Abstract
Innate lymphoid cells (ILCs) function in producing effector cytokines in response to pathogenic infections. However, the roles and related mechanisms of the ILC subpopulations, ILC1 and ILC2, which mirror Th1 and Th2 in adaptive immunity, remain unclear. In this study, we found the markedly elevated levels of the ILC1 transcription factor T-bet, the effector cytokine IFN-γ and the IL/receptor signaling molecules IL-12/IL-12R, which are indispensable for ILC1 differentiation, in the helper ILCs of chronic hepatitis B (CHB) patients. The elevated level of the ILC1 population was significantly associated with hepatic damage in CHB patients, and was not related to telbivudine treatment. In contrast, although we also observed elevated levels of ILC2-related factors, including IL-33, ST2, GATA3 and IL-13 in helper ILCs, the extent of elevation shown by each was lower than that shown by the ILC1-related factors. Furthermore, the activity of the ILC2s did not correlate with either HBV copies or liver damage. The findings of this study suggest potential pro-inflammatory roles for ILC1s in CHB pathogenesis, potentiating these cells and their related molecules as targets of diagnostic, prognostic and/or therapeutic strategies for hepatitis B.
Introduction
Hepatitis B virus (HBV) infection has been recognized as a pivotal etiological factor for development of chronic hepatitis B (CHB) and the related persistent inflammatory state of the liver that affects its structure and impairs its function. The underlying molecular mechanisms of the HBV-related liver inflammation primarily involves the host immune response to the HBV pathogen. 1 Although the CD8+ T cells that are considered the key cellular effectors mediating HBV clearance from the liver, the CD4+ Th cells are characterized as the master regulators of the adaptive immune response.2,3 Moreover, among the subpopulations of Th cells, Th1 and Th2 have been implicated in the pathogenesis of hepatic inflammation during HBV infection. 4
In addition to adaptive immunity, innate immunity also plays a role in the pathogenesis of hepatitis B. 4 Unlike the adaptive immune system, the innate immune system is involved in the development of mucosal-associated lymphoid tissues and serves as a rapid and early source of the effector cytokines that are typically associated with Th cell subsets in response to changes in the microenvironment caused by pathogenic infections, particularly via the innate lymphoid cells (ILCs). 5 ILCs primarily localize at mucosal barriers, such as in the lung, intestine and skin, and are capable of rapidly switching on responses to pathogens, even upon first exposure. 5
ILCs lack the known immune cell lineage markers and they were originally subdivided into three main groups according to their cytokine production profiles as corresponding to the Th1, Th2 or Th17 cell types. 6 Each ILC subset exerts different functions under physiological and pathological conditions, which are mediated by specific molecular profiles of cytokines and transcription factors (TFs). 7 ILCs of group 1 produce Th1-type cytokines, such as IFN-γ, with T-bet as the signature TF; the NK cells of this group are distinguished by their production of additional cytotoxic molecules, such as perforin and granzyme. The ILCs of group 2 produce the Th2-associated cytokines IL-5, IL-13 and/or IL-4, with GATA3 as the signature TF. Finally, ILCs of group 3 produce the Th17-associated cytokine(s) IL-17 and/or IL-22, with RORγt as the signature TF. While the NK cells are considered the cytotoxic effector cells of the ILCs (also termed killer ILCs), the collective ILCs of groups 1–3 are classified as helper-like ILCs that function according to their various effector cytokine profiles. 5
The ILCs combat pathogenic threats primarily by rapidly releasing a plethora of cytokines. The ILCs localized at mucosal surfaces act as sentinel cells,8,9 where release of their cytokine suite inhibits establishment and spread of infection, 10 and aids in the protection of epithelial tissue integrity. 8 As with any immune effector cell, uncontrolled activation and proliferation of ILCs can lead to damaging inflammation, as has been shown in gut, lung and skin.11–15 The pathogenic roles of ILCs have recently been expanded to liver tissue. Matsumoto et al. reported that the IL-22-producing ILC3s play a novel protective role in murine acute hepatitis. 16 However, ILCs may also play pivotal fuctions in chronic diseaes, for instance, McHedlidze et al. reported that IL-33-dependent ILC2s mediate hepatic fibrosis. 17 However, whether and how ILC1s and ILC2s contribute to heptatis B-induced hepatitc damage remains unclear.
To this end, the current study was designed to investigate the roles of ILC1s and ILC2s in hepatitis B by using serum samples taken from patients with CHB over the course of telebivudine treatment, and comparing the results with those from healthy control individuals.
Materials and methods
Patients
Clinical characteristics of the CHB patients (n = 30) at study enrollment.
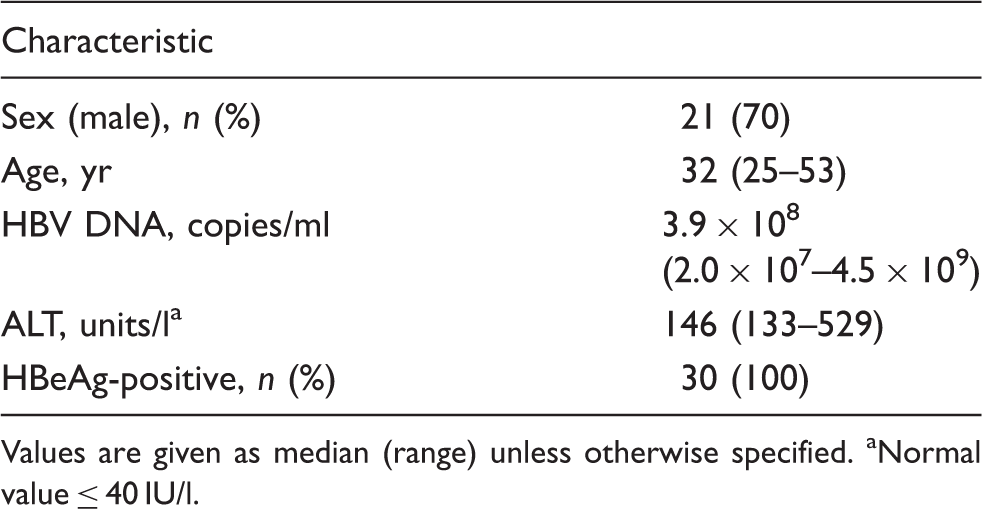
Values are given as median (range) unless otherwise specified. aNormal value ≤ 40 IU/l.
FACS
The total population of IL-7Rα (CD127)-positive helper ILCs was determined by detecting the Lin−CD45+CD127+ lymphocytes by FACS. 5 The individual heparinized blood samples from the CHB patients and healthy controls were processed with a Optiprep Nycoprep Lymphoprep Kit (Axis-shield, Oslo, Norway) to isolate PBMCs. The isolated PBMC samples were then prepared for FACS analysis by staining with the following lineage Ab mix: FITC-conjugated antihuman CD1a, CD3, CD11c, CD34, CD123, TCRαβ, TCRγδ, BDCA2, FcɛRI, CD19, CD14 and CD16 (eBioscience, San Diego, CA, USA); 18 peridinin-chlorophyll proteins conjugated to antihuman CD45 (eBioscience); phycoerythrin-conjugated antihuman CD127 (BD Biosciences, San Jose, CA, USA). The isotype control Ab was used in all cases. Sorting of Lin−CD45+CD127+ lymphocytes and FACS phenotype analysis were carried out with a FACSAria cell sorter (BD Biosciences) and FlowJo single cell analysis software (Tree Star, Inc., Ashland, OR, USA). The sorted cells were suspended in Trizol reagent (Invitrogen, Karlsruhe, Germany) and stored at −80℃ until RNA extraction.
Quantitative real-time RT-PCR
Primer sequences used in qRT-PCR.

F, forward; R, reverse.
ELISA
Plasma levels of IL-12, IL-33 (eBioscience), IL-13 and IFN-γ (Ucallm, Wuxi, China) were measured by ELISA kits, following the respective manufacturer protocols. Extinction spectra and cytokine concentrations were measured at 450 nm on an ABS-MONO spectrophotometer (Beckman, Krefeld, Germany) with the accompanying multimode analysis software. Standards and samples were run in duplicate.
Statistical analysis
Data are presented as mean ± SD (or SEM, where indicated). Comparisons between two groups were made using a t-test or a one-way ANOVA test, and comparisons between more than two groups were made using Tukey’s post-hoc test. All statistical methods were carried out by GraphPad Prism software, version 5.0 (GraphPad Software, San Diego, CA, USA). P-Values < 0.05 were considered statistically significant.
Results
CHB patients showed significantly increased numbers of ILC1s
We first determined the frequency of the total helper ILC (Lin–CD45+) population, including the CD127-positive cells (i.e. the ILC1, ILC2, ILC3 and LTi cells), as well as the CD127-negative cells [i.e. the killer ILCs (i.e. cNK cells)]. FACS analysis showed that, compared with the healthy control, the PBMCs from CHB patients had a significantly higher frequency of total helper ILCs, which, however, was almost restored to the level in healthy control after telbivudine treatment for 52 wk (Figure 1). The absolute count of total helper ILCs was also higher in the PBMCs of CHB patients than in healthy controls, and telbivudine treatment for 52 wk significantly decreased the ILC count in CHB patients, though it was still higher compared with healthy controls (Figure 1). These results suggest that ILCs might be involved in CHB pathogenesis.
Frequencies of helper ILCs in the total ILC population from CHB patients (n = 30) and healthy controls (HC; n = 28). (A) Representative results from FACS analysis of helper ILCs in lineage-negative total ILCs of PBMCs, with helper ILCs defined as Lin– (lineage marker-negative for CD1a, CD3, CD11c, CD34, CD123, TCRαβ, TCRγδ, BDCA2, FcɛRI, CD19, CD14 and CD16) but positive for CD45 and CD127. (B) Comparative statistical analysis of the detected frequencies of helper ILCs in the Lin–CD45+ total ILC populations of the respective groups. (C) Absolute counts of total helper ILCs per 1 × 106 of PBMCs in each group. Pre-: pre-telbivudine treatment; Post-: 52 wk post-telbivudine treatment. * P < 0.05; ** P < 0.01.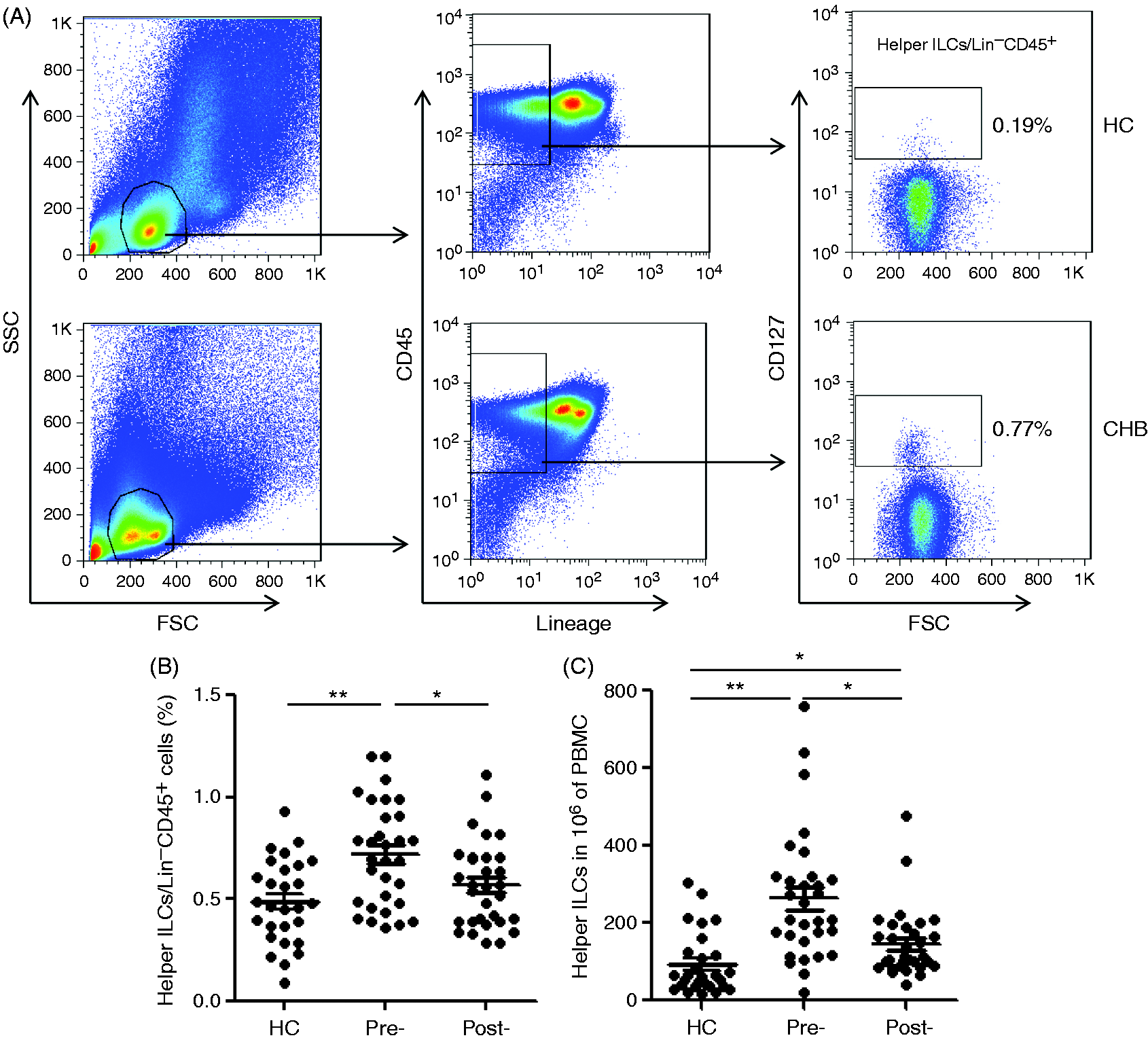
Considering the previous knowledge of Th1 and Th2 involvement in CHB pathogenesis, we next investigated whether and how the ILC1 and ILC2s, which mirror Th1 and Th2 cells in adaptive immunity, contribute to the pathogenic process of CHB. To this end, we measured the mRNA levels of signature TFs and receptors of ILC1s (T-bet and IL-12Rβ, respectively) and of ILC2s (GATA3 and ST2, respectively) in the FACS-isolated helper ILCs. While the mRNA levels of both of the signature TFs were elevated markedly in the helper ILCs from CHB patients compared with healthy controls, the increase in T-bet was more robust than that of GATA3. However, Telbivudine treatment restored the mRNA levels of both TFs to a normal level (Figure 2). The comparative analysis of the ILC1 and ILC2 receptors showed that the IL-12Rβ mRNA level was significantly higher in the helper ILCs from the CHB patients compared with the healthy controls but that the ST2 mRNA level was not significantly different between the two groups (Figure 2). While telbivudine treatment had no effects on ST2 expression, it markedly decreased IL-12Rβ mRNA level in CHB patients, which was still higher than that in the healthy controls (Figure 2).
mRNA levels of ILC1 and ILC2 signature TFs and receptors in the helper ILCs from CHB patients (n = 30) and healthy controls (HC; n = 28). Total ILCs (Lin–CD45+) were sorted from PBMCs of the respective groups by FACS, after which the helper ILCs were further sorted based upon CD127-positive expression. The relative mRNA level of the ILC1 and ILC2 TFs and receptors, T-bet/IL-12R and GATA3/ST2, respectively, were detected by qRT-PCR. Pre-: pre-telbivudine treatment; Post-: 52 wk post-telbivudine treatment. * P < 0.05; ** P < 0.01.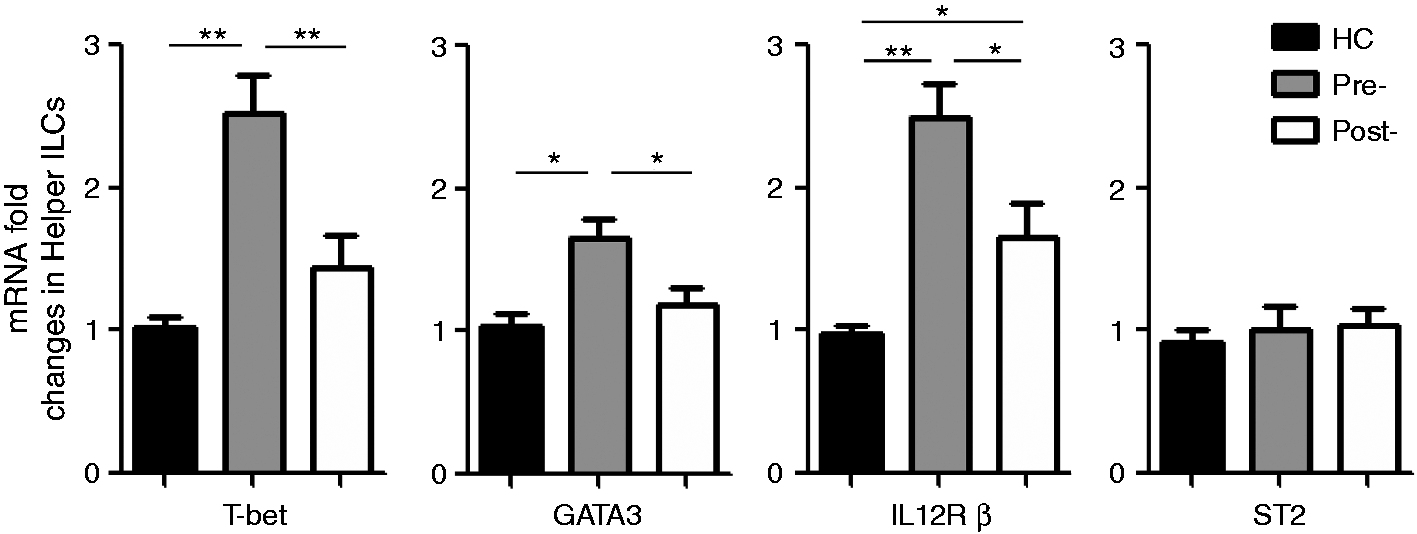
Helper ILCs from CHB patients produced more type I cytokines than type II cytokines
As type I and type II cytokines are known to be involved in CHB pathogenesis,
19
and helper ILCs are known to function primarily through their secreted cytokines,7,9,18 we investigated whether ILC1s and ILC2s contribute to the HBV-related alterations in type I and type II cytokine profiles. We found that the mRNA levels of IFN-γ and IL-13 were enhanced in the FACS-isolated helper ILCs from CHB patients compared with healthy controls but that the mRNA level of IL-4 did not differ between the two groups. Furthermore, the increase levels of type I cytokines were much higher than type II cytokines in helper ILCs from CHB patients. However, telbivudine treatment restored the mRNA levels of IFN-γ and IL-13 in CHB patients to normal levels (Figure 3A).
Levels of ILC1- and ILC2-related cytokines in helper ILCs, liver tissues and peripheral blood of CHB patients and healthy controls (HC). (A) Relative mRNA expression of ILC1- and ILC2-related cytokines in FACS-isolated helper ILCs in PBMCs from the CHB (n = 30) and HC (n = 28) groups, determined by qRT-PCR. (B) Relative mRNA expression of ILC1- and ILC2-related cytokines in liver tissues from the two groups, determined by qRT-PCR; CHB tissues were obtained by puncture and HC tissues were obtained by pruning of healthy liver tissue from organs used for transplantation. (C) Plasma ILC1- and ILC2-related cytokine level, detected by ELISA. Pre-: pre-telbivudine treatment; Post-: 52 wk post-telbivudine treatment. Data are presented as mean ± SD. *P < 0.05 and **P < 0.01.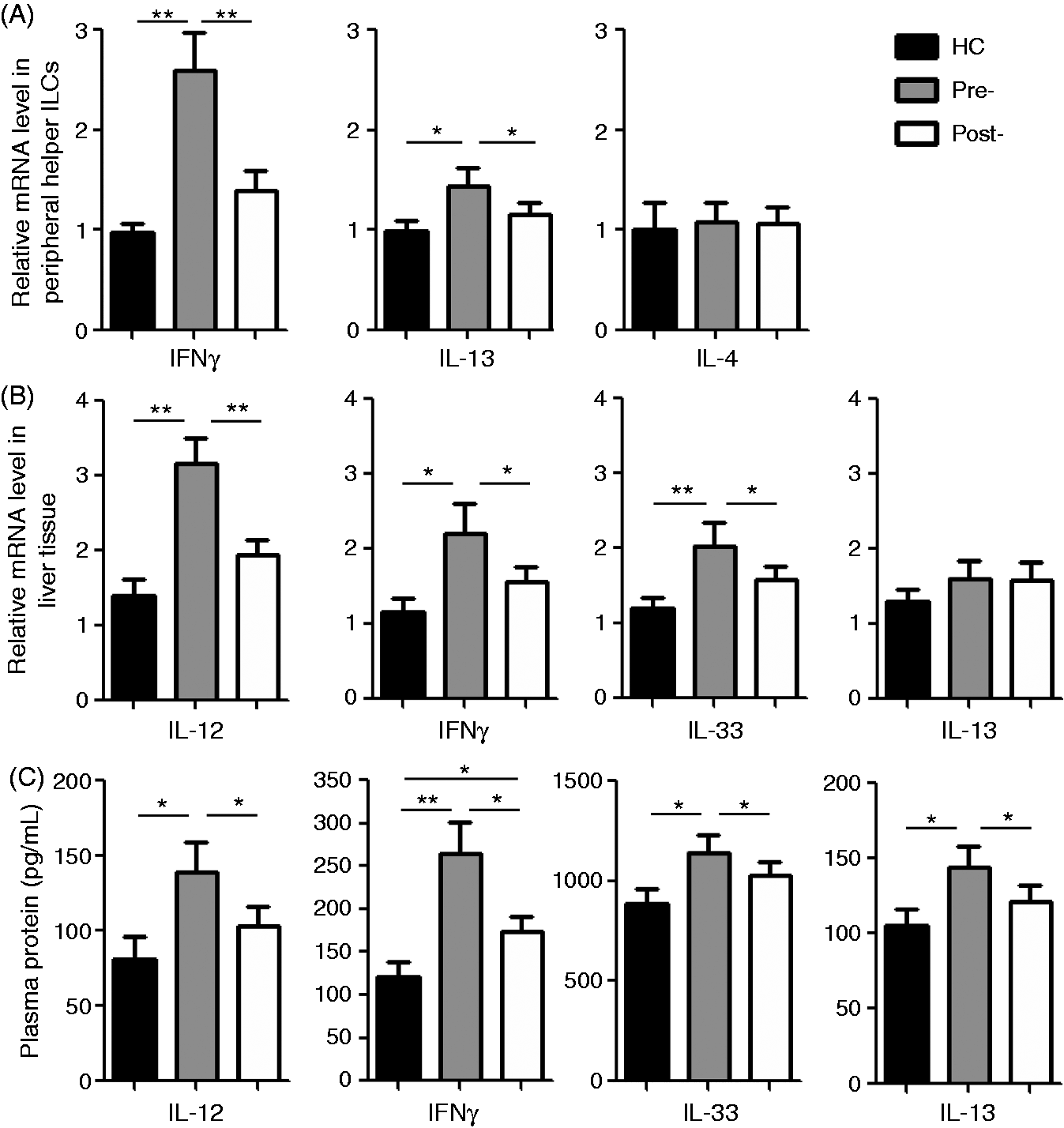
We further found that the expression levels of the type I cytokines (IL-12 and IFN-γ) and the type II cytokines (IL-33 and IL-13) were all increased in the CHB liver tissue (mRNA) and the plasma samples (protein), with the exception of IL-13 mRNA, which was not altered significantly in the liver tissues of CHB patients compared with that in healthy control tissues (Figure 3B, C). The CHB-related elevation in type I cytokine levels in the peripheral blood and the liver tissues was more robust than that of the type II cytokines (Figure 3). These results suggest the potential contribution of helper ILCs to the elevated type I and type II cytokine levels in the peripheral blood and the liver tissues. Again, telbivudine treatment could restore the expression levels of these cytokines to normal levels, with the exception of IFN-γ protein level in the plasma of CHB patients treated with telbivudine, compared with that in healthy controls (Figure 3B, C).
Aberrant increase in ILC1 activity was associated with hepatic injury during HBV infection
Because Th1 and Th2 cells can also produce type I and type II cytokines, respectively, we analyzed whether the cytokines produced by ILC1s and ILC2s contributed to liver injury in CHB patients. As expected, the mRNA levels of TFs T-bet and GATA3 were significantly correlated with the corresponding cytokines IFN-γ and IL-13 mRNA levels in isolated helper ILCs (Figure 4A, B). However, while the IFN-γ mRNA level in helper ILCs was markedly associated with the plasma ALT level in CHB patients (Figure 4C), the IL-13 mRNA level in helper ILCs did not show a correlation with the patients’ plasma ALT levels (Figure 4D). Thus, ILC1s, and not ILC2s, might contribute to the production of type I cytokines and liver injury during HBV infection.
Correlation between ILC1s/ILC2s and hepatic injury in CHB. Correlation analysis was conducted to determine the relationship between the cytokine IFN-γ mRNA level and the TF T-bet mRNA level in (A) helper ILCs or (C) the plasma ALT level in CHB patients, and between the IL-13 mRNA level and the TF GATA3 mRNA level in (B) helper ILCs or the (D) plasma ALT level in telbuvidine-naïve CHB patients at baseline (n = 30). The P-values for each correlation are indicated.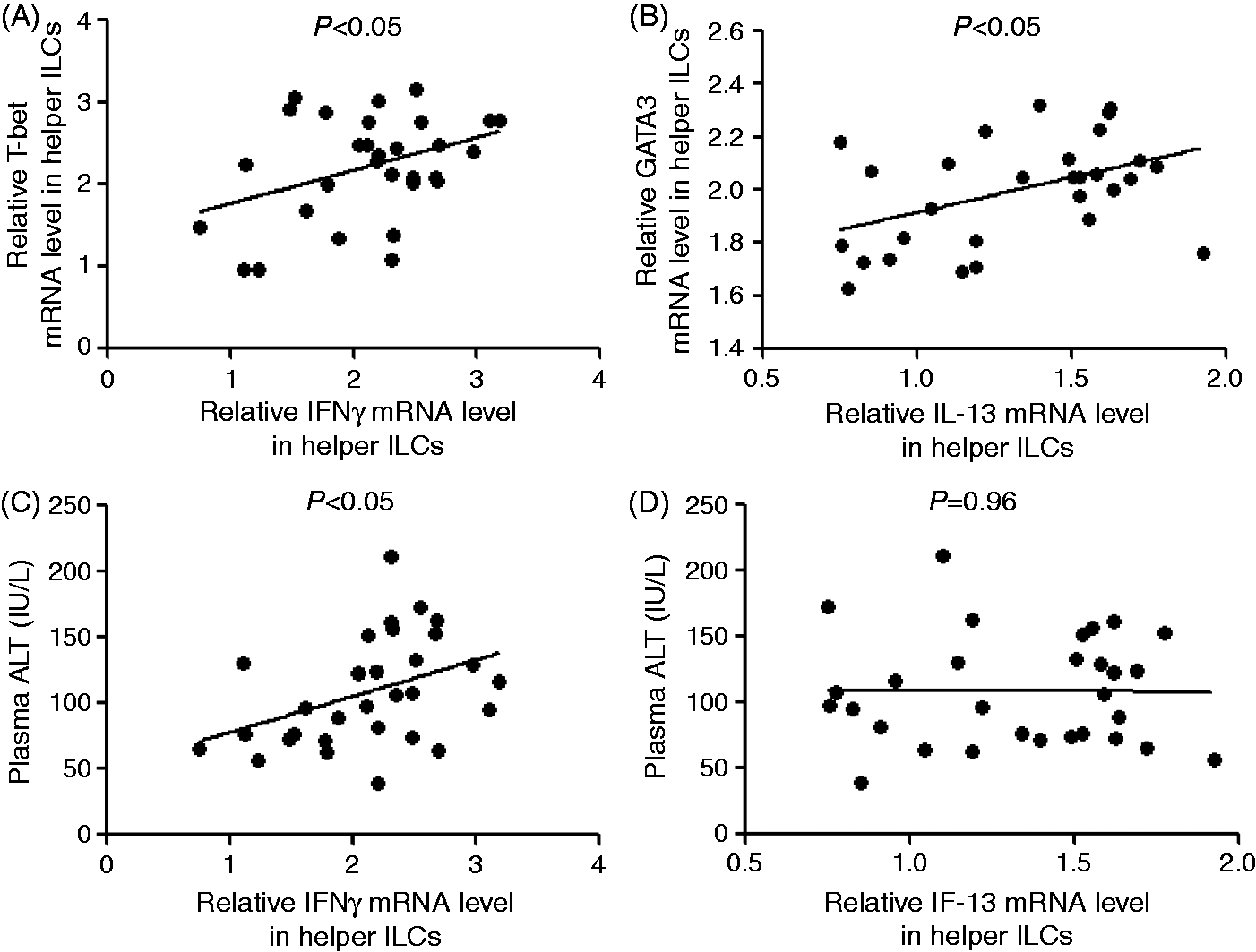
The antiviral activities of telbivudine involve inhibition of HBV DNA synthesis and have no effect on human DNA or DNA from other viruses.
20
Moreover, telbivudine ameliorates the virological and serological response and alters expression of the signature TFs and cytokines of various immune cells in CHB patients.21,22 Therefore, we examined whether the improvements in clinical parameters (integrity of liver function and structure, as well as viral load) induced by telbivudine treatment were associated with CHB-related alteration in the ILC1 and/or ILC2 populations. In agreement with our previously published findings,
22
the telbivudine treatment significantly suppressed HBV propagation and ameliorated hepatic injury, as evidenced in the present study by a gradual but marked decrease in HBV DNA copy number and ALT level observed over the treatment course (Figure 5A). Importantly, we also observed a significant decrease in the mRNA level of the ILC1-related cytokine IFN-γ in the FACS-sorted helper ILCs isolated from the CHB patients following telbivudine treatment; furthermore, the level of IFN-γ in the FACS-sorted helper ILCs was significantly correlated with the ALT level but not with the HBV DNA copy number during long-term telbivudine treatment (Figure 5B). No correlations were observed between the ILC2-related cytokine IL-13 in the FACS-sorted helper ILCs and either the ALT level or the HBV DNA copy number (Figure 5B).
Correlations between ILC1- and ILC2-related cytokines and clinical parameters of CHB and associations with long-term telbivudine treatment. (A) The relative mRNA levels of IFN-γ (ILC1-related) and IL-13 (ILC2-related) in the FACS-sorted helper ILCs, and the ALT levels and the HBV DNA copy number were measured in the CHB patients (n = 30) at the indicated treatment time points. (B) The correlation between the relative mRNA level of IFN-γ or IL-13 in the FACS-sorted helper ILCs and the ALT level or HBV DNA copy numbers from (A) was analyzed. Error bars indicate SEM.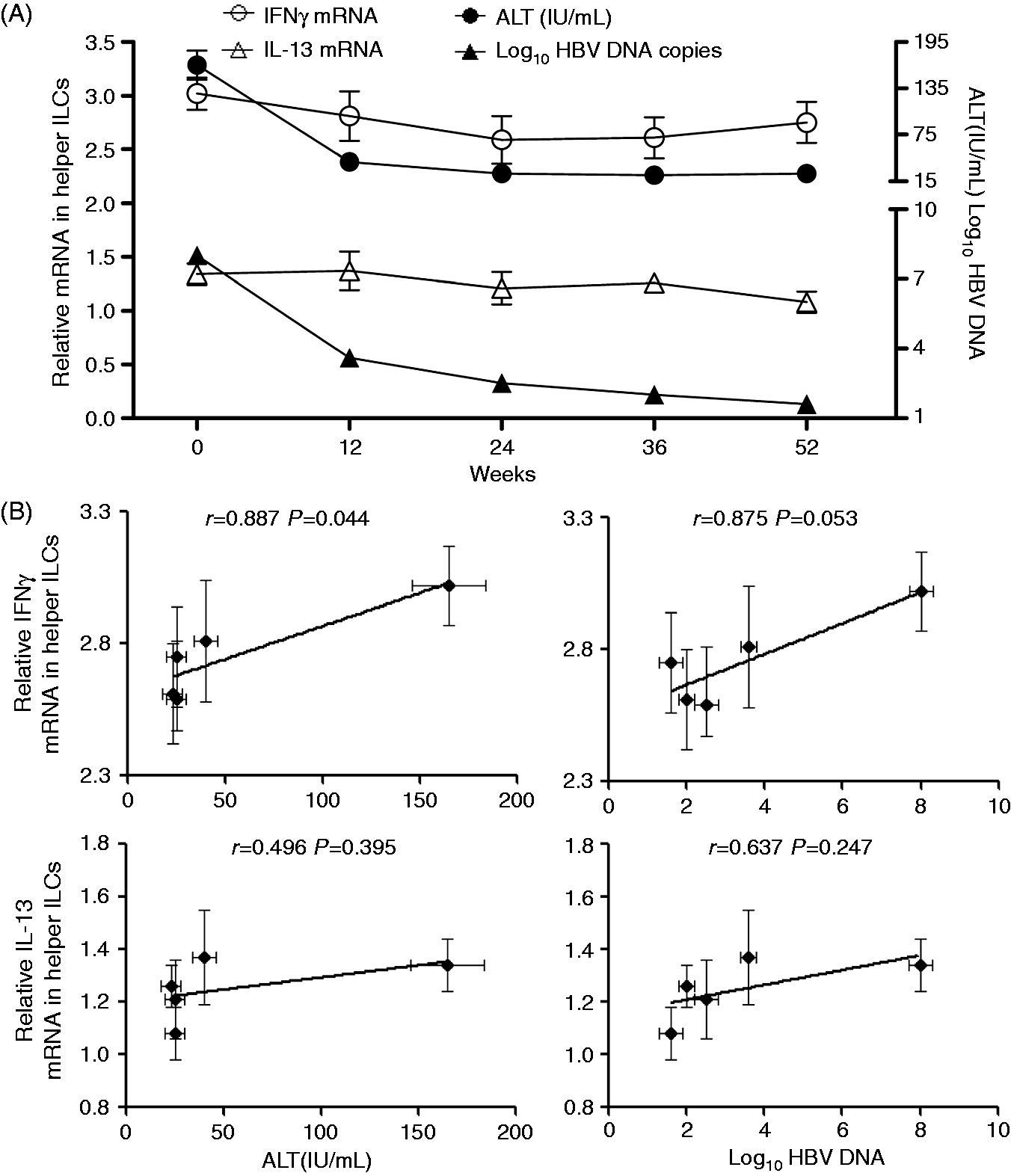
Discussion
Clearance of viral infections is mediated by both the innate and adaptive arms of the immune response; yet, these immune systems can also contribute to the pathogenesis that supports persistent hepatitis B infection and its pathogenic effects on liver structure and function.8,9,13,14 For instance, Th1 and Th2 cells have been implicated in the pathogenesis of hepatic inflammation during HBV infection; 4 however, the roles and related mechanisms of the ILC subpopulations ILC1 and ILC2, which mirror Th1 and Th2 in adaptive immunity, remain unclear. In the current study, we found that CHB patients have a greater frequency and count of peripheral ILC1s than of ILC2s, and that the helper ILCs from CHB patients produced more of the type I cytokine IFN-γ than of the type II cytokines such as IL-4 and IL-13. However, telbivudine treatment for 52 wk almost completely restored the frequency and count of helper ILCs, and the signature ILCTFs and cytokines in CHB patients. Furthermore, the CHB-related aberrant increase in ILC1s was found to be significantly associated with hepatic damage and the telbivudine-induced decrease in hepatic damage was found to be associated with the significantly decreased ILC1-related cytokine level, suggesting pathological roles for ILC1s in CHB pathogenesis.
Studies of group 1 ILCs have revealed their involvement in the pathogenic processes leading to liver injury by various stressor conditions, such as viral hepatitis infection, non-alcoholic fatty liver disease, alcoholic liver disease, cholestatic disease and autoimmune liver disease.23,24 The liver itself is enriched in NK cells, which are members of the group 1 ILCs. The main functions of NK cells are to kill target cells and to produce a variety of immunity-promoting cytokines and chemokines; among these, IFN-γ is the most prominent cytokine produced and contributes to many NK cell-mediated processes in the liver, such as those with antiviral, antifibrotic and antitumorigenic effects. 25 Besides these beneficial roles in host immunodefense, NK cells also play detrimental roles by enhancing hepatocellular damage and inhibiting liver regeneration.26,27 In addition to the NK cells, the other member of the group 1 ILCs, the ILC1 cells, also produce IFN-γ as their main effector cytokine, and this feature suggests the potential for ILC1s to be indirectly involved in liver injury as well, possibly by activating the NK cells. According to some, the observed accumulation of T-bet+IFN-γ+ ILC1s in the inflamed intestine of patients with inflammatory bowel disease suggests that intraepithelial ILC1s might initiate IFN-γ responses against pathogens, thereby contributing to the pathological processes upon dysregulation. 28 While such indirect evidence suggests that ILC1s are involved in liver injury, this theory needs to be confirmed in clinical studies and subjected to detailed investigations in animal models. In the current study, we observed a markedly elevated population and activity of ILC1s in CHB patients, as evidenced by elevated levels of the ILC1-related TF T-bet in helper ILCs, and the elevated effector cytokine IFN-γ and the signaling molecules IL-12/IL-12R, the latter of which is indispensable for differentiation of this particular cell type, in the helper ILCs and the blood and liver tissues of CHB patients. More importantly, we further observed that the CHB-related aberrant activity of the ILC1s was significantly associated with hepatic damage in the CHB patients, regardless of treatment (or treatment length) with telbivudine. These results collectively suggest that ILC1s might be substantially involved in the pathogenesis of CHB.
In contrast to ILC1s, ILC2s may play a protective role in liver injury. Volarevic et al. reported that the IL-33/ST2 axis suppresses liver injury induced by concanavalin A (ConA). 29 The ST2-deficient mice in that study developed significantly more severe hepatitis than their wild type ConA-induced counterparts, possibly as a result of the significantly higher number of mononuclear cells that were observed in the liver and the enhanced levels of pro-inflammatory cytokines that were found in the sera; however, when the wild type mice of that study were administered a single injection of IL-33, the liver injury was attenuated. 29 Arshad et al. also found that IL-33-deficient mice exhibited more severe ConA-induced liver injury than the wild type controls, suggesting a protective effect of IL-33 in ConA hepatitis. 30 In addition, Sakai et al also demonstrated that the IL-33/ST2 exerts protective effects on hepatocytes during hepatic ischemia/reperfusion (I/R) injury, thereby limiting liver injury and reducing the stimulus for inflammation. 31 The hypothesized protective function of IL-33/ST2 was further verified by Li et al. in a study of the mouse model of hepatic I/R injury. 32 These collective studies have demonstrated that the IL-33/ST2 axis can act as a protective mechanism against various types of hepatitis; however, whether ILC2s play a substantial role in these processes remains unclear because the IL-33/ST2 axis is related to both ILC2s and Th2 cells. 33 Nevertheless, a recent study by Liang et al. directly demonstrated that ILC2s are involved in acute liver injury by using an adenovirus (Ad)-induced hepatitis mouse model. 34 Specifically, the Ad-infection was shown to induce a steady increase in the hepatic expression of both IL-33 and its receptor ST2 during the first wk of infection. Treatment with exogenous IL-33 led to a substantial decrease in the model’s liver injury, which was accompanied by enhancement of both type 1 (IL-2 and IFN-γ) and type 2 (IL-5 and IL-13) immune responses to the infection. Interestingly, IL-33 was also demonstrated in that study to strongly induce the presence of novel nuocytes (the alternative name of ILC2s) in the livers and spleens of the Ad-infected mice, and this effect was accompanied by suppressed expression of pro-inflammatory cytokines, such as TNF-α, by hepatic T cells and macrophages. Thus, the hepatoprotective roles of IL-33 in an inflammatory condition might be, at least partially, mediated by the immunomodulatory roles of ILC2s. In the current study, we also observed CHB-related elevations in expression of the ILC2-related factors, including the signature TF GATA3 and effector cytokine IL-13, and the differentiation-related cytokine IL-33 and its receptor ST2. However, the observed elevations for each of these factors were lower than those observed for the ILC1-related factors in the CHB patients. Furthermore, the activity of ILC2s did not appear to be correlated with either HBV copy number or liver damage.
Altogether, the results from our study demonstrate the potential pro-inflammatory roles for ILC1s in CHB pathogenesis, in which the suppressive ILC2s do not counteract (inhibit) the elevated ILC1s activity or the consequent liver damage. Nevertheless, further investigations using suitable knock-out mouse models of the individual ILC subpopulations will help to more precisely clarify their particular roles and mechanisms in CHB, and may provide insights into related factors and pathways with potential as targets for molecular strategies that will improve management of this disease. In addition, the observations that telbivudine treatment significantly restored the elevated levels of ILC1s and ILC2s in CHB patients suggest the novel mechanisms underlying the therapeutic roles of telbivudine for CHB, in addition to its influence on the frequency and activity of Th17 and regulatory T cells in CHB patients. 22
Footnotes
Funding
This work was supported by grants from the Major State Basic Research Development Program of China (No. 2013CB531503), the National Natural Science Foundation of China (Nos. 31170839 and 30930086), the Program for Changjiang Scholars and Innovative Research Team in University (PCSIRT 10521) and the NSFC Major Collaborative Project (No. 81220108024).
Conflicts of interest
The authors do no have any potential conflicts of interest to declare.
