Abstract
Objective
A meta-analysis to compare the efficacy and safety of telbivudine (TBV) and lamivudine (LAM) in patients with chronic hepatitis B (CHB), assessed via changes in serum alanine aminotransferase (ALT) levels.
Method
The electronic literature databases PubMed®, Embase®, Web of Science, Cochrane Library, CISCOM, CINAHL, Google Scholar, China BioMedicine and China National Knowledge Infrastructure were searched for relevant studies. The effect of TBV and LAM treatment on serum ALT was assessed using standard mean differences (SMDs) and 95% confidence intervals (CI).
Results
The meta-analysis included six studies (TBV
Conclusion
Both LAM and TBV are effective in normalizing ALT levels in patients with CHB, but TBV may be a better choice due to its lower rates of drug resistance.
Introduction
Chronic hepatitis B (CHB) is a major public health threat worldwide, 1 and is one of the most frequent causes of hepatocellular carcinoma. 2 CHB is caused by hepatitis B virus (HBV), which is transmitted in bodily fluids (for example, via used hypodermic needles, blood transfusion, vertical transmission from mother to child during birth, and sexual transmission). 3 Untreated patients are vulnerable to liver-related sequelae. 4 An estimated 5% of the total worldwide population is infected with HBV, and approximately 500 000 patients annually die of HBV-related complications, including CHB. 5 The incidence of CHB varies widely, with the highest rates in the Asia–Pacific and sub Saharan Africa. 6 Risk factors for the development of CHB include vertical transmission of HBV from mother to infant, 7 age, alcohol use and HIV infection. 8
The antiviral nucleoside analogues telbivudine (TBV) and lamivudine (LAM) are promising treatments for CHB. 9 HBV DNA levels are the most critical clinical criterion for the judgement of antiviral treatment efficacy, and patients with persistently elevated HBV DNA have a heightened risk of cancers including hepatocellular carcinoma.10,11 The serum alanine aminotransferase (ALT) concentration is an important marker of CHB 12 and is commonly used to assess hepatocellular injury and liver health. 12 LAM effectively reduces serum viral load, improves ALT levels and liver fibrosis, and accelerates hepatitis B e antigen (HBeAg) seroconversion rates.13–15 Treatment with LAM reduces ALT levels in patients who were positive for hepatitis B surface antigen (HBsAg), suggesting that LAM can inhibit HBV transmission and normalize ALT. 16 TBV has been shown to be more effective than LAM in the treatment of patients with CHB,17–19 as well having a superior therapeutic response and resistance profile.18,20–23
The aim of the present meta-analysis was to compare the efficacy of TBV and LAM in the treatment of CHB (assessed via ALT levels and HBV DNA stratum).
Materials and methods
Data sources and keywords
Eligible studies published up to 30 June 2014 were identified by searching the electronic literature databases PubMed®, Embase®, Web of Science, Cochrane Library, CISCOM, CINAHL, Google Scholar, China BioMedicine (CBM) and China National Knowledge Infrastructure (CNKI). The following keywords and search strategies were used: (“hepatitis B surface antigens” OR “hepatitis B surface antigen” OR “Australia antigen” OR “HBsAg” OR “hepatitis B antigen” OR “hepatitis B virus”) AND (“alanine transaminase” OR “glutamic alanine transaminase” OR “alanine 2 oxoglutarate aminotransferase” OR “alanine aminotransferase” OR “glutamic pyruvic transaminase” OR “SGPT” OR “ALT” OR “GPT”) AND (“telbivudine” OR “beta-L-2′-deoxythymidine” OR “Tyzeka®” OR “TBV”) AND (“lamivudine” OR “2′,3′ Dideoxy 3′ thiacytidine” OR “3TC” OR “Epivir®” OR “GR109714X” OR “BCH189”). In addition, reference lists of relevant articles were searched to identify additional relevant papers. No language restrictions were set in this meta-analysis. When articles provided unclear additional data in their original publications, the first authors were contacted for clarification.
Selection criteria
To be included, studies had to fulfil all of the following criteria: (i) patients with CHB; (ii) human clinical cohort studies or diagnostic tests focused on CHB diagnosis with administration of LAM or TBV; (iii) pre and post-treatment data for serum ALT levels; (4) report adjusted standard mean differences (SMDs) and 95% confidence intervals (CI) for ALT serum level; (5) number of patients stated; and (6) >20 patients. When studies had populations overlapping >50% with two or more papers, the one with the most comprehensive population was included. When the same authors published more than one study with the same population, only the latest or most complete was included. All authors approved the final list of included studies.
Data extraction
Data were extracted from the included studies by two investigators (M.D. and G.M.X.) independently and in duplicate. If necessary, consensus was achieved by discussion and re-examination. The following data were extracted from eligible studies: surname of first author; date of publication; source of publication; study type; study design; sample size; age; sex; ethnicity and country of subjects; source of samples; detection method for ALT serum level; time of post-treatment sampling; serum ALT levels; and HBV DNA levels.
Quality assessment
Quality of the included studies was assessed by two investigators (F.L.W. and J.S.Z.) independently and in duplicate, using predefined criteria based on Newcastle-Ottawa Scale (NOS) criteria. 24 The NOS criteria are based on three aspects: (i) subject selection (0–4); (ii) comparability of subjects (0–2); (iii) clinical outcome (0–3). Total NOS scores range from 0 (lowest) to 9 (highest). According to the NOS scores, the included studies were classified as low quality (0–6) or high quality (7–9). Discrepancies regarding NOS scores were resolved by discussion and consultation with an additional reviewer (D.J.X.).
Statistical analyses
The effect size of each study was calculated using
Results
The study selection procedure is displayed in Figure 1. The literature search identified 132 papers on the basis of title and key words. After screening according to exclusion criteria, the final meta-analysis included six studies (TBV Flow diagram of the study selection process in a meta-analysis undertaken to compare the relative efficacy of telbivudine and lamivudine in the treatment of chronic hepatitis B infection (assessed via serum alanine aminotransferase levels). Characteristics of studies included in a meta-analysis undertaken to compare the relative efficacy of telbivudine and lamivudine in the treatment of chronic hepatitis B infection (assessed via serum alanine aminotransferase levels). TBV, telbivudine; LAM, lamivudine; M, male; F, female; NOS, Newcastle–Ottawa Scale; ACA, automatic biochemical analyser; ECLIA

Meta-analyses for the effects of TBV and LAM on serum ALT are shown in Figure 2. There was significant heterogeneity (TBV pretreatment vs post-treatment, 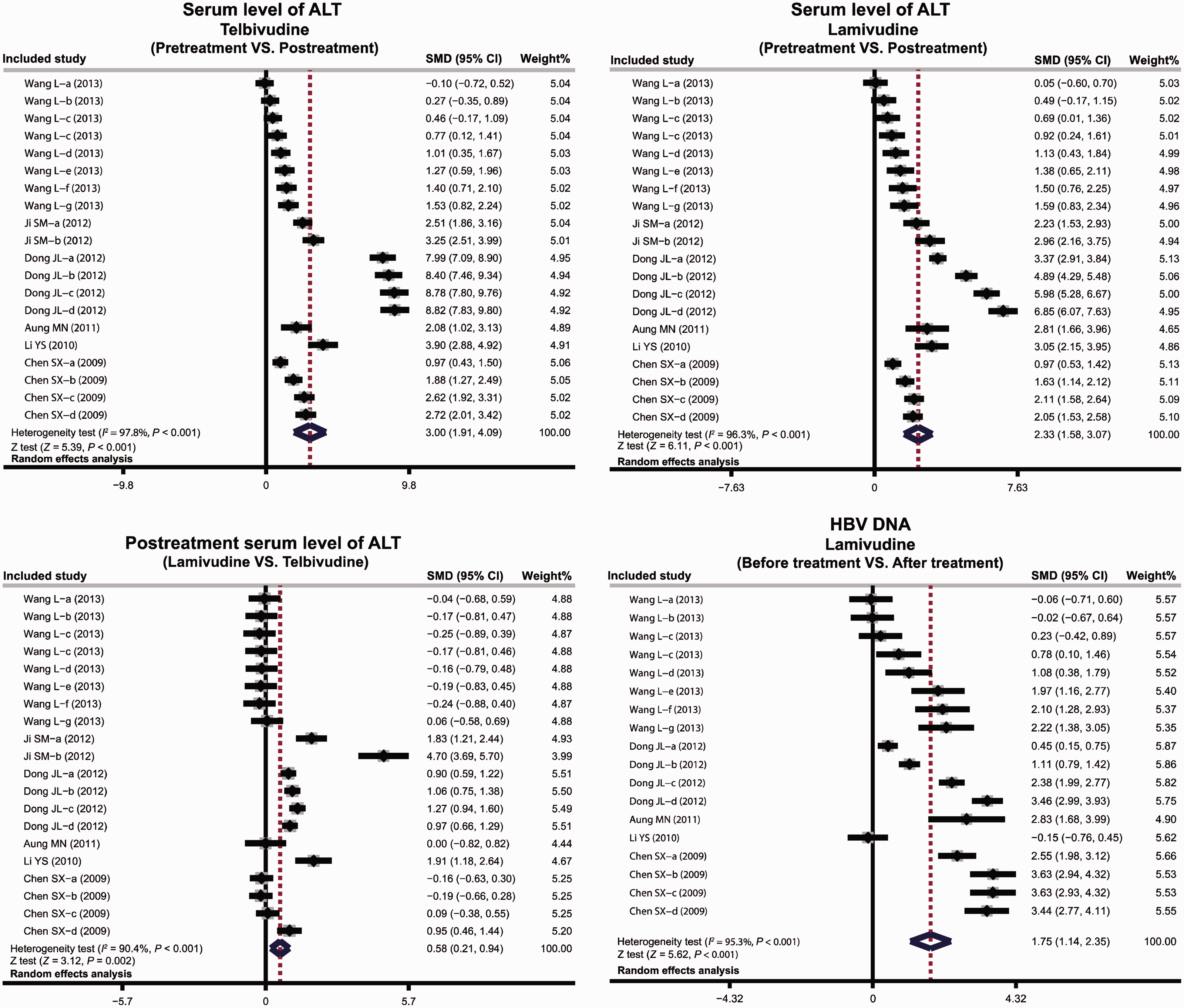
Subgroup analyses based on detection method are shown in Figure 3. For both TBV and LAM, post-treatment ALT levels were significantly lower than pretreatment values for all detection methods ( Subgroup analyses by method of serum alanine aminotransferase (ALT) analysis and time of post-treatment sampling for the effect of telbivudine and lamivudine on serum ALT levels in patients with chronic hepatitis B. Studies: Wang, 2013;
28
Ji, 2012;
32
Dong, 2012;
31
Aung, 2011;
29
Li, 2010;
33
Chen, 2009;
30
a–g, different study timepoints.
Subgroup analyses based on time of post-treatment sampling are shown in Figure 3. Time of post-treatment sampling was classified as <24 or ≥24 weeks. Post-treatment ALT levels were significantly lower than pretreatment values for both TBV and LAM, in both the <24 and ≥24 weeks subgroups (
Sensitivity analyses indicated that the meta-analysis was stable (Figure 4). Funnel plots and Egger’s test found strong evidence for publication bias in the comparison between pre-TBV and post-TBV treatment ALT levels (
Discussion
The current meta-analysis compared the relative efficacy of TBV and LAM in the treatment of CHB infection (assessed via serum ALT levels), and found that both drugs significantly reduced ALT compared with pretreatment levels. In addition, TBV resulted in a significantly greater decrease in ALT levels than LAM.
Studies have shown that LAM can improve liver histology and suppress viral DNA replication in patients with CHB after 1 year of treatment, with reversal of cirrhosis and fibrosis observed after 3 years. 34 Although LAM has a good safety profile in the treatment of HBV infection, resistance (and accompanying viral breakthrough, hepatitis flare and liver decompensation) is a growing problem. 35 TBV has lower resistance rates, causes viral suppression by inhibiting HBV DNA synthesis, 18 and has higher rates of survival, virological response, therapeutic response, HBeAg loss and ALT normalization than LAM.19,36 In addition, TBV has a high seroconversion rate and no carcinogenic, mutagenic or embryonic toxic effects. 37 Other nucleoside analogues include adefovir dipivoxil fumarate (ADF), entecavir (ETV) and tenofovir disoproxil fumarate (TDF). 38 Although ETV and TDF are first-line drugs for CHB, we evaluated LAM due to its clinical relevance and TBV due to its known effect on ALT.7,37,38
Although ALT levels are decreased by treatment with nucleoside analogues such as LAM and TBV, levels have been shown to increase after withdrawal of treatment.39,40 Studies have found that TBV provides greater and more consistent suppression of HBV DNA than LAM, which may be reflected in normalization of ALT levels.7,41 The between-drug difference in post-treatment ALT levels in the present analysis may be a result of LAM drug resistance. The current meta-analysis indicated that LAM treatment significantly decreased HBV DNA levels compared with baseline, which may lead to a reduction in HBV-related complications. 42 Our data are in accordance with others who found that TBV resulted in significantly greater decreases in serum ALT than LAM, as well as a significantly higher survival rate in patients with CHB. 20
We conducted subgroup analyses to determine whether detection method had any effect on our findings. In all subgroups (ACA, ECLIA and unknown), both TBV and LAM resulted in significant decreases in ALT levels, but the superiority of TBV could not be demonstrated when samples were analysed using ECLIA. This could be explained by differences between detection methods, but may also be due to the small number of studies involved. In addition, time of post-treatment sampling had no effect on the finding that TBV and LAM decreased ALT levels, but there was no significant between-drug difference when ALT was assessed at <24 weeks post-treatment. This finding suggests that the difference between LAM and TBV is not apparent in the early stages of treatment. Both TBV and LAM are capable of normalizing ALT levels in patients with CHB, but TBV is preferred because of its relatively low resistance levels.
The present study had several limitations. Since it was a cross-sectional retrospective study, our analysis did not take into consideration changes in viral load or treatment efficacy over time. In addition, due to limited data, it was not possible to analyse phenotypic or genotypic resistance, restricting our results to a narrow clinical application. We did not include some unpublished articles and abstracts due to incomplete data. Additionally, three of the six included studies were of low methodological quality due to insufficient data and lack of adjustment for confounding factors. This may be a source of heterogeneity and could affect our findings. Differences in included criteria, follow-up periods, treatment efficacy and severity of disease are also potential sources of heterogeneity. Although each study population was well defined and had similar inclusion criteria, there may be unknown factors that could have influenced our results. The participants included in this study did not differ significantly from other Asian population-based or hospital-based patients in terms of age/sex distribution, HBeAg and HBsAg status, or HBV DNA levels. The fact that all of the studies investigated Asian populations limits the applicability of these findings to other ethnicities. Finally, these findings may be influenced by the high levels of heterogeneity in our study. Further, larger scale clinical trials are required to contribute to the screening and development of individualized treatment schemes for patients with CHB.
In conclusion, both LAM and TBV are effective in normalizing ALT levels in patients with CHB, but TBV may be a better choice due to its lower rates of drug resistance.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
The study was supported by the Clinical Research Scheme of Heat Clearing and Ddetoxicating Fuzheng Quxie Treatment of HBeAg Negative Chronic Hepatitis B (20121139, Guangdong Province Pharmaceutical Administration). We would like to acknowledge the reviewers for their helpful comments on this paper.
