Abstract
Stress causes intestinal inflammation and barrier dysfunction. Corticotrophin-releasing factor (CRF)/CRF receptor (CRFR) signaling pathway has been shown to be important for stress-induced intestinal mucosal alteration.
Keywords
Introduction
Intestinal mucosa plays a vital role in the digestion and absorption of nutrients. It also provides a physical barrier to prevent the diffusion of pathogens, toxins and allergens from the external environment into tissues. 1 Dysfunction of intestinal mucosal barrier may lead to an increase in intestinal mucosal permeability, bacterial translocation and mucosal inflammation. 2
Various types of stresses, such as nutrient and growth factor deprivation, hemorrhagic shock, pathogenic bacteria toxins (e.g. endotoxin) and psychological stress, can cause damage to the intestinal barrier. 3 The precise mechanisms of stress-related disturbances in intestinal barrier have not been fully elucidated. They involve complex brain–gut interactions.4,5 The hypothalamus–pituitary–adrenal (HPA) has been extensively studied in the maintenance of stress-related behavioral mood change (anxiety and depression) and gastrointestinal parameters. 6 Several studies have demonstrated that corticotrophin-releasing factor (CRF) and adrenal glucocorticoid play a critical role in stress-induced intestinal disorders.7,8 Moreover, CRF mediates its effects via mast cell activation. 6 Activated mast cells release a variety of pro-inflammatory mediators such as prostaglandins, cytokines, tryptase and histamine, which influence intestinal barrier function. 7
Nutrients such as amino acids and fatty acids have been proposed as regulators to restore the intestinal barrier.9,10 It is well known that
LPS, a component of the cell wall of Gram-negative bacteria, is one of the most commonly used endotoxins in immunological challenge and intestinal damage in weanling piglets. 14 Peripheral administration of LPS can activate the HPA axis. 15 Furthermore, a well-characterized response to stress is activation of the HPA axis resulting in adrenal cortisol release. 16 Therefore, LPS is also a common tool used for inducing acute stress response and studying the effects of dietary regimes. 17
In this study, we hypothesized that ASN could maintain intestinal barrier function by regulating CRF/CRF receptor (CRFR) signaling pathway. We used a well-established piglet model to induce intestinal damage in weanling piglets by administering Escherichia coli LPS. The piglet model was considered one of the best animal models for studying nutritional and intestinal development because of their similarities to human in anatomy and digestive physiology. 18 Our objective was to determine whether dietary ASN supplementation could alleviate the damage of intestinal barrier function caused by LPS through modulation of CRF/CRFR signaling pathway.
Material and methods
Animals, diets, and experimental design
All procedures involving animal use and care were approved by the Animal Care and Use Committee of Wuhan Polytechnic University.
A total of 24 crossbred barrows [Duroc × Large White × Landrace; initial body mass (BM) of 7.37 ± 0.04 kg, weaned at 21 ± 1 d of age] were used in this study. Pigs were randomly allotted to four treatment groups with six replicates (pen) of one pig per replicate. The treatments were as follows: (1) non-challenged control (CONTR; pigs fed a control diet and injected with sterile saline); (2) LPS-challenged control [LPS, pigs fed control diet and challenged by injection with E. coli LPS, (E. coli serotype O55: B5; Sigma Chemical, St. Louis, MO, USA)]; (3) LPS + 0.5% ASN treatment (pigs fed a 0.5% ASN diet and challenged by injection with E. coli LPS); (4) LPS + 1.0% ASN treatment (pigs fed a 1.0% ASN diet and challenged by injection with E. coli LPS). The doses of ASN (
Ingredient composition of diets (as fed basis).
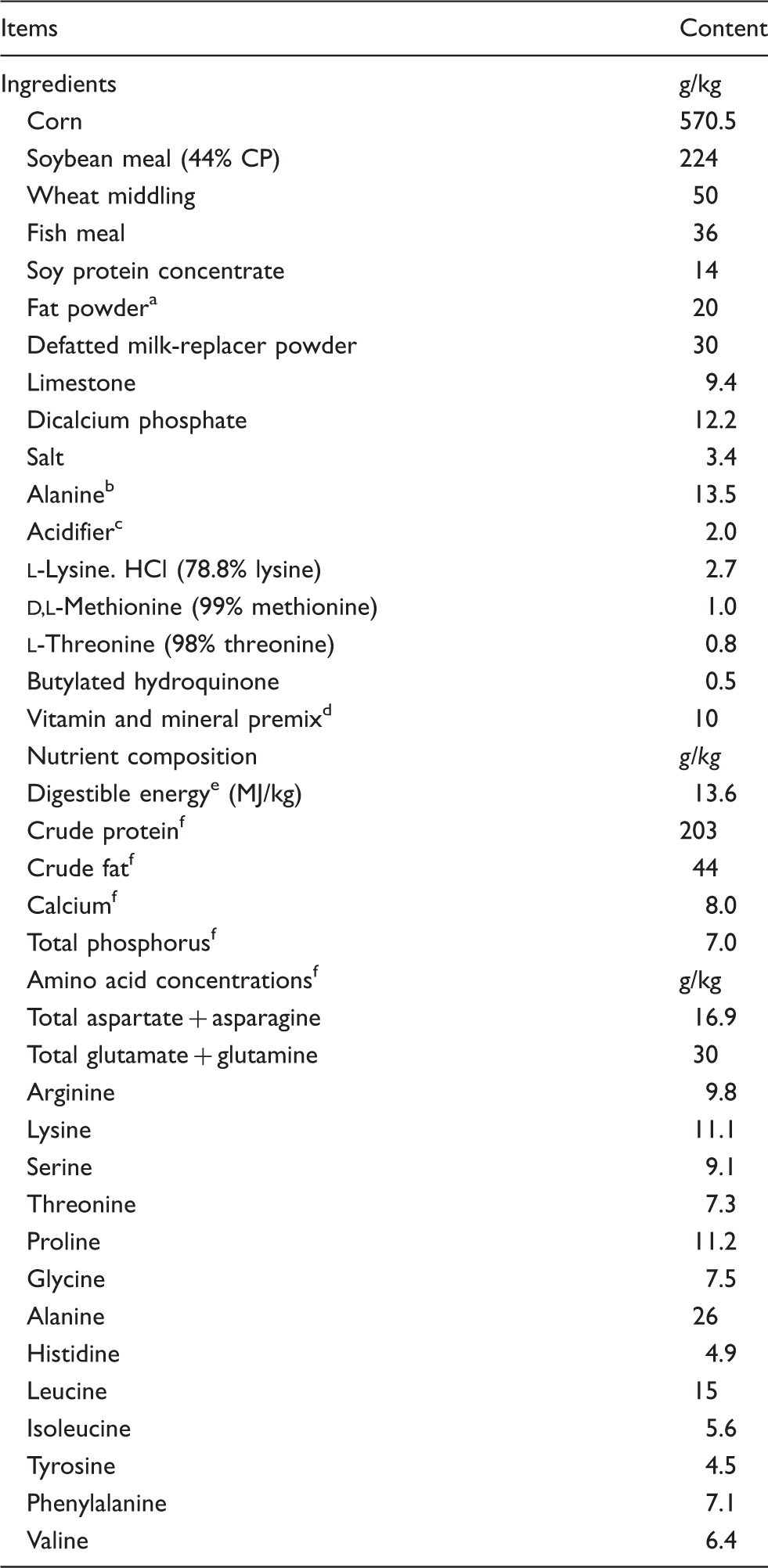
A rumen-stable fat powder, purchased from Berg + Schmidt (Hamburg, Germany). bIn the 0.5% ASN diet, 1.35% alanine was replaced with 0.5% ASN, 0.68% alanine and 0.17% cornstarch. In the 1.0% ASN diet, 1.35% alanine was replaced with 1.0% ASN and 0.35% cornstarch. All diets were isonitrogenous. cA compound acidifier including lactic acid and phosphoric acid, provided by Wuhan Fanhua Biotechnology Company (Wuhan, China). dThe vitamin and mineral premix provided the following amounts per kg of complete diet: retinol acetate, 2700 µg; cholecalciferol, 62.5 µg; dl-α-tocopheryl acetate, 20 mg; menadione, 3 mg; vitamin B12, 18 µg; riboflavin, 4 mg; niacin, 40 mg; pantothenic acid, 15 mg; choline chloride, 400 mg; folic acid, 700 µg; thiamin, 1.5 mg; pyridoxine, 3 mg; biotin, 100 µg; Zn, 80 mg (ZnSO4·7H2O); Mn, 20 mg (MnSO4·5H2O); Fe, 83 mg (FeSO4·H2O); Cu, 25 mg (CuSO4·5H2O); I, 0.48 mg (KI); Se, 0.36 mg (Na2SeO3·5H2O). eCalculated. fAnalyzed.
On d 19 of the trial, the challenged groups were injected i.p. with E. coli LPS at 100 µg/kg BM and the unchallenged group was injected with the same amount of 0.9% (wt/vol) saline. The LPS was dissolved in sterile 0.9% saline. The dose of LPS was selected in accordance with our previous experiment. 21
Sample collection
Four h after administration of LPS or sterile solution, all pigs were anesthetized by injection of sodium pentobarbital (80 mg/kg BM). The abdomen was opened through a midline incision and mesenteric lymph nodes (MLN), liver and spleen were harvested for determination of bacterial translocation. The jejunum and ileum were separated and flushed with ice-cold 0.9% NaCl solution. A 1–3-cm-long cross section of intestinal tissue was obtained from the mid-jejunum and mid-ileum, and placed in 4% paraformaldehyde in PBS for histological analysis. About 12 cm middle portions of jejunum and ileum were cut longitudinally and washed with ice-cold PBS. Mucosal samples were gently scraped with sterile glass slides from the connective tissue, immediately frozen in liquid nitrogen and then stored at −80℃ until further analysis.
Bacterial translocation
Bacterial translocation, which is associated with the failure of intestinal barrier function, has been described in detail previously. 22 Briefly, the collected MLN, spleen and liver were separately weighed and homogenized in 10 volumes of ice-cold sterile 0.9% saline. Fifty μl of tissue homogenates from different organs were uniformly smeared onto blood and MacConkey agar plates. The plates were examined after 48 h of incubation at 37℃ under aerobic conditions. The colony counting was performed in a blind manner. The viable bacterial counts were expressed as log 10 colony-forming units (CFU)/g of tissue mass. For each tissue, bacterial translocation was considered positive when the CFU was >100 per g of tissue.
Intestinal morphology
Fixed intestinal samples were dehydrated, cleared and embedded in paraffin. Three cross-sections (4 µm thick) for each intestinal sample were stained with hematoxylin and eosin. For quantification of mast cells, intestinal sections were stained with toluidine blue (pH 4.0). A total of 10 intact, well-oriented villi and their associated crypts from each segment were selected to determine the villus height and crypt depth. Villus:crypt ratio was calculated by dividing villus height by crypt depth. The same villus and crypt were used to measure the number of intraepithelial lymphocytes (IEL), goblet cells and intravillus lamina propria cell density. These variables were expressed per 100 enterocytes (IEL and goblet cells) and number of cells/mm2 (intravillus lamina propria cell density).
On basis of cellular and nuclear morphology and toluidine blue staining (for mast cell), the numbers of neutrophils and mast cells were counted at 40× magnification. Cell counts were conducted on six different fields per slides and six slides per treatment, and expressed as number of cells/mm2. All cell counts and intestinal morphologic measurements were performed by a blinded reviewer.
Western blot analysis
Briefly, the intestinal tissues (100–150 mg, n = 6) were homogenized and lysed in chilled lysis buffer. The homogenates were centrifuged at 12,000 g for 15 min at 4℃, and the supernatant was collected for Western blot and protein assay. Protein concentration was determined using the bicinchoninic acid protein assay kit (Applygen Technologies, Beijing, China). Intestinal mucosa proteins were separated by PAGE and transferred to polyvinylidene difluoride membranes. Membranes were blocked for non-specific binding for 1 h (3% BSA in TBS–Tween-20 buffer) at room temperature (21–25℃), and incubated overnight (12–16 h) at 4℃ with rabbit anti-human claudin-1 Ab [1:1000; #51-9000 (Invitrogen Technology, Danvers, MA, USAame dietary fat level (5% corn oil vs, 5%)] or mouse anti-β-actin Ab [monoclonal; 1:10000; #A2228 (Sigma Aldrich, St. Louis, MO, USA)]. The membranes were subsequently incubated with goat anti-rabbit (#ANT020) or mouse (#ANT019) immunoglobulin G HRP-conjugated secondary Ab (1:5000; AntGene Biotech, Wuhan, China) for 120 min at room temperature. Chemiluminescence signal was detected with enhanced Chemiluminescence Western blotting kit (Amersham Biosciences, Uppsala, Sweden), visualized using a Gene Genome bioimaging system. The bands were quantified by densitometric analysis using GeneTools software (Syngene, Frederick, MD, USA). The result was expressed as the abundance of claudin-1 protein to β-actin protein.
mRNA expression analysis by real-time PCR
Primer sequences used for real-time PCR.

Statistical analyses
Statistical analysis was carried out by ANOVA using the General Linear Model procedure in SAS (SAS Institute, Cary, NC, USA). Pig was the experimental unit. A post hoc LSD test was used to determine differences between treatments (CONTR vs. LPS, LPS vs. LPS + 0.5% ASN and LPS vs. LPS + 1.0% ASN) following ANOVA. Bacterial translocation (positive animals) data were analyzed by the one-way ANOVA and the Fisher’s exact test. Results were considered significantly different if P < 0.05. Instances in which 0.05 < P ≤ 0.10 were discussed as trends.
Results
Bacterial translocation
Effects of ASN supplementation on the incidences of bacterial translocation in weaned pigs challenged by E. coli LPS. a
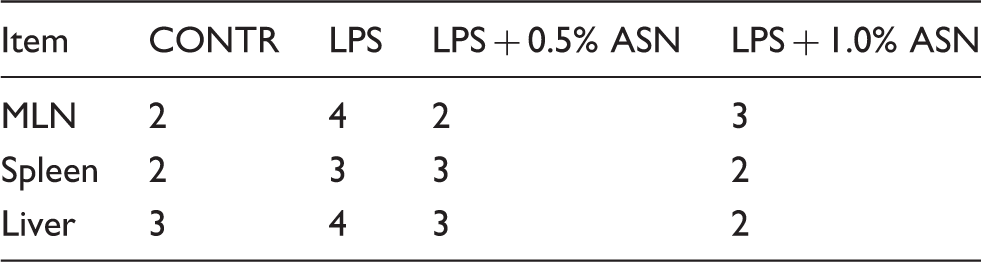
For each tissue, bacterial translocation was considered positive when the CFU were greater than 100 per g of tissue. n = 6 (one pig/pen). CONTR (non-challenged control): pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control): pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% ASN: pigs fed a 0.5% ASN diet and challenged with LPS; LPS + 1.0% ASN: pigs fed a 1.0% ASN diet and challenged with LPS.
Effects of ASN supplementation on translocation microorganisms of weaned pigs challenge by E. coli LPS. a
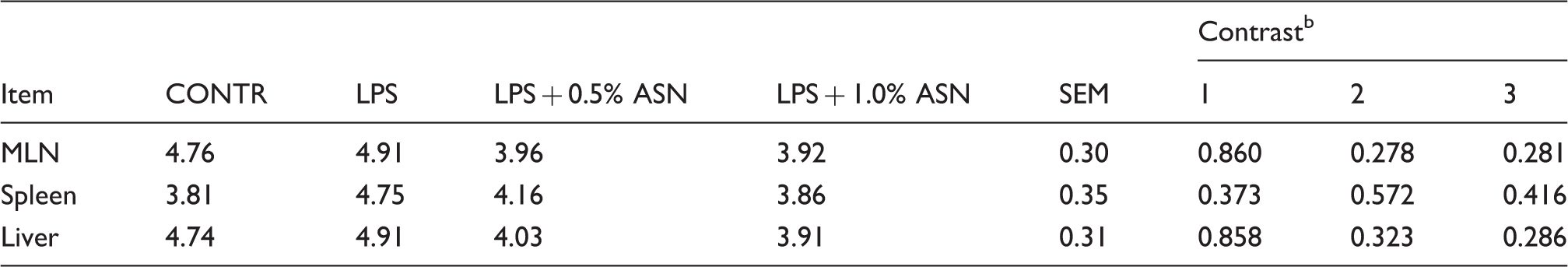
Values are means and combined SEM, n = 6 (1 pig/pen). The values were expressed in log10 (CFU)/g of the organ’s mass. CONTR (non-challenged control): pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control): pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% ASN: pigs fed a 0.5% ASN diet and challenged with LPS; LPS + 1.0% ASN: pigs fed a 1.0% ASN diet and challenged with LPS. bContrast: (1) CONTR vs. LPS; (2) LPS vs. LPS + 0.5% ASN; (3) LPS vs. LPS + 1.0% ASN.
Intestinal histology
The mucosal structure of intestine was analyzed by light microscopy. The intestinal mucosa in CONTR appeared intact and was in good condition (Figure 1a). Pigs challenged with LPS exhibited intestinal mucosal damage, including lifting of epithelium at the tip of the villus and villous atrophy (Figure 1b). ASN supplementation mitigated intestinal mucosal injury caused by LPS (Figure 1c, d). Pigs challenged with LPS had lower villus height in jejunum (P = 0.045) and ileum (P = 0.048) than those injected with saline (Table 5). Pigs fed ASN diets had an increased villus height in jejunum (1% ASN; P = 0.006) and ileum (0.5% ASN; P = 0.001) vs. pigs challenged with LPS. However, pigs challenged with LPS had lower crypt depth in ileum (P = 0.003) than pigs in the CONTR group. Supplementation of 0.5% ASN increased crypt depth in ileum (P < 0.001) vs. LPS-challenged pigs. Furthermore, pigs challenged with LPS had lower villus height/crypt depth in jejunum (P = 0.014) and ileum (P = 0.029) than pigs in the CONTR group. Supplementation of 1.0% ASN tended to increase villus height/crypt depth in jejunum (P = 0.093) and ileum (P = 0.062) compared with LPS-challenged pigs
Intestinal mucosal histological appearance of jejunum (hematoxylin and eosin). (a) Pigs fed a control diet and injected with sterile saline. No obvious damage was found. (b) Pigs fed control diet and injected with LPS. Intestinal mucosa was seriously damaged by LPS. Arrows represent the lifting of epithelium at the tip of the villus (a) and villous atrophy (b). (c) Pigs fed a 0.5% ASN diet and challenged with LPS. Intestinal mucosal damage was alleviated. (d) Pigs fed a 1.0% ASN diet and challenged with LPS. Intestinal mucosal damage was alleviated. Original magnification 100×. Scale bars = 82.6 µm. Effects of ASN supplementation on the intestinal morphology and tight junction protein expression in weaned pigs challenged by E. coli LPS.
a
Values are means and combined SEM, n = 6 (one pig/pen). CONTR (non-challenged control): pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control): pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% ASN: pigs fed a 0.5% ASN diet and challenged with LPS; LPS + 1.0% ASN: pigs fed a 1.0% ASN diet and challenged with LPS. bContrast: (1) CONTR vs. LPS; (2) LPS vs. LPS + 0.5% ASN; (3) LPS vs. LPS + 1.0% ASN.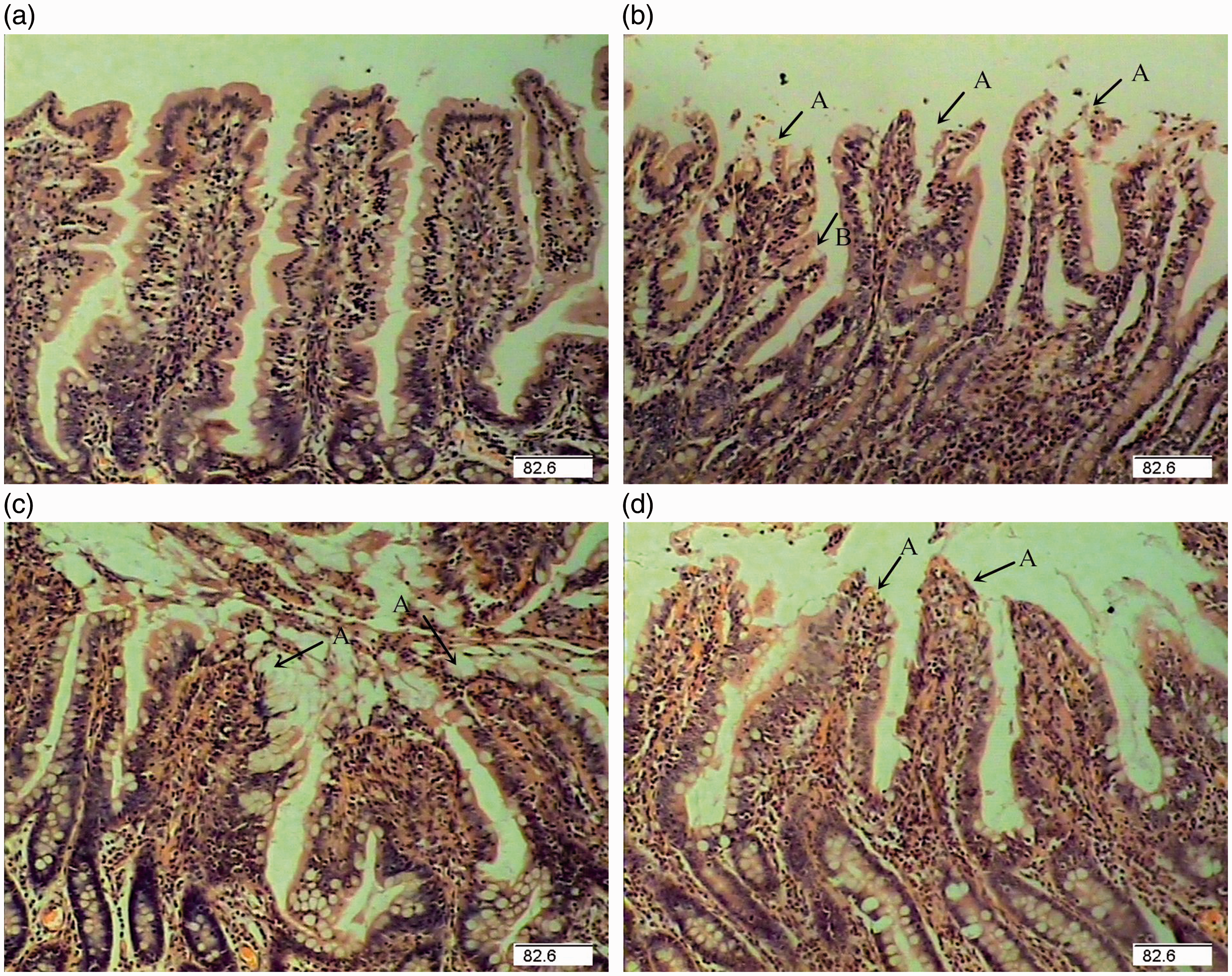
Intestinal tight junction protein claudin-1 expression
Western blotting analysis showed that pigs challenged with LPS had lower claudin-1 expression in the jejunal mucosa than pigs in CONTR group (P = 0.015) (Figure 2). However, 0.5% ASN supplementation increased claudin-1 expression in jejunal mucosa (P = 0.006) vs. LPS pigs. Neither LPS nor ASN supplementation affected claudin-1 expression in ileal mucosa.
Tight junction protein expression in the jejunum and ileum. The bands are representative Western blot images of claudin-1 and β-actin. Values are means and pooled SEM, n = 6 (one pig/pen). CONTR (non-challenged control) = pigs fed a control diet and injected with sterile saline (
 ); LPS (LPS challenged control) = pigs fed the same control diet and challenged with E. coli LPS (
); LPS (LPS challenged control) = pigs fed the same control diet and challenged with E. coli LPS ( ); LPS + 0.5% ASN = pigs fed a 0.5% ASN diet and challenged with LPS (
); LPS + 0.5% ASN = pigs fed a 0.5% ASN diet and challenged with LPS (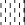 ); LPS + 1.0% ASN = pigs fed a 1.0% ASN diet and challenged with LPS (
); LPS + 1.0% ASN = pigs fed a 1.0% ASN diet and challenged with LPS ( ). Pigs challenged with LPS had lower claudin-1 expression in the jejunal mucosa than pigs in the CONTR group (P = 0.015). Supplementation of 0.5% ASN increased claudin-1 expression in jejunal mucosa (P = 0.006) vs. LPS pigs. Neither LPS nor ASN supplementation affected claudin-1 expression in ileal mucosa.
). Pigs challenged with LPS had lower claudin-1 expression in the jejunal mucosa than pigs in the CONTR group (P = 0.015). Supplementation of 0.5% ASN increased claudin-1 expression in jejunal mucosa (P = 0.006) vs. LPS pigs. Neither LPS nor ASN supplementation affected claudin-1 expression in ileal mucosa.
Intestinal immune cells and lamina propria cells
Effects of ASN supplementation on immune cells and lamina propria cells in the intestine of weaned pigs challenged by E. coli LPS. a
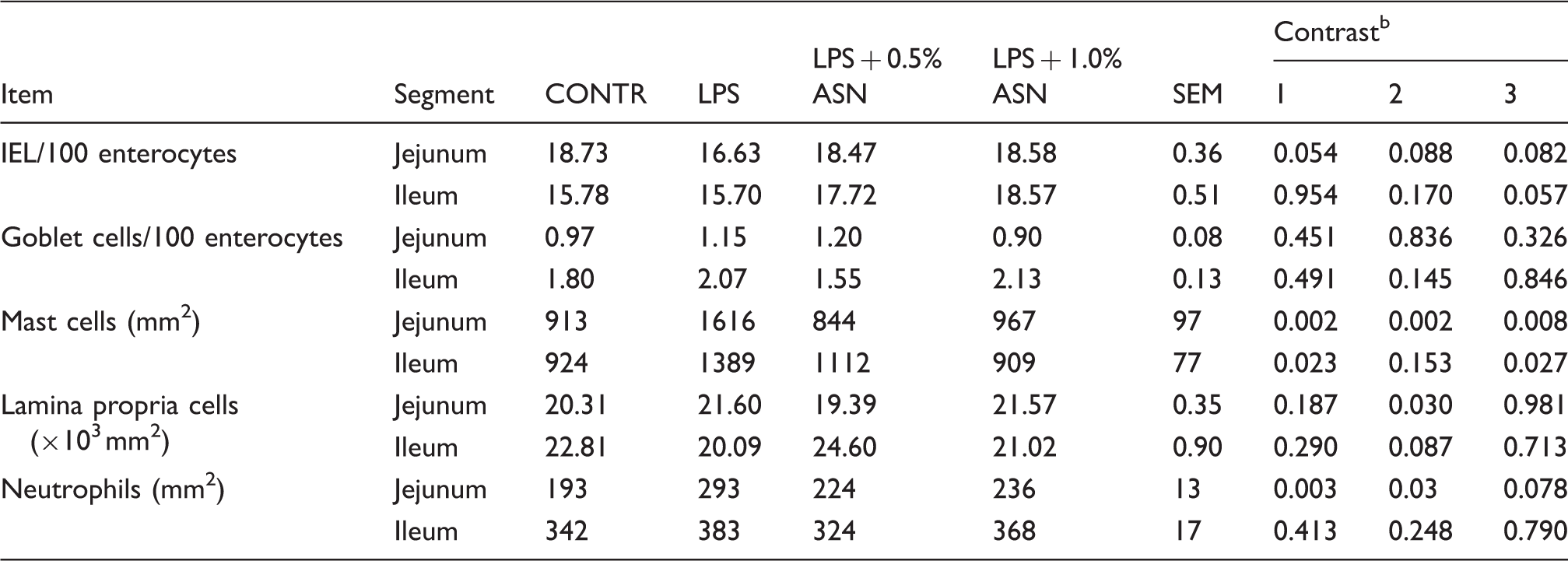
Values are means and combined SEM, n = 6 (one pig/pen). CONTR (non-challenged control): pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control): pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% ASN: pigs fed a 0.5% ASN diet and challenged with LPS; LPS + 1.0% ASN: pigs fed a 1.0% ASN diet and challenged with LPS. bContrast: (1) CONTR vs. LPS; (2) LPS vs. LPS + 0.5% ASN; (3) LPS vs. LPS + 1.0% ASN.
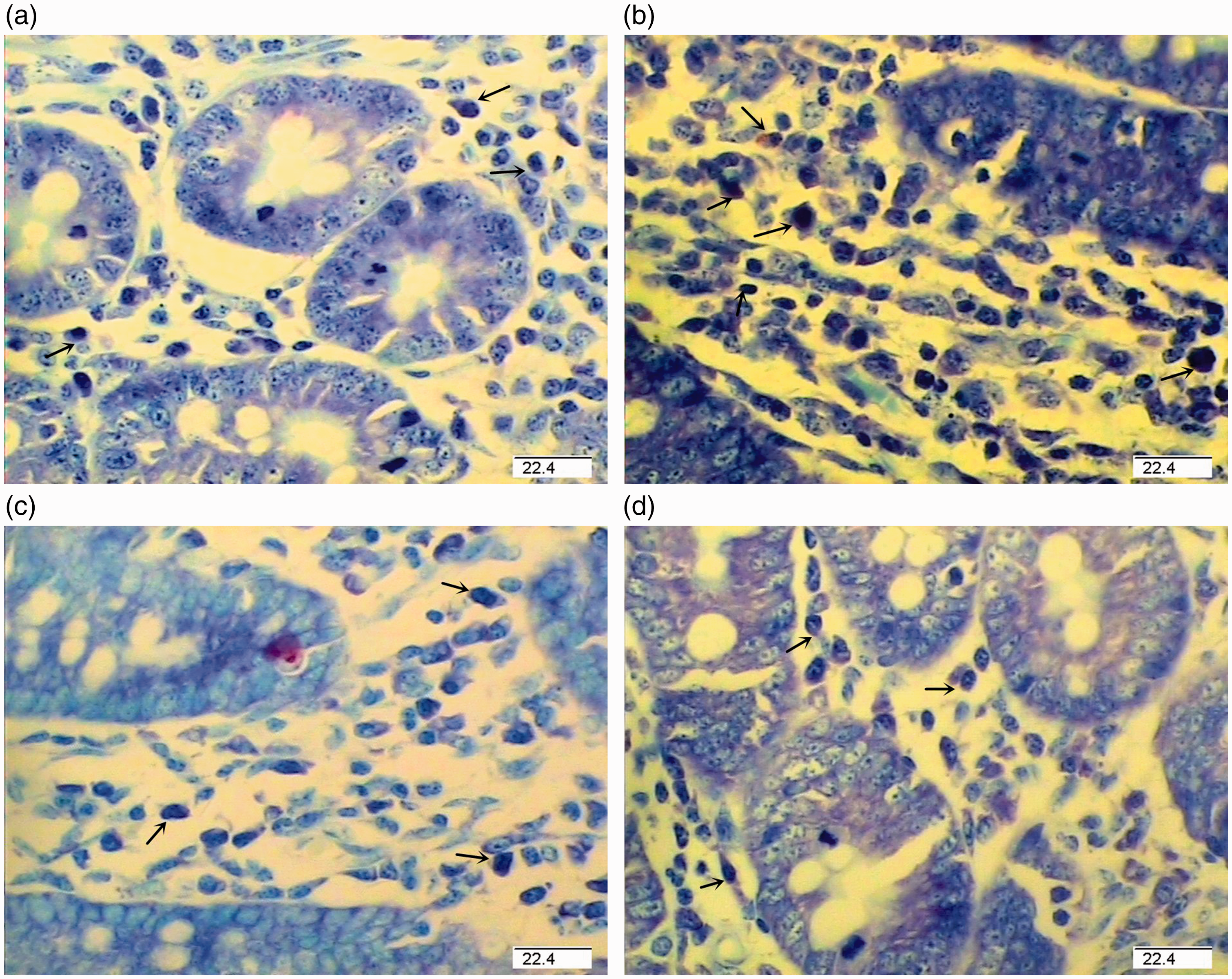
Photomicrographs of pig jejunal mucosa showing mast cells stained with toluidine blue. (a) Pigs fed a control diet and injected with sterile saline. (b) Pigs fed control diet and injected with LPS. (c) Pigs fed a 0.5% ASN diet and challenged with LPS. (d) Pigs fed a 1.0% ASN diet and challenged with LPS. Arrows indicate toluidine blue-positive mast cells. The numbers of mast cell in intestine were higher in LPS pigs (P < 0.05) than in CONTR pigs. Supplementation with 0.5% or 1.0% ASN decreased mast cell numbers compared with LPS pigs (P < 0.05). Original magnifications 400×. Scale bars = 22.4 µm.
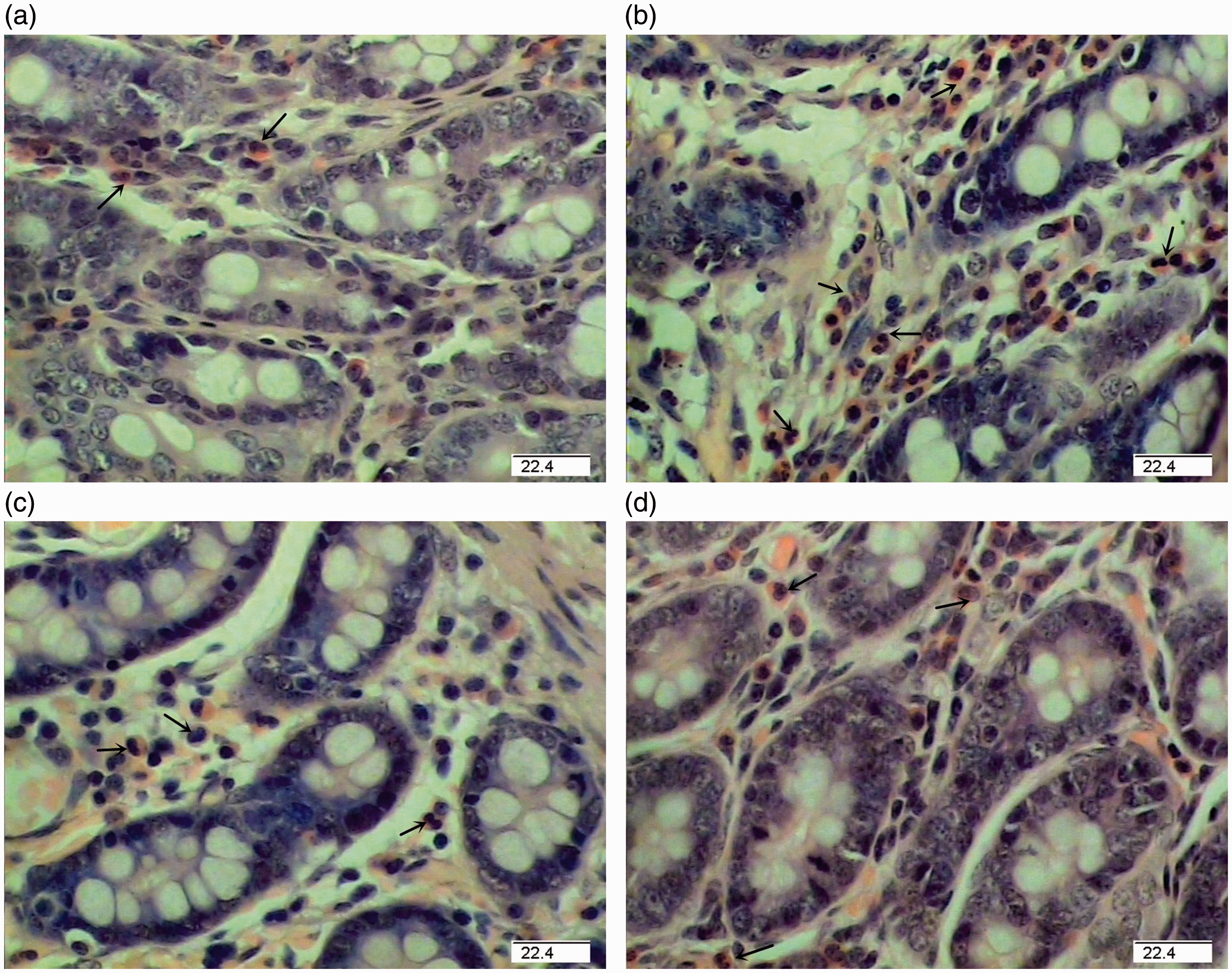
Representative photomicrographs of pig jejunal mucosa showing neutrophils (hematoxylin and eosin). (a) Pigs fed a control diet and injected with sterile saline. (b) Pigs fed control diet and injected with LPS. (c) Pigs fed a 0.5% ASN diet and challenged with LPS. (d) Pigs fed a 1.0% ASN diet and challenged with LPS. Arrows indicate neutrophils. Pig challenged with LPS had higher jejunal neutrophils numbers (P < 0.05) than CONTR pigs. Supplementation with 0.5% ASN decreased jejunal neutrophils numbers (P < 0.05) compared with LPS pigs. Original magnifications 400×. Scale bars = 22.4 µm.
CRFR1, CRF, glucocorticoid receptor and tryptase mRNA expression
Effects of ASN supplementation on mRNA expression of CRF, GR, tryptase and CRFR1 in the intestine of weaned pigs challenged by E. coli LPS. a
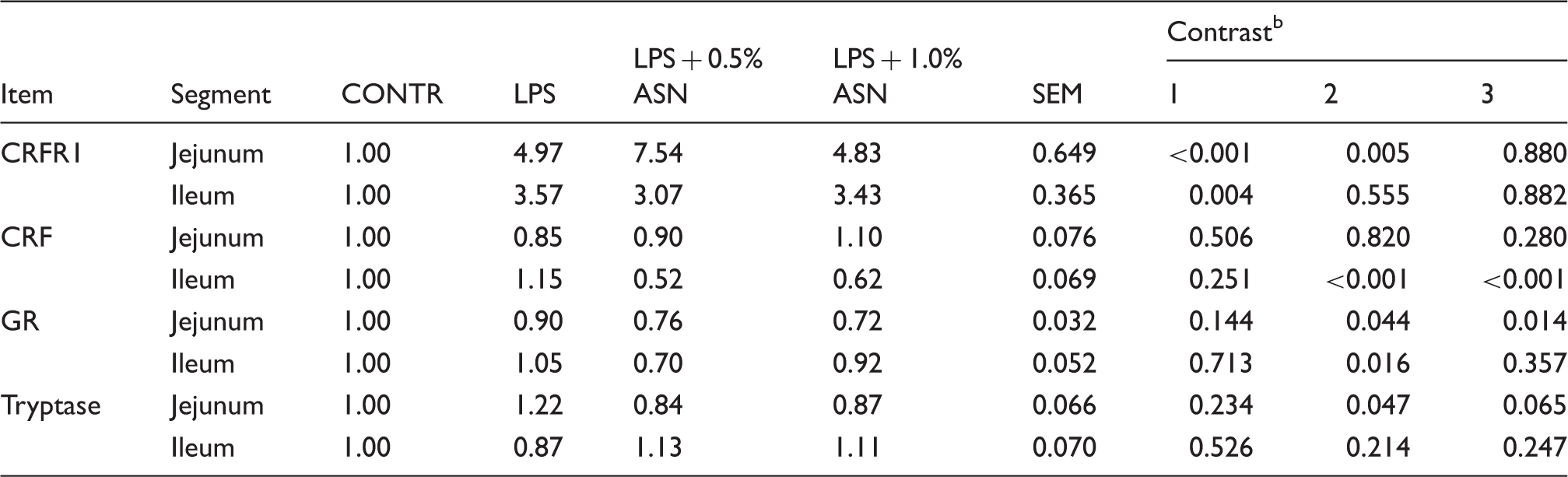
Values are means and combined SEM, n = 6 (one pig/pen). CONTR (non-challenged control): pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control): pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% ASN: pigs fed a 0.5% ASN diet and challenged with LPS; LPS + 1.0% ASN: pigs fed a 1.0% ASN diet and challenged with LPS. bContrast: (1) CONTR vs. LPS; (2) LPS vs. LPS + 0.5% ASN; (3) LPS vs. LPS + 1.0% ASN.
Discussion
Stress, in various forms, such as infection and chronic inflammatory stimulation, has been reported to impair intestinal barrier function. 3 In this study, we used a well-documented pig model to induce stress and intestinal damage by administering E. coli LPS. Our objective was to examine the protective effects and possible mechanisms of ASN on attenuating LPS stress-induced damage on intestinal barrier and function. In this study, we confirmed that LPS caused typical/classical morphologic changes, such as epithelium sloughing and villous atrophy, reduced villus height and villus height to crypt depth ratio. Generally, epithelium lifting and villous atrophy are accompanied by poor nutrient absorption and increased permeability on intestinal mucosa. Therefore, viable bacteria and their products in intestinal lumen pass through the intestinal wall to MLN and more distant sites, a process known as bacterial translocation. 24 Owing to individual animal variation, LPS only resulted in a numerical increase in bacterial translocation incidences and translocation microorganism. ASN supplementation mitigated the intestinal morphologic damage induced by LPS challenge, increased villus height and the ratio of villus height to crypt depth. Intestinal epithelial cell turnover and gut barrier functions are dynamic processes that are markedly affected by nutritional status, the route of feeding and the adequacy of specific nutrients in the diet. 25 Specifically, it needs adequate amounts of ‘fuel’, like amino acids and Glc to maintain and repair intestinal integrity and function under stress conditions. 26 In our study, dietary ASN might be converted to aspartate (ASP) and glutamate for the major fuels of the small intestinal mucosa. Furthermore, ASN is a powerful stimulator of ornithine decarboxylase and cell proliferation in a variety of cell types, including colonic cells. 13 We also found that 0.5% ASN increased the crypt depth compared with the LPS group. A deeper crypt may indicate faster tissue turnover to permit renewal of the villus, 27 and the intestinal response is to try to compensate for lifting or atrophy of villus due to LPS challenge. Therefore, ASN protected and maintained the integrity of intestinal mucosa. It is critical for maintaining gut barrier function.
Tight junctions between intestinal epithelial cells play a critical role in intestinal barrier function. Tight junctions are organized by four types of transmembrane protein, occludin, claudin, tricellulin and junctional adhesion molecules, and many other regulatory proteins.21,28,29 Of them, the valuable and essential ones are occludin, claudins and ZO-1, which participate in tight junction structural integrity. 30 In the present study, LPS reduced the expression of tight junctional protein claudin-1. ASN supplementation enhanced the expression of claudin-1 in jejunum of pigs challenged with LPS. Our results suggested that ASN supplementation might help to repair the tight junction damage of pigs challenged with LPS, and restore the intestinal barrier function.
The intestinal barrier includes physical diffusion barrier, physiological and enzymatic barriers, and immunological barriers. 3 IELs are one of the components of the intestinal immunological barrier and might serve as a first line of defense against intestinal pathogens. 31 It is well known that LPS can cause intestinal barrier dysfunction and induce inflammatory response. 32 In the present study, LPS increased neutrophil infiltration and decreased IEL numbers in the intestine. However, ASN supplementation tended to increase IEL numbers and decrease neutrophil infiltration in jejunum. Currently, there are a few studies on the regulation of IEL through dietary ASN supplementation. Kandil et al. 33 reported that ASN could stimulate proliferation of a porcine jejunal cell line through the stimulation of ornithine decarboxylase. In addition, a previous study in mice showed that the decrease in IEL number was associated with decreasing tight junction integrity in intestine. 34 IELs play protective roles in defense against infection, repair of injury of mucosal barrier and maintenance of the barrier function in the intestine. 35 Our results indicated that ASN maintained intestinal barrier integrity by inducing the proliferation of IELs and alleviating the intestinal inflammatory response.
Although it is known that stress impairs intestinal barrier function, the precise mechanism has not been fully elucidated. Several studies have demonstrated that stress-induced intestinal barrier dysfunction is due to the activation of CRF/CRFR pathway in the intestine.7,8 CRF produced in the central nervous system and peripheral tissue in response to stress has been shown to play a central role in stress-induced intestinal pathophysiology. Wang et al.
36
found that weaning stress disrupted the epithelial barrier and increased mRNA and protein abundance of CRF in jejunum. Overman et al.
7
reported that in vitro CRF exposure to ileal mucosa increased intestinal paracellular permeability. In fact, CRF activity is mediated by activation of seven specific transmembrane G-protein-coupled receptors known as CRFR1 and CRFR2.
37
Moeser et al.
38
showed that weaning-induced intestinal dysfunction was mediated by the activation of peripheral CRFR. Smith et al.
8
demonstrated that CRFR1 likely mediated the damage of intestinal barrier function and hypersecretion in early-weaned pigs. In agreement with these findings, our study showed that LPS increased mRNA abundance of CRFR1 in intestine. In addition, GR is a downstream factor of the CRF/CRFR signaling pathway. Several studies have shown that dysfunction of the intestinal barrier induced by stress might be mediated by GR and adrenal corticosteroids.39,40 However, we found that LPS did not affect mRNA the abundance of CRF and GR. It is a complex process for activation of the CRF/CRFR pathway. It needs more factors to participate. Interestingly, our data showed that ASN supplementation decreased mRNA abundance of CRF in ileum and mRNA abundance of GR in jejunum. Similarly, Erwan et al.
41
reported that central injection of
In recent years, there has been increasing evidence that stress enhances gut mucosal permeability by a mast cell-dependent mechanism.42,43 Our study showed that LPS stress increased mast cell number in intestinal mucosa. Similarly, Zhong et al. 44 found that the number of submucosal mast cells was increased after stress. Studies have shown that the inflammatory infiltrate is dominated by increasing populations of activated mast cells in patients with irritable bowel syndrome.45,46 Activated mast cells release tryptase. 47 Tryptase is the most abundant secretory granule-derived serine proteinase contained in mast cells that leads to the activation of sensory neurons and the regulation of mucosal permeability. 48 In the present study, ASN ameliorated the increase in mast cell number and abundance of tryptase mRNA in jejunum and ileum, which were induced by LPS. It is possible that ASN inhibited the mast cell activation, which resulted in a decrease in mast cell number and abundance of tryptase mRNA in pigs challenged with LPS. The exact mechanisms are not understood. Further investigation is needed.
In conclusion, LPS stress impairs intestinal barrier function, which can be alleviated by ASN. Dietary supplementation with ASN helps maintain intestinal mucosa homeostasis, which is associated, in part, with regulation of the CRF/CRFR1 signaling pathway and mast cell activation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the National Natural Science Foundation of China (31372318 and 31422053) and the State’s Key Project of Research and Development (2016YFD0501210).
