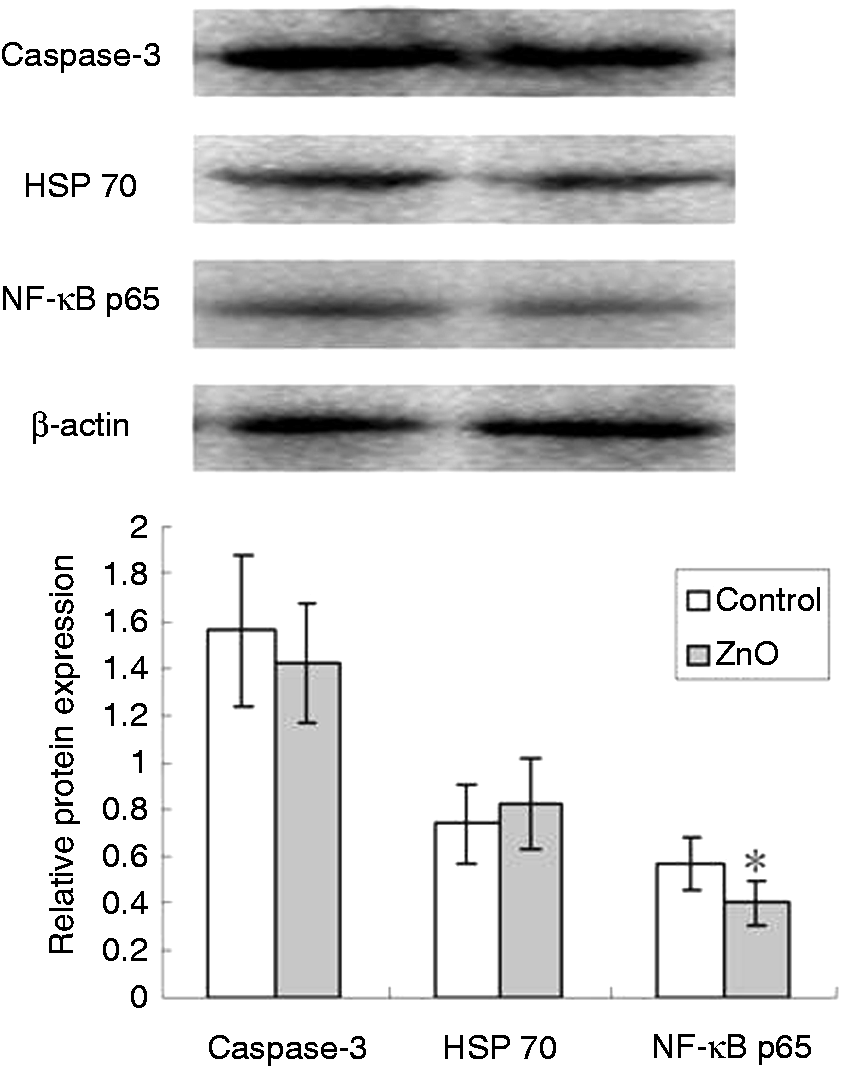
Discussion
Weaning piglets are abruptly forced to combine stressors, such as removal from sow and littermates, transportation to a new environment and abrupt changes in diet. 4 Growth retardation, intestinal disorders and post-weaning diarrhea are common problems in weaned piglets.1–4 Barnett et al. 22 reported that most pigs began scouring 4–5 d post-weaning; scouring peaked at d 8 post-weaning and returned to normal after d 15 post-weaning. A large body of evidence has demonstrated that supplementing nursery pigs with 2000–4000 mg Zn/kg from ZnO alleviates post-weaning diarrhea and improves growth performance.1,9,10,13 As previously reported, the present study showed that dietary addition of 2200 mg Zn/kg from ZnO decreased post-weaning scour scores, and increased daily gain and feed intake in newly weaned pigs.
Compromising alterations in villus crypt structure, such as villus atrophy and crypt hyperplasia, are generally seen in newly weaned piglets.4,5 The minimal value of villous height:crypt depth ratio occurs at about 6 d after weaning. 5 In the present study, the shorter villus and deeper crypt observed in the control treatment verified the weanling-induced intestinal structural injury. Our data indicating that ZnO supplementation increased the villus height:crypt depth ratio at the jejunal mucosa is consistent with the previous reports.9,13
Apart from the function of the digestion and absorption of nutrients, the small-intestinal epithelium serves as a barrier against noxious antigens and pathogens. 3 Impaired intestinal barrier function may promote the translocation of bacteria, and the entering of allergenic compounds from the gut into the body. 3 This results in immunologic responses and an increased susceptibility to infections. 3 A large body of evidence has demonstrated that the small-intestinal barrier and absorptive functions deteriorate within a short time of weaning.1–4 Using either the Ussing chamber technique or orally administered marker probes, the paracellular barrier function is reported to be decreased when pigs are weaned at 3 wks of age or earlier.1–4 Plasma DAO has been suggested as a quantitative and sensitive circulating marker for monitoring the extent of intestinal mucosal injury.13,23 DAO is found exclusively in the villi of the small intestine. When intestinal mucosal integrity is damaged, an increased amount of DAO is released into blood.13,23 Considerable research has shown that Zn played a role in maintaining epithelial barrier integrity and function.9–13 In the present study, ZnO supplementation improved the intestinal barrier, manifested by decreased plasma DAO activity in weaned pigs, which was consistent with previous reports. 13 Endotoxin (LPS), which is part of the cell wall of Gram-negative bacteria, is released into the circulation when the bacteria overgrow or the intestinal barrier is destroyed. 17 It is widely accepted that endotoxin is closely related to the development of post-weaning diarrhea. 5 Our observations that ZnO supplementation decreased plasma endotoxin concentration supported the improved intestinal barrier.
Weaning-associated intestinal inflammation occurs in various animal species. 8 In mice, weaning induced an increase in the number of specific cytokine-secreting intestinal lymphocytes, and changed the distribution of lymphocyte subsets in the small intestine.24,25 In rats, weaning was associated with increased IL-1 expression in the intestine. 26 In piglets, weaning anorexia contributed to local inflammation in the small intestine. 7 The intestinal expression of pro-inflammatory cytokines was up-regulated by weaning in the piglets.4,8 Cytokines play an important role in the immune and inflammatory responses.4,8 They also participate in the regulation of the intestinal barrier integrity.4,27 Researches in vitro and in vivo show that over-production of pro-inflammatory cytokines has negative effects on intestinal integrity and epithelial function.4,18,27 Most pro-inflammatory cytokines, such as TNF-α, IFN-γ and IL-6, induce a pathologic opening of the intestinal tight junction and increase intestinal epithelial permeability.4,9,27 The barrier disruptive actions of TNF-α and IFN-γ have been well-established.28,29 TNF-α can act synergistically with INF-γ to induce disruption in tight junctions.9,27 Controlling the release of intestinal pro-inflammatory cytokines may alleviate intestinal disorders induced by weaning.4,8 In vitro, ZnO reduced inflammatory response of porcine epithelial IPEC J2 cells to enterotoxigenic Escherichia coli infection, 30 and counteracted the up-regulated the expression of the inflammatory IL-8 and TNF-α mRNA levels caused by E. coli K88 in Caco-2 enterocytes. 12 In the present study, ZnO supplementation decreased intestinal mRNA expressions of pro-inflammatory cytokines and chemokines in piglets, indicating that the weaning-induced inflammation was minimized by ZnO supplementation. Our observations were supported by Sargeant et al., 14 who reported that the chemokine CXCL2 was down-regulated in ZnO-treated piglets by transcriptome analysis. Moeser et al. 31 reported that mucosal mast cell activation was a critical event mediating the intestinal barrier dysfunction in early-weaned pigs. Mast cell activation leads to degranulation and release of several mediators, such as histamine, cytokines and chemokines, which injure the intestinal structure and increase intestinal permeability. 32 The stem cell factor is a chemotactic molecule for mast cells and induces their degranulation. 33 Ou et al. 33 reported that ZnO supplementation inhibits the expression of stem cell factor in small intestine, leading to reductions in the number of mast cells and histamine release. It could be suggested that ZnO improves intestinal integrity partially by inhibiting the expression of genes associated with inflammation in weaned pigs.
To elucidate the mechanisms by which ZnO alleviates the intestinal inflammatory response, we examined the role of TLR and NLR signaling pathways. TLRs and NODs are two major forms of innate immune sensors, which play a crucial role in the activation of the innate system as a result of the recognition of PAMPs in the gut. 34 Of them, TLRs are membrane-anchored proteins, whereas NODs are cytoplasmic proteins. 15 TLRs activate immune responses by sensing microbial structures, such as bacterial LPS, viral RNA and endogenous ‘danger’ molecules released by damaged host cells. 35 Among the TLRs family, TLR4 is responsible for recognizing endotoxin (LPS) from Gram-negative bacteria. Among the NLRs family, NOD1 and NOD2 possess the ability to connect with LPS and peptidoglycan, and to transduce a TLRs-independent signal.18,34 MyD88 is an adapter protein that mediates signal transduction for most TLRs, and leads to activation of NF-κB and production of pro-inflammatory cytokines and chemokines. 35 Interaction of PAMPs with TLRs or some NLRs triggers multiple downstream intracellular signals that lead to the activation of NF-κB, which then induces activation of inflammatory genes. 34 The TLR4–Myd88 signaling pathway has been shown to be involved in intestinal inflammation.18,19,34 Juul-Madse et al. 36 reported that TLR4 + cells were markedly enhanced by weaning in piglets. To our knowledge, the research on ZnO regulating the TLR and NLR signaling pathways is very limited. In the present study, we demonstrated, for the first time, that the intestinal mRNA levels of TLR4 and its downstream signals, including MyD88, IRAK1 and TRAF6, were decreased, and the expressions of intestinal pro-inflammatory cytokines and chemokines were decreased simultaneously in the ZnO-supplemented piglets. Although NF-κB p65 mRNA abundance was not affected by ZnO supplementation, NF-κB p65 protein expression was down-regulated by ZnO. NLRs transmit a TLR-independent signal, which also leads to NF-κB activation via the adaptor molecule RIPK2, and stimulates the synthesis of pro-inflammatory mediators.18,34 However, we found that ZnO supplementation had no effect on intestinal expressions of NOD1 and NOD2, or their adaptor molecule RIPK2. The results indicated that the protective effects of ZnO on intestinal integrity were closely related to decreasing the expressions of genes associated with inflammation through inhibiting the TLR4–MyD88 signaling pathways.
It has been suggested that the successful prophylactic use of ZnO in preventing diarrhea may be owing to its antibacterial activity. 1 In vitro studies show that ZnO inhibits the growth of Staphylococcus aureus and E. coli. 1 In piglets, high doses of ZnO reduced bacterial translocation from the small intestine to the mesenteric lymph node, and decreased the viable count of E. coli and Clostridium spp. in the intestine.10,13 ZnO protected cells from enterotoxigenic E. coli (ETEC)-induced damage by inhibiting bacterial adhesion and internalization. 12 It has been reported that weaning alters intestinal microbiota and increases the E. coli:Lactobacilli ratio.37,38 Weaning removes young pigs from passive immune protection from the milk of the sow and increases their susceptibility to ETEC infection. 32 The Gram-negative bacterium E. coli is considered to be one of the main causes of post-weaning diarrhea in pigs. 36 TLR4 recognizes endotoxin from Gram-negative bacteria. 34 In the present study, ZnO supplementation decreased plasma endotoxin concentration. The influences of ZnO on TLR4–MyD88 signaling pathway in weanling pigs might be related to the altered intestinal microbiota and decreased endotoxin concentration. Moreover, it was reported that ZnO supplementation altered ETEC gene expression of factors involved in the initiation of an immune response, such as LPS or toxin. 30 The production of several bacterial toxins, including Shiga and Diptheria toxins, is regulated by Zn concentration. 30
HSPs play a central role in the protection of cells, tissues or organs subjected to various types of stressors. 39 HSP 70 protects against apoptosis, endotoxins, reactive oxygen species, radiation and ischemia. 39 It was reported that HSP acted as inhibitors of NF-κB-mediated cytokine expression. 30 There is some evidence to suggest that the reduced inflammatory response with Zn treatment is via the increased expression of stress response proteins. 30 Klosterhalfen et al. 40 found that treatment with Zn2+ induced expression of HSP70, and reduced the plasma TNF-α, IL-1β and IL-6 response to LPS in pigs. Van Molle et al. 41 reported that ZnSO4-treated mice were protected from TNF-induced inflammation, and ZnSO4 treatment induced HSP70 expression in murine small intestine. However, unlike previous findings, the present study showed that ZnO supplementation had no effect on intestinal caspase-3 and HSP 70 protein expressions. David et al. 39 found that weaning modulated the expression of HSP in the intestinal tract of pigs according to spatial-temporal patterns. The expression of HSP 70 was transiently increased between 24 and 48 h post-weaning in the mid-jejunum. 39 The discrepancy regarding the effects of ZnO on stress response proteins might be related to their spatial-temporal patterns.
In summary, dietary addition of 2200 mg Zn/kg from ZnO ameliorated weaning-associated growth retardation and intestinal injury. The improved intestinal barrier in pigs fed ZnO was accompanied by reduced intestinal mRNA expression of TLR4, MyD88, IRAKs and TRAF6, as well as reduced intestinal protein expression of NF-kB p65, leading to reduced intestinal pro-inflammatory cytokines and chemokines. The results indicated that the protective effects of ZnO on intestinal integrity were closely related to decreasing the expressions of genes associated with inflammation through inhibiting the TLR4–MyD88 signaling pathways.
Footnotes
Funding
This research was jointly supported by Zhejiang Provincial Qianjiang Talent project of China (No. 2013R10036), National Natural Science Foundation of China (No. 31072039), the “948” Project from the Ministry of Agriculture (No. 2011-G7), and program for Changjiang Scholars and Innovative Research Team in University (No. IRT1040).
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
