Abstract
Objective
To investigate the clinical characteristics of hepatitis B surface antigen (HBsAg) seroclearance in patients with chronic hepatitis B virus (HBV) infection.
Methods
Patients with chronic HBV infection who achieved sustained virological response (SVR) within 6 years of ceasing formal antiviral treatment were assessed for HBsAg seroclearance (defined as loss of serum HBsAg on repeated testing for a period of >6 months), using enzyme immunoassays. Phase of HBV infection and liver function (serum alanine aminotransferase [ALT] and aspartate aminotransferase [AST] levels) and HBV DNA levels were also assessed.
Results
In total, 272 patients with chronic HBV and SVR were included; HBsAg seroclearance was achieved in 42 patients and not achieved in 230 patients. Serum HBsAg and ALT levels, ratios of HBsAg to HBV DNA and ratios of AST to ALT were significantly different between patients achieving, and not achieving, HBsAg seroclearance. The area under the receiver operating characteristic (ROC) curve of HBsAg levels for predicting the likelihood of HBsAg seroclearance was 0.85; the cut-off value was 203.86 IU/ml.
Conclusions
These data demonstrate that HBsAg seroclearance was independently associated with host immunity, serum HBsAg level, serum ALT level, serum HBsAg to HBV DNA ratio and timing of drug therapy within the course of chronic HBV infection.
Keywords
Introduction
Hepatitis B virus (HBV) infection is one of the most serious global health problems, with 350–400 million people infected worldwide. 1 Approximately 1 million deaths occur annually, due to the long-term complications of HBV infection, which include cirrhosis, liver failure and hepatocellular carcinoma. 2 Among the clinical parameters of HBV infection, hepatitis B surface antigen (HBsAg) has been regarded as a potential predictor of response to chronic HBV treatment and as a partial surrogate marker of HBV covalently closed circular DNA (cccDNA). 3 HBV cccDNA, located in hepatocytes, is the intracellular template of HBV DNA, therefore HBsAg is one of the subviral replication products of cccDNA. Obtaining liver tissues from patients in order to detect intrahepatic cccDNA levels in the clinical setting is difficult, consequently serum HBsAg is regarded as a surrogate marker of this molecule. HBsAg seroclearance is one of the ultimate goals of anti-HBV therapy, therefore studies have investigated the predictive value of HBsAg seroclearance.4,5 However, to date, the clinical characteristics of HBV patients who can achieve HBsAg seroclearance have not been fully elucidated. In the present study we evaluated patient characteristics (including phase of chronic HBV infection and liver function) by investigating the differences in clinical parameters between those who had achieved HBsAg seroclearance and those who had not, within 6 years of cessation of antiviral therapy.
Patients and methods
Patients and treatments
Patients with chronic HBV infection who had achieved a sustained virological response (SVR; defined as undetectable serum HBV DNA for a period of at least 6 months), 6 within a 6-year time period between July 2006 and July 2012, were eligible for study inclusion. The study was carried out in the Infectious Diseases Department of the Shenzhen Third People’s Hospital, Shenzhen, Guangdong, China.
Patients were excluded if they were coinfected with hepatitis delta virus, hepatitis C virus, or human immunodeficiency virus; tests for these viruses were undertaken using standard laboratory methods. Patients with Wilson’s disease, autoimmune hepatitis or primary biliary cirrhosis, and those with a substantial intake of alcohol (20 g per day for female patients; 30 g per day for male patients) were also excluded.
Hepatitis B surface antigen seroclearance was defined as loss of serum HBsAg on repeated testing for a period of >6 months and during all subsequent follow-ups until time of analysis. 7 During the study period, patients were treated with nucleoside analogues (adefovir, 10 mg/day or lamivudine, 100 mg/day), either as monotherapy or with pegylated interferon-α (100 µg/week) for >6 months, according to clinical practice guidelines. 7
All patients were HBV treatment naïve before enrolment, had serum HBV DNA levels ≥100 copies/ml, and had been persistently HBsAg positive for >6 months. Patients gave verbal consent to enter the study and the study itself was approved by the Ethics Committee of Shenzhen Third People’s Hospital
Laboratory measurements
Serum HBsAg and Hepatitis Be antigen (HBeAg) levels were quantified through enzyme immunoassays (ARCHITECT platform; Abbott Laboratories, Abbott Park, IL, USA) according to the manufacturer’s instructions.
Serum alanine aminotransferase (ALT) was measured using the ALAT (GPT) FS kit and aspartate aminotransferase (AST) levels were measured using the ASAT (GOT) FS kit (both from DiaSys Diagnostic Systems, Holzheim, Germany), according to the manufacturer’s instructions. Serum HBV DNA levels were quantified using the Cobas® TaqMan® real time–polymerase chain reaction analyser (Roche Diagnostics, Indianapolis, IN, USA), as previously described. 8 As HBV genotyping can influence HBV replication in patients with chronic HBV infection, HBV genotyping (B/C) was performed by restriction fragment length polymorphism 9 using the restriction enzyme Tsp509I (obtained from MBI Fermentas, Canada) in 120 patients. Not all patients could be tested, for various reasons including cost and insufficient HBV DNA samples. Patients were categorized according to their phase of chronic HBV infection, as follows: immune tolerance (IT); immune clearance (IC); HBeAg negative low-replicative (LR); HBeAg negative hepatitis (ENH).
Statistical analyses
Continuous variables were expressed as the mean (range) and were analysed using nonpaired Student’s
Results
Clinical parameters of patients with chronic hepatitis B virus (HBV) infection, stratified according to those who did not achieve hepatitis B virus surface antigen (HBsAg) seroclearance (HBsAg seropositive) and those who did achieve HBsAg seroclearance (HBsAg seronegative).

Data presented as
ALT, alanine aminotransferase (normal range 5–40 U/l); AST, aspartate aminotransferase (normal range 5–40 U/l).
Patients were categorized according to their phase of chronic HBV infection, as follows: immune tolerance (IT); immune clearance (IC); HbeAg-negative low-replicative (LR); HbeAg-negative hepatitis (ENH). HBsAg negative: 0–0.05 IU/ml; HBV DNA-negative: <100 copies/ml.
Nonpaired Student’s
χ2-test.
Mann–Whitney
Patients not achieving HBsAg seroclearance were categorized according to their phase of chronic HBV infection (IT, IC, LR, ENH), as shown in Table 1. There was a significant difference in the distribution of the phase of chronic HBV infection between the group achieving seroclearance and the group not achieving seroclearance. (
The area under the ROC curve of HBsAg levels for predicting the likelihood of HBsAg seroclearance was 0.85 (95% CI: 0.77–0.93; Receiver operating characteristic curve of serum hepatitis B virus surface antigen (HBsAg) levels for predicting the likelihood of HBsAg seroclearance in patients with chronic hepatitis B virus infection. Patients with chronic hepatitis B infection whose serum HBsAg levels were <203.86 IU/ml (defined as the cut-off value; HBsAg negative was defined as <0.05 IU/ml) may likely achieve HBsAg seroclearance in the following 6 years. The cut-off value had a sensitivity of 83.9%, specificity of 81.0%, positive predictive value of 83.3% and negative predictive value of 86.1%.
Cumulative rate analysis showed that the SVR rate was significantly higher in patients achieving HBsAg seroclearance compared with those not achieving seroclearance. In the group that went on to eliminate HBsAg, 100% of patients had achieved SVR by 53 months, compared with 99.1% achieving SVR by 60 months, in those who did not go on to achieve HBsAg seroclearance ( Cumulative rates of sustained virological response (SVR) according to hepatitis B surface antigen (HBsAg) seroclearance, in patients with chronic hepatitis B (CHB) virus infection: 100% of those who eliminated HBsAg achieved SVR by 53 months; 99.1% of those who did not go on to achieve HBsAg seroclearance achieved SVR by 60 months (
In patients achieving HBsAg seroclearance, a serum ALT level >80 U/l was significantly associated with HBsAg seroclearance: HBsAg seroclearance cumulative percentage rates at 12, 24, 48 and 60 months were 68.18%, 77.27%, 86.36% and 95.45%, respectively, in patients with serum ALT levels ≥80 U/l, and 15%, 30%, 60% and 95%, respectively, in patients with serum ALT levels <80 U/l ( Cumulative rates of hepatitis B virus surface antigen (HBsAg) seroclearance according to liver function, in patients with chronic hepatitis B (CHB) virus infection. HBsAg seroclearance cumulative percentage rates at 12, 24, 48 and 60 months were 68.18%, 77.27%, 86.36% and 95.45%, respectively, in patients with serum alanine aminotransferase (ALT) levels ≥80 U/l, and 15%, 30%, 60% and 95%, respectively, in patients with serum ALT levels <80 U/l (P = 0.01).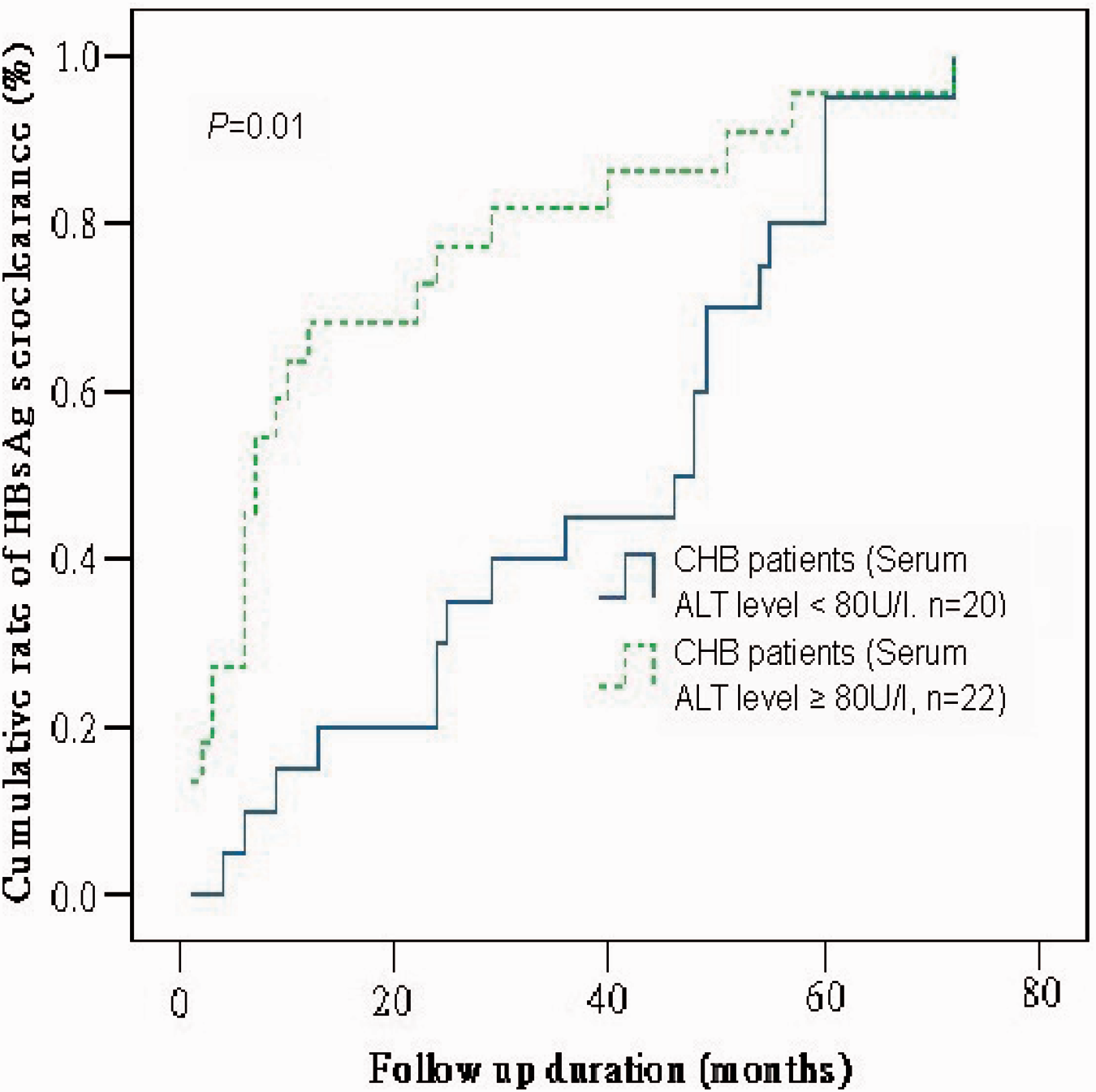
Discussion
Hepatitis B serum antigens are generated from two HBV mRNA transcripts, with subsequent translation resulting in small, medium and large surface envelope proteins. Newly synthesized envelope protein interacts with mature HBV nucleocapsids at the endoplasmic reticulum, prior to being secreted from the hepatocyte. 10 Studies have documented that patients who achieve HBsAg seroclearance before the development of cirrhosis have a considerably more favourable long-term prognosis compared with those who remain HBsAg positive, even when HBV DNA can still be detected within their hepatocytes. 11
In our study, serum HBsAg levels in patients achieving HBsAg seroclearance were significantly lower than those in patients not achieving HBsAg seroclearance. This finding suggests that transcription of cccDNA for surface envelope proteins in the former may be suppressed by various intrinsic factors, such as degree of cccDNA methylation or acetylation of the surrounding histones.12–14 While our observation is consistent with other preliminary studies, our cut-off value for the serum HBsAg level was higher than that reported elsewhere (203.86 IU/ml in the present study, versus 100 IU/ml elsewhere).15,16 This may be due to our recruitment of greater numbers of patients who went on to achieve HBsAg seroclearance.
As HBV genotype can influence HBV replication in patients with chronic HBV infection, HBV genotyping was performed. 9 However, no significant difference was found in the genotyping of the two groups and we believe this had no influence on the results.
Our data indicate that the immunity of patients achieving HBsAg seroclearance may be more robust than that of patients not achieving HBsAg seroclearance. On one hand, the percentage of patients achieving HBsAg seroclearance who were in the IC phase of HBV infection was significantly greater than the percentage in IC who did not achieve HBsAg seroclearance (47.62% versus 21.30%). In addition, the percentages of patients in the IT and ENH phases were also substantially higher in those in the seropostive group (32.74% and 33.04%, respectively) compared with those in the seronegative group (14.29% and 23.81%, respectively). Patients with chronic HBV infection who transform spontaneously into the IC phase have been described as generally having a ‘potent’ immune response. 17 Conversely, it was confirmed that the function of natural killer cells may be impaired in IT phase, 18 and the emergence of viral mutations (such as BCP/PC variants in ENH phase) may result in immune escape. 19 It was also suggested that increased serum ALT levels and a low AST/ALT ratio predict potent anti-HBV immunity in patients with chronic HBV; 20 these traits were both identified in the patients achieving HBsAg seroclearance in our study.
Contrary to our data, other research has indicated that there was no significant difference in serum ALT levels between patients achieving, and not achieving, HBsAg seroclearance. 16 This discrepancy may be due to the fact that other research has focused on spontaneous HBsAg seroclearance and did not take into account the effects of drug therapy. Pharmacotherapeutic choices may be influential in determining the rate of HBsAg seroclearance in patients with chronic HBV infection. Compared with combination therapy of nucleoside analogue and interferon-α, nucleoside analogue therapy alone has a limited impact on HBsAg seroclearance. 21 Prolonged drug use may lead to mutations and systemic side-effects, and premature termination of drug therapy can result in resurgence of serum HBsAg and HBV-DNA levels. Otherwise, cccDNA is the intracellular template for HBsAg production and, although no available drugs can completely eliminate existing cccDNA in the nuclei of the infected hepatocyte, there may be differential treatment effects at different hepatocyte proliferation phases (for example the initial proliferation, exponential proliferation and growth confluency phases). 22
Several lines of evidence have suggested that HBV replication is highly dependent on the growth status of hepatocytes; clinical specimens have shown low levels of intrahepatic or serum HBV DNA during severe acute hepatitis at the time of active liver regeneration, representing a stage of rapid cell growth. 23 Cell culture data indicate that lamivudine can significantly eliminate cccDNA in the exponential proliferation phase compared with other phases, possibly due to a dilution effect. 24 Another study demonstrated that high serum ALT levels may predict damage to hepatocytes, resulting in rapid cell proliferation. 25 Cumulative rate analysis in our study demonstrated that patients achieving HBsAg seroclearance also achieved SVR earlier than those who did not. In the former, patients with a higher serum ALT level (≥80 U/l) acquired HBsAg seroclearance earlier than those with a lower serum ALT level <80 U/l, indicating that drug therapy during the rapid cell proliferation period may have a positive effect in achieving HBsAg seroclearance.
The serum HBsAg/HBV DNA ratio reflects the association between HBsAg production and HBV replication in patients with chronic HBV. Research has found that the ratio generally ranged between 0.5 and 0.6, and was significantly higher in HBeAg-negative patients than in HBeAg-positive patients. 10 This may be due to the fact that HBsAg production is preserved under preferential immunity pressure in HBeAg-negative patients; 3 alternatively it may be due to HBV integration in the host genome. 20 One study observed that a large number of HBV integration events was positively associated with serum HBsAg levels according to whole genome sequencing. 26 Another paper reported that HBeAg-negative patients usually had a longer HBV infection history, and were expected to have more extensive HBV DNA integration, compared with HBeAg-positive patients. 27 While the serum HBsAg to HBV DNA ratio in patients not achieving HBsAg seroclearance in our study (0.58) was similar to that observed by others (0.5–0.6), it was significantly lower in patients achieving HBsAg seroclearance (0.35). This may be due to enhanced host immunity in relation to the control of subviral production, or because fewer HBV integration events occurred in patients achieving HBsAg seroclearance than in those not achieving HBsAg seroclearance, as the proportion of HBeAg negative patients was significantly higher in the latter group, in our study.
In conclusion, our study identified several clinical characteristics of patients with chronic HBV infection that may help to predict those most likely to achieve HBsAg seroclearance after antiviral treatment. These characteristics include having a vigorous immune response, having low serum HBsAg levels (cut-off value, 203.86 IU/ml), having high serum ALT levels and having a low serum HBsAg to HBV DNA ratio. Appropriate antiviral therapy during the rapid cell-proliferation period may also be helpful in achieving HBsAg seroclearance.
Footnotes
Acknowledgements
The authors thank XC Chen and M Wang from the Institute of Hepatology, Shenzhen Third People’s Hospital for their technical assistance.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
