Abstract
Stringently defined otitis-prone (sOP) children represent a new classification of the otitis-prone condition. Previous studies showed dysfunction in Ab, B-cell memory and T-cell memory responses. We sought to determine whether there are defects in numbers, phenotype and/or function of professional APC in the peripheral blood of sOP infants. APC phenotypic counts, MHC II expression and intracellular cytokine levels were determined in response to TLR7/8 (R848) stimulation by flow cytometry. Innate immune mRNA expression was measured using RT-PCR and cytokines were measured using Luminex technology. Significant (
Keywords
Introduction
Acute otitis media (AOM) is a common upper respiratory infection that accounts for the most antibiotic prescriptions among childhood illnesses,
1
and represents a substantial healthcare burden worldwide, with >$5 billion spent annually in the USA alone.
2
AOM is caused most commonly by
Since defining the sOP child, we have been intensively studying their immune profile, focused on the adaptive response. We have shown that sOP children have diminished adaptive immune responses to
Importantly, we have also shown that sOP children frequently develop non-protective Ab titers after a primary series of pediatric vaccines.
18
Specifically, non-protective responses were seen to diphtheria toxoid (DT), tetanus toxoid (TT), pertussis toxoid (PT), filamentous hemagglutinin (FHA), pertactin (PRN) diphtheria, tetanus, acellular pertussis (DTap), hepatitis B, polio 3 and
In the work described in this paper, we sought to determine if there were differences in the innate immune responses of sOP infants by analyzing the peripheral blood APC numbers, their activation and/or function compared with NOP infants. Since the yield of PBMCs from infants is limited, we combined phenotyping and intracellular cytokine experiments and used a single TLR agonist (TLR7/8 agonist: Resiquimod, R848), which can simultaneously activate all three APC subsets.
Materials and methods
Subjects
Infants (6–9 mo old) who had completed their primary series of vaccinations by 6 mo of age were involved in the study. The infants were from a cohort recruited as part of the prospective study of immunity to OM (NIDCD R0108671). Children were enrolled at the age of 6 mo from a middle class, suburban socio-demographic, representative of the whole population at a private pediatric practice (Legacy Pediatrics, Rochester, NY, USA) and followed prospectively until 30 mo of age. Upon enrollment detailed demographic, epidemiologic and AOM risk factor data were collected for every child. At the age of 6 mo, children were enrolled and had a nasal wash, nasopharyngeal swab, throat culture and blood obtained at pre-scheduled 6, 9, 12, 15, 18, 24 and 36 mo health visits. In addition, whenever a child was ill with AOM, middle ear fluid was collected under local anesthesia along with other clinical samples. Middle ear fluid was obtained by tympanocentesis to assure the diagnosis of AOM, to confirm the presence of otopathogens in middle ear fluid and to provide the potential therapeutic benefit. All clinically diagnosed viral upper respiratory tract infections (URI) during visits were recorded in the database. All the nasal wash and middle ear fluid samples collected have been tested for the presence of most common otopathogens (NT
PBMC sample processing and in vitro stimulation
Heparinized venous blood was drawn and PBMCs isolated from buffy coat using Ficoll gradient and stored in liquid nitrogen until use. After thawing and washing, cell were counted and rested overnight in culture medium [RPMI-1640 (Life Technologies, Grand Island, NY, USA) supplemented with 10% FBS, 2 mM
Flow cytometry analysis
Post stimulation, cells were stained with live-dead fixable aqua dead cell stain (Life Technologies) for 30 min. Cells were then washed and surface stained with various cell surface Abs in the presence of Fc receptor blocking solution (human TruStain FcX; Biolegend, San Diego, CA, USA) for 20 min in the dark at 4℃ to identify monocytes, conventional dendritic cells (cDCs) and plasmacytoid dendritic cells (pDCs), referred to as APCs herein. For intracellular staining, IL-12p40/70 (C11.5; BD Biosciences, San Jose, CA, USA), TNF-α (MAb11) and IFN-α2b (7N4-1; BD Biosciences) Ab cocktail was used for 30 min in the dark at room temperature (20–22℃) after cells were fixed and permeabilized using BD Cytofix/Cytoperm Fixation and perm solution for 20 min. After the wash steps, cells were acquired on a BD LSR II flow cytometer and data analyzed using FlowJo software (FLOWJO, Ashland, OR, USA).
Cytokine measurement
Supernatants stored at −80℃ were thawed at room temperature and cytokine and chemokines were measured from undiluted samples using Bio-Plex Pro Human Cytokine Group I 12-plex assay kit (Bio-Rad Laboratories, Hercules, CA, USA) according to manufacturer’s instructions. Prepared samples were run on a Bio-Plex 200 system with Luminex xMAP technology (Bio-Rad) and the analysis was performed using Excel (Microsoft, Redmond, WA, USA) and GraphPad Prism (GraphPad Software, La Jolla, CA, USA). IFN-α levels from supernatants were determined by human IFN-α multi-subtype ELISA Kit (Life Technologies).
RNA extraction and quantitative real-time PCR
RNA extraction was performed using Qiagen RNeasy Mini kit (Qiagen Biosciences, Germantown, MD, USA). Criteria for inclusion in downstream applications was based on OD 260/280 of >1.8 and the absence of visible degradation. RNA was reverse transcribed to cDNA using RT2 First Strand Kit (Qiagen) according to the manufacturer’s instructions. For the innate immune response genes, human primers for each specific TLR, intracellular signaling molecules and cytokines were purchased from SA Biosciences (Valencia, CA, USA). Real-time PCR assays were run for each sample containing 50–100 ng of cDNA as a template and specific primers using SYBER Green dye on an iCycler IQ (Bio-Rad).
Real-time data and statistical analysis
For real-time analysis, average
Results
Study population
Clinical and demographic characteristics of study subjects.
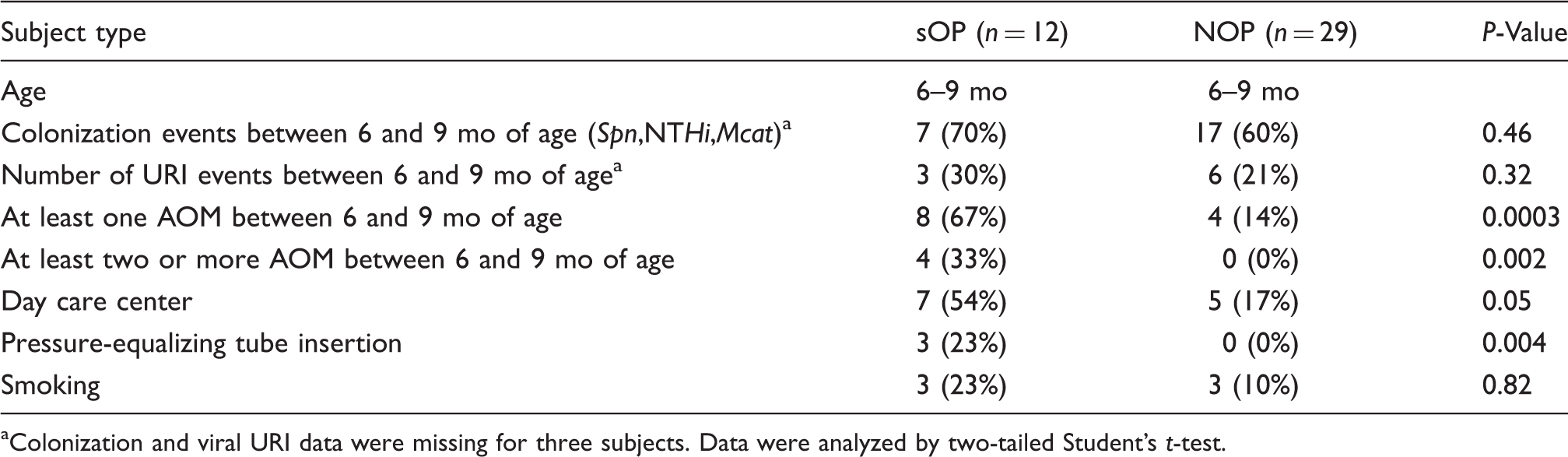
Colonization and viral URI data were missing for three subjects. Data were analyzed by two-tailed Student’s
Monocytes and cDCs
To determine whether the previously observed adaptive immune deficiency of inadequate B- and T-cell responses in sOP infants may be, in part, due to low numbers of professional APCs (monocytes, cDCs and pDCs) in human peripheral blood, we compared quantitative differences in APC phenotype subsets in PBMCs between sOP and NOP infants. Based on previous studies,20,23,24 we defined monocytes as HLA-DR+CD14+CD16+ cells, cDCs as HLA-DR+CD14lowCD16lowCD11chiCD303− and pDCs as HLA-DR+CD14lowCD16lowCD11c−CD303+ (Figure S1). Live cell adjusted cell counts in sOP ( Phenotypic counts of APCs in the peripheral blood of sOP (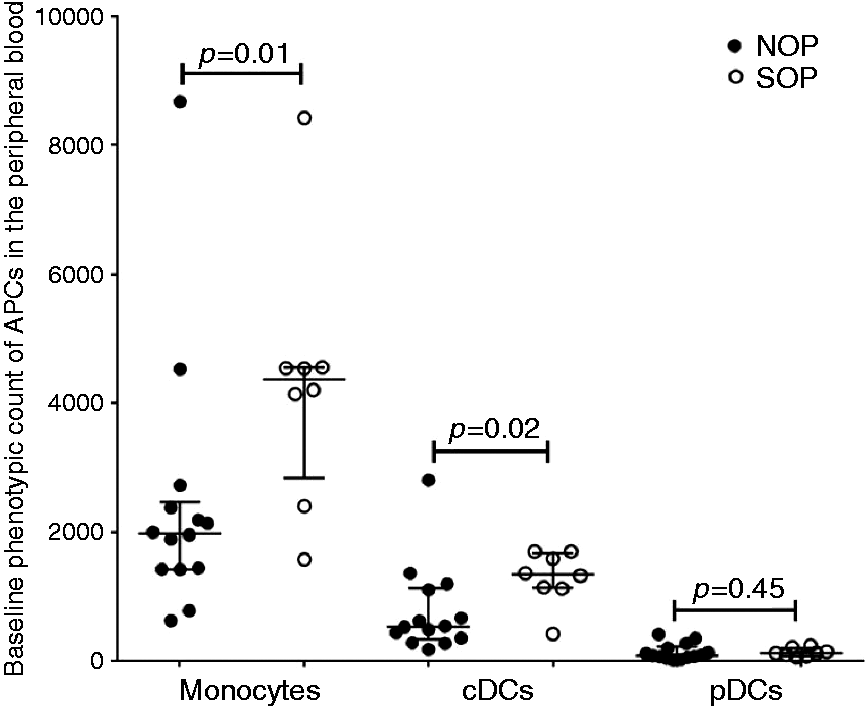
MHC II and CD86 expression
To test whether the professional APCs from sOP infants have diminished antigen presenting and co-stimulatory capabilities compared with NOP infant APCs, we tested both the baseline and activated levels of MHC II and CD86 on monocytes, cDCs and pDCs. Both the baseline and activated MHC II and activated CD86 expression levels of sOP ( APC activation and cytokine secretion of sOP (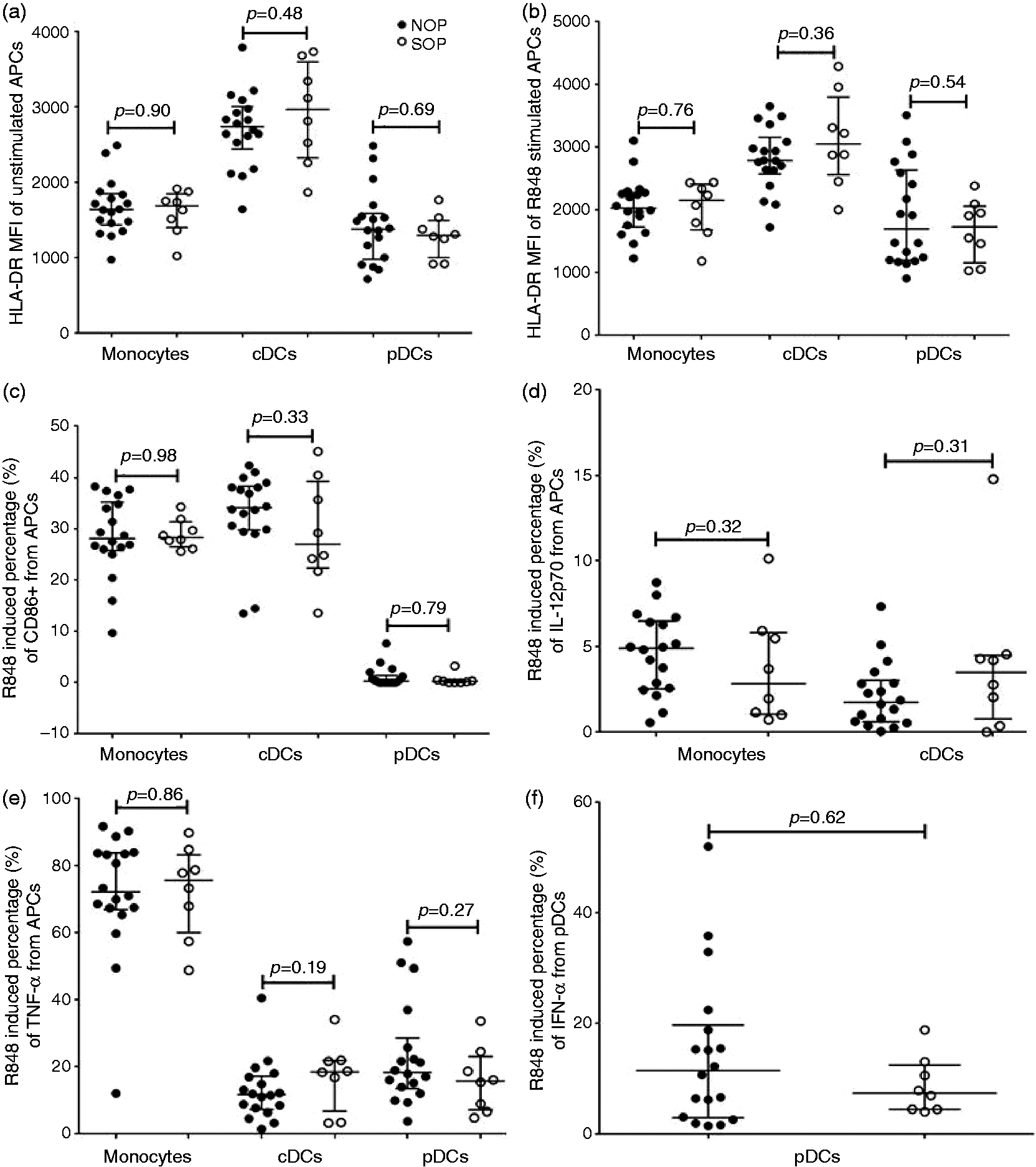
Innate cytokine secretion
We analyzed intracellular APC (6 h R848 stimulation), as well as total PBMC supernatant (24 h R848 stimulation without BFA) cytokine levels to further determine whether sOP APCs function similarly to NOP APCs. For the intracellular cytokine experiments, we concentrated on IL-12, TNF-α and IFN-α based on prior studies and preliminary work.
20
We found that intracellular IL12p40/70 and TNF-α levels from monocytes and cDCS were not significantly different between sOP (
Next, we measured the total innate associated cytokine/chemokine levels from PBMCs to get a global view of cytokine-chemokine differences between sOP and NOP upon TLR7/8 stimulation. We found that none of the 11 different innate associated cytokine/chemokine levels tested, IL-1β, IL-6, IL-8, IL-10, IL-12p70, TNF-α, IFN-γ, CCL2, CCL4, CCL5 and CXCL10, were significantly different between sOP ( Comparison of PBMC-derived total innate related cytokine/chemokine secretion from sOP (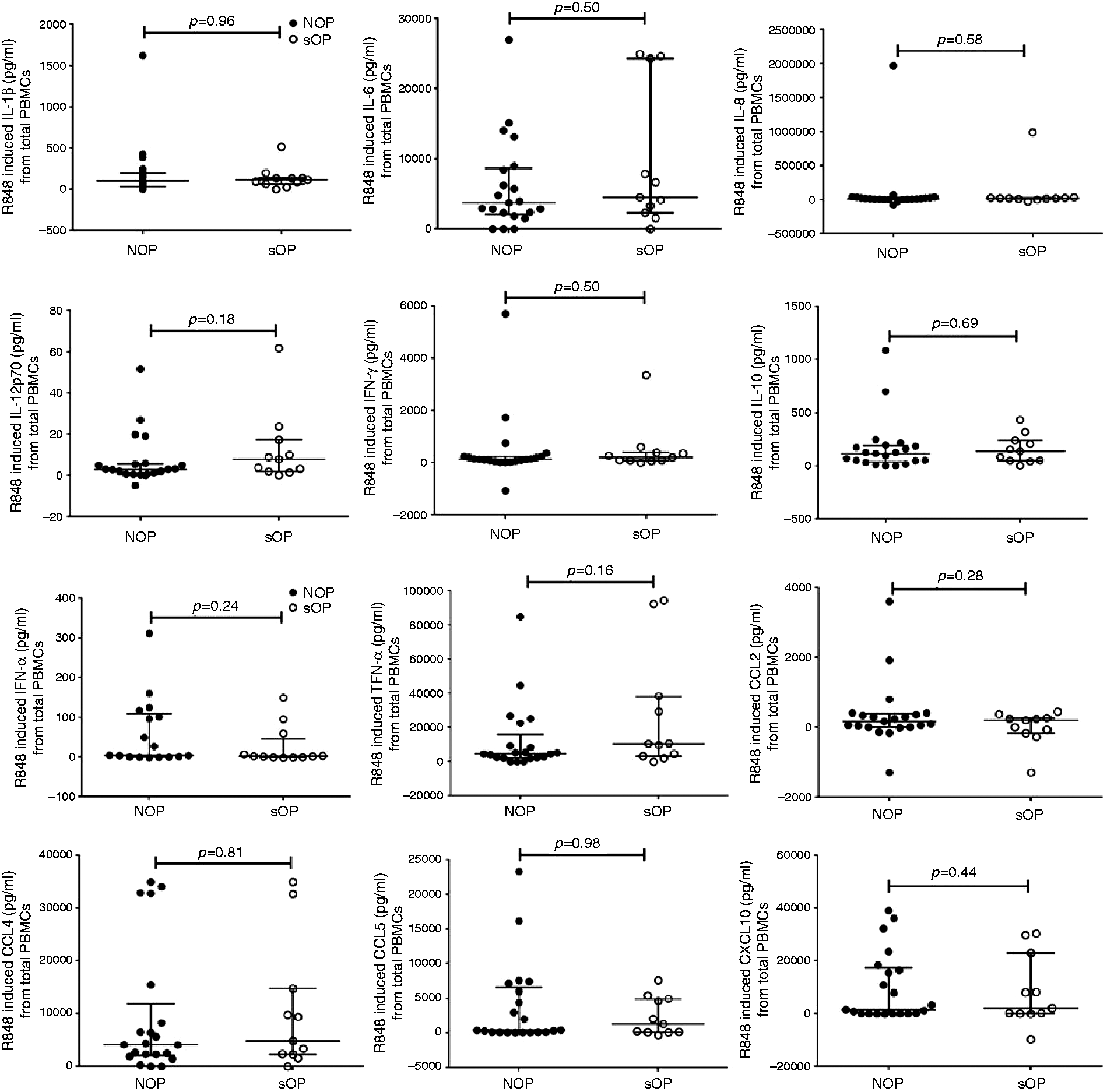
TLRs and intracellular innate transcription factors
Our previous studies have demonstrated differential regulation of TLRs and intracellular transcripts during AOM with concurrent viral infection.25–27 TLR expression levels dramatically affect innate sensing of viral and bacterial pathogens and might affect T- and B-cell responses. Therefore, we sought to determine whether the baseline or activated (24 h R848 stimulation) TLR and intracellular innate signaling molecule expression levels were different between sOP and NOP infants. We found that baseline and activated expression of TLR2, 4, 7 and 8, and intracellular signaling molecules such as MyD88, TRIF, IRAK4, IRF3 and IRF7, as well as IL-12p35 and IL-10, were not significantly different in sOP ( Comparison of the PBMC mRNA fold-change of innate gene transcripts upon 24 h stimulation with R848 (1 µg/ml) between sOP (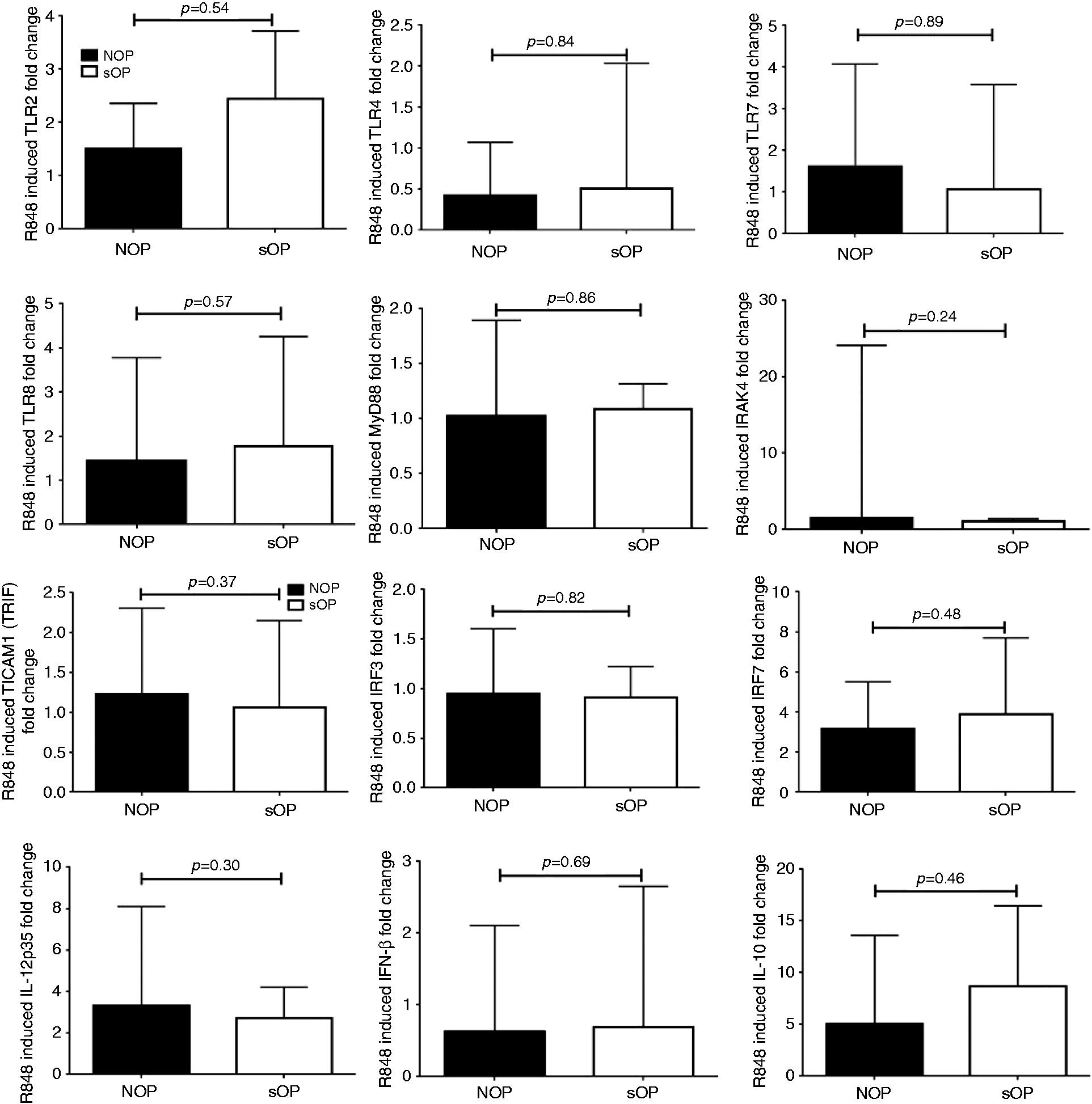
Discussion
Over the last several years, we have identified a new population of children we term as sOP with clear, unique adaptive and innate immunologic deficiencies associated with their propensity to recurrent AOM.5,9,12,18,19 Our prior work has established dysfunction in multiple immune cells that play a key role in immunological control of bacterial and viral infections.9,12,19 Here, for the first time, we assessed the systemic innate response at the single-cell level in sOP and NOP children. We used a synthetic imidazoquinoline TLR7/8 agonist—resiquimod, R848 (1 µg/ml)—because human pDCs express TLR7, whereas monocytes and cDCs express TLR8, 28 enabling simultaneous assessment of all three professional APC subsets. R848 consistently yielded the strongest PBMC response in our hands compared with other TLR agonists and provided an objective assessment of basic APC responses in peripheral blood of sOP and NOP infants as an unrelated stimulant instead of common otopathogen antigens.
We found that infants who are sOP have higher frequencies of monocytes and cDCs than NOP infants. The presence of significantly increased numbers of monocytes and cDCs in the blood of sOP infants is consistent with the existence of a pro-inflammatory state in sOP children between recurrent AOMs. The presence of elevated levels of pro-inflammatory cytokines/chemokines at the mucosa, as previously determined,1,25–27 may be recruiting increased numbers of innate cells including professional APCs to the site of inflammation.
We found that APCs from sOP and NOP infants show similar levels of resting and activated MHC II and CD86 expression. The result suggests that APCs of sOP infants respond equally well to unrelated TLR agonist stimulation in terms of MHC II or CD86 expression or cytokine secretion. Therefore, the migrating blood APCs recruited to mucosa in sOP infants might not be functionally different from APCs of NOP infants.
In addition, we found that sOP and NOP infants produce similar levels of innate associated cytokines upon activation. Baseline cytokine/chemokine levels, as well as expression levels of TLRs and intracellular signaling molecules from PBMCs in response to TLR stimulation, further support the notion that sOP APCs might not be defective in the systemic compartment.
Weakened adaptive immunity in sOP children could affect the control of viral replication, as well as nasopharyngeal bacterial colonization that may allow prolonged innate inflammatory dominance. Prior studies from our laboratory have shown that at the onset of AOM, the mucosal sites of infection such as nasopharynx and middle ear show enhanced pro-inflammatory cytokine/chemokine secretion (IL-6, IL-1β, CCL2) compared with peripheral blood.26,27 However, when we previously compared the PBMC transcriptome profile of the same child at the onset of AOM compared with a time shortly before the onset of AOM, we found genes associated with bacterial defenses, specifically β defensins and complement pathway genes in general, were up-regulated, while genes associated with cell-mediated immune responses, pro-inflammatory cytokine responses (IL-2 and IL-1β) and bacterial endocytosis were down-regulated.26,27 Therefore, host immune responses clearly diverge in the mucosal and blood compartments emphasizing the differences between local and systemic immune response that coordinate anti-bacterial host response against OM.
Our previous studies have demonstrated lower neutrophil chemoattractants such as IL-8 and CXCL5 in the nasopharynx of the sOP child. 9 Diminished neutrophil recruitment will further aggravate the bacterial load. Also, concurrent viral (RSV) infection of neutrophils has been shown to impair neutrophil phagocytic function during AOM regardless of OP status. 12 The resultant elevated mucosal inflammation and cytokine milieu might be contributing to mucosal attenuation, if any, of APC immune response in sOP children.
Our study has limitations inherent to clinical studies in humans. Using tympanocentesis as a diagnostic method might influence the current episode and development of further AOM episodes by improving the quality of treatment. Therefore, the results from our unique cohort may not be comparable with findings from clinically diagnosed OP children. The difficulty of making a correct diagnosis of AOM is something that flaws most studies done in this area and, until now, has created conflicting results in identifying otitis proneness due to immunological deficiencies. To reduce heterogeneity in the comparisons we sought, samples from children were selected when they were of similar age, same ethnicity and similar AOM risk factor environment. The age range of subjects was chosen specifically for the immunological question we sought to answer as 6–9 mo is the age when infants are most susceptible to recurrent OM. Despite the 9 yr of prospective collection of samples from >700 children, this allowed the study of a relatively small number of subjects.
Nevertheless, we can conclude that the enhanced monocyte and cDC numbers in 6–9-mo-old sOP infants indicate probable mechanisms to compensate for diminished neutrophil recruitment, enhance tissue repair and antigen presentation to improve adaptive T- and B-cell response. The non-difference, however, in APC activation, cytokine secretion and transcription factor expression between groups could suggest a lack of defect or lack of the evidence for a defect in the limited samples tested. Considering the self-limiting characteristic of otitis proneness, it would be interesting to compare the ontogeny of mucosal and systemic innate immune response prospectively in sOP children to understand the development of compensatory innate immune mechanisms.
Future studies from our laboratory will focus on the activation and function of APC subsets at the mucosal site of infection, especially in the nasopharynx of sOP children. Studies will also address the differential roles played by the first responders of the immune system (such as neutrophils and APCs) to counter concurrent bacterial and viral infection leading to recurrent AOM. Moreover, in sOP children, chronic exposure of recruited T cells to microbial antigens in the nasopharynx might also result in T-cell exhaustion and attenuated adaptive immune response, which needs to be further investigated.
Footnotes
Funding
This work was supported by the National Institutes of Health (NIH) [National Institute on Deafness and Other Communications Disorders (NIDCD)] grant R01 08671.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Acknowledgements
We thank Karin Pryharski and Jareth Wischmeyer for technical help, and Drs. Robert Zagursky and Matt Morris for critical reading of the manuscript.
