Abstract
Invariant NK T (iNKT) cells perform numerous immunoregulatory functions. In mice, they express a unique and invariant Vα14-Jα18 rearrangement of α chain in their TCR recognizing glycolipid Ags presented by CD1d. This recognition results in the rapid release of both Th1- and Th2-type cytokines, making them early mediators of the immune response. Owing to their rapid activation and genetic rigidity of their TCR, iNKT cells share characteristics with innate lymphocytes. Therefore, we investigated whether iNKT cells could be induced to express TLRs, a class of pathogen recognition receptor. Mouse iNKT cells were stimulated with anti-CD3 monoclonal Ab and IFN-α, resulting in an increase in the transcription of TLRs 3, 5, 7 and 9, and increased surface expression of TLR3. These cells were subsequently stimulated with TLR ligands, resulting in an increase in the production of IFN-γ, IL-4 and TNF-α. Supernatants from these cells also increased macrophage production of IL-6 and prostaglandin E2, and increased their phagocytic activity and CD80 expression. These supernatants also reduced vesicular stomatitis virus-GFP replication in fibroblasts. This study demonstrates the role of IFN-α in iNKT cell activation, as well as the direct modulatory effects of TLR ligands on iNKT cell function, including antiviral activity.
Introduction
Invariant NK T (iNKT) cells are a class of double-negative (DN) or CD4+ T lymphocytes that express the invariant Vα14-Jα18 rearrangement of α chain in their TCR in mice (Vα24-Jα18 in humans).1,2 iNKT cells recognize glycolipid Ags presented in the context of the MHC class I-like molecule CD1d, and are capable of rapidly producing significant quantities of both Th1- and Th2-type cytokines, including IFN-γ and IL-4, 3 shortly after glycolipid stimulation.
iNKT cells express PRRs, such as the TLRs, which recognize PAMPs.4–7 An anti-inflammatory role for direct stimulation of TLR3 of iNKT cells has been suggested in the mouse liver, 6 as well as increased IFN-γ production by TLR4-stimulated iNKT cells in the mouse lung. 7 However, relatively little work has been done to elucidate how TLR expression and functionality in iNKT cells are influenced by other signals, including cytokines, such as IL-12 and IL-18.
In contrast, it has been established that TLR ligands (TLR-Ls) activate APCs to stimulate indirectly iNKT cells through the production of co-stimulatory cytokines, such as IL-12 and IFN-α.8–11 The production of IL-12 and IL-18 by TLR-stimulated APCs, which leads to increased T cell activation, has been well documented.12–15 However, recent studies of conventional CD4+ and CD8+ T cells, CD4+CD25+ regulatory T cells and γδ T cells have demonstrated a potential role for APC-independent activation through direct TLR stimulation.16–24
Biologically, type I IFNs, especially IFN-α and IFN-β, are indispensable in eliciting many antiviral responses. This is supported by the fact that mice with defective type I IFN signaling not only have impaired innate antiviral responses, but also have deficient T cell responses to lymphocytic choriomeningitis virus, vesicular stomatitis virus (VSV) and Vaccinia virus. 25 Type I IFNs also act directly on iNKT cells, increasing their activation by TLR-stimulated dendritic cells (DCs); 26 therefore, we sought to investigate the role of type I IFNs in iNKT cell activation with respect to their functional expression of TLRs. Existing evidence suggests that TLR2 expression in naïve T cells is inducible by co-stimulation with IFN-α. TLR2 is also constitutively expressed by memory CD4+ T cells. 16 In fact, type I IFNs have also been shown to have numerous direct effects on the functionality and survival of conventional CD4+ T cells, particularly relating to the Th1 response. 27 In addition, CD4+ T cell activation and survival can be enhanced through direct TLR activation,28,29 and co-stimulation of CD4+ T cells with anti-CD28 Ab and CpG oligodeoxynucleotides (ODNs) can enhance their ability to promote Ag-specific Ab responses. 30 Considering the above evidence, and given the rapid nature of the iNKT cell response, it is conceivable that iNKT cells may also respond to direct TLR stimulation through modulation of their cytokine output. Indeed, our previous findings have indicated that direct co-stimulation of mouse iNKT cells with TLR ligands affects their cytokine output, including their ability to activate neighboring cells, such as DCs. 4
The expression and function of TLRs in iNKT cells are poorly understood, particularly after stimulation with IFN-α, which has been shown to have numerous effects on T cell activity, including TLR expression and IFN-γ production.16,27,31 The objective of this study was to investigate the role of TLR expression and function in iNKT cells primed with IFN-α. Owing to their role in detecting viral-derived nucleic acid PAMPs, the TLRs investigated were TLRs 3, 7 and 9. 32 We hypothesized that previously reported TLR expression and cytokine responses can be significantly enhanced by exogenous IFN-α. To that end, we observed that, following treatment with IFN-α, DN32.D3 cells, a well-characterized mouse iNKT cell hybridoma, exhibited the ability of prolonged cytokine production in a Th1-biased manner, as measured by the ratio of IFN-γ to IL-4 in the cell culture supernatant. IFN-α-treated DN32.D3 cells also demonstrated an enhanced ability to respond to direct treatment with TLR ligands. We also demonstrated the potential for iNKT cells exposed to TLR agonists and IFN-α to act as indirect mediators of innate responses of other cell types, such as macrophages and fibroblasts. Therefore, iNKT cells may have the capacity to respond directly to certain TLR ligands in an IFN-α-dependent manner.
Materials and methods
Cell lines
Mouse Vα14-Jα18+iNKT cell hybridomas DN32.D3, N38.2C12 and N38.3C32,33 were maintained in RPMI 1640 medium (Gibco, Carlsbad, CA, USA) supplemented with 10% heat-inactivated FBS (Gibco), 1% penicillin/streptomycin (Pen/Strep), 1% Minimum Essential Medium (MEM) non-essential amino acids (Gibco), 0.1% 0.1 M sodium pyruvate (Gibco) and 0.00035% 2-mercaptoethanol (Fluka, St. Louis, MO, USA). iNKT cell hybridomas were used as a pure source of iNKT cells as opposed to bulk populations containing iNKT cells and other cell types, such as DCs or macrophages, that express high levels of TLRs. Off-target TLR activation of even a small number of APCs could have greatly hindered our objective to investigate direct TLR activation of iNKT cells; thus, it was favorable to use a pure iNKT cell hybridoma population.
The mouse monocyte/macrophage cell line RAW264.7 was maintained in complete RPMI 1640 with 10% FBS and 1% Pen/Strep. The mouse fibroblast cell line L-929 was kindly provided by Dr. Douglas Hodgins (University of Guelph, Guelph, ON, Canada) and were maintained in complete RPMI. Vero cells were maintained in EMEM balanced with Earle’s salts (Hyclone, Rockford, IL, USA) supplemented with 10% FBS and 1% Pen/Strep. 293T cells were maintained in DMEM (Gibco, Carlsbad, CA, USA) supplemented with 10% FBS and 1% Pen/Strep.
Virus
VSV expressing enhanced GFP (VSV-GFP) was propagated using 293T cells. 34 Titration of VSV-GFP was performed by plaque assay using Vero cells.
Stimulation of iNKT cell hybridomas
For stimulation of the TCR signaling pathway of DN32.D3 cells, anti-CD3 mAb (Clone eBio500A2; eBioscience, San Diego, CA, USA) was used to coat the wells of 48-well cell culture plates at 4℃ overnight. Concentrations of 10, 100 and 1000 ng/ml were tested in combination with 100 or 1000 U/ml of recombinant (r) IFN-α2 (Cat#14-8312-80; eBioscience) to determine the optimal concentration required for maximum expression of TLR by iNKT cells. Wells were then washed twice with PBS immediately before iNKT cell hybridomas were seeded at a density of 5 × 105 cells/well. For iNKT cell stimulation with α-galactosylceramide (α-GalCer) (Cedarlane Labs, Brockville, ON, Canada), medium was supplemented with 500 ng/ml of α-GalCer. α-GalCer and TLR ligands were suspended in the cell culture medium at the indicated concentrations. Cultures were then incubated at 37℃ in 5% CO2 for the indicated treatment period.
DN32.D3 iNKT cells were stimulated with TLR ligands 24 h after stimulation with anti-CD3 mAb and rIFN-α2, an active form of IFN-α (eBioscience). Cells were treated with TLR ligands at the following concentrations used previously: 4 25 µg/ml TLR3 ligand polyinosinic-cytidylic acid [poly (I:C)] (Sigma, St. Louis, MO, USA); 1 µg/ml TLR7/8 ligand R848; 10 µg/ml TLR9 ligand ODN2395 (CpG); 10 µg/ml TLR9 ligand control ODN2395C (CpGCx) (Invivogen, San Diego, CA, USA).
To assess cytokine production by iNKT cells, they were simultaneously stimulated with anti-CD3 mAb and rIFN-α2 for 24 h. Cells were then transferred to new plates that were not coated with anti-CD3 mAb, and then cultured in the presence of TLR ligands for 18 h. Supernatants were then collected for cytokine quantification by ELISA.
In experiments in which DN32.D3 cell supernatants were used to stimulate other cells, DN32.D3 cells were simultaneously stimulated with anti-CD3 mAb and rIFN-α2 for 24 h. Cells were then transferred to fresh culture plates that were not coated with anti-CD3 mAb, and then cultured in the presence of TLR ligands for 4 h. TLR ligands were then removed by washing the cells twice with PBS. Cells were then cultured for another 24 h before supernatants were collected for treating either RAW264.7 cells or L-929 cells.
RNA extraction, cDNA synthesis and real-time PCR
Total RNA was isolated from iNKT cell hybridomas using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). RNA concentration and quality were quantified by NanoDrop ND2000 (Thermo, Rockford, IL, USA). cDNA synthesis was carried out according to the Super Script First-Strand protocol (Invitrogen). cDNA samples were diluted 1:10 prior to real-time PCR.
Real-time PCR was performed according to the protocol for the Light Cycler 480 (Roche, Laval, QC, Canada). Pfaffl’s formula was used to calculate the relative expression of target genes.
35
Efficiency of the PCR amplification was calculated using the following formula: Efficiency (E) = 10−1/slope. E-Values were obtained from standard curves generated from serial dilutions of cDNA samples that were positive for the target gene. Relative gene expression of each sample was calculated using Pfaffl’s formula:
Stimulation of RAW264.7 macrophages
RAW264.7 macrophages were stimulated with supernatants derived from DN32.D3 cells stimulated with anti-CD3 mAb and rIFN-α2, and then pulsed with TLR ligands for 4 h, washed and cultured for another 24 h. Macrophages were incubated with a 1:1 ratio of growth medium and iNKT cell supernatants for 18 h. Supernatants were then collected for analysis by ELISA/enzyme immunoassay (EIA) or cells were collected for analysis by flow cytometry (described below).
ELISA
Ready-Set-Go sandwich ELISA kits for mouse IFN-γ, IL-4, TNF-α and IL-6 were purchased from eBioscience. ELISAs were performed according to the protocols supplied by the manufacturer. Plates were coated with capture Ab overnight (18 h) at 4℃. Plates were then washed and blocked with blocking buffer from the manufacturer. Samples and standards were diluted and added to the plate, and then incubated overnight at 4℃. The following day, plates were washed and detection Ab was added for 1 h, followed by streptavidin and then TMB substrate. The enzymatic reaction was stopped using a sulfuric acid stop solution according to manufacturer’s protocol. Plates were read spectrophotometrically at 450 nm with an absorbance plate reader (Powerwave XS; BioTek Instruments, Winooski, VT, USA).
Prostaglandin E2 EIA
The inflammatory mediator prostaglandin E2 (PGE2) produced by RAW264.7 macrophage cells was measured using the EIA kit obtained from Cayman Chemical (Ann Arbor, MI, USA). As per the protocol provided by the manufacturer, culture supernatant or PGE2 standard was loaded onto goat anti-mouse IgG-coated strip wells. PGE2–acetylcholinesterase-conjugated tracer was then added to each of the wells containing samples or standards, followed by PGE2 mAb. Strip wells were incubated overnight at 4℃. Following the overnight incubation, wells were washed with wash buffer. Ellman’s reagent was then added to each well. The plate was then allowed to develop in the dark at room temperature (23℃). The absorbance of each sample was measured at 412 nm using the Powerwave XS (BioTek Instruments) plate reader.
Flow cytometry
To analyze surface expression of TLR3 by DN32.D3 cells, cells were stimulated with α-GalCer or anti-CD3 mAb and rIFN-α2 for 24 h. Cells were washed with PBS and then re-suspended in FACS buffer (PBS with 0.1% sodium azide and 1% BSA). Cells were first treated with anti-CD16/32 (eBioscience) to block possible nonspecific binding to Fc receptors that may be expressed by iNKT cells, as both NK and T cells can express Fc receptors.36,37 After two washes, cells were stained with unconjugated rat anti-mouse TLR3 IgG2a Ab (clone 313129; R&D Systems, Minneapolis, MN, USA) or rat IgG2a isotype control (eBioscience) at room temperature for 1 h. This was followed by two additional washes in FACS buffer, then the addition of phycoerythrin (PE)-conjugated mouse anti-rat IgG2a (clone R2a-21B2; eBioscience) for 1 h at room temperature. Cells were fixed with 2% paraformaldehyde (Fisher, Rockford, IL, USA) after two final washes in FACS buffer. Data were acquired using a FACScan flow cytometer (BD Biosciences, Mississauga, ON, USA). Data were analyzed using FlowJoversion 7.6.5 flow cytometry analysis software (Tree Star, Ashland, OR, USA).
For the detection of CD80 on RAW264.7 cells, cells were collected and washed in PBS and re-suspended in FACS buffer. Cells were then treated with anti-CD16/32, and washed and re-suspended in FACS buffer. Cells were stained with FITC-conjugated Armenian hamster anti-mouse CD80 (clone 16-10A1; eBioscience). Cells were washed and re-suspended in FACS buffer and fixed with 2% paraformaldehyde. Data were collected with a FACScan flow cytometer (BD Biosciences). Data analysis was performed using FlowJo flow cytometry analysis software (Tree Star).
Phagocytosis assay
Phagocytic activity of RAW264.7 cells was measured using a Phagocytosis Assay Kit (Cayman Chemical), as per the manufacturer’s instructions. Fluorescence was measured using a fluorescent plate reader (GloMax-multi microplate multimode reader; Promega, Madison, WI, USA) with an excitation of 490 nm and fluorescence detected at 535 nm. To control for background fluorescence, the fluorescence values from bead-negative cell cultures were averaged and subtracted from the values obtained from experimental samples.
VSV-GFP antiviral bioassay
L-929 cells were seeded in 12-well plates and grown to confluence. They were then treated with DN32.D3 cell supernatants or rIFN-α2 for 6 h. L-929 cells were then infected with VSV-GFP at a multiplicity of infection (MOI) of 1 and incubated for 12 h before they were washed and re-suspended in FACS buffer prior to analysis by flow cytometry. Data were analyzed with FlowJo software (Tree Star). Negative control cells, which did not receive any virus, were not fluorescent, while cells populations that were supportive of VSV-GFP replication fluoresced in the green spectrum. Cells that were pre-treated with rIFN-α2 prior to infection as a positive control demonstrated dose-dependent antiviral activity.
Statistical analysis
Statistical significance was examined using the Student’s t-test. In addition, statistical significance was also examined using ANOVA followed by Dunnett’s post hoc test, which corroborated the results from the t-test; therefore, only statistical significance from the t-test is shown in the appropriate figures. Comparisons were considered statistically significant when P ≤ 0.05. Each experiment was repeated at least 4–6 times in its entirety or partially; in each experiment, samples were tested in triplicate. The data shown in each figure are representative of one experiment.
Results
Anti-CD3 stimulation of NKT cell hybridomas induces the expression of IFN-α receptor mRNA
To investigate the possibility that activated iNKT cells could become susceptible to type I IFN stimulation, we first asked whether three different iNKT cell hybridomas—N38-2C12, N38-3C3 and DN32.D3—expressed the type I IFN receptor, IFNAR. Paget et al.
26
have demonstrated a key role for IFN-α/β receptor alpha chain (IFNAR1) in the iNKT cell response to type I IFN derived from TLR-stimulated DCs; thus, we decided to focus on IFNAR1 gene expression by iNKT cell hybridomas. Each hybridoma was stimulated with three doses of plate-bound anti-CD3 mAb and two different doses of mouse rIFN-α2. There was significant up-regulation of IFNAR1 expression after treating two of the three iNKT cell lines with 100 ng/ml anti-CD3 mAb. There was further dose-dependent up-regulation of IFNAR1 expression with the addition of rIFN-α2 (Figure 1A). This suggested that IFN-α stimulation of iNKT cells may operate in a positive feedback loop in which activation of the IFNAR1 results in its elevated expression, and also that triggering CD3 can lead to increased IFNAR1 expression.
IFNAR1 and TLR expression by iNKT cell hybridomas in response to anti-CD3 mAb and rIFN-α2. N38-2C12 (2C12), N38-3C3 (3C3) and DN32.D3 (DN32) iNKT cell hybridomas were simultaneously stimulated with rIFN-α2 or medium control and either immobilized anti-CD3 mAb (A–E, G) or solubilized αGalCer (F) in the cell culture medium for 18 h. (A–E) Real-time PCR was performed on cDNA samples to quantify relative gene expression. (F, G) DN32.D3 cells were cultured for 18 h with either (F) α-GalCer (500 ng/ml) or (G) anti-CD3 mAb (1000 ng/ml) in the presence of rIFN-α2 (1000 U/ml). Surface expression of TLR3 was analyzed using flow cytometry. The dead cells, as well as doublets, were excluded by adjusting the scatter voltage and gating strategy using height and width of the forward and side scatters. Data shown represent results from one experiment. Bars are representative of the mean value of the triplicate cultures. Error bars represent the SEM. *Statistical significance determined by Student’s t-test (P ≤ 0.05). #Statistically significant effect (P ≤ 0.05) due to anti-CD3 mAb alone.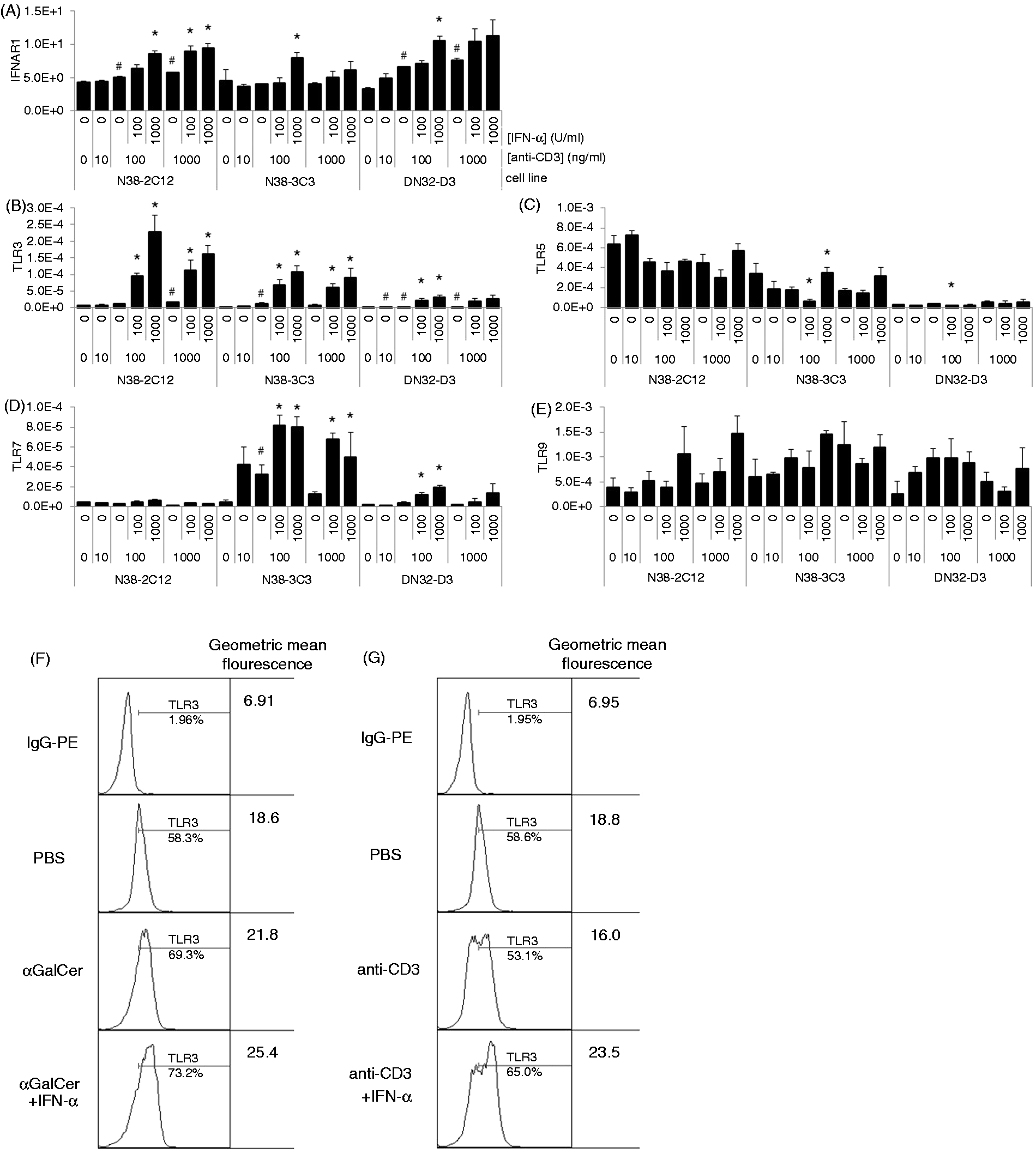
IFN-α significantly enhances the expression of select TLRs in iNKT cell hybridomas
To confirm the expression of TLR transcripts by iNKT cells, we stimulated N38-2C12, N38-3C3 and DN32.D3 cells with anti-CD3 mAb. Transcripts for TLRs 3, 5, 7 and 9 were found in all three hybridomas, supporting previously reported TLR expression in DN32.D3 cells, 4 although the transcription of TLR genes was not significantly affected by the dose of anti-CD3 mAb (Figure 1B–E). TLR3 gene expression by all three hybridomas was significantly affected by rIFN-α2, with 1000 U/ml of rIFN-α2 having a significant effect on all three cell lines (Figure 1B). We observed TLR7 gene expression to be significantly affected by rIFN-α2 in two of the three cell lines when co-stimulated with 100 ng/ml anti-CD3, particularly at doses of 100 and 1000 U/ml rIFN-α2 (Figure 1D). We observed an effect of rIFN-α2 on TLR9 gene expression (P < 0.05); however, there were no significant differences between rIFN-α2 treatment and control groups (Figure 1E). TLR5 gene expression was not significantly affected by rIFN-α2 stimulation (Figure 1C).
As robust TLR3 transcription was observed in all three cell lines when co-stimulated with anti-CD3 mAb and rIFN-α2, we investigated the effect that rIFN-α2 might have on TLR3 protein expression. We investigated TLR3 surface expression, which has previously been found to be increased in poly (I:C)-treated iNKT cells. 6 TLR3 surface protein was found to be expressed in DN32.D3 cells stimulated with either α-GalCer or anti-CD3 mAb. This expression was further enhanced by the addition of rIFN-α2 (Figure 1F, G).
IFN-γ and IL-4 production by DN32.D3, N38-2C12 and N38-3C3 cells is dependent on the dose of anti-CD3 mAb and IFN-α
In order to assess the ability of iNKT cell hybridomas to produce cytokines, we co-stimulated iNKT hybridomas with anti-CD3 and rIFN-α2 for 18 h and analyzed the culture supernatant by ELISA. We observed a significant up-regulation of IFN-γ by DN32.D3 iNKT cells after treatment with rIFN-α2, particularly with 1000 U/ml of rIFN-α2. A similar trend was observed in the other two cell lines, although the observed changes did not reach statistical significance (Figure 2A). In contrast, IL-4 production was not significantly affected by rIFN-α2, except in DN32.D3 cells treated with 1000 U/ml rIFN-α2 and 1000 ng/ml of anti-CD3 mAb (Figure 2B). IL-2 was also analyzed and was found to be highly inducible by anti-CD3 mAb, confirming our previous findings;
4
however, no statistically significant effect due to rIFN-α2 was observed (data not shown). In order to quantify relative levels of IFN-γ and IL-4, an IFN-γ:IL-4 ratio was calculated by dividing the amount of IFN-γ in one supernatant sample by the amount of IL-4 in the same sample, as determined by ELISA. rIFN-α2 treatment had a significant effect on the IFN-γ:IL-4 ratio in two out of three cell lines, particularly with 1000 U/ml of rIFN-α2, suggesting that stimulation of IFNAR1 may favor an increase in IFN-γ production by iNKT cells (Figure 2C). Thus, IFN-α treatment leads to enhanced TLR expression, and can also decrease IL-4 production bias, including increasing the relative IFN-γ production. As the DN32.D3 iNKT cell hybridoma has been investigated in previous studies and has been shown to have a robust cytokine response to both TCR and TLR stimuli, we further investigated the potential effects of rIFN-α2 on this cell line.
Cytokine production by rIFN-α2-treated iNKT cell hybridomas. Anti-CD3 mAb treated N38-2C12, N38-3C3 and DN32.D3 Vα14-Jα18 iNKT cell hybridomas were cultured in the presence of rIFN-α2 or medium control and plate-bound anti-CD3 mAb for 18 h. (A–C) IFN-γ and IL-4 in supernatants were quantified by ELISA. (D) Bars are representative of the mean value of the triplicate cultures. Error bars represent the SEM. *Statistically significant effect of rIFN-α2 compared with cells treated with only the same dose of anti-CD3 mAb, as determined by Student’s t-test (P ≤ 0.05). #Statistically significant effect (P ≤ 0.05) due to anti-CD3 mAb alone compared with PBS-only controls.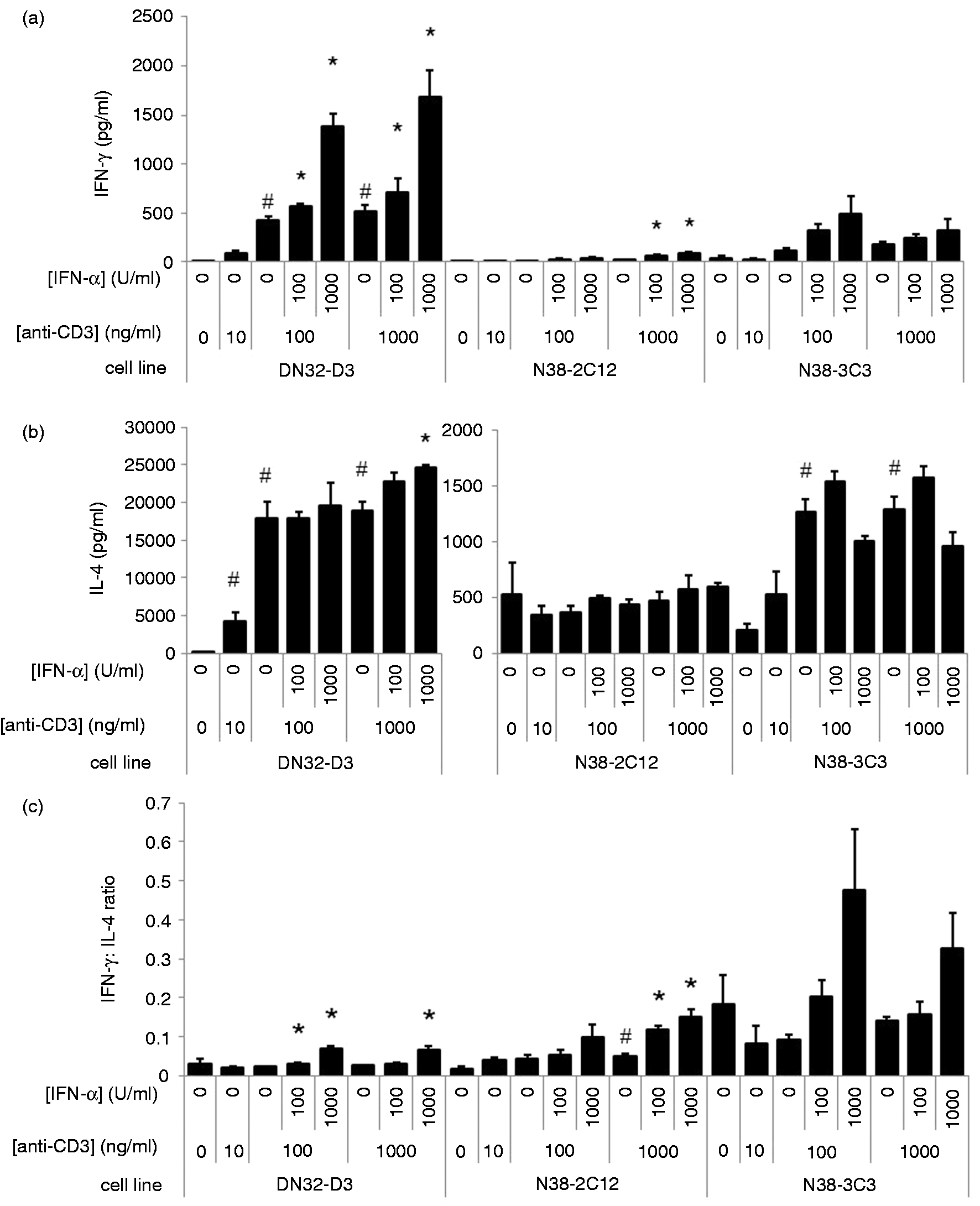
IFN-γ, IL-4 and TNF-α production is enhanced in iNKT cells treated with IFN-α and anti-CD3 mAb followed by TLR-Ls
To determine the functionality of iNKT cell-expressed TLR proteins, DN32.D3 cells were stimulated with anti-CD3 mAb and rIFN-α2 for 18 h. These iNKT cells were then incubated in fresh culture plates lacking anti-CD3 mAb and rIFN-α2 in medium containing one of four TLR-Ls or controls [poly (I:C), R848, ODN2395, ODN2395C and PBS control] for another 24 h. The dose of each TLR ligand used has been described in an earlier study.
4
In addition to the previously described up-regulation of IFN-γ and IL-4 (Figure 2), a significant up-regulation of TNF-α production was observed following the treatment of iNKT cells with anti-CD3 and rIFN-α2 (Figure 3A–C). Stimulation of iNKT cells with TLR-Ls was found to have a significant effect upon production of IFN-γ and IL-4 by these anti-CD3- and IFN-α2-treated cells. The increase in IFN-γ by TLR-Ls was found to be statistically dependent upon the addition of rIFN-α2 to the culture supernatant, regardless of the dose of anti-CD3, suggesting that the effects of TLR-Ls on IFN-γ production are dependent on the effects of rIFN-α2. In contrast, while all TLR-Ls enhanced IL-4 and TNF-α production by 10 or 1000 ng/ml anti-CD3 + 1000 U/ml rIFN-α2 co-stimulated iNKT cells, only CpG significantly enhanced IL-4 and TNF-α production by iNKT cells co-stimulated with 100 ng/ml of anti-CD3 + 1000 U/ml rIFN-α2 (Figure 3B, C). In addition, TLR-Ls also had a significant effect on the IFN-γ:IL-4 ratio when iNKT cells were stimulated with 10 ng/ml anti-CD3, indicating that TLR-Ls can shift the balance between IFN-γ and IL-4 production, especially when iNKT cells were pre-treated with 10 ng/ml of anti-CD3 (P < 0.05) (Figure 3D). These results suggest that TCR and IFNAR1 stimulation work in tandem to sensitize iNKT cells to stimulation with TLR ligands.
Cytokine production after TLR ligation of DN32.D3 cells pre-treated with rIFN-α2 and anti-CD3 mAb. iNKT cells were simultaneously treated with rIFN-α2 or medium control and anti-CD3 mAb for 18 h. Cells were then washed and plated in fresh culture plates in growth medium supplemented with TLR ligands for 24 h. Supernatants were collected and IFN-γ, TNF-α and IL-4 were quantified by ELISA. Bars are representative of the mean value of the triplicate cultures. Error bars represent the SEM. *Statistically significant effect due to TLR-L treatment compared with cells treated with the same dose of anti-CD3 mAb and rIFN-α2, as determined by Student’s t-test (P ≤ 0.05). #Statistically significant effect (P ≤ 0.05) due to rIFN-α2 alone compared with PBS-only controls.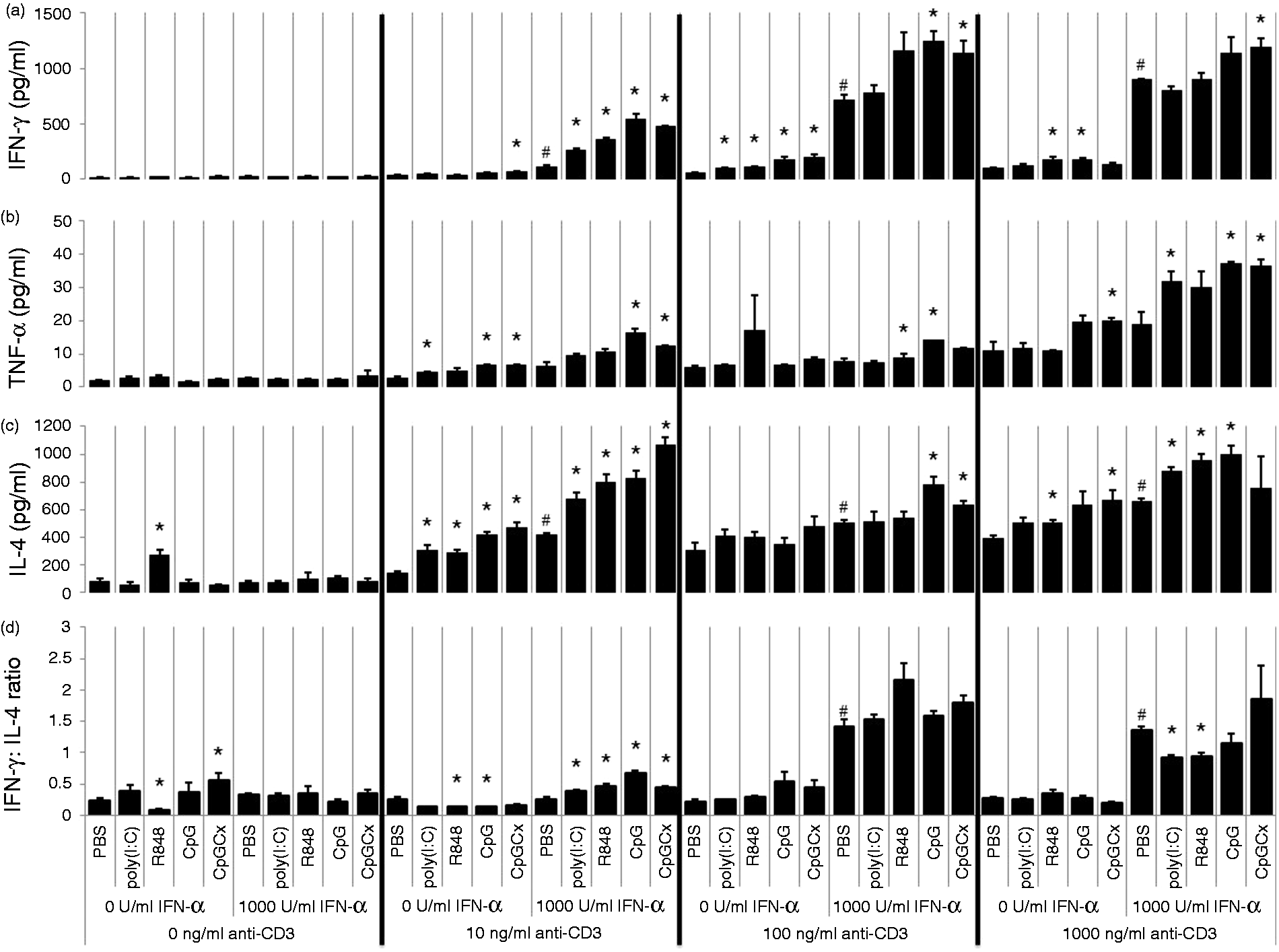
IFN-γ production by iNKT cells is prolonged after treatment with IFN-α and anti-CD3 mAb
We next sought to determine the impact that cytokines from TLR-treated iNKT cells have on other cell types. However, when assessing the ability of iNKT cells to communicate with other cell types through secreted cytokines, we found it necessary to ensure that the supernatants used were free of contaminating TLR-Ls, as they are capable of eliciting responses in many cell types and could mask the effects of iNKT cell-derived cytokines. To address this problem, iNKT cells were stimulated with anti-CD3 mAb and rIFN-α2 for 18 h before they were moved to a fresh culture plate lacking anti-CD3 mAb, and incubated with TLR-Ls for 4 h. The cells were then washed to remove TLR-Ls and incubated in standard growth medium for another 24 h before culture supernatants were collected. As expected, overall IFN-γ production was lower than in experiments in which iNKT cells were incubated with TLR-Ls for a full 24 h; however, IFN-γ production was sustained even after the removal of stimuli, and iNKT cells that were pulsed with TLR-Ls continued to produce significantly more IFN-γ than control cells that were likewise stimulated with anti-CD3 mAb and rIFN-α2, but not exposed to TLR ligands (Figure 4A). Thus, after transient exposure to TLR ligands, iNKT cells continued to produce cytokines that could potentially influence the function of other cell types. Therefore, all further experiments were carried out using iNKT cells pre-treated with anti-CD3 mAb and rIFN-α2 prior to the addition of TLR ligands, followed by removal of TLR ligands from the culture supernatant in order to minimize the possible effects of contaminating TLR ligands.
Activation of RAW264.7 cells with TLR ligand-treated DN32.D3 cell culture supernatants. iNKT cells were simultaneously treated with rIFN-α2 or medium control and 100 ng/ml of anti-CD3 mAb for 18 h. Cells were then washed and plated in fresh culture plates in growth medium supplemented with TLR ligands for 4 h. Cells were washed and then cultured for another 24 h in growth medium. (A) IFN-γ from these supernatants was quantified by ELISA. RAW264.7 cells were cultured in the presence of the DN32.D3 supernatants for 24 h and analyzed for (B) IL-6 by ELISA and (C) PGE2 by EIA. (D) In a separate experiment, RAW264.7 cells were cultured with TLR-treated DN32.D3 supernatants and FITC-conjugated latex microbeads. Phagocytic activity was quantified with a fluorescence plate reader. Bars are representative of the mean value of the triplicate cultures. Error bars represent the SEM. *Statistically significant effect due to TLR-L treatment compared with cells treated with the same dose of anti-CD3 mAb and rIFN-α2, as determined by Student’s t-test (P ≤ 0.05). #Statistically significant effect (P ≤ 0.05) due to rIFN-α2 alone compared with PBS-only controls. RFU: relative fluorescent units.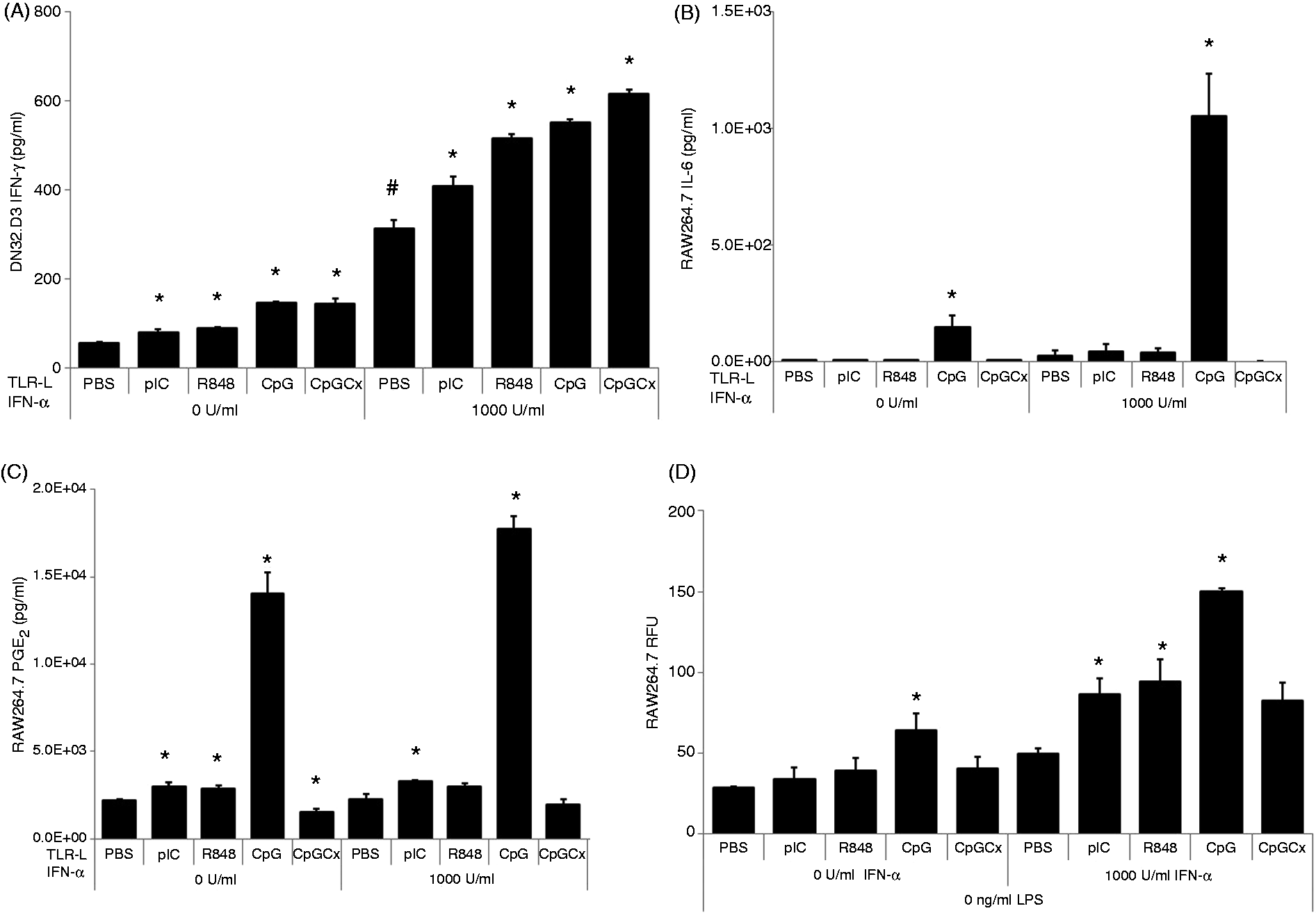
Macrophages are activated by supernatant from TLR-treated iNKT cells
To examine the possibility that TLR-activated iNKT cells can activate macrophages, iNKT cell culture supernatants (Figure 4A) were used to stimulate the mouse macrophage cell line RAW264.7 for 24 h. Supernatant derived from iNKT cells treated with TLR-Ls with or without IFN-α pre-treatment showed a significantly greater capacity than PBS controls to stimulate macrophages (Figure 4B–D). IL-6 and PGE2 were measured as different indicators of macrophage activation as they could be potentially produced in response to iNKT cell-derived cytokines, such as IFN-γ or TNF-α.38,39 Mouse macrophages showed enhanced secretion of IL-6 and PGE2 after treatment with supernatant from TLR-L-treated iNKT cells (Figure 4B, C). Supernatant from CpG-stimulated iNKT cells had the most potent effects on macrophages among all the TLR-Ls tested, despite the fact that CpG-stimulated and other TLR-L-stimulated iNKT cells produced similar amounts of IFN-γ (Figure 4A).
To investigate the ability of iNKT cells to induce phagocytic activity in macrophages, this parameter was assessed in macrophages after stimulation with supernatant from TLR-stimulated iNKT cells. In addition to iNKT cell supernatant, macrophages were cultured in the presence of fluorescent latex beads; their uptake of these beads was an indicator of phagocytic activity. Poly (I:C), R848 and CpG significantly affected the ability of iNKT cells to induce latex bead uptake by macrophages, with CpG having the greatest effect (Figure 4D).
Next, we sought to determine if supernatant from TLR-treated iNKT cells could induce macrophages to express elevated levels of CD80, which is important for T cell co-stimulation. Macrophages stimulated with supernatant from TLR-L-treated iNKT cells expressed higher amounts of CD80 on their cell surface (Figure 5). Supernatant from CpG-treated iNKT cells, which were first co-stimulated with anti-CD3 + rIFN-α, was the most potent in inducing CD80 expression on macrophages, boosting expression from 8% by supernatant from PBS-treated iNKT cells to 23% by the CpG-treated iNKT cells (Figure 5).
CD80 expression by RAW264.7 cells after treatment with TLR-triggered DN32.D3 cell culture supernatants. DN32.D3 cells were simultaneously treated with rIFN-α2 or medium control and 100 ng/ml anti-CD3 mAb for 18 h. Cells were then washed and plated in fresh culture plates in growth medium supplemented with TLR ligands for 4 h. Cells were washed and then cultured for another 24 h in growth medium. Supernatants were collected and then RAW264.7 cells were cultured in the presence of the DN32.D3 supernatants for 24 h. Cells were stained with anti-CD80 mAb, and cell populations were analyzed using flow cytometry. The dead cells, as well as doublets, were excluded by adjusting the scatter voltage and gating strategy using height and width of the forward and side scatters. Positive staining of the remaining population was determined by the upper two percentile of unstimulated control cells. The dotted line represents the isotype control. The gray line represents CD80 expression by RAW264.7 cells stimulated with supernatant from DN32.D3 cells stimulated with anti-CD3 alone. The black line represents the expression due to treatment with supernatant from rIFN-α2 - and/or TLR-L-treated DN32.D3 cells. MFI: median fluorescent intensity.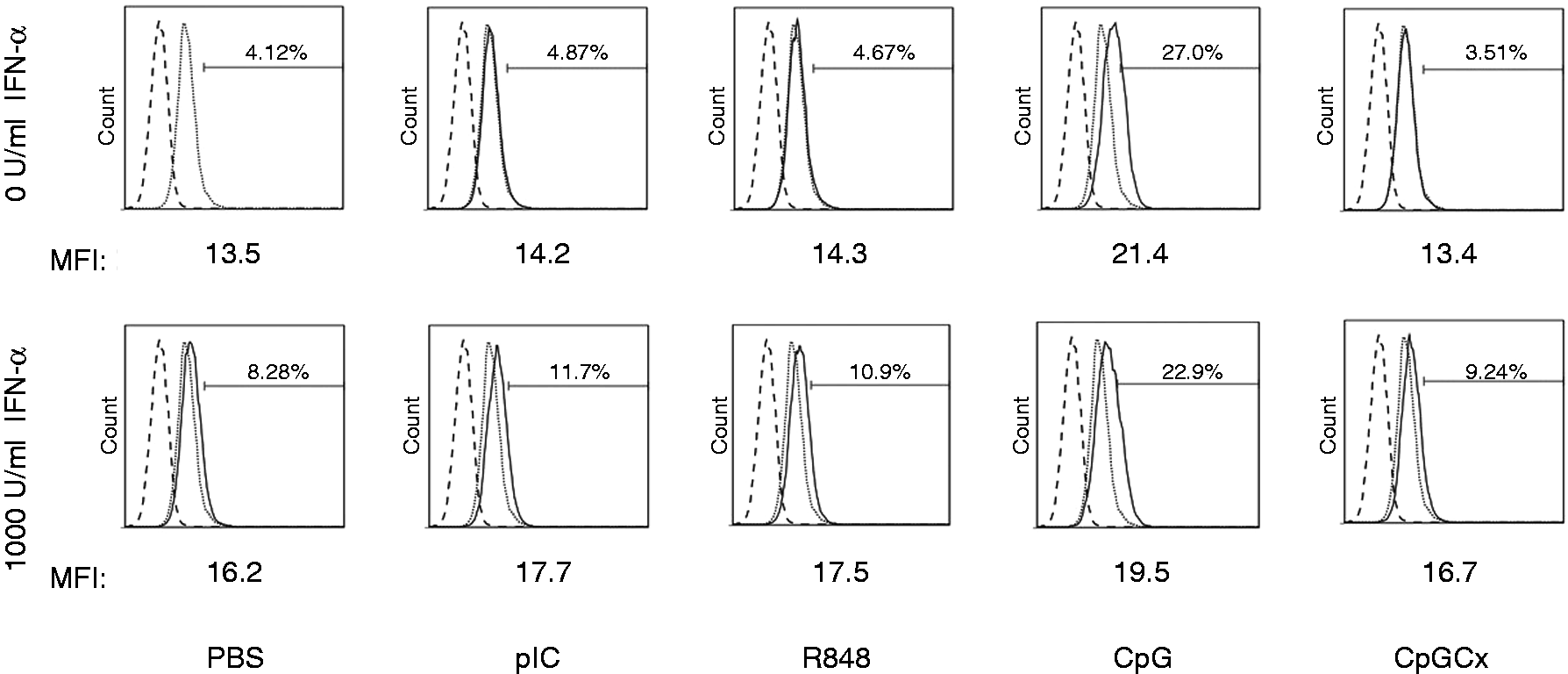
Antiviral activity of iNKT cells is modulated by TLR ligands
As iNKT cells are involved in early cytokine responses to some viral infections,
40
these cytokines may be involved in mediating antiviral activity in neighboring cells. Culture supernatant from iNKT cells treated with anti-CD3 mAb and poly (I:C) or R848 had negligible antiviral activities in the VSV bioassay. Culture supernatant from iNKT cells treated with anti-CD3mAb and CpG or CpGCx had modest protective effects, similar to direct stimulation of fibroblasts with 1 U/ml rIFN-α2 (Figure 6). Anti-CD3 mAb- and rIFN-α2-treated iNKT cells produced supernatant that was more protective, roughly equivalent to 10 U/ml of rIFN-α2 (51.2% of cells were fluorescent) (Figure 6). This protective effect was somewhat enhanced in supernatants from iNKT cells treated with poly (I:C) (45.1%), R848 (45.1%), CpG (40.0%) or CpGCx (43.5%). The results demonstrate the capacity for anti-CD3 mAb- and rIFN-α2-treated iNKT cells to activate innate antiviral mechanisms, as well as a possible role for TLR-Ls in acting as a third signal, in addition to TCR triggering and IFN-α, which can promote the antiviral responses of iNKT cells.
Antiviral activity of cell culture supernatants from rIFN-α2 - and TLR ligand-treated DN32.D3 cells. DN32.D3 cells were simultaneously treated with rIFN-α2 or medium control and 100 ng/ml anti-CD3 mAb for 18 h. Cells were then washed and plated in fresh culture plates in growth medium supplemented with TLR ligands for 4 h. Cells were washed and then cultured for another 24 h in growth medium. Supernatants were collected and used to treat L-929 cells for 6 h. L-929 cells were then infected with VSV-GFP at MOI = 1 for 12 h and then analyzed by flow cytometry. Some L-929 cell cultures were treated directly with rIFN-α2 instead of DN32.D3 supernatants as a positive control for antiviral activity. Cell populations infected with VSV-GFP were detectable owing to the expression of viral enhanced GFP. As a control, L-929 fibroblasts were cultured with increasing concentrations of rIFN-α2 (left panels) to inhibit viral replication. Fibroblasts treated with supernatant derived from DN32.D3 cells stimulated as above are represented in the right grid. Gate for fibroblasts positive for VSV-GFP replication was set based on the upper 2% of the virus-negative control. The experiment was performed three times in its entirety or partially. Data shown are representative of one experiment.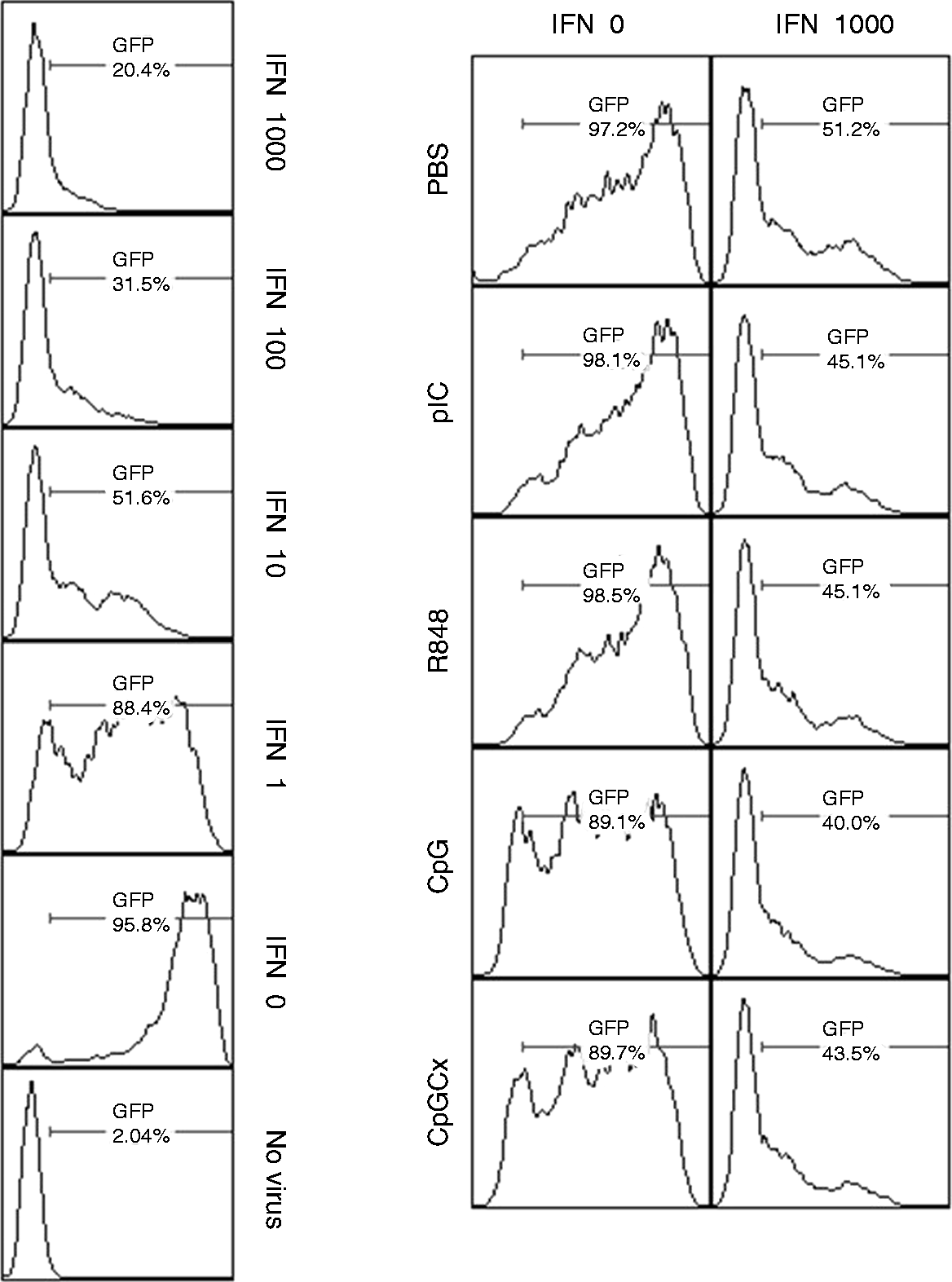
Discussion
This study demonstrates the inducible expression of TLRs by multiple iNKT cell lines following pre-treatment with IFN-α and anti-CD3 mAb. Recent studies have demonstrated that primary murine iNKT cells are, indeed, capable of expressing functional TLR3 and TLR4 protein; for instance, activation of TLR3 expressed by iNKT cells was suggested to enhance iNKT cell-mediated suppression of hepatic γδ T cells. 6 In a more recent study, direct stimulation of sorted iNKT (α-GalCer/CD1d tetramer+) cells with LPS resulted in increased IFN-γ production, but suppressed IL-4 production. 7 It was also suggested that iNKT cells constitutively express TLR4; however, this is uncertain as the cells were sorted using α-GalCer/CD1d tetramer, which can, at least partially, stimulate the Vα14-Jα18 TCR, casting doubt on whether TLR4 expression is truly constitutive or is actually inducible by TCR stimulation. 7 In fact, this is partly why, in this study, iNKT cell hybridomas were used as a model for iNKT cells—they can be cultured in the absence of TCR stimuli and without any requirement for accessory cells that might express high levels of TLRs themselves.4,41
We observed that IFN-α stimulation alone was insufficient to induce TLR expression by iNKT cell hybridomas DN32.D3, N38-2C12 and N38-3C3. However, when combined with TCR triggering, IFN-α significantly enhanced the expression of some, but not all, TLR genes. It has been established that at least some of the effects of direct stimulation of T cells through IFNAR1 are dependent on molecules that are also involved in the TCR signaling cascade, such as Zap70, Lck and CD45, 42 and it has been proposed that IFNAR1 and TCR signaling are linked through ERK1/2 MAPK signaling. 43
We observed that anti-CD3 and IFN-α co-stimulation induced the expression of two RNA-sensing TLRs associated with viruses, namely TLRs 3 and 7. The expression of TLR9, which recognizes CpG DNA, was not significantly affected by IFN-α treatment. However, the lack of modulation of expression of TLR9 did not appear to hinder the direct effects of the TLR9 ligand, CpG ODN2395, on DN32.D3 iNKT cells.
As the expression of TLR3 and TLR7 was dependent on IFN-α co-stimulation, it should also be noted that IFNAR1 expression was significantly increased in iNKT cell hybridomas by the triggering of CD3 alone. This suggests that iNKT cells become more sensitive to type I IFN stimulation upon TCR activation, which could further sensitize them to stimulation with TLR-Ls.
Primary human iNKT (Vα24 -Vβ11+) cells have been shown to express nearly all known TLR genes, with the exception of TLR8 5 . However, these primary human iNKT cells were not able to respond directly to TLR-Ls following activation with sub-optimal concentrations of IL-2, and a combination of anti-CD3 and anti-CD28 mAbs. Rather, TLR-Ls were shown to be crucial in activating APCs, which could then activate iNKT cells. This has also been demonstrated in a mouse model in which iNKT cell activation was shown to depend on a combination of glycolipid Ag presentation and type I IFN secretion by TLR-activated DCs, rather than stimulation through their own TLRs.26,44 Similar to primary human iNKT cells, mouse DN32.D3 iNKT cells did not significantly increase cytokine production in response to most TLR ligands after pre-stimulation with anti-CD3 mAbs alone, as reported previously. 4 However, peptidoglycan, a NOD ligand, was found to have a suppressive effect on DN32.D3 cells, which significantly decreased production of IL-2, IL-4 and IFN-γ. In addition, treatment with CpG ODN2395 resulted in a two- to threefold increase in IL-2 production, but had no effect on the levels of IL-4 or IFN-γ. 4 The lack of responsiveness to most TLR ligands may be attributed to the fact that these cells were lacking IFNAR1 stimulation, which has been shown to have significant effects on the expression of some TLRs in conventional T cells. 16 Hence, we report a significant effect of IFN-α on iNKT cell behavior. In this regard, the production of IFN-γ and IL-4 was increased in DN32.D3 cells stimulated with anti-CD3 mAb and rIFN-α2. However, IFN-γ production had a greater relative increase than IL-4 when compared with DN32.D3 cells stimulated with anti-CD3 mAb alone. This suggests that type I IFNs serve to depolarize iNKT cell cytokine production, shifting the balance between IFN-γ and IL-4 production. Using this model, we also demonstrate that a consequence of iNKT cell activation with type I IFN and TCR agonists may include increased TLR expression and function.
Previous work has demonstrated that iNKT cell activation can be mediated by type I IFN in the context of a mouse cytomegalovirus (MCMV) infection model. 26 In this model, DCs infected with MCMV were stimulated, through TLR9, to produce IFN-β. This IFN-β enabled iNKT cells to produce IFN-γ, but not IL-4, altering the cytokine output of iNKT cells towards a Th1-like response. In 2007, Paget et al. 26 also observed that a combination of type I IFN-mediated stimulation and the recognition of host glycolipids presented by infected DCs synergistically activates iNKT cells.
The effects of type I IFNs on iNKT cell activation are somewhat similar to those upon conventional T cell activation, as IFN-α has been shown to have a profound influence on the behavior of conventional CD4+ T cells, promoting a Th1 phenotype in the biased production of IFN-γ over IL-4. 27 In turn, this could have an impact on their ability to respond to TLR ligands. Indeed, there is evidence that TLR expression and function differ between subtypes of CD4+ T cells. For instance, TLR2 ligands have been shown to activate directly Th1 cells, but not Th2 cells. 29 In addition, naïve CD4+ T cells activated with anti-CD3 mAb and IFN-α have been demonstrated to express increased TLR2 and TLR4 at the mRNA and protein levels. 16 Thus, type I IFNs have been demonstrated to promote the Th1 phenotype, as well as TLR expression and function in conventional CD4+ T cells.
An important finding of the present study was that culture supernatants of iNKT cells that were co-stimulated with anti-CD3 and IFN-α before they were treated with TLR ligands induced the activation of macrophages, as judged by an increase in IL-6 and PGE2 output, uptake of fluorescent beads and surface expression of CD80. The potent activation of macrophages by CpG-stimulated iNKT cells after co-stimulation with anti-CD3 and IFN-α is of particular interest as supernatant from the iNKT cells demonstrated only modest increases in IFN-γ production compared with anti-CD3 and IFN-α co-stimulated iNKT cells treated with poly (I:C), R848 or non-CpG ODN 2395C. Despite this, we observed that CpG-stimulated iNKT cell supernatant was able to activate macrophages to a significantly greater extent compared with other TLR ligands. This was somewhat unexpected because IFN-γ is a major inducer of classical macrophage activation; 39 thus, we initially predicted that the degree of macrophage activation would correspond more closely with IFN-γ levels measured in the supernatant.45–47 This suggests the possibility of the existence of another mechanism by which macrophages are activated in addition to iNKT cell-derived IFN-γ, such as TNF-α or IL-4. 48
Indeed, macrophages can be alternately activated by IL-4, resulting in reduced antimicrobial activities and increased function conducive to tissue repair. 39 Upon activation, DN32.D3 cells produce appreciable quantities of IL-4 in addition to IL-2 and IFN-γ. The ratio of IFN-γ to IL-4 was modulated by TLR activation; thus, it is possible that this ratio may affect the manner in which RAW264.7 cells were activated. However, this does little to explain the particularly potent effects of CpG on iNKT cells in activating macrophages.
Another possible explanation of the impact of CpG on macrophage activation is induction of TNF-α production from iNKT cells. CD3-triggered iNKT cells co-stimulated with IFN-α and then treated with CpG produced significantly higher quantities of TNF-α than similarly co-stimulated cells treated with other TLR ligands, especially when sub-optimal concentrations of anti-CD3 mAb were used. In classical macrophage activation, IFN-γ potentiates the activation of macrophages by TNF-α. 39 This TNF-α can either be exogenous or produced locally as a result of TLR activation of macrophages, acting in an autocrine/paracrine manner. Therefore, it is plausible that IFN-γ and TNF-α work in concert and account for the differences observed between CpG-treated iNKT cells and those treated with other TLR ligands.
iNKT cell supernatants also exhibited significant antiviral properties. L-929 fibroblasts were rendered resistant to VSV-GFP infection by supernatants from rIFN-α2 and TLR ligand-treated DN32.D3 cells. The antiviral activity of iNKT cell supernatants was comparable with mid-range doses of direct rIFN-α2 stimulation. Therefore, it is possible that iNKT cells produce further type I IFNs in response to IFN-α stimulation. Notably, it was observed by others that hepatic iNKT cells could produce IFN-β, contributing to the priming of anti-inflammatory IL-10-producing CD8+ T cells; 49 however, a model that demonstrates a direct innate antiviral role for iNKT cell-produced type I IFNs has not yet, to our knowledge, been described. Other antiviral cytokines produced by iNKT cells, such as TNF-α, which was significantly enhanced by CpG stimulation, could also have contributed to the antiviral activity we observed. In fact, TNF-α has been shown to inhibit the replication of several viruses, including influenza A virus and hepatitis B virus.50,51 The present findings suggest a previously overlooked potential role for the activation of iNKT cells with IFN-α and TLR ligands in the context of innate antiviral responses.
Primer sequences for target genes.

Footnotes
Acknowledgements
We would like to thank Leah Read and Michael St Paul for technical advice and assistance, and Dr. Byram Bridle for editorial input. We also acknowledge Drs. Mansel Griffiths, Ali Ashkar and Byram Bridle for providing various reagents.
Funding
This work was supported by an Ontario Graduate Scholarship awarded to A Ian Villanueva.

