Abstract
Pro-inflammatory cytokine release (IL-8, IL-6, TNF-α, IL-1β) by peripheral blood neutrophils, isolated from periodontitis patients (before/after therapy) and matched controls, was determined after 18 h culture in the presence/absence of Escherichia coli LPS, opsonised Staphylococcus aureus, heat-killed Fusobacterium nucleatum and Porphyromonas gingivalis. All cultures demonstrated differences in the amounts of each cytokine detected (P < 0.0001), with a clear release pattern (IL-8 > IL-6 > TNF-α = IL-1β). Median cytokine release from unstimulated patient neutrophils was consistently, but non-significantly, higher than from control cells. Stimulated cytokine release from untreated patient neutrophils was also consistently higher than from control cells. This hyper-reactivity was significant for all tested cytokines when data for all stimuli were combined (P < 0.016). In terms of individual stimuli, significant hyper-reactivity was detected with LPS (IL-8), F. nucleatum (IL-8, TNF-α), opsonised S. aureus (IL-8, TNF-α, IL-1β) and P. gingivalis (IL-8, IL-1β). Cytokine production by patient neutrophils did not reduce following successful non-surgical periodontal therapy and, except for responses to F. nucleatum, the cytokine hyper-reactivity detected pre-therapy was retained. These data demonstrate that chronic periodontitis is characterised by neutrophils that constitutively exhibit cytokine hyper-reactivity, the effects of which could modulate local and systemic inflammatory-immune responses and influence the risk and severity of periodontitis-associated systemic inflammatory diseases.
Introduction
Chronic periodontitis is one of the most prevalent chronic inflammatory diseases of humans and in its most severe form is the sixth most prevalent disease, affecting 11.2% of the global population and representing a substantial public health burden. 1 The pathogenesis of chronic periodontitis is associated with a dysregulated host inflammatory-immune response to intra-oral plaque bacteria, 2 which is responsible for the majority of periodontal tissue damage. 3 A hyper-inflammatory neutrophil phenotype is considered central to the pathogenesis of chronic periodontitis, 4 and also many of the systemic inflammatory diseases associated with periodontitis, including type 2 diabetes and cardiovascular disease.5,6 As such, neutrophilic ‘hyper-inflammation’ may represent one causal mechanism by which periodontitis contributes to co-morbidity.
Coordination of inflammatory-immune responses including neutrophil function involves the release of locally produced and systemically circulating cytokines. 7 Originally, monocytes and lymphocytes were regarded as the predominant source of pro-inflammatory cytokines in inflammatory lesions with neutrophils thought to produce only IL-8. 8 However, neutrophils themselves have been shown to release physiologically relevant concentrations of various cytokines including IL-8, IL-6, TNF-α and IL-1β,9–11 and therefore play an active role in coordinating the inflammatory immune response, in addition to their traditional antimicrobial activity. 12 Given the large numbers of neutrophils in peripheral blood and that they represent the dominant inflammatory cell in the periodontal tissues during periodontitis, neutrophil cytokine release is likely to impact significantly on the host response locally and systemically, with the latter increasing the risk of co-morbidity.
There have been few studies of neutrophil-derived cytokines in the pathogenesis of periodontitis and related systemic disease, despite their potential importance. The majority of studies investigating cytokines in chronic periodontitis have been performed on plasma, gingival crevicular fluid (GCF) and tissue biopsy samples, and have generally shown higher levels in disease. To date, there have been only three published studies investigating peripheral blood neutrophil cytokine release in patients with chronic periodontitis.13–15 Two studies demonstrated no significant difference in neutrophil IL-8, IL-1β and TNF-α release in chronic periodontitis compared with healthy controls in the absence or presence of granulocyte macrophage colony-stimulating factor or opsonised Staphylococcus aureus.13,14 By contrast, the third study reported significantly reduced neutrophil IL-8 release from patient neutrophils compared with control cells in the presence and absence of LPS, phytohaemagglutin, zymosan A, Porphyromonas gingivalis or Aggregatibacter actinomycetemcomitans. 15 All these studies used short culture times to determine cytokine production (≤ 5 h), which may have been too short to determine neutrophil cytokine production accurately. Although the specific mechanisms involved in neutrophil cytokine release are incompletely defined, 16 neutrophils are not known to store cytokines and time would be required for the synthesis and activation of pro-cytokine molecules following stimulation and prior to release. 17
Type 2 diabetes displays a unique bi-directional relationship with chronic periodontitis, namely that chronic periodontitis is a risk factor for type 2 diabetes and vice versa, 18 with periodontal treatment reliably shown to improve glycaemic control. 19 Peripheral blood neutrophils isolated from patients with type 2 diabetes have recently been shown to release greater amounts of IL-8, IL-1β and TNF-α when cultured for 18 h, in the presence and absence of LPS, than neutrophils isolated from healthy controls. 5 To date, there have been no studies investigating cytokine release, in the presence or absence of recognised periodontal pathogens, within an extended (18 h) culture period, from neutrophils isolated from patients with chronic periodontitis. Furthermore, there have been no studies on the effects of successful periodontal therapy on neutrophil cytokine production in periodontitis.
Here we determined neutrophil cytokine release following 18 h culture in the absence of and following stimulation with Escherichia coli LPS, opsonised S. aureus and the periodontal pathogens, Fusobacterium nucleatum and P. gingivalis. In addition, the effect of non-surgical periodontal therapy and stabilisation of periodontal disease on neutrophil cytokine release was also investigated. Given the increasing awareness of the ability of neutrophils to secrete cytokines and co-ordinate the inflammatory-immune response, we hypothesised that increased cytokine release from peripheral blood neutrophils may be involved in the pathogenesis of chronic periodontitis and have a potential role in associated systemic diseases.
Materials and methods
Ethical approval
Ethical approval was granted by the West Midlands Research Ethics Committee (REC reference no. 10/H1208/48). National Health Service (NHS) permission was granted by the Birmingham Community Healthcare NHS Trust on behalf of the Birmingham and the Black Country Comprehensive Local Research Network (BBC CLRN) Research Management & Governance (RM&G) Consortium Office (NHS R&D reference no. R&D1398; RM&G reference no. BCHCTDent265.1398.P). The study was adopted onto the National Institute of Health Research Network Portfolio (NIHR UKCRN Study ID 10318). The study was also registered on ClinicalTrials.gov (identifier: NCT01233765). The study was classified as a clinical study and came under the Department of Health Research Governance Framework for Health and Social Care.
Volunteer recruitment
Volunteers diagnosed with chronic periodontitis (n = 20) were recruited from patients referred to the Birmingham Dental Hospital for clinical evaluation and treatment [12 men and 8 women; mean ± SD age 46 ± 8 yr (range 37–61 yr)]. Chronic periodontitis was defined as the presence of at least two non-adjacent sites with probing pocket depths > 4 mm, along with radiographic bone loss ≥ 30% of the root length (non-first molar or incisor sites) in accordance with the consensus criteria of the European Federation of Periodontology. 20 Age- and sex-matched periodontally healthy control volunteers (n = 20) were recruited from staff of the Birmingham Dental Hospital & School [12 men and 8 women; mean ± SD age 46 ± 8 yr (range 32–62 yr)]. There were no significant differences in patient and control volunteer age, as determined by a Mann–Whitney U-test. Control volunteers had no evidence of attachment loss, no probing pocket depths > 4 mm and whole-mouth bleeding scores < 10%. All volunteers were never-smokers and systemically healthy, as confirmed by a detailed medical history questionnaire.
Periodontal assessment
Clinical measures included probing pocket depth and recession using a PCP-UNC-15 periodontal probe (Hu-Friedy Manufacturing Co., Chicago, IL, USA). Bleeding on probing was recorded in conjunction with the probing pocket depth measurements. Gingival inflammation was measured using the Löe gingival index. 21 The presence of dental plaque was measured using the modified Quigley and Hein plaque index. 22 All clinical measures were recorded on six sites per tooth (three measurements on each of the buccal/labial and palatal/lingual thirds) by a single calibrated examiner at baseline and 2 mo post-therapy.
Non-surgical periodontal therapy
Patient volunteers received tailored oral hygiene instruction and conventional non-surgical periodontal therapy by a single operator. This consisted of scaling and root surface debridement of all periodontal pockets > 4 mm performed under local anaesthesia on a quadrant-by-quadrant basis within a maximum of 4 wk.
Neutrophil isolation
Peripheral blood was obtained from volunteers with chronic periodontitis and age-/sex-matched controls before (baseline; n = 20) and following conventional non-surgical periodontal therapy (review; n = 19). Blood was obtained between 09:00 and 10:00 am from a patient and a matched healthy control volunteer within 30 min of each other. Blood was collected from the antecubital fossa into Vacutainers (Greiner, Bio-One Ltd, Stonehouse, UK) containing lithium heparin (17 IU/ml) as the anticoagulant. Neutrophils were isolated using a discontinuous Percoll gradient (δ = 1.079: 1.098), followed by erythrocyte lysis (0.83% NH4Cl containing 1% KHCO3, 0.04% Na2EDTA·2H2O and 0.25% BSA; 20 min). Isolated cells were > 99% polymorphonuclear cells morphologically and devoid of erythrocytes and monocytes. The absence of monocytes in cells prepared using this method has been confirmed previously within our laboratory by gene expression analyses. 23 Giemsa-stained cell smears also confirmed the presence of > 99% neutrophils in the cell preparation. 24 Neutrophil isolation was performed at room temperature within a biological safety cabinet (Jouan Microbiological Safety Cabinet BS 5726) using aseptic technique.
Preparation of bacterial stimuli
Fusobacterium nucleatum (Fn; ATCC 10953) and P. gingivalis (Pg; ATCC 33277) were grown anaerobically at 37℃ as previously described. 25 Staphylococcus aureus (NCTC 6571) was grown in air on mannitol salt agar and inoculated into tryptone soya broth. Bacteria were washed three times in sterile PBS and heat treated (100℃ for 10 min) prior to dilution with sterile PBS to give a final suspension of 8.5 × 109 and 4.12 × 109 for F. nucleatum and P. gingivalis, respectively, and stored at –80℃. The number of F. nucleatum and P. gingivalis in cell suspensions was determined by measuring the OD at 600 nm using conversion factors previously determined by the Forsyth Institute, Boston, MA, USA (OD1.0600 nm = 1.62 × 109 for F. nucleatum and 1.69 × 109 for P. gingivalis). 25 The bacterial density of S. aureus suspensions was determined by plate colony counting. Opsonised S. aureus was prepared as previously described, 26 and stored as a 1.1 × 1011 cells/ml suspension at –80℃. Escherichia coli serotype O26:B6 LPS (Sigma L5543; Sigma, St Louis, MO, USA) was supplied as a 1 mg/ml, 0.2 µm filtered, aqueous solution as confirmed by the product information sheet.
Neutrophil culture
Freshly isolated neutrophils were immediately washed at room temperature in PBS prior to re-suspension in RPMI-1640 (Sigma R8758) supplemented with 0.3 g/l glutamine, 2.32 g/l HEPES, 2 g/l sodium bicarbonate, 100 µg/ml streptomycin, 100 IU/ml penicillin and 10% heat-inactivated FBS. Neutrophils were cultured at 2.5 × 106 cells/ml (18 h at 37℃ and 5% CO2) in the presence of LPS (5 µg/ml), F. nucleatum (MOI 100:1), P. gingivalis (MOI 100:1), opsonised S. aureus (MOI 300:1) or additional RPMI as negative control. Neutrophil viability was determined immediately before and after 18 h culture by trypan blue dye exclusion.
Determination of neutrophil cytokine release
Following 18 h of culture, neutrophil cell culture supernatants were collected aseptically and stored at –80℃. Commercial ELISA kits (R&D Systems, Abingdon, UK) were used to measure IL-8, IL-6, TNF-α and IL-1β levels within the neutrophil culture supernatants. All samples were allowed to equilibrate to room temperature before use. Standards and samples were assayed in duplicate according to the manufacturer’s instructions. All cytokine data were calculated to the nearest whole value in pg/ml due to the reported ELISA assay sensitivities and inter-assay variation (typically > 1 pg/ml), as specified within the product information sheets.
Data handling and statistical analyses
Data were recorded and manipulated in Microsoft Excel and evaluated using GraphPad Prism 5 (version 5.04; GraphPad Software, La Jolla, CA, USA). Data distribution was determined using the Kolmogorov–Smirnov test. Differences in probing pocket depths, the only dataset found to be normally distributed, were determined by ANOVA followed by Tukey–Kramer multiple comparisons test. All other statistical comparisons were performed using non-parametric methods, as indicated in the text and/or Figures/Tables. Except for probing pocket depths, all data were reported as median and interquartile range (IQR). In order to assist graphical representation of the cytokine data, ‘outliers’ were determined (calculated as > 1.5 × IQR), 27 and plotted outside the boxplot, with the most extreme values above the y-axis maximum. All cytokine concentrations, including outlying values, were included in all statistical analyses.
Results
Longitudinal changes in clinical measures
Clinical measures of patients with chronic periodontitis before (baseline; n = 20) and 2 mo after (review; n = 19) non-surgical periodontal therapy and matched healthy controls (n = 20).
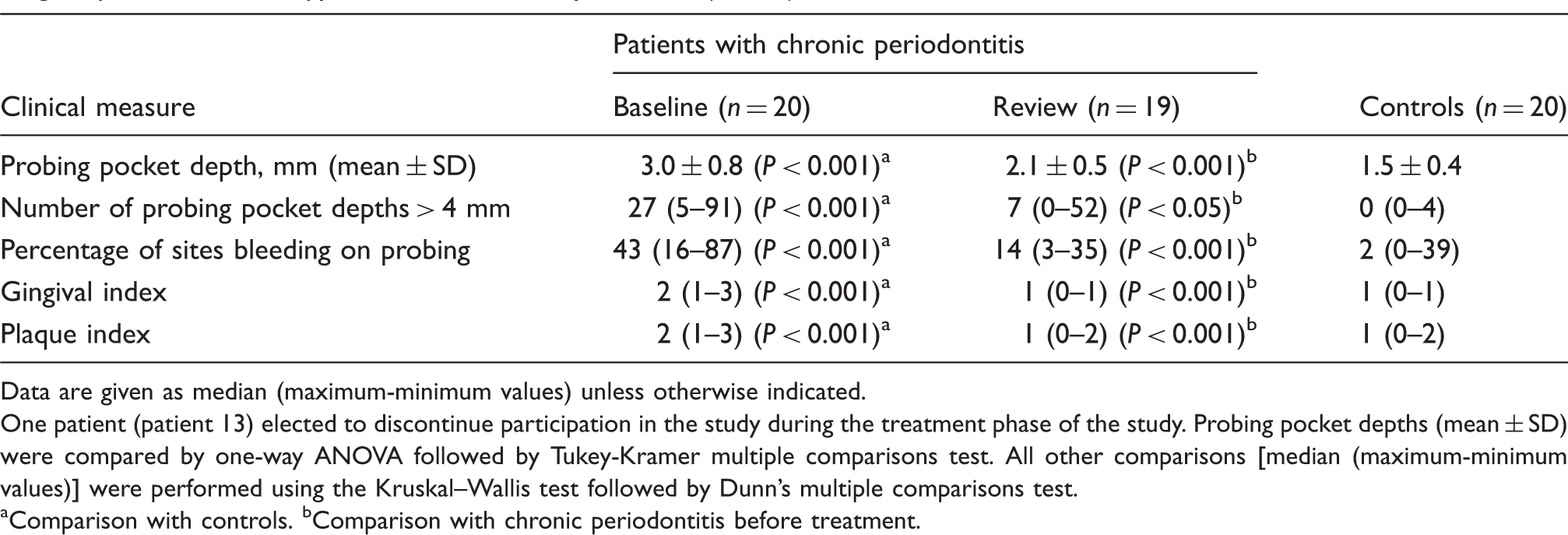
Data are given as median (maximum-minimum values) unless otherwise indicated.
One patient (patient 13) elected to discontinue participation in the study during the treatment phase of the study. Probing pocket depths (mean ± SD) were compared by one-way ANOVA followed by Tukey-Kramer multiple comparisons test. All other comparisons [median (maximum-minimum values)] were performed using the Kruskal–Wallis test followed by Dunn’s multiple comparisons test.
Comparison with controls. bComparison with chronic periodontitis before treatment.
Neutrophil viability following culture
There were no significant differences in neutrophil viability following 18 h of culture between patient (mean 97.7 ± 2.3%; range 94.1–100%) and control cells (mean 97.5 ± 1.8%; range 93.5–100%; data not shown). However, neutrophil viability in the presence of opsonised S. aureus (mean 95.4 ± 2.8%; range 90.9–100%) was significantly lower than in cultures containing additional RPMI or other stimuli (mean 98.9 ± 0.4%; range 98.4–99.2%; P < 0.05, Friedman/Dunn’s test).
Unstimulated neutrophil cytokine release
Unstimulated IL-8, IL-6, TNF-α and IL-1β release (pg/ml) from patient (n = 19) and control (n = 19) neutrophils at baseline and review.
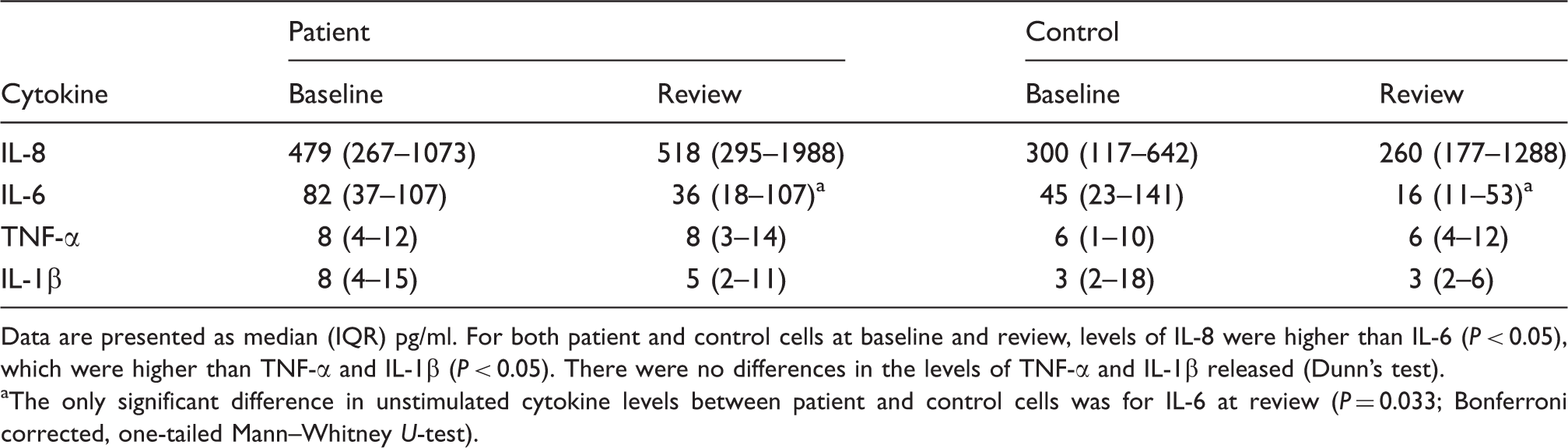
Data are presented as median (IQR) pg/ml. For both patient and control cells at baseline and review, levels of IL-8 were higher than IL-6 (P < 0.05), which were higher than TNF-α and IL-1β (P < 0.05). There were no differences in the levels of TNF-α and IL-1β released (Dunn’s test).
The only significant difference in unstimulated cytokine levels between patient and control cells was for IL-6 at review (P = 0.033; Bonferroni corrected, one-tailed Mann–Whitney U-test).
Stimulated neutrophil cytokine release
Neutrophils demonstrated a positive cytokine response to all stimuli employed [IL-8, TNF-α and IL-1β (P < 0.0004); IL-6 (P < 0.0012); Figures 1–4)].
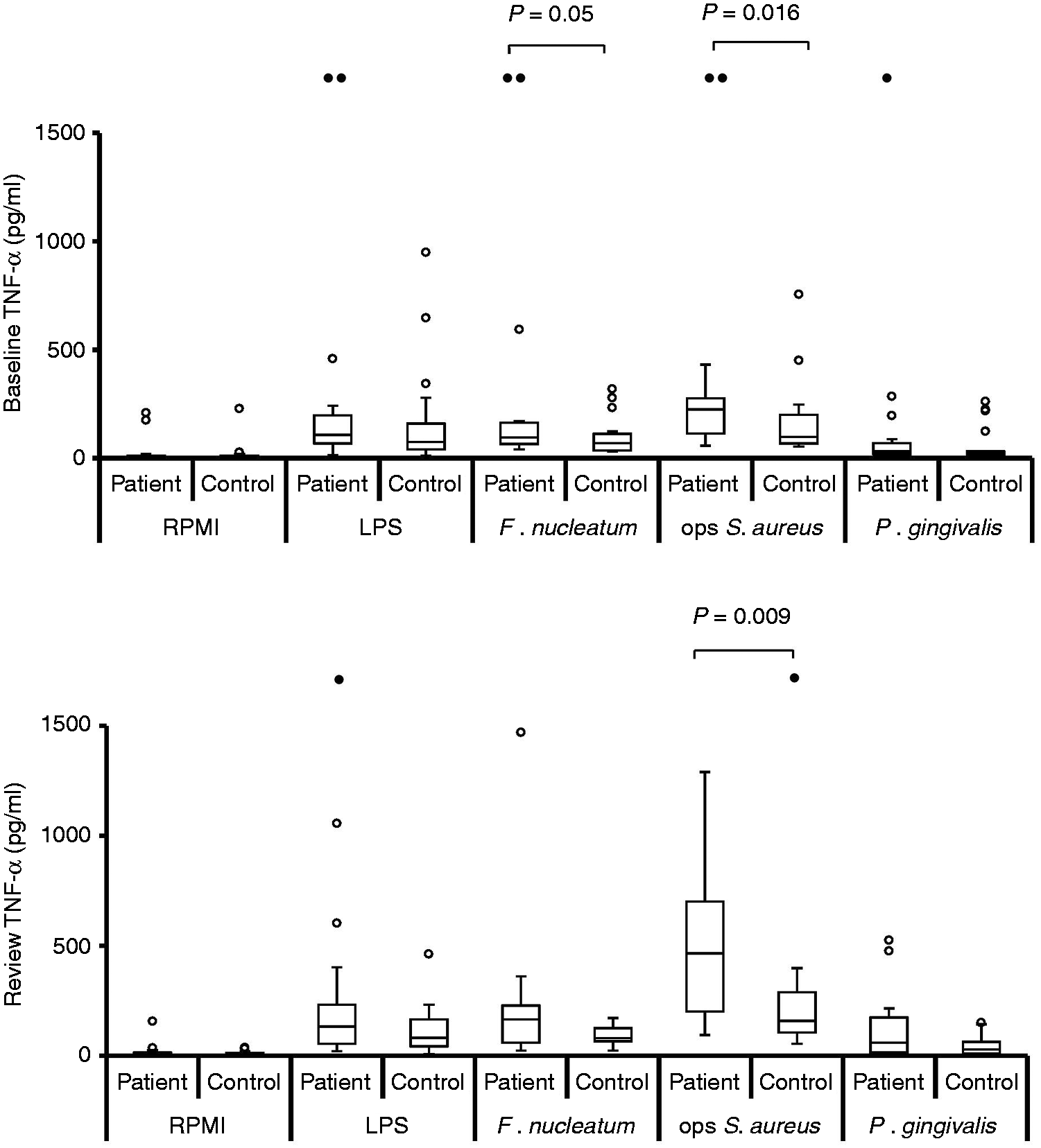
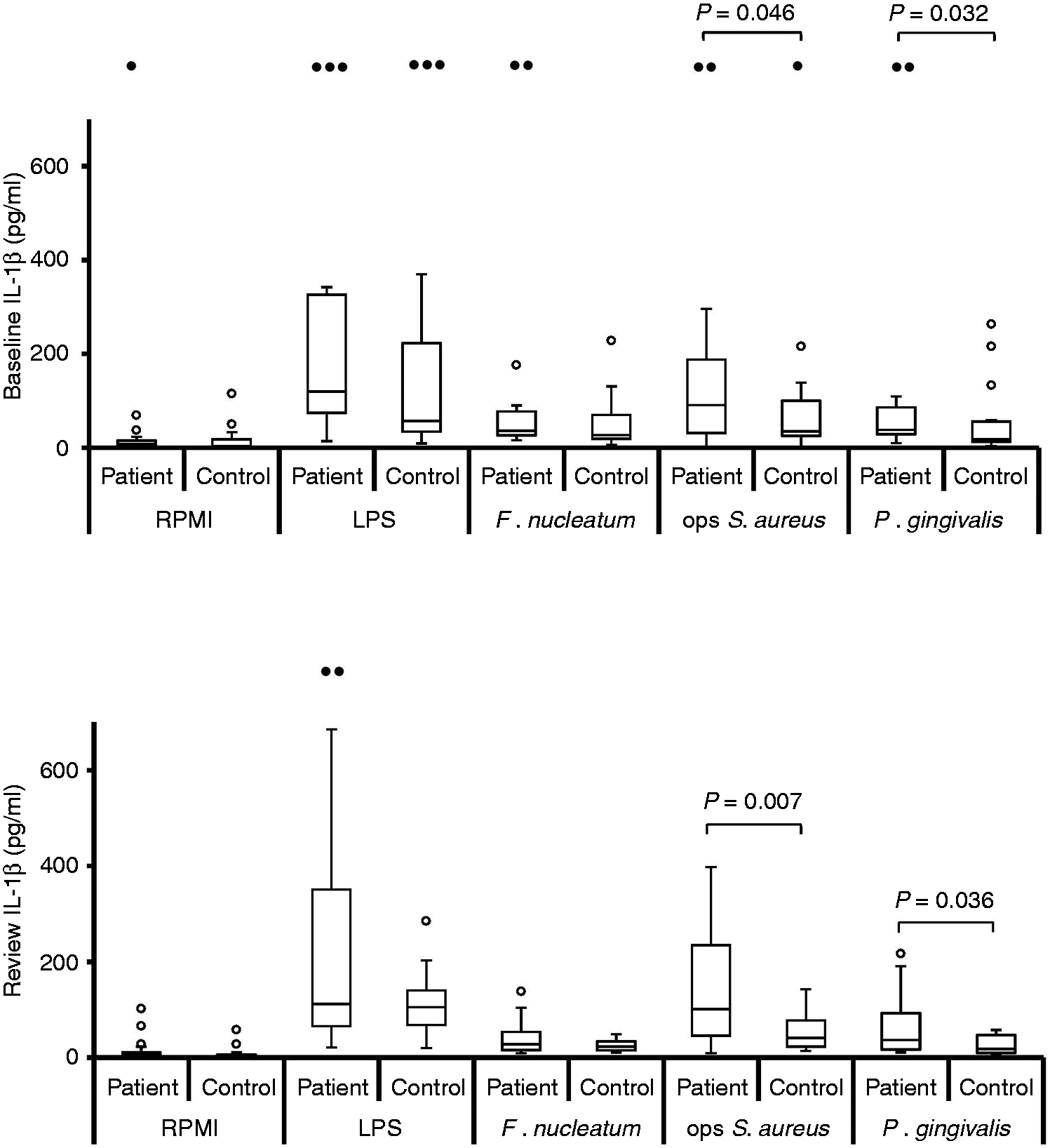
Stimulated IL-8 production by neutrophils at baseline, pre-treatment [n = 19; n = 18 for opsonised (ops) S. aureus] and review, post-treatment (n = 19). Box and whisker plots showing median, IQR, maximum and minimum. Outliers (values > 1.5 × IQR) are shown as open circles (○) or, if above the y-axis maximum, solid circles (•). P-Values calculated using Bonferroni corrected one-tailed Mann–Whitney U-test. Stimulated levels of IL-8 were significantly greater than those for RPMI control for all stimuli, at baseline and review, for both patient and control groups (P < 0.0004; Bonferroni corrected, one-tailed Wilcoxon test). Stimulated IL-6 production by neutrophils at baseline, pre-treatment [n = 19; n = 18 for opsonised (ops) S. aureus] and review, post-treatment (n = 19). Box and whisker plots showing median, IQR, maximum and minimum. Outliers (values > 1.5 × IQR) are shown as open circles (○) or, if above the y-axis maximum, solid circles (•). P-Values calculated using Bonferroni corrected, one-tailed Mann–Whitney U-test. Stimulated levels of IL-6 were significantly greater than those for RPMI control for all stimuli, at baseline and review, for both patient and control groups (P < 0.0012; Bonferroni corrected, one-tailed Wilcoxon test). Stimulated TNF-α production by neutrophils at baseline, pre-treatment [n = 19; n = 18 for opsonised (ops) S. aureus] and review, post-treatment (n = 19). Box and whisker plots showing median, IQR, maximum and minimum. Outliers (values > 1.5 × IQR) are shown as open circles (○) or, if above the y-axis maximum, solid circles (•). P-values calculated using Bonferroni corrected, one-tailed Mann–Whitney U-test. Stimulated levels of TNF-α were significantly greater than those for RPMI control for all stimuli, at baseline and review, for both patient and control groups (P < 0.0004; Bonferroni corrected, one-tailed Wilcoxon test). Stimulated IL-1β production by neutrophils at baseline, pre-treatment [n = 19; n = 18 for opsonised (ops) S. aureus] and review, post-treatment (n = 19). Box and whisker plots showing median, IQR, maximum and minimum. Outliers (values > 1.5 × IQR) are shown as open circles (○) or, if above the y-axis maximum, solid circles (•). P-Values calculated using Bonferroni corrected, one-tailed Mann–Whitney U-test. Stimulated levels of IL-1β were significantly greater than those for RPMI control for all stimuli, at baseline and review, for both patient and control groups (P < 0.0004; Bonferroni corrected, one-tailed Wilcoxon test).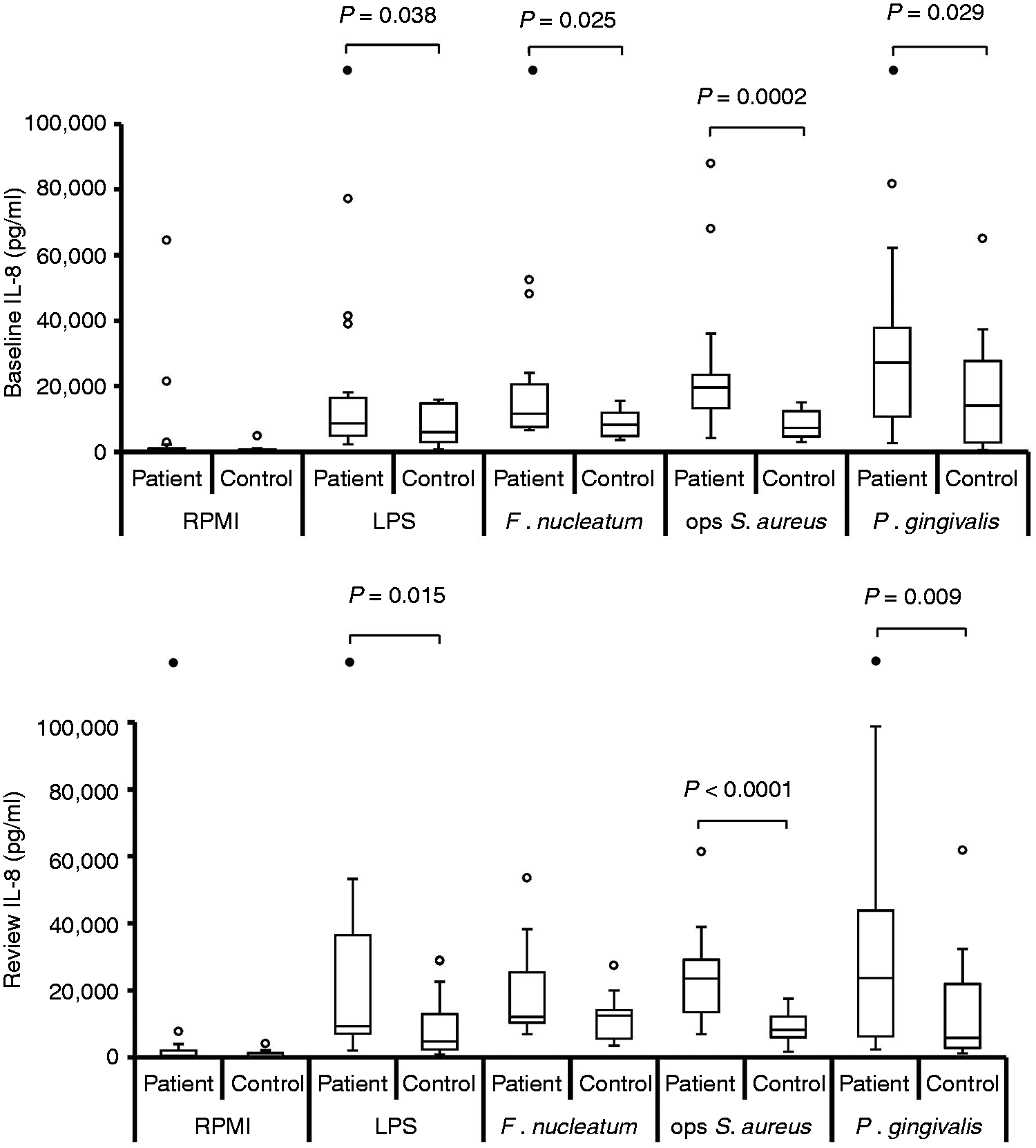
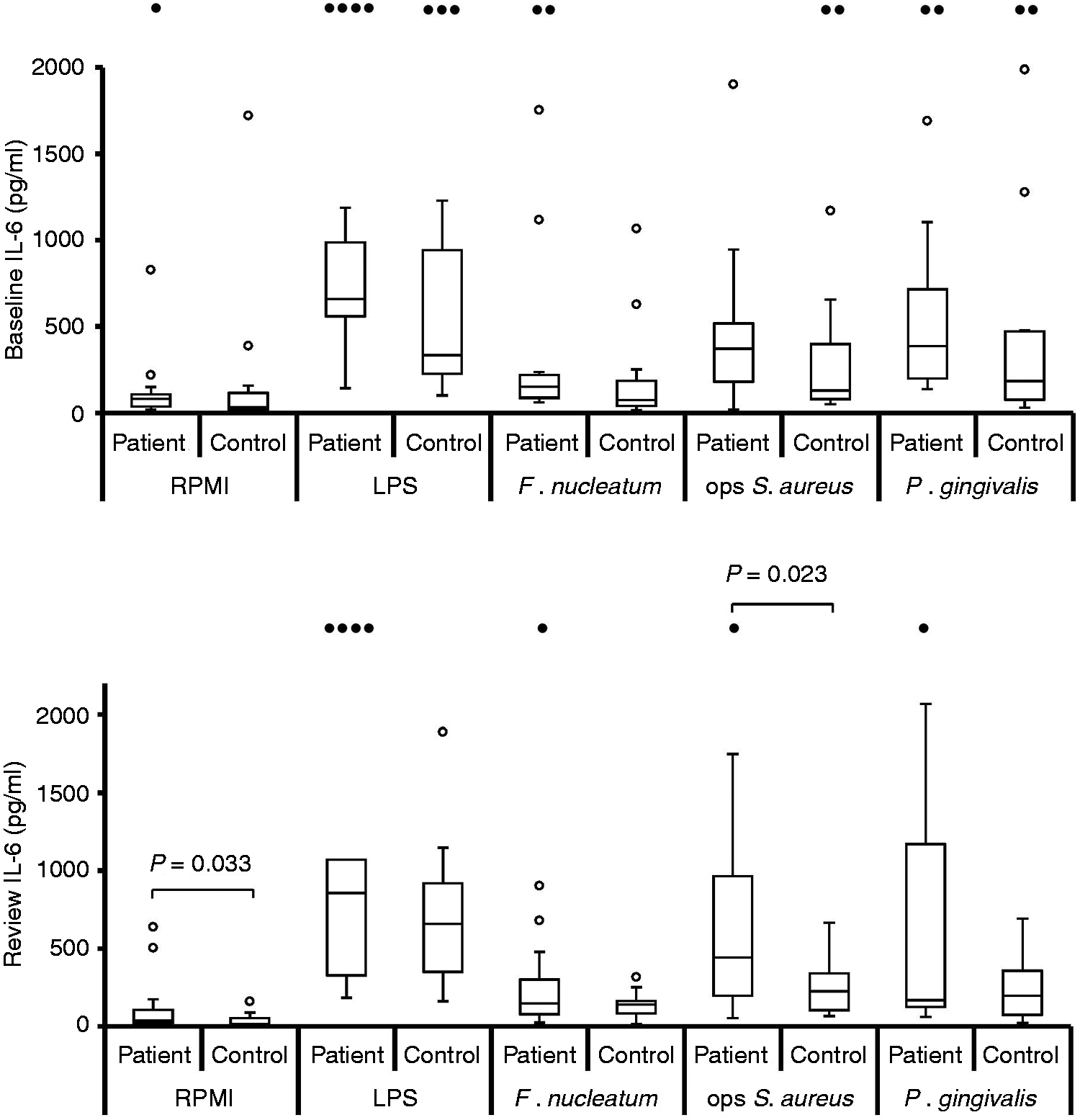
Stimulated IL-8, IL-6, TNF-α and IL-1β release (pg/ml) from patient (n = 19) and control (n = 19) neutrophils at baseline and review in the presence of E. coli LPS (5 µg/ml), F. nucleatum (MOI 100:1), IgG-opsonised S. aureus (MOI 300:1) and P. gingivalis (MOI 100:1).
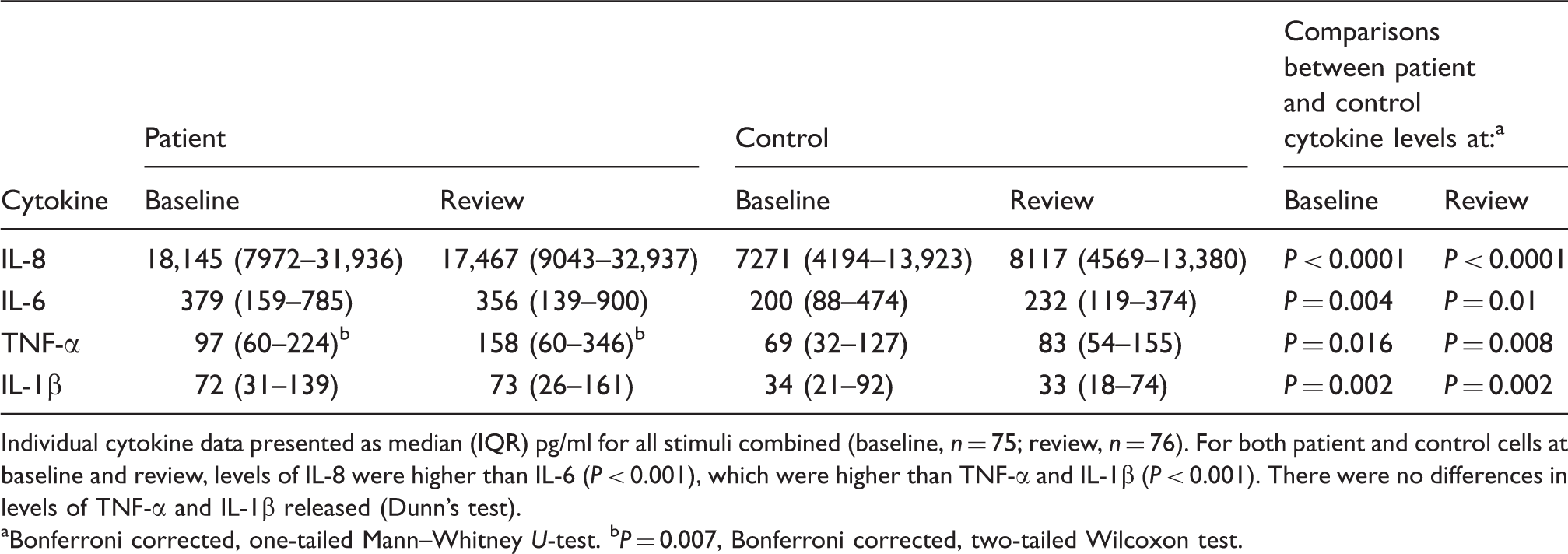
Individual cytokine data presented as median (IQR) pg/ml for all stimuli combined (baseline, n = 75; review, n = 76). For both patient and control cells at baseline and review, levels of IL-8 were higher than IL-6 (P < 0.001), which were higher than TNF-α and IL-1β (P < 0.001). There were no differences in levels of TNF-α and IL-1β released (Dunn’s test).
Bonferroni corrected, one-tailed Mann–Whitney U-test. bP = 0.007, Bonferroni corrected, two-tailed Wilcoxon test.
Differences in cytokine release between patient and control neutrophils at baseline and review in response to individual (n = 19; n = 18 for opsonised (ops) S. aureus at baseline) and combined stimuli (n = 75 at baseline and n = 76 at review) (↑, patient > control; ↔, patient = control; Bonferroni corrected, 1-tailed Mann-Whitney) and change in cytokine release post-therapy (↑, increase; ↓ decrease; ↔, no change at review; Bonferroni corrected 2-tailed Wilcoxon).
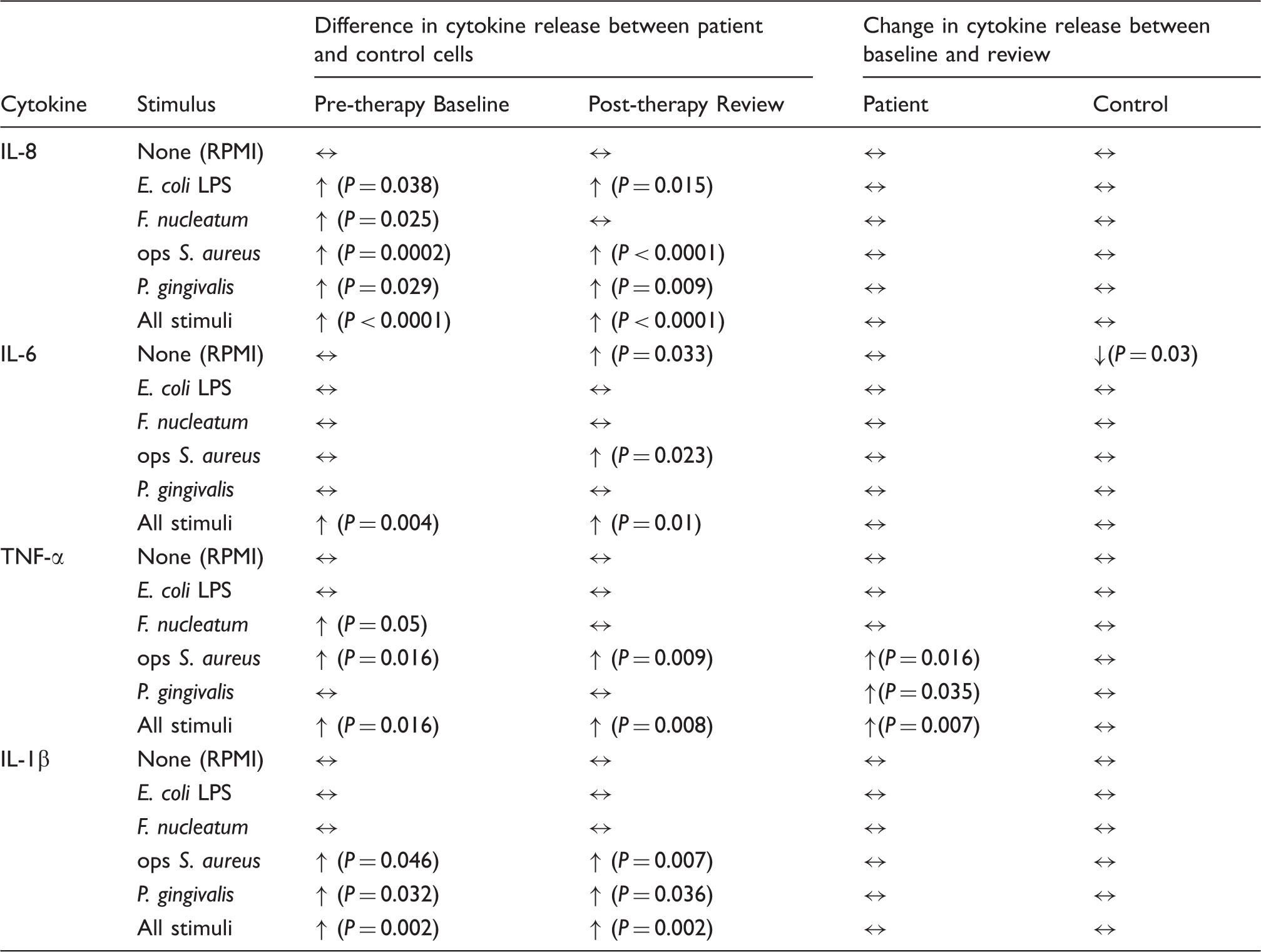
At both baseline and review the median levels of all four cytokines released were highest from patient cells, irrespective of which stimulus was used (Figures 1–4). When analysing the data for individual stimuli, several therapy-/stimulus-related differences between cytokine release from patient and control cells were detected (Figures 1–4 and summarised in Table 4). Patient neutrophils released greater amounts of IL-8 and TNF-α than controls in the presence of the periodontopathogen F. nucleatum at baseline (P ≤ 0.05), with no significant differences at post-treatment review. However, this apparent therapy-related change was not reflected by a significant reduction in cytokine production by patient cells post-therapy. By contrast, heightened FcγR-stimulated neutrophil IL-8, TNF-α and IL-1β release was a consistent feature of periodontitis neutrophils at both baseline (P < 0.05) and review (P < 0.023). Furthermore, greater release of IL-8 and IL-1β by patient cells was also a consistent feature after stimulation with P. gingivalis (baseline P < 0.032; review P < 0.036), as was production of IL-8 in response to E. coli LPS (baseline P = 0.038; review P = 0.015).
Release of IL-8, IL-6 and IL-1β by patient cells was unaffected by therapy. However, TNF-α release by patient cells in response to opsonised S. aureus (P = 0.016) and P. gingivalis (P = 0.035) was significantly higher at the post-therapy review, as was TNF-α production when combining the data for all stimuli (P = 0.007; Table 4).
Discussion
The data from this study demonstrate, for the first time, that peripheral blood neutrophils from patients with chronic periodontitis release greater amounts of cytokines (IL-8, IL-6, TNF-α and IL-1β) compared with those from healthy individuals, and exhibit a hyper-reactive phenotype in terms of cytokine production in response to a variety of stimuli, including the periodontitis-associated periodontal bacteria P. gingivalis and F. nucleatum. Furthermore, this is the first longitudinal intervention study analysing the impact of therapeutic reductions in periodontal inflammation on the ability of peripheral blood neutrophils to generate cytokines in the presence and absence of periodontally relevant stimuli. In most instances, the observed cytokine hyper-reactivity of patient neutrophils and their ability to release cytokines was not altered by successful non-surgical therapy. Together, these novel data are consistent with current literature highlighting a dysregulated neutrophil phenotype in chronic periodontitis pathogenesis and are significant because cytokines regulate many different aspects of the inflammatory-immune response known to be altered in periodontitis 28 including neutrophil reactive oxygen species (ROS) generation, extracellular trap (NET) release and directional chemotactic accuracy.29–32
Although median levels of unstimulated cytokine release were highest for patient cells, the difference, compared with control cell levels, was not significant for either the combined or individual cytokine data, except for IL-6 at review. This latter result was due to a significant decrease in control neutrophil IL-6 release at review compared with baseline, despite no known intervention between the sampling visits. Thus, our 18h culture data do not provide evidence that unstimulated peripheral blood neutrophils from periodontitis patients are hyperactive in terms of baseline, unstimulated cytokine release and agree with two reports using shorter (1–5 h) culture times.13,14 By contrast, the reported 4–6 times lower unstimulated release of IL-8 from patient cells compared with control neutrophils, where none of the cells responded to stimuli including E. coli LPS and zymosan A, 15 is not supported. However, further studies on larger numbers are required to determine whether the generally higher median release of cytokines by patient cells reported here would become statistically significant. This is of potential importance because even small differences in ‘baseline’, unstimulated cytokine release may be biologically important in the pathogenesis of periodontitis, given the large numbers of systemic and locally recruited neutrophils within the periodontium and the chronic nature of the disease.
Although cytokine hyperactivity (increased cytokine release in the absence of stimulation) was not statistically significant, greater stimulated IL-8, IL-6, TNF-α and IL-1β release from patient compared with control neutrophils was a consistent feature when comparing data for all stimuli combined, demonstrating that patient cells are hyper-reactive in terms of cytokine production. While these analyses of cytokine responses to multiple stimuli suggest that patient neutrophils will be hyper-reactive to the many host and plaque-associated factors known to be simultaneously present within periodontal tissues and blood, 33 it is important to remember that our data are based on summation of cytokine release stimulated by individual stimuli rather than responses to the combined stimuli.
The pattern of stimulated cytokine responses by neutrophils from patient cells varied according to the individual stimulus and cytokine investigated. Untreated patient neutrophils released significantly greater amounts of IL-8 in response to all tested stimuli compared with control cells. By contrast, there were no significant differences in stimulated IL-6 release between patient and control cells for any of the stimuli. TNF-α and IL-1β release by patient cells showed an intermediate response profile showing hyper-reactivity to opsonised S. aureus (TNF-α and IL-1β), F. nucleatum (TNF-α) and P. gingivalis (IL-1β).
The FcγR-stimulated IL-8, TNF-α and IL-1β hyper-reactivity presented in this study contrasts with the lack of significant differences between periodontitis and control neutrophils previously reported. 14 However, the authors of the mentioned study utilised a 5h culture time and detected only low levels of FcγR-stimulated IL-8 (range 16–64 pg/ml) and TNF-α (range 13–248 pg/ml). By comparison, high levels of cytokine were detected in this study (stimulated ranges: IL-8, 496–118,466 pg/ml; TNF-α, 2–8326 pg/ml) using an 18h culture period, a time known to correspond to that required for the majority of stimulated neutrophil IL-8, IL-6, TNF-α and IL-1β to be released (16–18 h).34,35 Similarly, the significantly greater E. coli LPS- and P. gingivalis-stimulated IL-8 release from patient compared with control neutrophils presented here contrasts with a study reporting lower E. coli LPS and P. gingivalis-stimulated IL-8 release from periodontitis neutrophils. 15 However, this latter study only used a 1h culture time and failed to demonstrate stimulation of control neutrophils, which produced extremely high levels of IL-8 (∼800 ng) in the absence of stimulation.
This is the first known study to investigate the effect of successful periodontal treatment on peripheral blood neutrophil cytokine release. There were no detectable changes in IL-8, IL-6 or IL-1β production by patient neutrophils at the post-therapy review, indicating that any neutrophil cytokine hyperactivity/hyper-reactivity detected was constitutive rather than being secondary to the inflammatory process. Indeed, significant differences between patient and control cell release of IL-8, IL-6 and IL-1β detected pre-therapy were also demonstrated post-therapy, except for F. nucleatum-stimulated IL-8. This finding contrasts with data on FcγR-stimulated neutrophil ROS hyper-reactivity in periodontitis, which is not detectable following treatment. 30 In contrast to IL-8, IL-6 or IL-1β, a therapy-related increase in TNF-α release by patient cells was detected when analysing responses to opsonised S. aureus, P. gingivalis and all stimuli combined. Although this change had no positive effect on TNF-α hyper-reactivity detected at pre-therapy baseline, it is difficult to explain why reducing the inflammatory burden to the body, by reducing the periodontal bacterial load and inflammation, should apparently increase stimulated TNF-α production by patient neutrophils.
In terms of periodontitis, an important and novel finding was the consistent IL-8 and IL-1β hyper-reactivity of patient cells to P. gingivalis, a bacterium strongly associated with chronic periodontitis. 36 Porphyromonas gingivalis LPS is structurally distinct from other sources of LPS, 37 induces cytokine production via different signalling mechanisms, 38 and is heterogenous, with some P. gingivalis LPS molecules binding to TLR2, others to TLR4 and, possibly, also to receptor antagonists.39,40 Taken together, a constitutive defect in TLR receptor or downstream signalling relating to IL-8 and IL-1β release may explain the persistent hyper-reactive P. gingivalis-stimulated cytokine release from patient compared with control neutrophils detected in this study.
In contrast to the response to P. gingivalis, neutrophil hyper-reactivity to F. nucleatum, a bacterium known to be important in plaque associated with periodontitis,36,41 was detected before treatment (IL-8 and TNF-α) but could not be demonstrated post-therapy. As patient levels of both cytokines were not lowered after treatment, this apparent loss of hyper-reactivity cannot be attributed to reduced periodontal bacterial load and inflammation. Further studies are obviously required to determine the significance of these observations.
Dysregulated neutrophil cytokine release, namely cytokine hyper-reactivity to periodontal pathogens and FcγR activation, could have a number of different consequences on the inflammatory-immune response in relation to the pathogenesis of chronic periodontitis. Greater neutrophil IL-8, IL-6 and TNF-α release may result in neutrophil priming for greater ROS release.42,43 Increased extracellular ROS release may then result in collateral host tissue damage as a result of osteoclast activation causing bone resorption in addition to degradation of type I collagen of the periodontal ligament.44,45 Neutrophil ROS and IL-8 release will also result in NET formation,46,47 which, if excessive, may result in the generation of auto-Abs, 48 along with impairing the inflammatory-immune response by sequestering local cytokine dissemination. 49 Heightened release of IL-8, IL-6, TNF-α and IL-1β by neutrophils could also trigger neutrophil antimicrobial enzyme and granule protein release with the potential to cause destruction of adjacent periodontal connective tissue. 50
In addition to the above, there are a variety of indirect effects of dysregulated neutrophil cytokine release that could impact on the pathogenesis of chronic periodontitis. For example, neutrophil IL-8 release is likely to affect concurrent neutrophil chemotaxis and recruitment within the periodontal tissues. 51 Our group has demonstrated that peripheral blood neutrophils from patients with periodontitis display impaired directional chemotactic accuracy, 32 and, in this patient and control cohort, we have shown a moderate positive correlation between neutrophil IL-8 release and chemotactic index towards fMLP, suggesting that neutrophil IL-8 release may assist with neutrophil chemotaxis towards bacterial-derived chemo-attractants (Ling MR, Chapple IL, Roberts HM, Matthews JB, unpublished observations). In addition, any adaptive immune response will be heightened by IL-8, IL-6 and TNF-α, resulting in T-cell activation which is also directly influenced by IL-6. 52 The generation of Abs and bacterial opsonisation will result in FcγR-stimulated neutrophil activation, thereby further exacerbating the inflammatory-immune response in relation to the persistent FcγR-stimulated IL-8, IL-6, IL-1β and TNF-α release, and FcγR-stimulated neutrophil ROS hyper-reactivity in chronic periodontitis. 30
Overall, the data presented in this study demonstrate that neutrophils from untreated periodontitis patients are hyper-reactive to a variety of disease-associated stimuli with regard to IL-8, IL-6, TNF-α and IL-1β release compared with cells from healthy control individuals. Furthermore, production of cytokines by patient cells was not reduced after successful non-surgical periodontal therapy and, except for responses to F. nucleatum, cytokine hyper-reactivity detected pre-therapy was shown to be retained. These data suggest that neutrophil cytokine hyper-reactivity is not secondary to the inflammatory reaction within the periodontal tissues during periodontal disease but is the result of constitutive differences in neutrophils isolated from controls and periodontitis patients. Such a difference in neutrophil reactivity could modulate both local and systemic inflammatory-immune responses and influence the risk and severity of periodontitis, as well as periodontitis-associated systemic inflammatory disease.
Footnotes
Funding
This work was supported by the Oral & Dental Research Trust (grant number 14945).
Conflict of interest
The authors do not have any potential conflict of interest to declare.
