Abstract
We recently reported our findings from a longitudinal, prospective study where we identified 10% infants who were low vaccine responders (LVR) at age 9–12 mo following routine primary series vaccine schedule. We found multiple cellular deficiencies in LVR children, including low number of memory B cells, reduced polyclonal stimulation of naïve/memory T cell response and suboptimal APC response. These children outgrew their poor vaccine response by the time they received booster doses of vaccine. Studies in human infant innate immunity are rare because of the unique challenges in specimen collection. As innate immunity instructs adaptive immunity, we hypothesized that the primary immune defect lies with innate immunity and in this study we sought to determine the ontogeny of innate immune response in LVR children between 6 and 36 mo of age. Interestingly, suboptimal APC response observed in LVR children at 6–9 mo of age characterized by significantly (P < 0.05) low basal MHC II expression, low R848 induced IRF7 fold change, as well as low IFN-α, IL-12p70 and IL-1β levels, came to parity with normal vaccine responders by 12–15 mo of age, suggesting that the observed immune deficiency in LVR children may be the result of delayed maturation of immune system.
Introduction
For the last few years our laboratory has conducted a clinical field study and identified a population of infants who respond with sub-optimal immunity to routine pediatric vaccines after their primary series at 2, 4 and 6 mo of age; we call these infants low vaccine responders (LVRs). 1 We have defined LVRs as those infants with Ab levels below protection to > 50% of vaccine Ags tested when measured after the primary vaccine series by collecting blood samples at 9–12 mo of age. Among the vaccine Ags tested, chemically detoxified pertussis (PT) elicited the weakest serum IgG titers in LVR children followed by pertactin (PRN) and filamentous hemagglutinin (FHA), which is very significant given the resurgence of pertussis among vaccinated infants. 1 LVR infants experience more frequent middle-ear infections, lobar pneumonia and upper respiratory viral infections (Pichichero et al., unpublished data). Until now it has been widely accepted that 5–15% of infants develop sub-protective Ab level to various vaccines; LVR infants therefore may become vulnerable to potential life-threatening vaccine-preventable infections as their protection is limited by herd immunity.
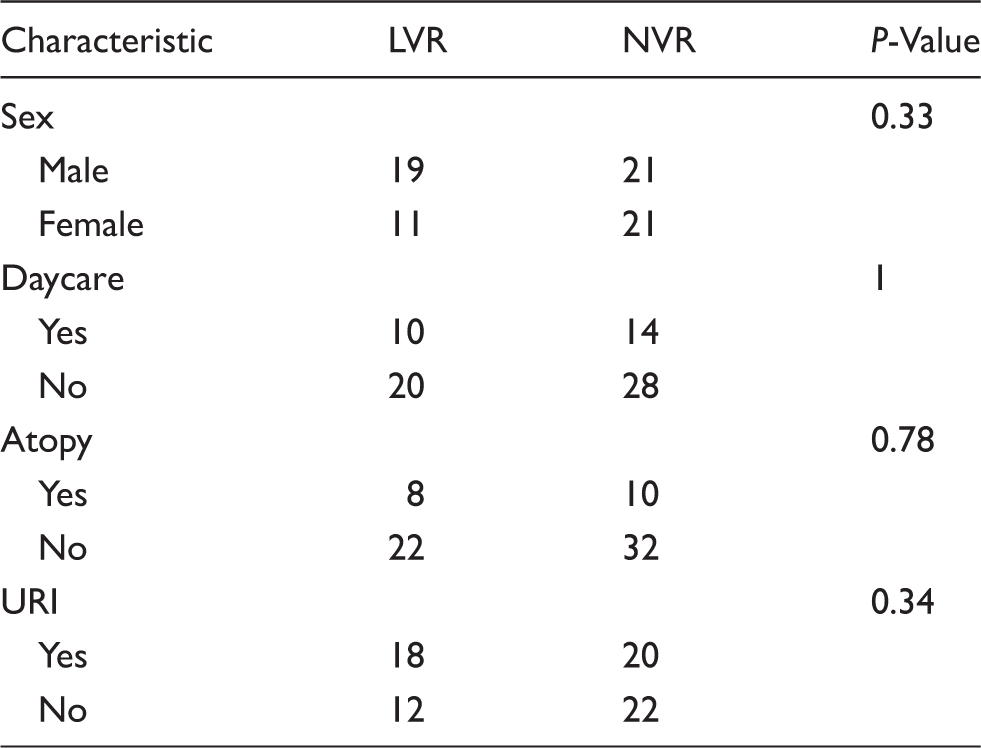
Clinical and demographic characteristics of children in this study.
Suboptimal activation and functional immaturity of professional APCs in LVRs may directly contribute to defective T-cell function, B-cell memory and reduced vaccine responses. However, the contributions of specific immune cells and their interactions to the broad defects in innate and adaptive arms of immunity of LVR infants remain unknown. Owing to the unique challenges in specimen collection, human infant innate immune response studies are rare and a dearth in knowledge exists as to how the innate immunity regulates T- and B-cell responses. We hypothesized that the low innate immune response in infants at 6–9 mo of age is a maturational delay causing downstream T-cell activation defects leading to poor T-cell help to B cells and low Ab response. Therefore, in this prospective longitudinal study, we sought to determine the maturational profile of professional APCs from 6–36 mo old LVR children compared with NVRs based on their activation and cytokine response to TLR stimulation.
Material and methods
Subjects
Healthy children were enrolled from a middle-class, suburban socio-demographic population in Rochester, NY, in a prospective, longitudinal study. Upon completion of their primary series of vaccinations, children entered the study and blood collected at 6, 9, 12, 15, 18, 24 and 30–36 mo of age. The blood samples analyzed in the current study started with their first healthy visit at 6 mo of age, then 9, 12, 15, 24 and 36 mo of age, and based on the availability of suitable and sufficient quantities of PBMCs. Written informed consent was obtained from parents of the children in association with a protocol approved by the Rochester General Hospital Institutional Review Board.
Vaccinations, minimal protective Ab levels, group definition
All children received CDC-recommended, age-appropriate vaccinations with USFDA-approved products. The minimum protective Ab level for diphtheria toxoid (DT) and tetanus toxoid (TT) when measured by an ELISA method is 0.1 IU/ml, for Haemophilus influenza type b polysaccharide, PRP is 0.15 μ/ml, for Streptococcus pneumoniae (Spn)-conjugated polysaccharides is 0.35 μ/ml, for polio using a microneutralizaton assay is >1:8 titer, and for HepB is >10 mIU/ml. 6 A correlate of protection for acellular pertussis vaccine Ags has not been established; however, a titer of 8 ELU/ml has been proposed for PT, PRN and FHA. 7
LVRs were defined as infants who had sub-protective Ab levels at 9–12 mo to > 50% of vaccines tested. NVRs were defined as infants (9–12 mo) who had sub-protective Ab levels to < 50% of vaccines tested.
PBMC sample processing and in vitro stimulation
Heparinized venous blood was drawn and PBMCs isolated from buffy coat using Ficoll gradient, according to manufacturer’s instructions. Cells were washed and re-suspended in recovery cell culture freezing media (Life Technologies, Grand Island, NY, USA) and frozen in liquid nitrogen until use. Prior to stimulation, frozen PBMCs were quick-thawed in a 37℃ water bath followed by slowly adding culture medium [RPMI-1640 supplemented with 10% of FBS, 2 mM l-glutamine, 100 U/ml penicillin and 100 µg/ml streptomycin (Sigma Aldrich, St. Louis, MO, USA)]. After thawing and washing, cells were counted and rested for overnight (16 h, 200 µl/well at 2 × 106 cells/ml) in 96-well flat-bottom plates at 37℃ in 5% CO2 incubator. For intracellular cytokine assays, total PBMCs were stimulated with 1 µg/ml of TLR 7/8 agonist R848 (Invivogen, San Diego, CA, USA) for 6 h with 10 µg/ml brefeldin-A (BFA; Cell signaling, Danvers, MA, USA) for the last 5 h at 37℃ in 5% CO2 incubator. These cells were then processed for flow cytometry analysis. An identical set of samples were incubated for 24 h at 37℃ in 5% CO2 incubator with or without R848 (1 µg/ml) in 96-well round-bottom plates without adding BFA. After 24 h culture, cells were spun and supernatants were collected and stored at –80℃ until Luminex analysis. 8 Cell pellets were stabilized using RNAlater (Sigma Aldrich, St Louis, MO, USA) and stored at –20℃ until total RNA was extracted.
Flow cytometry analysis
Post-stimulation, cells were washed once before incubating them with live–dead fixable aqua dead cell stain (Life Technologies) in 1% PBS–FBS buffer for 30 min. Cells were then washed and surface stained with various cell surface Abs [CD14 (M5E2), CD16 (3G8), CD303 (201 A) and lineage markers: CD3 (HIT3a), CD56 (HCD56), CD19 (HIB19), CD20 (2H7); all from Biolegend (San Diego, CA, USA) along with HLA-DR (G46-6; BD Biosciences, San Diego, CA, USA) and CD11c (BU15; AbD Serotec)] in the presence of Fc receptor blocking solution (human TruStain FcX; Biolegend) for 20 min in the dark at 4℃. For intracellular staining, IL-12p40/70 (C11.5; BD Biosciences), TNF-α (MAb11) and IFN-α2 b (7N4-1; BD Biosciences) Ab cocktail was used for 30 min in the dark at room temperature (20–22℃) after cells were fixed and permeabilized using BD Cytofix/Cytoperm Fixation and perm solution for 20 min. After the wash steps, cells were acquired on a BD LSR II flow cytometer and data analyzed using FlowJo software (FLOWJO, Ashland, OR, USA).
Cytokine measurement
Supernatants stored at –80℃ were thawed at room temperature and cytokine and chemokines were measured from undiluted samples using Bio-Plex Pro Human Cytokine Group I 12-plex assay kit (Bio-Rad Laboratories, Hercules, CA, USA) according to the manufacturer’s instructions. Prepared samples were run on a Bio-Plex 200 system with Luminex xMAP technology (Bio-Rad) and the analysis was performed using Excel (Microsoft, Redmond, WA, USA) and GraphPad Prism (GraphPad Software, La Jolla, CA, USA). IFN-α levels from supernatants were determined by human IFN-α multi-subtype ELISA Kit (Life Technologies). The cytokines/chemokines tested with their detection limits were as follows: IL-1β, 0.5 pg/ml; IL-6, IL-8, IL-10, IP-10 and IL-12p70, 2 pg/ml; IFN-γ and MCP-1, 1.5 pg/ml; MIP-1α, 0.07 pg/ml; MIP-1β, 0.7 pg/ml; RANTES, 1 pg/ml; TNF-α, 4 pg/ml; and IFN-α, 12.5 pg/ml.
RNA extraction and quantitative real-time PCR
RNA extraction was performed using Qiagen’s RNeasy Mini kit (Qiagen iBiosciences, Germantown, MD, USA), following the manufacturer’s instructions. To remove traces of DNA if any, a DNA digestion step was performed using DNase I enzyme from Qiagen. Criteria for inclusion in downstream applications was based on OD 260/280 of > 1.8 and the absence of visible degradation. RNA was reverse transcribed to cDNA using RT 2 First Strand Kit (Qiagen) according to the manufacturer’s instructions. For the innate immune response genes, human primers were purchased from SA Biosciences (Valencia, CA, USA). Real-time PCR assays were run for each sample containing 50–100 ng of cDNA as a template and specific primers using SYBER Green dye on an iCycler IQ from Bio-Rad. GAPDH gene expression was used as a control to normalize the data. The results presented represent fold change given by the equation 2−ΔΔ C t, a convenient way to analyze the relative changes in gene expression from real-time quantitative PCR experiments.9,10
Statistical analysis
Data were analyzed by Fisher’s exact test and two-tailed Mann-Whitney U-tests (non-parametric data) and P < 0.05 was considered significant.
Results
APC MHC II expression differences between LVR and NVR children was only observed at 6–9 mo of age
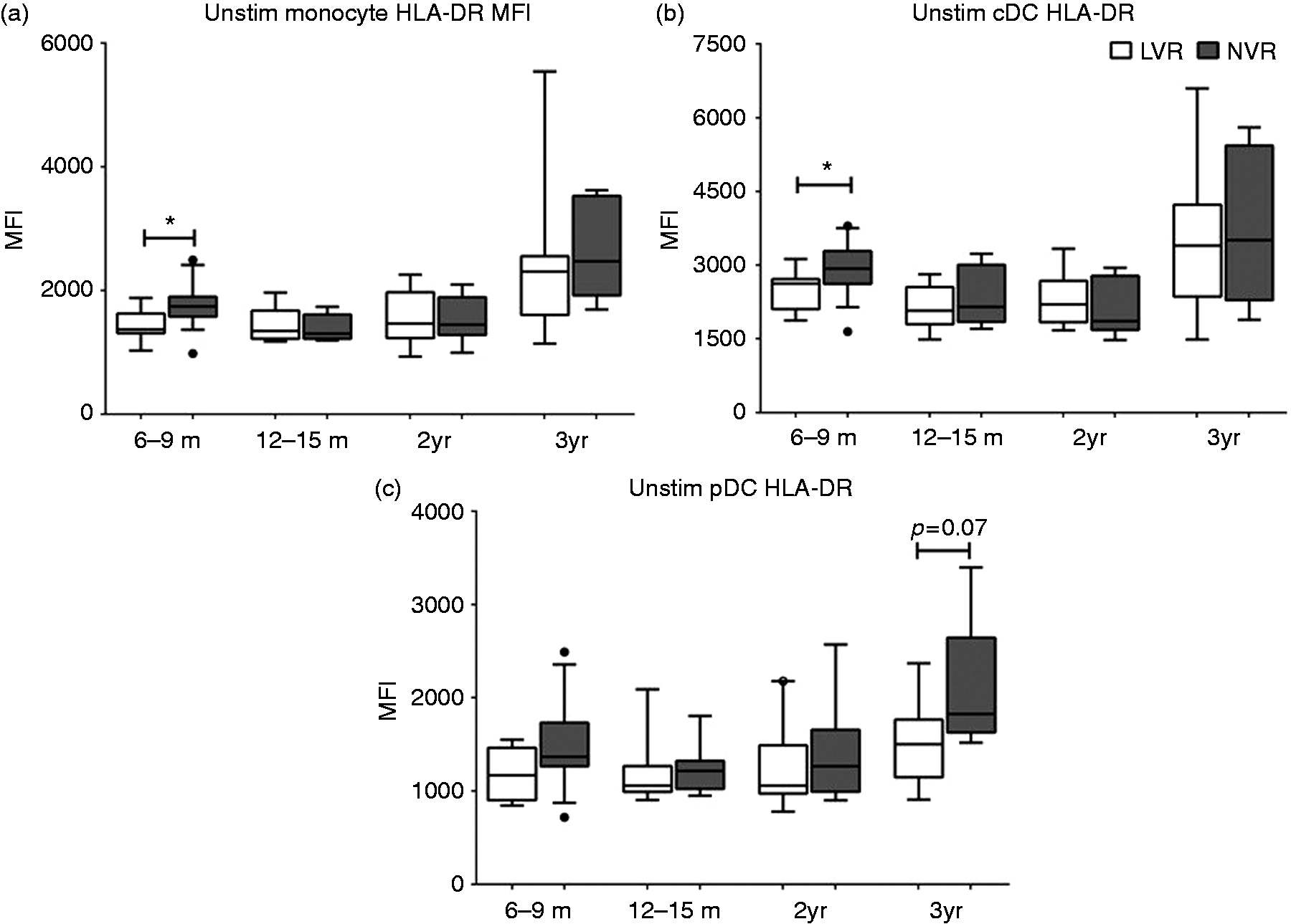
We first analyzed quantitative differences in APC phenotype subsets between LVR and NVR subjects at all health visits spanning 6–36 mo of age (Figure 1). No significant differences in total frequency of APC subsets—monocytes (HLA-DR+ CD14+ CD16+), cDCs (HLA-DR+CD14lowCD16lowCD11chiCD303-) or pDCs (HLA-DR+ CD14lowCD16lowCD11c– CD303+)—were observed between LVR and NVR children at any of the time points tested. We previously described lower baseline MHC II expression on monocytes and cDCs of 6–9-mo-old LVR infants compared with NVR infants.
1
Here, we analyzed whether the MHC II expression differences persist as the LVR children get older compared with NVR children, using prospectively collected samples. Interestingly, HLA-DR expression levels of monocytes and cDCs did not show significant differences between LVR and NVR children starting from 12 mo to 36 mo (Figure 2a, b). Although median pDC-MHC II levels were lower in LVR infants, the levels were not significantly different from NVR at any of the time points tested (Figure 2c).
Gating strategy for simultaneous analysis of the activation of three different APC types upon stimulation with TLR7/8 agonist (R848, 1 µg/ml). (a) Initial gating of total events based on forward (FSC) and side scatter (SSC) properties followed by gating out dead cells, doublets and HLA–DR–, lineage+ (CD3, CD19, CD20, CD56) cells. (b) Monocytes were defined as HLA-DR+CD14+CD16+ cells, conventional DCs (cDCs) as HLA-DR+CD14lowCD16lowCD11chiCD303– and plasmacytoid DCs (pDCs) as HLA-DR+CD14lowCD16lowCD11c–CD303+. MHC II expression on peripheral blood APC subsets from LVR and NVR children assessed by flow cytometry. (a) baseline monocyte HLA-DR MFI (b) baseline cDC HLA-DR MFI (c) baseline pDC HLA-DR MFI. The number of LVR/NVR subjects used for 6–9 mo, 12–15 mo, 2 yr and 3 yr time points are as follows: LVR (n = 9, 8, 9 and 7); NVR (n = 17, 7, 8 and 7). Statistical significance was assessed by Mann–Whitney U-test and data show the box and whisker plot with range for each APC subset.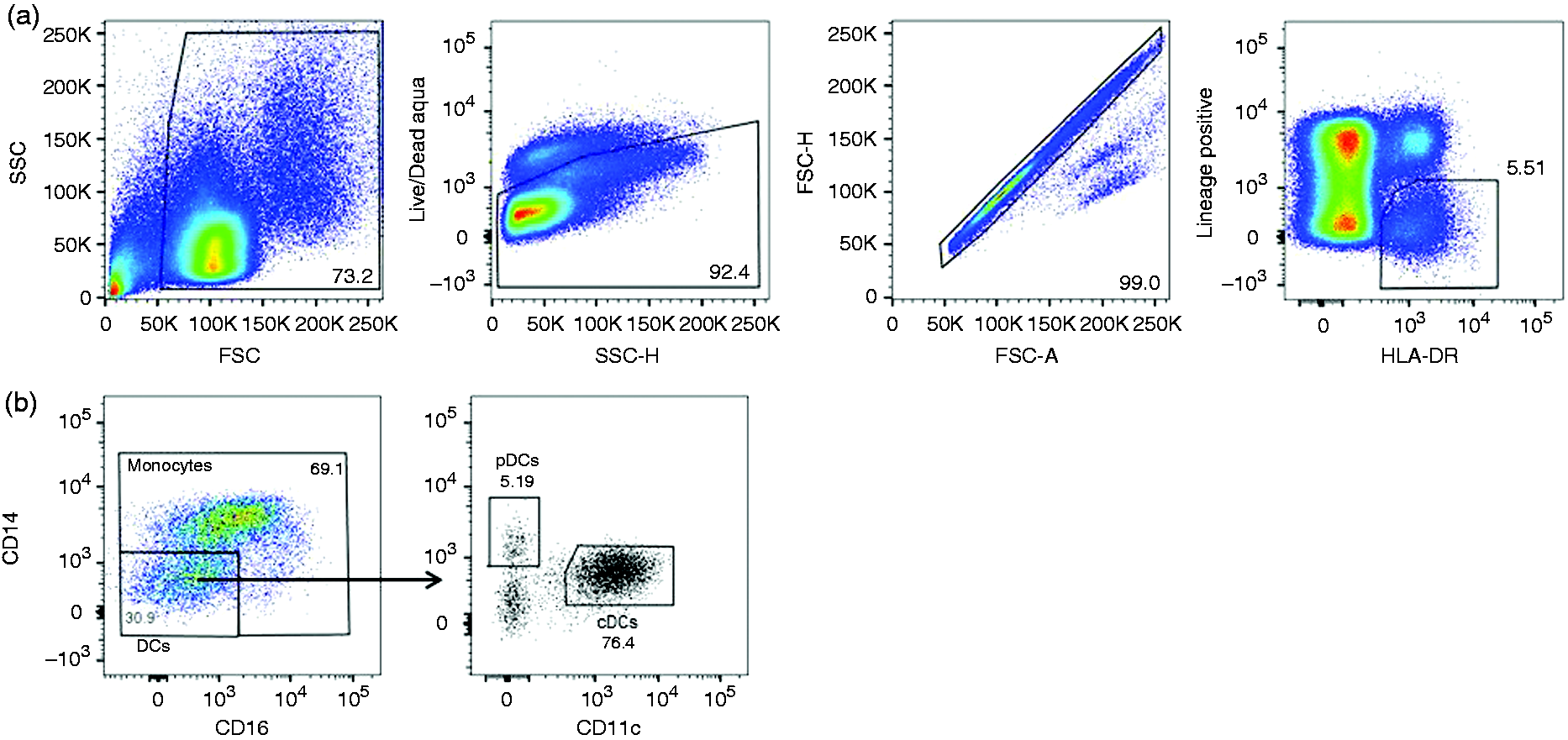
Longitudinal TLR7/8 mediated Th1 and pro-inflammatory cytokines levels from LVR and NVR children
We analyzed intracellular APC IL-12, TNF-α and IFN-α cytokine levels after 6 h of R848 stimulation and cytokine levels from the total PBMC supernatant after 24 h of R848 stimulation without BFA. We found that intracellular IL12p40/70 and TNF-α levels from monocytes and cDCs were not significantly different between LVR and NVR children at the time points tested. Intracellular IFN-α response from pDCs were also not significantly different between LVR and NVR children at any time point tested.
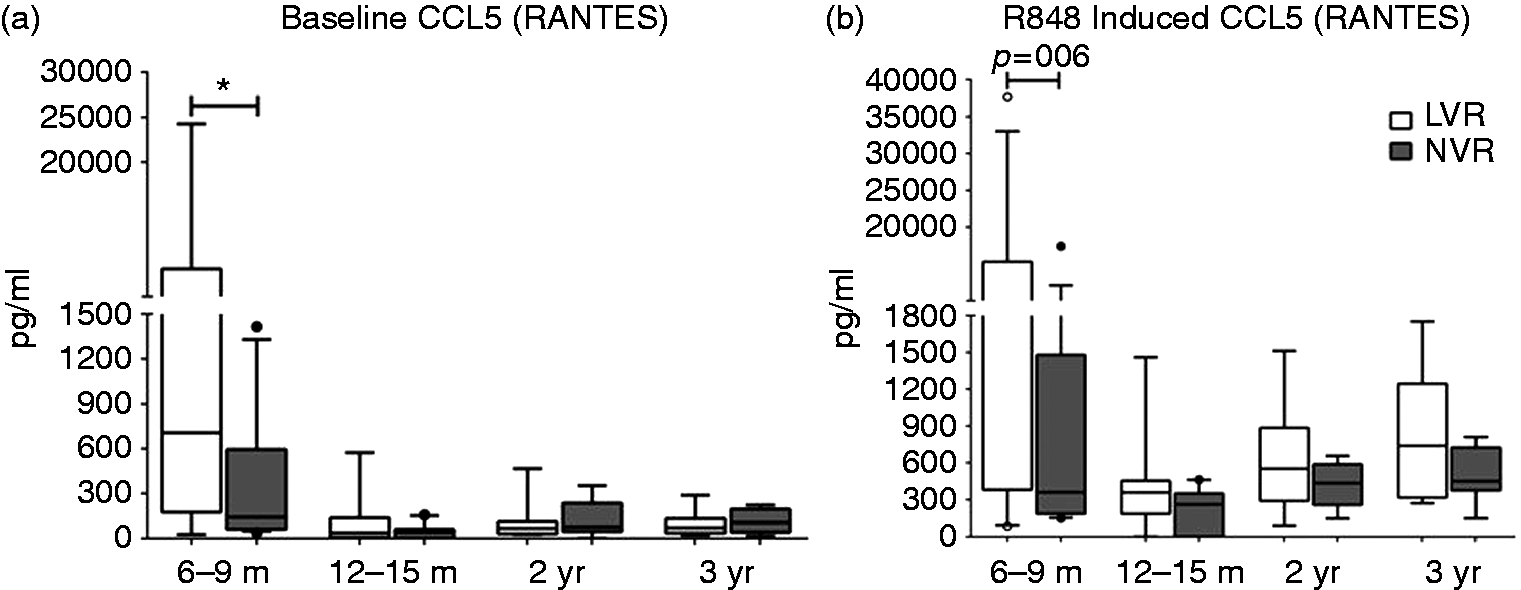
However, when the levels of secreted cytokines/chemokines from total PBMCs stimulated with R848 were measured, LVR children at 6–9 mo of age secreted significantly (P < 0.05) lower quantities of Th1-polarizing cytokines, such as IFN-α and IL-12p70, compared with NVR children (Figure 3a, b). They also secreted lower levels of IL-1β than NVR infants at 6–9 mo of age (Figure 3c). The defective Th1 and pro-inflammatory cytokine production from LVR children seems to be mitigated as the children age, as evidenced by similar levels of IFN-α, IL-12p70 and IL-1β levels between LVR and NVR children at 12–36 mo of age (Figure 3a–c). Interestingly, pro-inflammatory IL-1 β and TNF-α levels from LVR children were significantly (P < 0.05) elevated compared with NVR children at 12–15 mo and 12–24 mo of age, respectively (Figure 3c, d). In contrast, LVR infants had significantly (P < 0.05) higher baseline RANTES (CCL5) levels (Figure 4a) compared with NVR infants and a trend (P = 0.06) toward higher levels (Figure 4b) upon R848 stimulation at 6–9 mo. However, LVR and NVR children did not have significantly different baseline or activated RANTES secretion from 12–36 mo of age (Figure 4a, b).
R848-induced (24 h) total cytokine secretion (pg/ml) from peripheral blood supernatants of LVR and NVR children. (a) Multi-subtype IFN-α (b) IL-12p70, (c) IL-1β and (d) TNF-α. The number of LVR/NVR subjects used for 6–9 mo, 12–15 mo, 2 yr and 3 yr time points are as follows: IFN-α (LVR: n = 15, 8, 8 and 6; NVR: n = 14, 10, 8 and 7); IL-12p70, IL-1β and TNF-α (LVR: n = 17, 8, 8 and 6; NVR: n = 15, 10, 8 and 7). Unstimulated controls were used to subtract the background levels of individual cytokines. Statistical significance was assessed by Mann–Whitney U-test and data show the box and whisker plot with range. (a) Baseline and (b) R848-induced RANTES secretion from peripheral blood supernatants of LVR and NVR children. The number of LVR/NVR subjects used for 6–9 mo, 12–15 mo, 2 yr and 3 yr time points are as follows: LVR (n = 17, 8, 8 and 6); NVR (n = 15, 10, 8 and 7). Statistical significance was assessed by Mann–Whitney U-test and data show the box and whisker plot with range.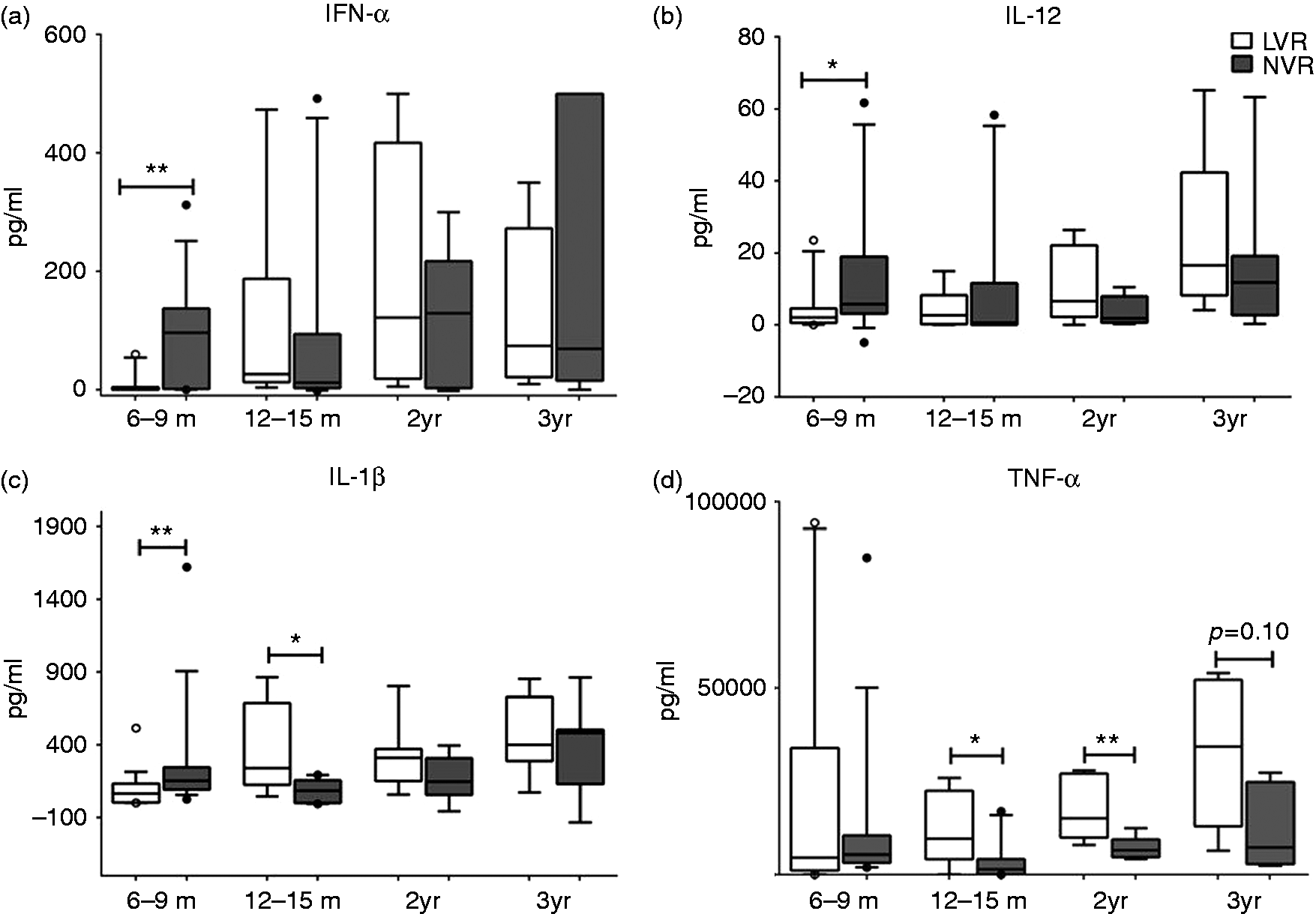
Reduced TLR7/8 mediated IRF7 expression between LVR and NVR children observed only at 6–9 mo
We previously reported that LVR infants have significantly lower IRF7 expression levels compared with NVR infants upon R848 stimulation at 6–9 mo of age.
1
In order to determine whether the observed mitigation in Th1-polarizing cytokine secretion differences between LVR and NVR children by 12 mo of age (Figure 3a–c) was related to overcoming the activated expression defects in key intracellular signaling molecules involved in the TLR7/8-MyD88-IRF7 pathway, we evaluated the mRNA baseline and stimulated expression levels of MyD88 and IRF7. We did not measure significant differences in baseline or activated MyD88 expression levels of LVR and NVR children except for a higher trend with NVR children (P = 0.07) at 12–15 mo of age (Figure 5a). However, we observed that by 12–15 mo of age activated LVR: IRF7 expression level reached similar levels to those of NVR children and the recovery persisted up to the last longitudinal sample tested in 36-mo-old children, correlating with the cytokine data (Figure 5b). We did not observe significant differences in baseline or activated expression levels of TLR7 or TLR8 (data not shown).
Comparison of the PBMC mRNA fold-change of innate gene transcripts (a) MyD88 and (b) IRF7 upon 24 h stimulation with R848 (1 µg/ml) between LVR and NVR children. Data expressed as the change in fold induction of genes of interest compared with corresponding unstimulated PBMC. The number of LVR/NVR subjects used for 6–9 mo, 12–15 mo, 2 yr and 3 yr time points are as follows: LVR (n = 14, 7, 8 and 5); NVR (n = 14, 7, 8 and 8). Statistical significance was assessed by Mann–Whitney U-test and data show the box and whisker plot with range.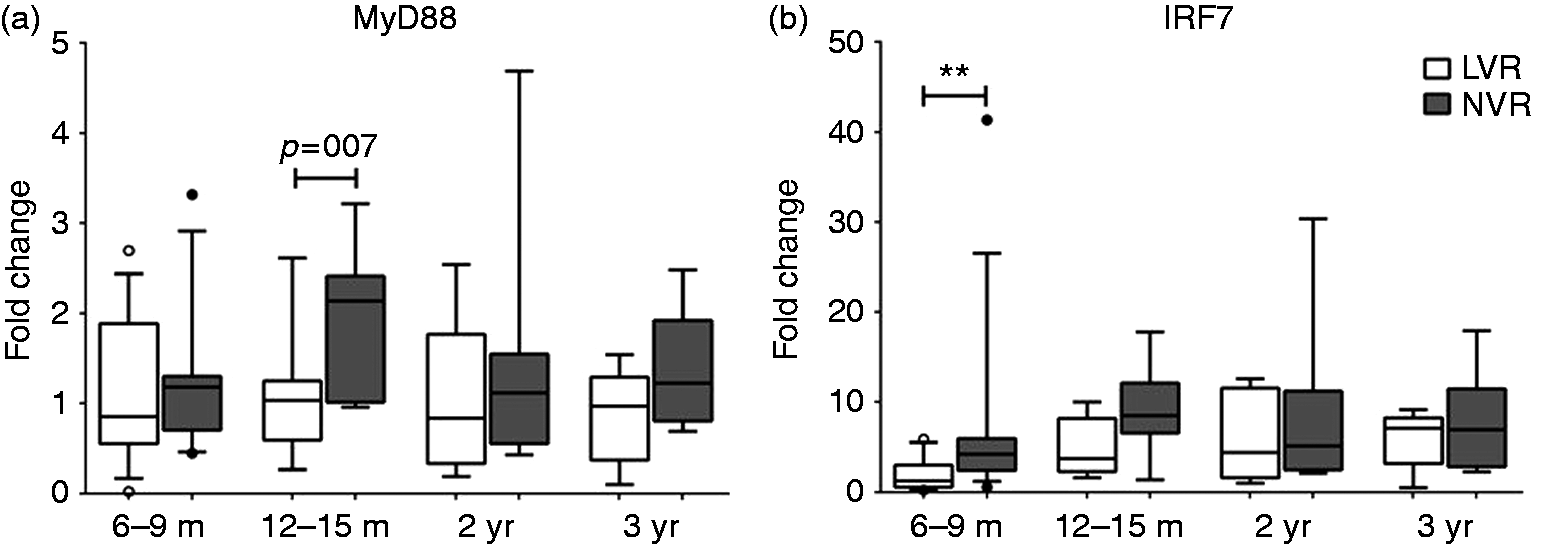
Discussion
We previously described a group of children who respond with sub-protective Ab levels to multiple vaccine Ags in the first two years of life. 1 While multiple factors such as hereditary, environmental and history of frequent infections could contribute to this immune phenotype, we discovered that specific immune defects in T cells, B cells and APCs exist in these children at 6–12 mo of age compared with NVR children. At 6–9 mo of age APCs from LVR children exhibit significantly lower surface expression of MHC II, intracellular IRF7 and the Th1 cytokines IFN-α, IL-12p70 and IL-1β, which could contribute to T- and B-cell activation defects and poor Ab response (Surendran et al., 2016). 1 It is increasingly appreciated that an important element of vaccine efficacy is the ability of a given vaccine to activate the innate immune system to enhance APC function and Th1-polarizing adaptive responses.11,12 However, LVR children generally outgrow their poor vaccine response and infection proneness by the time they received booster doses of vaccines, though it is unknown whether this is the result of delayed maturation of immune response or unidentified compensatory mechanisms. Therefore, as a first step, it was critical to determine the ontogeny of immune responses, especially innate immunity, in these children so as to identify potential mechanisms and solutions to mitigate the low vaccine response.
In this longitudinal study, we discovered that the diminished Ag-presenting capacity demonstrated by low MHC II, as well as defective ability to generate Th1 polarizing signals by LVR children at 6–9 mo compared with their NVR peers, is mitigated as early as 12–15 mo of age. Considering the fact that the vaccine Ab response is also regained in these children by 2 yr, makes it attractive to speculate of a delayed maturation of innate, and, subsequently, adaptive responses might be helping LVR children to overcome suboptimal Ab response. Monocyte MHC II levels at 12–15 mo NVR infant were significantly (P = 0.005) lower than 6–9 mo NVR infants. Although not significantly different, similarly lower median MHC II levels were also observed with cDC and pDC subsets at 12–15 mo. Twelve–15-mo-old infants also show lower median cytokine responses compared with 6–9-mo-olds. Rowe et al. have reported an early initiation followed by decline and later resurgence of cytokine levels (especially IFN-γ) in infants from 2–6 to 12–18 mo old. 13 However, there are no similar published reports on this type of change in MHC II levels among young children, and this will require further investigation in the future.
Upon activation, DCs efficiently process and present Ags in the context of MHC II and upregulate expression of co-stimulatory molecules, as well as increase production of Th1-polarizing IL-12p70 or IFN-α/β. 14 R848 is known to induce IL-12 and IFN-α from monocytes/cDCs and pDCs, respectively. 15 Suboptimal IRF7 transcription may contribute to the observed defects in the production of essential immunomodulatory cytokines, such as IFN-α and IL-12,16,17 by LVR children at 6–9 mo of age. However, by 12–15 mo, APCs from LVR children seem to have MHC II expression comparable with NVR children, consistent with similar levels of IRF7 and Th1-polarizing cytokines from NVR and LVR children throughout the duration of study. Interestingly, LVR children secreted higher amounts of pro-inflammatory cytokines (IL-1β and TNF-α) compared with NVRs starting from 12 mo of age raising the possibility of a compensatory inflammatory response. Multiple studies have reported that viral URI, particularly with influenza, rhino and respiratory syncytial viruses, result in elevated serum and nasopharyngeal levels of RANTES (CCL5).18–20 Although the LVR study population we used was not significantly different in their viral URI status compared with NVR children, heightened baseline secretion of RANTES by LVR children at 6–9 mo of age, while their immune response was suboptimal, may suggest either subclinical viral infections or compensatory immune response to ward off viral infections by recruiting cytotoxic T cells. We tend to believe it is the latter since the baseline LVR-RANTES levels drops, becoming similar to NVR children once the LVR APC response was regained by 12–15 mo of age.
The importance of the TLR pathway for host defense in infants is obvious in the clinical consequences of TLR pathway defects. Defects of signaling molecules downstream of TLRs, including IRAK4, MyD88 and IRF7, result in selective predisposition to streptococcal or staphylococcal pyogenic infections and viral infections, respectively.21–23 We did not observe significant differences in basal or activated TLR7/8 expression levels in PBMCs isolated from LVR and NVR infants at any of the time points tested. This suggests that the lower IFN-α, IL-12p70 and IL-1β cytokine levels from R848-induced LVR PBMCs at 6–9 mo of age might be due to signaling defects downstream to TLRs. Among the major intracellular signaling molecules tested that are downstream to various TLRs, only IRF7 levels were significantly lower in R848 induced LVR PBMCs. Hence, the significantly low IFN-α level in R848-induced LVR PBMCs at 6–9 mo of age may be a direct result of suboptimal IRF7 transcription. Gautier et al. 16 demonstrated that IL-12p70 production in response to TLR activation was dependent on production of IFN-α/β that regulated IL-12p35 mRNA accumulation. Interestingly, we observed a correlation of R848-induced increased IL12p70 secretion in 6–9-mo-old NVR children with higher fold change in IRF7 expression compared with LVR children. DC–TLR engagement by R848 induces endogenous IFNs mediated by IRF7/8 that further synergize with the NF-κB pathway for possible IL-12p35 synthesis leading to IL-12p70 secretion.16,24 Once the LVR children overcome the activated IRF7 expression defects by 12–15 mo, the IFN-α and IL-12p70 cytokine responses were also recovered pointing towards a delayed innate immune maturation in LVR children.
In the USA and other countries where parent refusal of vaccines has increased and herd immunity may become threatened, the implications of our findings are obvious. Understanding the mechanisms of how suboptimal innate response from LVR children can be mitigated sooner and elucidating the parallels to neonatal immune responses will help us in improving the vaccine response in neonates and young infants. Judicious targeting of multiple TLR agonists on APCs is one such avenue to pursue to overcome the APC tardiness in neonates and LVR children. The possibilities of T- or B-cell intrinsic defects leading to low vaccine response cannot yet be ruled out. Therefore, future research from our laboratory will investigate the ontogeny of T- and B-cell responses in LVR children, as well as the interplay of innate and adaptive immunity to identify additional mechanisms contributing to the LVR immune defect.
Footnotes
Acknowledgement
We thank Karin Pryharski and Jareth Wischmeyer for their technical help, and Dr Robert Zagursky for his helpful comments on the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NIH R01DC008671 awarded to Dr Pichichero. NS is supported by NIH NIAID 5 R03 AI117700-02.
