Abstract
TLR2 associates with TLR1 and recognizes microbial lipoproteins. Pam3CSK4, a triacylated lipoprotein, is anchored to the extracellular domain of TLR1 and TLR2 and induces pro-inflammatory signals. Here we show that C4b binding protein (C4BP), which is a complement pathway inhibitor, is a TLR2-associated molecule. Immunoprecipitation assay using anti-TLR2 mAb shows that C4BP binds to TLR2. In C4BP-deficient mice, Pam3CSK4-induced IL-6 levels were increased compared with wild type mice. In C4BP-expressing cells, Pam3CSK4-induced IL-8 production was reduced depending on the C4BP expression levels. These results reveal the important role of C4BP in negative regulation of TLR1/2-dependent pro-inflammatory cytokine production. Furthermore, using a fluorescent conjugated Pam3CSK4, we show that C4BP blocks the binding of Pam3CSK4 to TLR1/2. Finally, we show that exogenous C4BP also inhibits Pam3CSK4-induced signaling leading to IL-8 production. Our results indicate C4BP binding to TLR2 and consequent neutralization of its activity otherwise inducing pro-inflammatory cytokine production. C4BP is a negative regulator of TLR1/2 activity.
Introduction
TLRs play a crucial role in innate and adaptive immunity by recognizing diverse pathogen-derived molecules. TLRs are type I transmembrane glycoproteins. 1 Binding of ligands to the extracellular domains of TLRs causes rearrangement of the receptor complexes, 2 and triggers immune response signaling. Cell surface TLRs, including TLR1/TLR2, TLR2/TLR6 and TLR4/MD-2, recognize microbial membrane lipids, whereas TLR3, TLR7, TLR8 and TLR9 localize to intracellular organelles and recognize microbial nucleic acids.3–5 TLRs recognize not only exogenous molecules, but also endogenous molecules such as heat shock proteins, 6 high mobility group box 1, 7 soluble hyaluronan, 8 necrotic cells, 9 and so on. TLRs are important for tissue protection against pathogens, but an exaggerated response can be harmful because TLR activation by TLR ligands results in the production of pro-inflammatory cytokines such as TNF-α and IL-6. Un-regulated activation of TLRs participates in the onset of numerous diseases, such as chronic inflammation, sepsis and autoimmune disease.10,11 Therefore, TLRs activity must be highly regulated. It has been reported that deficiency or mutation of a TLR regulator, such as NOD2, results in dysregulated TLR signaling, thereby leading to disease. 12
C4b binding protein (C4BP), a plasma protein synthesized in the liver, 13 is a complement regulatory protein. C4BP is a functional cofactor for factor I-dependent degradation of C4b and C3b,14,15 and a major soluble inhibitor of the classical and lectin complement pathways. 16 Thus, C4BP protects the host’s own tissue from complement attack.
Previously, we isolated MD-2 as a TLR4-associated molecule. 17 MD-2 interacts with a leucine-rich repeat (LRR) of TLR4, and TLR4/MD-2 complex works as an LPS receptor. As the extracellular LRR of TLR2 is similar to TLR4, we hypothesized that some molecule binds to TLR2 and works as a functional molecule of the TLR2 response. In the present study, we show that C4BP binds to TLR2 and inhibits TLR1/2 ligand binding and reduces cytokine production.
Materials and methods
Reagents and plasmids
Palmitoyl-3-cysteine-serine-lysine-4 (Pam3CSK4) was purchased from EMC Microcollections (Tübingen, Germany). Human TNF-α recombinant protein was purchased from eBioscience (San Diego, CA, USA). Anti-mouse TLR2 mAb (#1-10, rat IgG2a/κ) for Western blot analysis was established in University of Tokyo (K. Miyake) by immunizing a rat with a fusion protein in which a cytoplasmic portion of mouse TLR2 was fused with glutathione S-transferase. The cDNA encoding mouse TLR2-GFP or mouse C4BP-2HA was cloned into pMXpuro (pMXpuro-mTLR2-GFP and pMXpuro-mC4BP-2HA). For transfection reagent, Polyethylenimine ‘Max’ was purchased from Cosmo Bio (Tokyo, Japan).
Cells and mice
A20 cells were cultured in RPMI-1640 medium containing 10% FBS, 50 μM 2-mercaptoethanol and penicillin–streptomycin–glutamine (ThermoFisherScientific, Waltham, MA, USA). HEK293T cells were cultured in DMEM containing 10% FBS and penicillin–streptomycin–glutamine. HEK293T cells stably expressing mouse TLR2 (293-mTLR2) and control HEK293T cells (293-vec) were established by transfection with pMXpuro-mTLR2-GFP or pMXpuro, respectively. Those cells were cultured in complete DMEM medium supplemented with puromycin (1 µg/ml). HEK293 cells stably expressing mouse TLR1 and mouse TLR2 (293-mTLR1/2) was purchased from InvivoGen (San Diego, CA, USA). 293-mTLR1/2 cells were cultured according to manufacturer’s instructions. C57BL/6 C4BP-deficient mice were provided by Dr MI Nonaka. 18
Detection and microsequencing of 25-kDa molecule
Cell surface biotinylation of A20 cells were performed as described previously. 19 Briefly, cells (∼5 × 107 cells/ml) were suspended in saline containing 100 mM HEPES (pH 8.0), and then biotinylated with sulfosuccinimidobiotin (Pierce, Rockford, IL, USA) at a final concentration of 0.5 mg/ml. After incubation for 30 min, cells were lysed and immunoprecipitated with anti-mouse TLR2 mAb (CB225) 20 or control anti-mouse mAb, respectively. The immunoprecipitates were subjected to Western blot analysis with HRP-conjugated streptavidin (GE Healthcare, Little Chalfont, UK). The co-precipitated protein was extracted, and its N-terminal amino acid sequence was determined by APRO Science (Tokushima, Japan).
Determination of cytokine concentrations
The concentration of mouse IL-6 in serum or of human IL-8 in culture supernatants was determined by ELISA with the mouse IL-6 (eBioscience) or the human IL-8 (BioLegend, San Diego, CA, USA) ELISA kit.
Immunoprecipitation
For analysis of binding of mouse C4BP to mouse TLR2, 293-mTLR2 and 293-vec were transfected with pMXpuro-mC4BP-2HA or pMXpuro. After incubation for 2 d, these cells (1 × 107 cells) were lysed in 1 ml of a lysis buffer [150 mM NaCl, 20 mM Tris-HCl (pH 7.4), 1 mM EDTA, 10% glycerol, 1% Triton X-100 and 1 × cOmplete (Roche Diagnostics, Mannheim, Germany)]. Cell lysates were then incubated with anti-GFP polyclonal Ab (pAb) (MBL, Aichi, Japan)-coupled beads at 4℃ for 2 h. For determination of C4BP expression levels in culture supernatants, 1 ml culture supernatants were incubated with anti-HA mAb (3F10; Roche Diagnostics)-coupled beads at 4℃ for 2 h. Those beads were washed three times with the washing buffer [150 mM NaCl, 20 mM Tris-HCl (pH 7.4), 1 mM EDTA, 10% glycerol, and 0.1% Triton X-100] and boiled with Laemmli sample buffer (BioRad, Hercules, CA, USA). The eluted proteins were subjected to Western blot analysis.
Detection of Pam3CSK4 binding
293-mTLR1/2 cells in 12-well plate were transfected with 600 ng of the pMXpuro-mC4BP-2HA or pMXpuro. HEK293T cells were used as negative control cell line for 293-mTLR1/2 cells. At 24 h post-transfection, transfected cells and HEK293T cells were treated with 5 µg/ml rhodamine-labeled Pam3CSK4 (InvivoGen) or medium alone. After 30 min, these cells were washed with PBS and fixed by 1% paraformaldehyde. Prepared samples were analyzed by LSM 710 confocal microscopy (ZEISS, Oberkochen, Germany). Using image analysis software Image J (http://rsb.info.nih.gov/ij/), intensity of Pam3CSK4 Rhodamine was quantified.
Flow cytometry
293-mTLR1/2 cells were transfected with a pMXpuro-mC4BP-2HA or pMXpuro. After 24 h, these cells were incubated with biotinylated anti-TLR2 mAb (CB225) or anti-TLR1 mAb (TR23) 21 for 15 min at 4℃ followed by incubation with of APC-conjugated streptavidin (BioLegend) for 15 min at 4℃. The stained cells were examined by the flow cytometer, FACSCantoII (BD Bioscience, Franklin Lakes, NJ, USA). Analysis was conducted with FlowJo 10.1r7 software (Tree Star Inc., Ashland, OR, USA).
Exogenous addition of C4BP
HEK293T cells were plated at 10 ml culture medium in a 10-cm dish and transfected with the pMXpuro-mC4BP-2HA or pMXpuro (10 µg/dish). At 2 d post-transfection, the C4BP-containing or not containing culture supernatant was collected and then 0.25 ml of it was added into 293-mTLR1/2 cells in 0.25 ml culture medium in a 24-well plate (final concentration: 50%). These cells were then stimulated with the indicated amount of Pam3CSK4 4 h after addition of supernatant.
Western blot analysis
Samples were resolved by SDS-PAGE (8%, 12% or 4–15%) and then electroblotted onto a membrane filter (Immobilon-P; Millipore, Billerica, MA, USA). The membrane was blocked in TBST containing 5% skim milk for 1 h and incubated overnight at 4℃ with biotin–anti-HA mAb (3F10; Roche Diagnostics), anti-GFP rabbit pAb, anti-TLR1 rabbit pAb (H-90; Santa Cruz, Dallas, TX, USA), anti-TLR2 rat mAb (#1-10), anti-NF-κB p65 rabbit mAb, anti-phospho-NF-κB p65 rabbit mAb, anti-p38 MAPK rabbit mAb or anti-phospho-p38 MAPK rabbit mAb (Cell Signaling, Beverly, MA, USA). The membrane was then incubated at room temperature for 1 h with HRP-conjugated streptavidin (GE Healthcare), anti-rat IgG (Invitrogen, Carlsbad, CA, USA) or anti-rabbit IgG pAb (BD Biosciences or Cell Signaling). Immunoreactive bands were visualized using the enhanced chemiluminescence ECL substrate (GE Healthcare) and a LAS4000 imaging system (GE Healthcare). Band intensity was quantified by ImageQuant TL software (GE Healthcare).
Statistical analysis
Student’s t-test was used to evaluate the significance of difference between groups. A value of P < 0.05 was considered statistically significant. All data are presented as mean ± SD.
Results
C4BP binds to TLR2
To search for TLR2-associated molecules, we conducted cell-surface biotinylated immunoprecipitation assay on mouse B-cell lymphoma A20 cell line, which expresses TLR2 endogenously. As MD-2 bound to the extracellular domain of TLR4, we expected that the TLR2-associated molecule would also be a cell-surface binding molecule. To examine the cell-surface binding molecule with TLR2 on A20 cells, biotinylated A20 cells were lysed and immunoprecipitated with anti-TLR2 mAb. As shown in Figure 1a, a prominent 25-kDa molecule was detected in TLR2 immunoprecipitate. We confirmed by Western blot analysis that the 100-kDa molecule was endogenous TLR2 using anti-TLR2 mAb (#1-10) (Supplementary Figure S1).
Mouse C4BP binds to TLR2. (a) Cell surface proteins of A20 cells were biotinylated as described in the ‘Materials and methods’. These cells were then lysed and subjected to immunoprecipitation with anti-TLR2 or control mAbs, followed by Western blot analysis with streptavidin–HRP. TLR2 and 25-kDa molecule (arrow) are indicated. (b) The determined sequence of the 25-kDa molecule (arrow) is indicated. The homology score between bovine and mouse C4BP α-chain was analyzed using Genetyx version 11 software (Genetyx Corporation, Tokyo, Japan). (c) TLR2-expressing and/or C4BP-expressing HEK293T cells were lysed and the lysates were immunoprecipitated with anti-GFP mAb, followed by Western blot analysis with anti-HA or anti-GFP Abs. Expression levels of C4BP in the supernatants were determined by immunoprecipitation and Western blot analysis with anti-HA mAb.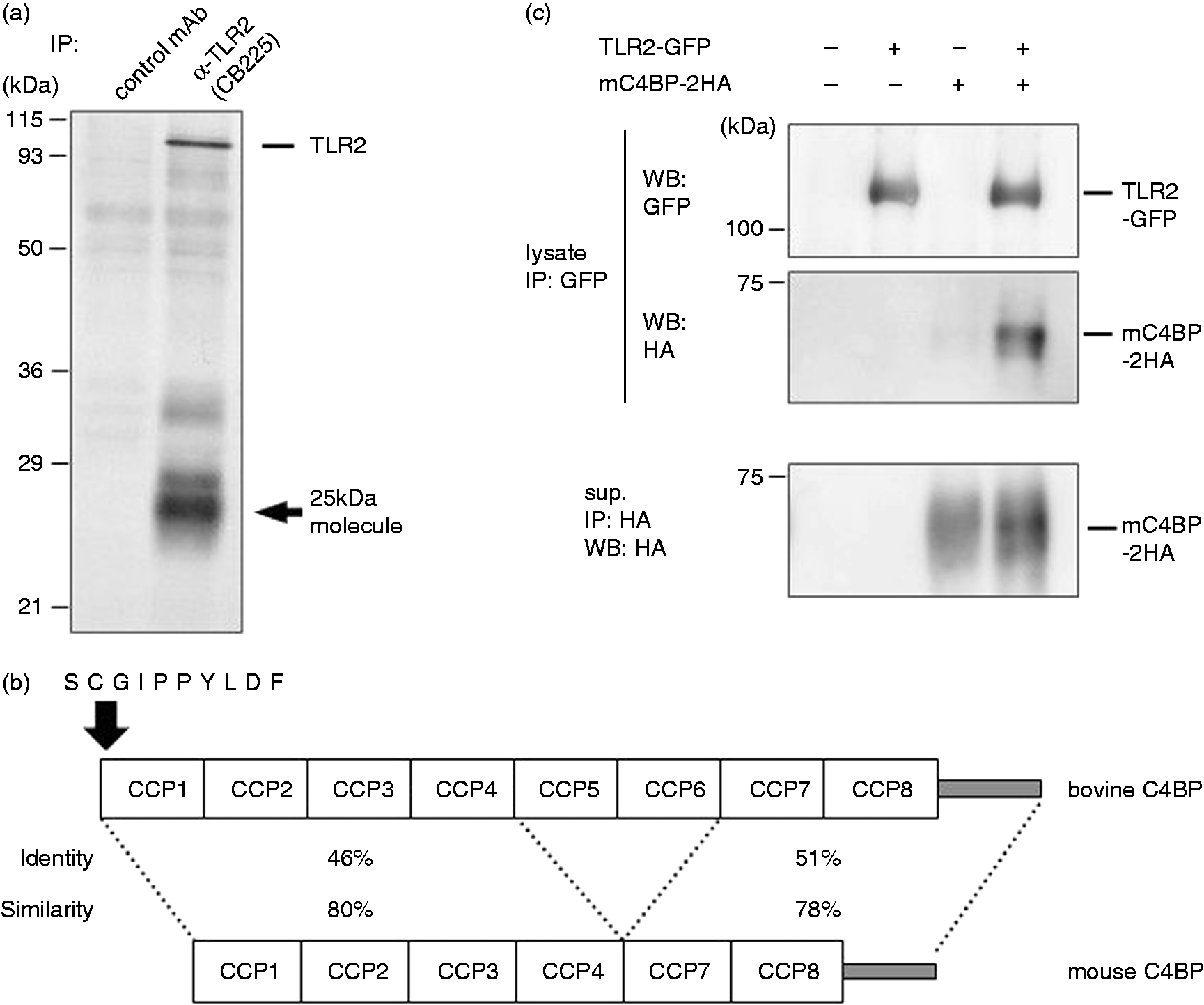
A total of 10 amino acid residues of N-terminal from the 25-kDa molecule were successfully determined, and it was completely identical to that of bovine C4BP (Figure 1b). This bovine C4BP was considered to be derived from FBS, because C4BP is a plasma protein synthesized in the liver, and A20 did not express C4BP (Supplementary Figure S2). The major form of bovine C4BP is composed of seven identical α-chains and one β-chain, and the α-chain contains eight complement control protein (CCP) domains. The determined sequence is in agreement with 1–10 amino acids of the CCP1 (Figure 1b). Because bovine C4BP α-chain is approximately 70 kDa in size, co-precipitated molecule (25 kDa) is possibly due to non-specific degradation, through manufacturing process or heat inactivation. These data corresponded to a previous report showing that 23-kDa fragment of bovine C4BP in FBS was co-immunoprecipitated with CD40. 22
Although mouse C4BP lacks both CCPs 5 and 6, mouse and bovine C4BP are both highly conserved (Figure 1b). Thus, we expected that mouse C4BP also binds to TLR2. Immunoprecipitation assay was performed on mouse TLR2-expressing and/or mouse C4BP-expressing HEK293T cells. As shown in Figure 1c, HA-tagged mouse C4BP was co-precipitated with GFP-tagged mouse TLR2. These results show that mouse C4BP, which will subsequently be termed C4BP, is a cogent candidate for mouse TLR2 binding proteins.
C4BP-deficient mice showed increased IL-6 production in response to Pam3CSK4
TLR2 in association with TLR1 or TLR6 is essential for recognizing bacterial lipoproteins and lipopeptides.23,24 These lipoproteins are anchored to the cell membrane via conserved N-termini modified by lipid chains and induce strong pro-inflammatory signals in immune cells. To study the contribution of C4BP to TLR2 response, we examined the responsiveness of C4BP-deficient mice to Pam3CSK4, which is one of the triacylated lipoproteins and TLR1/2 ligand. We injected the Pam3CSK4 in mice, and analyzed the serum IL-6 production. Serum IL-6 levels were significantly increased in C4BP-deficient mice compared with wild type mice (Figure 2). This result raises the possibility that the C4BP plays important roles in blocking Pam3CSK4-driven pro-inflammatory cytokine production.
C4BP knockout increases serum IL-6 in response to Pam3CSK4. C4BP-deficient mice (n = 8) and wild type mice (n = 9) were injected i.p. with 100 µg Pam3CSK4. After 1 h, sera were collected and the levels of IL-6 were analyzed by ELISA. (**P < 0.01 vs. wild type). Horizontal bars represent median values.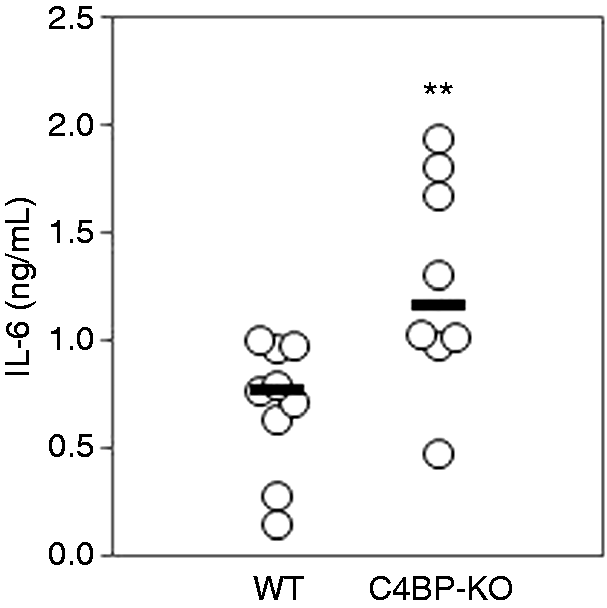
C4BP inhibits Pam3CSK4-driven IL-8 production
Next, we analyzed the IL-8 production in Pam3CSK4-stimulated 293-mTLR1/2 cells (Figure 3). In this experiment, 293-mTLR1/2 cells were used, as they are cells that can be transfected at a high efficiency. Furthermore, the C4BP expression level in 293-mTLR1/2 cells seemed to be physiological rather than supraphysiological because C4BP in the plasma level is very high (in humans, 200 µg/ml
25
). Depending on the C4BP expression levels, Pam3CSK4-driven IL-8 production was reduced (Figure 3a). We excluded the possibility that C4BP affects TLR1 and TLR2 expression by Western blot analysis (Figure 3b). No substantial changes in the expression of TLR1 and TLR2 were observed by increasing the expression of C4BP. Furthermore, we examined the IL-8 production in response to TNF-α, which is TLR-independent inducer of IL-8 (Figure 3c). No significant difference in IL-8 production was observed. Together, these results indicate that C4BP inhibits Pam3CSK4-driven pro-inflammatory cytokine production.
C4BP inhibits Pam3CSK4-driven IL-8 production. (a) 293-mTLR1/2 cells in a 6-well plate were transfected with various amounts of the pMXpuro-mC4BP-2HA (0, 0.5, 1 and 1.5 µg). The total amount of transfected DNA was equalized by pMXpuro. At 2 d post-transfection, these cells were stimulated with the indicated concentration of Pam3CSK4 for 4 h. The levels of IL-8 in the supernatants were determined by ELISA. [*P < 0.05, **P < 0.01 vs. C4BP (-)]. (b) Transfected cells in Figure 2a were lysed in lysis buffer, and expression levels of TLR1 and TLR2 were confirmed by Western blot with anti-TLR1 mAb or anti-TLR2 mAb, respectively. Percentages to the band intensity of C4BP (-) cells are indicated. Expression levels of C4BP in the supernatants were determined by immunoprecipitation and Western blot analysis with anti-HA mAb. (c) 293-mTLR1/2 cells in a 6-well plate were transfected with 1.5 µg of the pMXpuro-mC4BP-2HA or pMXpuro. At 2 d posttransfection, these cells were stimulated with the indicated concentration of TNF-α for 4 h. The levels of IL-8 in the supernatants were determined by ELISA.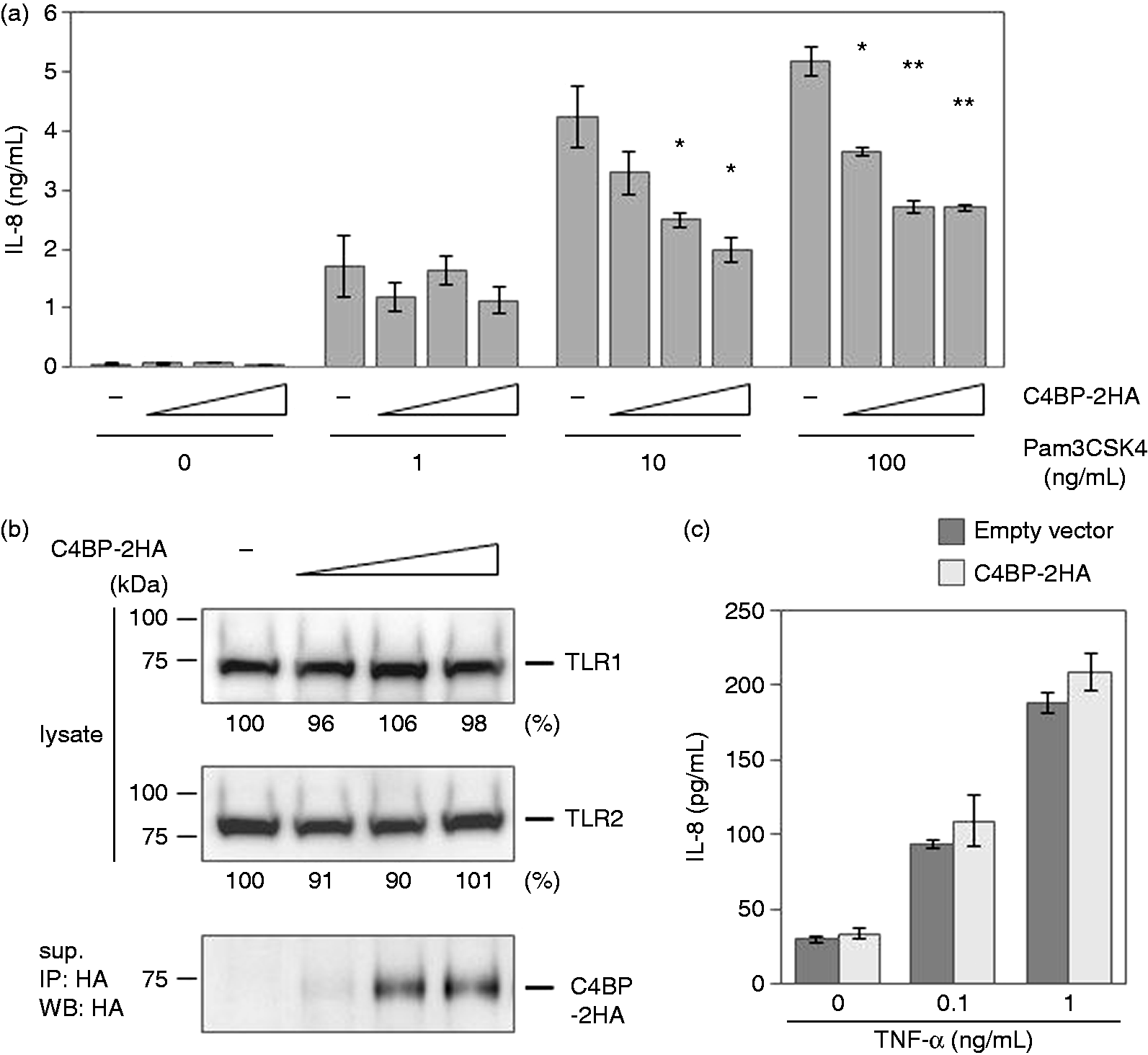
C4BP inhibits the binding of Pam3CSK4 to TLR1/2
To elucidate the inhibitory mechanism of C4BP, we analyzed the binding of ligand to TLR1/2 using rhodamine-labeled Pam3CSK4. As shown in Figure 4a, Pam3CSK4 fluorescence in C4BP-expressing 293-mTLR1/2 cells was significantly reduced by 50% compared with that in vector-transfected 293-mTLR1/2 cells. This result suggests that C4BP inhibits the binding of Pam3CSK4 to TLR1/2. We excluded the possibility that C4BP affects cell surface expression of TLR1 and TLR2 by flow cytometric analysis (Figure 4b). No substantial changes in the cell surface expression of TLR1 and TLR2 were observed by the expression of C4BP. As C4BP binds to TLR2, C4BP probably masks the Pam3CSK4 binding site on TLR1/2. There remains a possibility that alterations in levels of other lipopeptide-binding proteins in the serum might be involved in the reduction of binding of Pam3CSK4 to the cells.
C4BP blocks the binding of Pam3CSK4 to TLR1/2. 293-mTLR1/2 cells were transfected with the pMXpuro-mC4BP-2HA or pMXpuro. (a) Transfected cells and HEK293T cells were treated with Pam3CSK4 rhodamine or medium alone for 30 min. These cells were analyzed by confocal microscopy (left panel). Five areas of intensity of Pam3CSK4 rhodamine for each sample were quantified and divided by each cell numbers (right panel). The results are expressed as percentages of the relative intensity of empty vector-transfected 293-mTLR1/2 cells (***P < 0.005 vs. empty vector). (b) Transfected cells were stained with anti-TLR2 and TLR1 mAb and analyzed by flow cytometry. Relative mean florescence intensities (stained/non-stained) are indicated.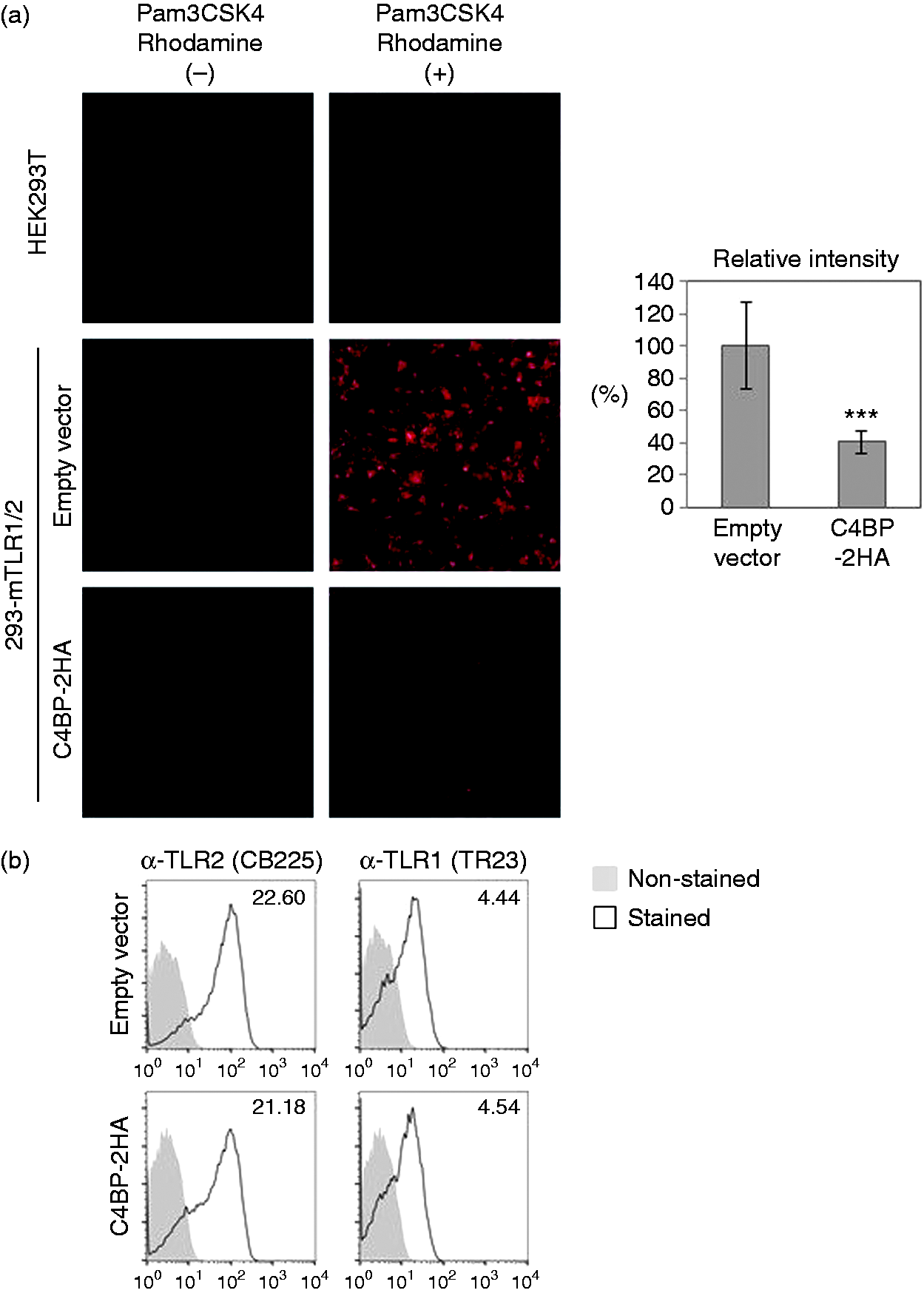
Exogenous C4BP inhibits Pam3CSK4-driven TLR1/2 signaling
As C4BP inhibits the binding of Pam3CSK4 to TLR1/2, exogenous C4BP may also inhibit the TLR1/2 response. We thus examined the effect of exogenous C4BP on TLR1/2-dependent pro-inflammatory cytokine production (Figure 5a). We used C4BP-expressing HEK293T or empty vector-transfected HEK293T cells and collected those supernatants. C4BP-containing or not containing supernatant was added to the 293-mTLR1/2 cells exogenously and then the cells were stimulated with Pam3CSK4. By adding the C4BP-containing supernatant, approximately 30% of Pam3CSK4-driven IL-8 production was reduced in 293-mTLR1/2 cells, suggesting that exogenous C4BP is sufficient for inhibition of TLR1/2 response in 293-mTLR1/2 cells. We also examined the IL-8 production in response to TNF-α (Figure 5b). No significant difference in IL-8 production was observed by addition of C4BP. Furthermore, we examined the effects of exogenous C4BP on Pam3CSK4-driven signaling (Figure 5c). NF-κB and p38 MAPK are important transcription factors that induce pro-inflammatory cytokines such as IL-6 and IL-8. Stimulation of TLR1/2 leads to phosphorylation of p65 subunit of NF-κB and p38 MAPK, and thereby facilitates transcriptional activity. Phosphorylation of both p65 subunit of NF-κB and p38 MAPK was significantly inhibited in 293-mTLR1/2 cells by adding exogenous C4BP. This result suggests that exogenous C4BP inhibits TLR1/2 signaling. Thus, C4BP appears to inhibit TLR2 activation on the cell surface of 293-mTLR1/2 cells. These results support the hypothesis that C4BP masks TLR2 and inhibits its activity.
Exogenous C4BP inhibits TLR1/2 signaling. (a, b) C4BP-containing or not containing supernatant of HEK293T cells (C4BP-2HA sup. or control sup.) was added to the 293-mTLR1/2 cells for 4 h, and these cells were stimulated with the indicated amounts of (a) Pam3CSK4 or (b) TNF-α for 24 h. The levels of IL-8 in the supernatants were determined by ELISA (*P < 0.05, ***P < 0.005 vs. control sup.). (c) C4BP-2HA sup. or control sup. was added to the 293-mTLR1/2 cells for 4 h. After stimulation with 100 ng/ml Pam3CSK4 for 30 or 60 min, the cells were lysed in Laemmli sample buffer and the lysates were subjected to Western blot analysis with the indicated Abs (NF-κB p65, phospho-NF-κB p65, p38 MAPK, or phospho-p38 MAPK) (left panel). Phosphorylation levels are expressed as a fold increase over that of un-stimulated cells (-) (*P < 0.05 vs. control sup.) (right panel).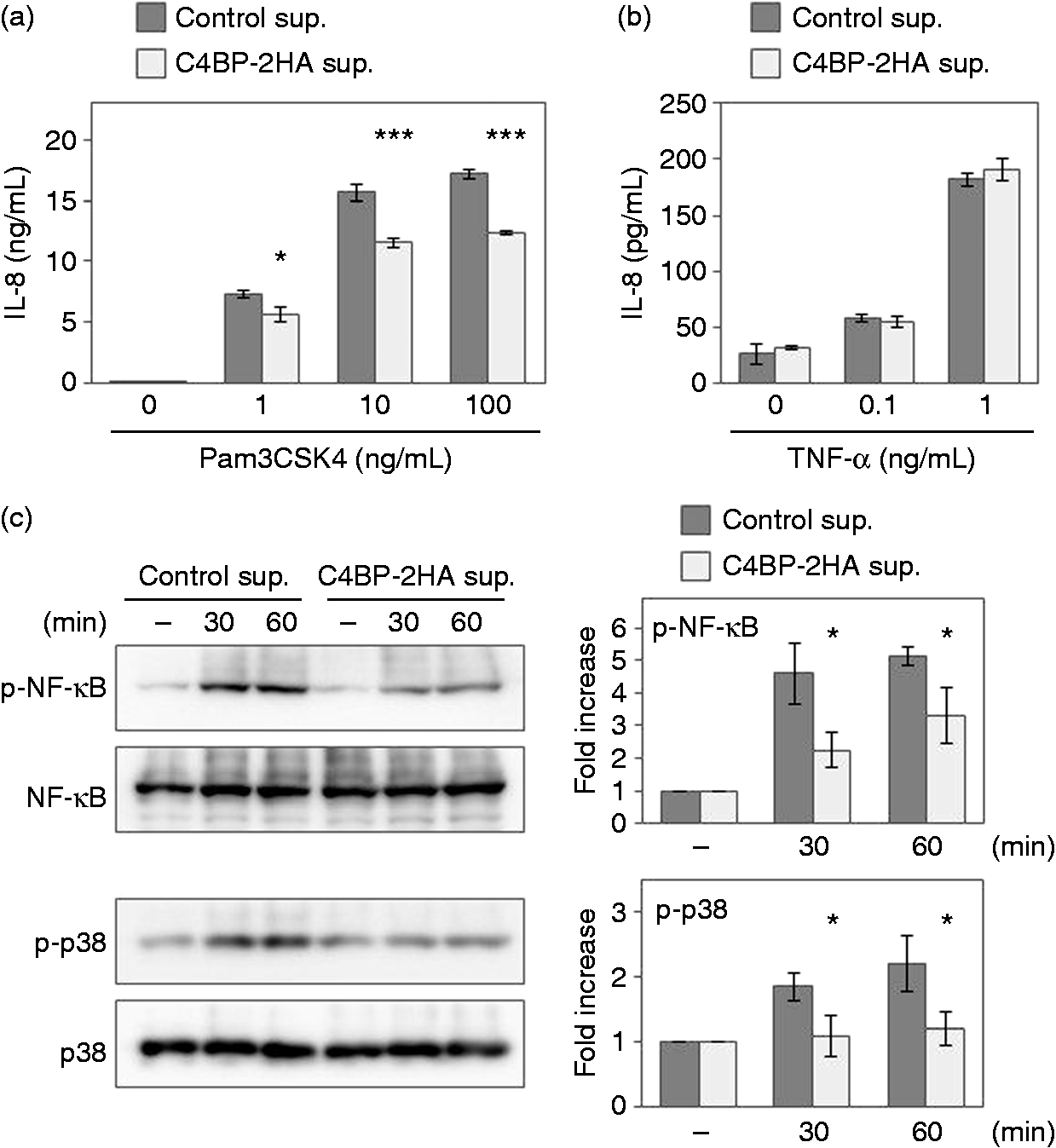
Discussion
In the present study, we, for the first time, have demonstrated that C4BP binds to TLR2 and inhibits the binding of ligand to TLR1/2. The binding of C4BP leads to the inhibition of ligand-driven TLR1/2 signaling and cytokine production. Furthermore, we have shown inhibitory effects on TLR1/2 signaling and on pro-inflammatory cytokine production by exogenous C4BP. Our previous studies have shown several TLR accessory molecules that directly interact with TLRs, such as MD-2 17 or PRAT4A. 26 MD-2 is an LPS receptor component, and PRAT4A is a chaperone of TLRs except TLR3. These molecules positively work in TLR response. However, this study suggests that C4BP negatively works in TLR1/2 response.
C4BP is known as a classical and lectin complement pathways inhibitor. C4BP also has many other roles such as B-cell activation, 22 regulation of blood coagulation 27 and removal of apoptotic cells. 28 In the present study, we found a novel function of C4BP. In steady state, 200 µg/ml C4BP is contained in human plasma, 25 and continuously prevents immunoreactions for the host tissue in the complement pathway. Furthermore, C4BP is classified as an acute-phase reactant because its level is elevated (up to 400%) during inflammation.29,30 Since C4BP inhibited TLR1/2 response, C4BP might be important for regulating inflammatory processes. Therefore, C4BP might continuously control two kinds of innate immunity called the complement system and the TLR1/2 response.
In addition to analysis of the blocking mechanism using the expression and exogenous addition of C4BP in 293-mTLR1/2 cells, we examined the responsiveness of C4BP-deficient mice to TLR1/2 ligand. Therefore, it is likely that C4BP also works in vivo as a negative regulator of pro-inflammatory cytokines. This raises the possibility that C4BP is involved in diseases caused by uncontrolled activation of TLR2. Indeed, it has been reported that single nucleotide polymorphisms (SNPs) in C4BP in women result in dysregulated complement activity, thereby leading to miscarriage. 31 We might be able to identify the risk of onset caused by dysregulated TLR2 by checking SNPs and mutation of C4BP.
The present study provides evidence of bovine C4BP binding to mouse TLR2. Therefore, we needed to exclude the possibility that inhibition of the TLR1/2 response seen in the presence of mouse C4BP could be attributed to bovine C4BP. We found that vector-transfected 293-mTLR1/2 cells showed Pam3CSK4 fluorescence in the presence of FBS (Figure 4, left panel). We further showed that the mouse C4BP inhibits the TLR1/2 response under FBS containing medium. These findings suggest that inhibition of the TLR2 response is attributable to the property of mouse C4BP itself but not to the presence of bovine C4BP.
In summary, we have demonstrated that C4BP binds to TLR2 and inhibits the binding of Pam3CSK4 to TLR1/2, thereby inhibiting Pam3CSK4-driven signaling. Despite the wide range of ligands recognized by TLRs, the receptors share a common structural framework in their extracellular, ligand-binding domains. 32 Further study is required to investigate the possibility that C4BP also binds to other TLRs.
Footnotes
Acknowledgements
We thank F Nagaoka, M Naruse, T Miyake and Y Nakagomi for excellent technical assistance and helpful discussions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported, in part, by the Grants-in-Aid for Scientific Research (C) from the Ministry of Education, Culture, Sports, Science and Technology of the Japanese Government (23590564 and 15K09491); a Grant for Joint Research Project of the Institute of Medical Science, the University of Tokyo (I.D.No.340 and I.D.No.356); a Grant from Aichi Cancer Research Foundation; a Grant from Okinaka Memorial Institute for Medical Research; and a Grant from AiKei-Kai Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
