Abstract
The distal compartments of the udder are the first to interact with invading pathogens. The regulatory and effector functions of two major teat regions [Fürstenberg's rosette (FR); teat cistern (TC)] are largely unknown. The objective of this study was to establish an in vitro model with explants of the FR and the TC to analyse their response towards Escherichia coli LPS and Staphylococcus aureus lipoteichoic acid (LTA). Quantitative stereological analysis confirmed differences in the cellular composition of FR and TC explants. Chemokine (CXCL8, CCL5, CCL20) and TNF-α mRNA were expressed at low levels in both locations. Explant stimulation with LPS increased the mRNA abundance of all tested chemokines and TNF-α. Stimulation with LTA only induced CCL20 and CXCL8. LPS- and LTA-stimulated explant supernatants contained CXCL8 and CXCL3. Supernatants significantly attracted neutrophils in vitro. Compared with TC, the FR showed high constitutive mRNA expression of S100 proteins (A8, A9, A12). In the TC, both LPS and LTA significantly induced S100A8, whereas S100A9 and S100A12 expression was only induced by LPS. The novel model system underpins the role of the teat for recognising pathogens and shaping a pathogen- and location-specific immune response.
Introduction
Bovine mastitis is a harmful yet common disease in the dairy industry and is primarily caused by bacterial infections. 1 The predominant pathogen involved in cases of severe clinical mastitis is Escherichia coli (E. coli), whereas subclinical mastitis is frequently caused by Staphylococcus aureus (S. aureus).2,3 Differences in the pathogenesis are reflected by different gene expression patterns in host tissues. 4 Generally, E. coli intramammary infections (IMI) result in a much higher number of regulated genes compared with IMI caused by S. aureus. 5
Studies on host–pathogen interactions in the bovine udder have focused mainly on lobulo-alveolar tissue and mammary epithelial cells (MEC).6–8 The role of the teat in mastitis pathogenesis has received less attention. 9 Mastitis pathogens predominantly invade the mammary gland via the teat canal and initially encounter the surface of the Fürstenberg's rosette (FR) and the teat cistern (TC). The FR is a small region (2–3 mm) between the streak canal and the TC. A characteristic feature of the FR is the highly folded mucosal surface and the presence of leukocytes. 10 Whether the FR is just part of the physical barrier inhibiting bacterial entry into the teat or whether it has a distinct immunological role is not fully understood. The differential response of the FR and the TC has recently been studied in an in vivo model after experimental E. coli challenge. 9 Twelve hours after intramammary challenge, the most pronounced host response was found in the teat and gland cistern. Overall, these observations underpin the hypothesis that the teat fulfils an important sentinel function and possibly triggers the clinical course of mastitis.
In order to analyse the PAMP-specific response patterns in the FR and TC, we set up an explant culture system for bovine teat tissue, in which we tested the response to highly purified LPS and lipoteichoic acid (LTA) in vitro. These PAMPs were obtained from E. coli and S. aureus, and have been previously used for in vivo studies and are known to cause clinical or subclinical mastitis respectively.11,12 Analysis of PAMP-specific responses was based on frequently upregulated genes in PAMP-stimulated bovine mammary cells and udder parenchyma.7–9,13 In addition to the inflammatory cytokine TNF-α, most of the regulated factors were chemokines, including the CC-motif ligands 5 and 20 (CCL5 and CCL20 respectively), and the CXC-motif ligands 8 (CXCL8; IL-8) and 3 (CXCL3; GRO-γ). Another set of early regulated genes during bovine mastitis codes for S100 calcium-binding proteins with reported inflammatory, antimicrobial and chemotactic functions.8,14
We focused on three S100 proteins, namely S100A8 [calgranulin A; myeloid-related protein 8 (MRP8)], S100A9 (calgranulin B; MRP14) and S100A12 [calgranulin C; extracellular newly identified RAGE binding protein (EN-RAGE)]. They were repeatedly shown to be upregulated in pathogen-challenged bovine mammary tissue and cells.8,9,15 Whether the major locations of the teat (FR, TC) display pathogen-specific differences in their response is currently unknown. This was addressed with the presented FR and TC explant culture system.
Material and methods
Source of tissues
Tissue was obtained from 21 clinically healthy lactating dairy cows at the abattoir. A thorough examination of the udder and collection of sterile milk samples were performed immediately before slaughtering. The California Mastitis Test (CMT; WDT, Garbsen, Germany) was carried out for semiquantitative determination of the somatic cell count in milk according to the manufacturer’s instructions. Only cows with no signs of clinical mastitis and no or mild gelling in the CMT were chosen for tissue sampling. Milk samples were transported on ice to the laboratory and were plated on Columbia Sheep Blood Agar, Violet Red Bile Agar and Edwards Agar (all from Oxoid, Wesel, Germany) and incubated for 48 h at 37℃. Tissue from udder quarters that showed bacterial growth of Streptococcus spp., S. aureus or Enterobacteriaceae in milk samples were excluded from the study. Immediately after slaughtering, the entire udder was dissected from the animal and was thoroughly cleaned with water, followed by disinfection with 70% ethanol (Roth, Karlsruhe, Germany). After cutting off the teats, a Vasoflo-T. mandrin (Dispomed, Gelnhausen, Germany) was inserted in the teat canal to avoid damage before opening the teat longitudinally with a sterile scalpel. The opened teat was pinned in the lamina muscularis with sterile cannulas on a polystyrene plate covered with sterile aluminium foil.
From each teat, four tissue samples of the FR and four tissue samples of the TC were collected within 20 min after slaughtering (Figure 1). Sample collection was carried out using a BARD magnum Biopsy Instrument and a 12-Gauge Core Tissue Biopsy Needle (both BARD, Covington, GA, USA). Samples were immediately placed in 4℃ PBS and stored on ice until further processing. Explants were prepared under sterile conditions. To ensure approximately equal sample sizes we prepared cubus-shaped explants (2 × 2 × 2 mm, 6–10 mg), including the mucosal epithelium after removing the main part of connective tissue.
Experimental set-up: 22 udder quarters from six animals were sampled. Four tissue samples were taken from the TC and FR respectively. After sample preparation, two tissue explants/location were stimulated for 3 h with either 1 µg/ml LPS or 10 µg/ml LTA, and two remained unstimulated. The mRNA was extracted from two pooled explants/location either stimulated or unstimulated.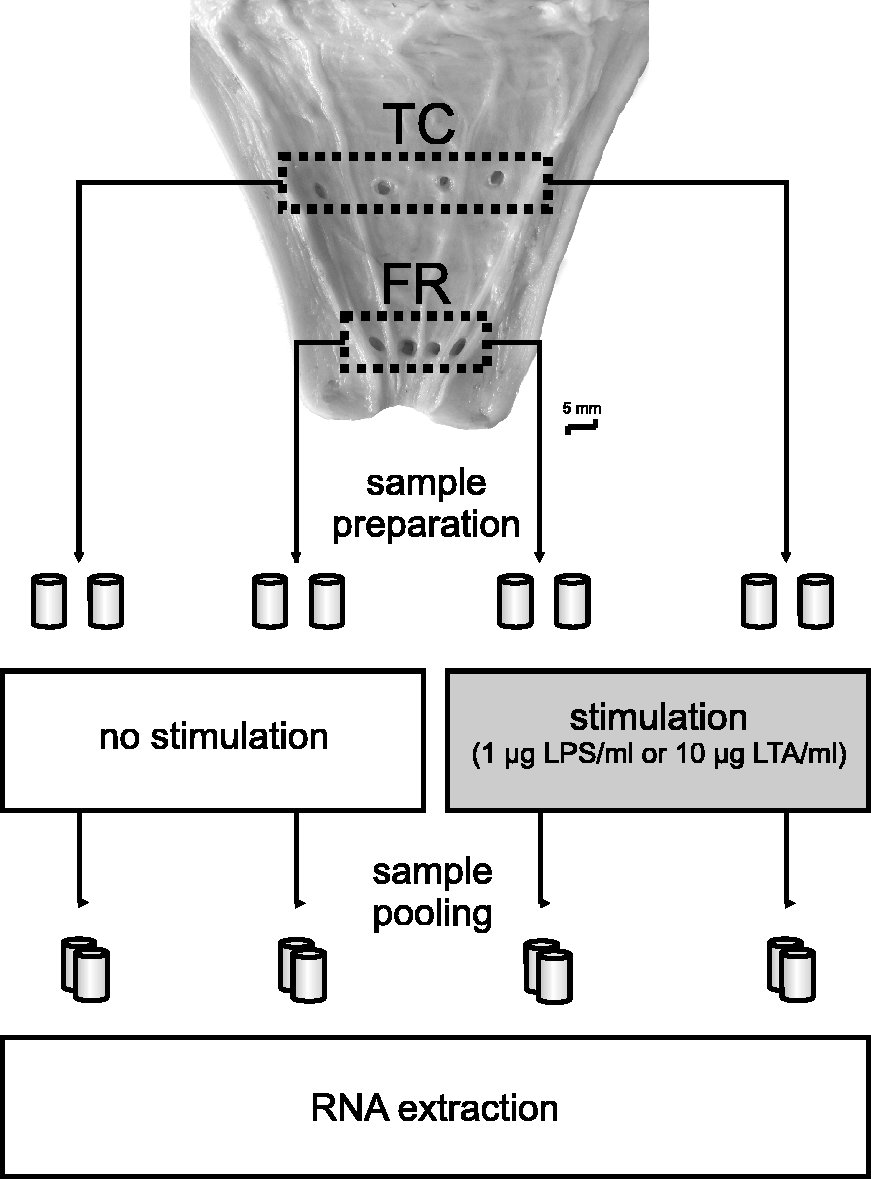
In total, 336 explants from 11 cows (eight Brown Swiss, three Holstein) were used for RT-qPCR; 304 explants from 10 cows (nine Brown Swiss, one Fleckvieh) were used to generate culture supernatants for chemotaxis assays and ELISA; 64 explants (two Brown Swiss cows) were examined for cellular composition in quantitative stereological analysis. Different breeds were randomly assigned to different treatment groups.
LPS and LTA preparation
LPS from the mastitis-causing E. coli strain 1303 was prepared as described. 16 Briefly, it was grown in Luria–Bertani medium in a 10 -l fermenter (BIOFLO 110; New Brunswick Scientific, Enfield, CT, USA). LPS was isolated from the dry bacterial cells utilising the hot phenol/water procedure, 17 and purified by incubation with DNase, RNase and Proteinase K, followed by ultracentrifugation. In order to obtain a highly pure preparation that is devoid of contaminating TLR2 ligands (lipopeptides/lipoproteins), LPS was re-extracted with phenol in the presence of triethylamine and deoxycholate. 18
LTA was isolated from disrupted cell pellets of S. aureus 1027 mastitis isolate (sequence type, ST13312), as described by Morath et al. 19 LTA was recovered from aqueous phase after n-butanol extraction and purified on a HiPrep column (16 × 100 mm, bed volume 20 ml) of octyl-sepharose (GE Healthcare, Little Chalfont, UK) by hydrophobic interaction chromatography (gradient of 15–60% n-propanol). Highly pure LTA was obtained after treatment with 1% H2O2 for 24 h at 37℃. 20
Explant culture
Single tissue explants were incubated in 24-well dishes (Greiner, Frickenhausen, Germany) in 1 ml DMEM-F12 (Sigma-Aldrich, Steinheim, Germany) supplemented with insulin (10 µg/ml), hydrocortisone (0.5 µg/ml), penicillin G (100 µg/ml) streptomycin (100 µg/ml) and amphotericin B (2.5 µg/ml) (all from Invitrogen, Karlsruhe, Germany), as described by Rabot et al. 21 Initially, all explants were incubated at 4℃ for 1 h. In two of four FR/TC explants, teat culture medium was replaced with medium containing 1 µg LPS/ml or 10 µg LTA/ml. The other two FR/TC explants served as unstimulated controls. For real-time RT-qPCR analysis, explants were incubated at 37℃ and 5% CO2 for 3 h. After incubation, explants were immediately placed in RNAlater (Sigma-Aldrich) and stored at 4℃ for 18 h. After removal of RNAlater, explants were stored in 1 ml Cryotubes (Roth) at −80℃.
For the generation of culture supernatants, explants were incubated at 37℃ and 5% CO2 for 18 h. Supernatants were stored at −80℃ in 1.5 ml aliquots (Greiner). For quantitative stereological analysis, explants were stored for 24 h in paraformaldehyde (4%) before embedding in paraffin (both SAV LP, Flintsbach a. Inn, Germany).
Quantitative stereological analysis of the tissue composition of explants from the TC and FR
Volume densities of muscular tissue, connective tissue, epithelium and CD11a/18-positive leukocytes in explants of the TC and the FR were estimated by quantitative stereological analysis. 22 Formalin-fixed and paraffin-embedded FR/TC explants were completely cut into consecutive sections (∼4 µm). Every 30th–32nd section was selected for subsequent quantitative stereological analyses by systematic random sampling. Every 30th section (17 ± 3 per explant) was stained with Masson–Trichrom to distinguish between musculature and connective tissue. 23 Every 62nd and 63rd section (9 ± 1 per explant) was used for immunohistochemical identification of cytokeratin-positive epithelium, or of CD11a/18-positive leukocytesrespectively.24,25 Primary Abs were directed against cytokeratin (mouse anti-human cytokeratin, clones AE1/AE3; Dako, Carpinteria, CA, USA) and CD11a/18 (mouse anti-CD11a/18, clone BAT75A; VMRD, Pullman, WA, USA). HRP-coupled rabbit antimouse Ig (Dako, Glostrup, Denmark) served as secondary Ab. Diaminobenzidine was used as chromogen and Mayer’s hemalum as nuclear counterstain, following standard protocols (described in detail in the Supplementary Material). The volume densities of muscular tissue and of connective tissue in the explants were calculated as the quotient of the cumulative areas of the muscular, or connective tissue, and the total section areas of the teat explants determined in Masson–Trichrom stained sections by point counting (1766 ± 732 and 1,931,369 ± 615,851 points per explant respectively).22,26 Analogously, the volume density of the epithelium in the explants was assessed by automated measurement of total tissue and cytokeratin-positive area (VIS-Visiopharm Integrator System v3.4.1.0, newCAST software; Visiopharm A/S, Hørsholm, Denmark). A detailed description of the quantitative stereological analyses is provided in the Supplementary material).
Total RNA extraction and reverse transcription
Total RNA was extracted using the RNeasy Mini Kit (Qiagen, Hilden, Germany), according to the manufacturer’s instructions. Briefly, tissue was homogenised in 1.5-ml tubes using micropestles (Eppendorf, Hamburg, Germany) and lyses buffer containing β-mercaptoethanol (Sigma-Aldrich). Subsequently, total RNA was bound to a silica-membrane spin column system and purified, including the elimination of genomic DNA with the RNase-Free DNase Set (Qiagen). The quality of total RNA was assessed in the automated electrophoresis Experion system, using Experion RNA StdSens Chips (both Bio-Rad Laboratories, Munich, Germany). Measured RNA quality indicators (RQI) were between 6.5 and 10 (mean = 8.7). There was no correlation between RQI and quantification cycle (Cq) values. Reverse transcription of mRNA into cDNA was carried out using oligo-(dt)12–18 primers and Superscript II reverse transcriptase (both Invitrogen) according to the manufacturer’s recommendations. To quantify the amount of cDNA, the optical density at 260 nm was determined with a photometer (Eppendorf). DNA integrity was verified by the OD260/280 nm absorption ratio between 1.7 and 2.1, and the cDNA concentration was calculated and adjusted to 200 ng/µl.
Quantification by RT-qPCR
Oligonucleotide primers for RT-qPCR.

Upper line: forward primer, lower line: reverse primer.
Separation of blood polymorphonuclear leukocytes for chemotaxis assays
Blood samples were taken from two healthy, lactating, multiparous Holstein cows at the vena jugularis externa using the BD Vacutainer Blood Selection Set and heparinised Vacutainer Tubes (Becton Dickinson, Heidelberg, Germany). The blood was mixed 1:1 with PBS, layered on Biocoll (Biochrom, Berlin, Germany) and centrifuged at 10℃ for 30 min at 1300 g. Blood plasma and interphase were discharged by pipetting. The remaining erythrocytes were lysed twice with distilled water. Afterwards, polymorphonuclear leukocytes (PMN) were washed in PBS and, after centrifugation at 4℃ for 8 min at 220 g, they were resuspended in DMEM-F12. PMN were counted with a Sysmex pocH-100iV Diff (Sysmex, Kobe, Japan) and diluted with DMEM-F12 to a final concentration of 5 × 106 PMN/ml.
Chemotaxis assays
The in vitro chemotaxis assays were performed in 10-well transmigration chambers using a 3 -µm pore membrane (both NeuroProbe, Gaithersburg, MD, USA). Lower chamber wells were filled with 300 µl explant culture supernatants that had been gained 18 h after incubating explants in the presence or absence of 1 µg/ml LPS or 10 µg/ml LTA respectively. Cell culture medium (DMEM-F12) served as a negative control and recombinant human IL-8 (rhIL-8; CellConcept, Umkirch, Germany) in the concentration of 100 ng/ml in DMEM-F12 served as a positive control within the chemotaxis assay. Approximately 130 µl Percoll was layered at the bottom of the lower well to prevent the adhesion of migrated PMN to the walls of the well. Upper chamber wells were filled with 200 µl PMN suspension (5 × 106cells/ml), which had been generated from blood PMN as described above. Then transmigration chambers were incubated for 2 h at 37℃ and 5% CO2. After incubation, cell suspensions were completely retrieved from the lower and upper wells. Cell suspensions from the lower wells were immediately transferred to precooled (4℃) TruCount® tubes (Becton Dickinson), and quantified by flow cytometry as described in the next section.
Flow cytometric determination of migrated PMN
The numbers of migrated PMN were determined by flow cytometric acquisition (FACScan; Becton Dickinson). PMN were morphologically identified in forward versus side scatter dot plots (for detailed gate settings see the Supplementary Material, Figure G). Acquisition was stopped automatically when 6000 TruCount® beads were identified in green fluorescence (FL-1) versus orange fluorescence (FL-2). Data analysis was performed with the FCS Express software (Version V3; De Novo Software, Los Angeles, CA, USA). The number of migrated PMN was determined by relating the number of acquired PMN events to the total number of TruCount® beads in the sample. To avoid day- and blood donor-dependent variability, the relative PMN migration rate (PMN %) was calculated by setting the number of migrated PMN in the positive controls (100 ng/ml rhIL-8) equal to 100%. The mean absolute migration rate (related to input of cells in the upper chamber well) of all positive controls was 74% (coefficient of variation = 14%).
ELISA
The response of tissue explants to LPS and LTA was quantified at the protein level for TNF-α, CXCL3 and CXCL8 as described.29,30 Standard curves were obtained by diluting recombinant bovine TNF-α, CXCL3 or CXCL8 in PBS supplemented with 5 mg/ml gelatin. The lower limits of detection were 40, 300 and 20 pg/ml for TNF-α, CXCL3 and CXCL8 respectively.
Statistical analysis
Statistical significances of differences in mRNA expression (log fold expression), as well as differences in relative rate of PMN migration and in CXCL8 and CXCL3 concentrations between FR and TC, and between control and LPS/LTA stimulation were tested by a mixed model (ProcMixed in order to correct for repeated measures within each quarter), performed with SAS (version 9.2; SAS Institute, Cary, NC, USA). Differences of the volume densities of the tissues investigated in quantitative stereological analysis were also tested for significance by a mixed model. Before analysing, the logarithms of the values were taken to obtain normal distribution. The values of the unstimulated trials were tested for outliers. Values in the unstimulated trials that were outside a range of 3 × interquartile range (IQR) from the first and third quartile, respectively, were judged as outliers and the values (stimulated and unstimulated) of these quarters were excluded from analysis. P-Values < 0.05 were considered significant.
Results
FR and TC explants differ in tissue composition
TC explants displayed significantly higher volume densities of muscular tissue (TC: 40% ± 9%; FR: 25% ± 6%; P < 0.01), whereas FR explants displayed higher volume densities of connective tissue (FR: 56 ± 5%; TC: 42% ± 7%; P < 0.01). FR explants also had significantly higher volume densities of epithelium (FR: 7% ± 4%; TC: 4% ± 3%; P < 0.01) and of CD11a/18-positive cells (FR: 0.03% ± 0.02%; TC: 0.007% ± 0.006%; P < 0.01; Supplementary Figure F).
Compared with LTA, LPS elicits a higher expression of inflammatory and chemotactic mediators in teat explants
After explant stimulation with 1 µg/ml LPS TNF-α mRNA, copy numbers were increased 5.8-fold in the FR (P < 0.01) and 10.6-fold in the TC (P < 0.01). There was no significant induction of TNF-α in LTA-stimulated explants (Figure 2).
Fold expression of TNF-α, CCL20, CXCL8 and CCL5 mRNA measured by RT-qPCR in explants of FR (white) and explants of the TC (grey) after stimulation for 3 h with 1 µg/ml LPS or 10 µg/ml LTA, and in control explants without stimulation [control (ctrl), median = 1]; n = 22 udder quarters from six animals. Asterisks mark statistically significant differences (P < 0.05).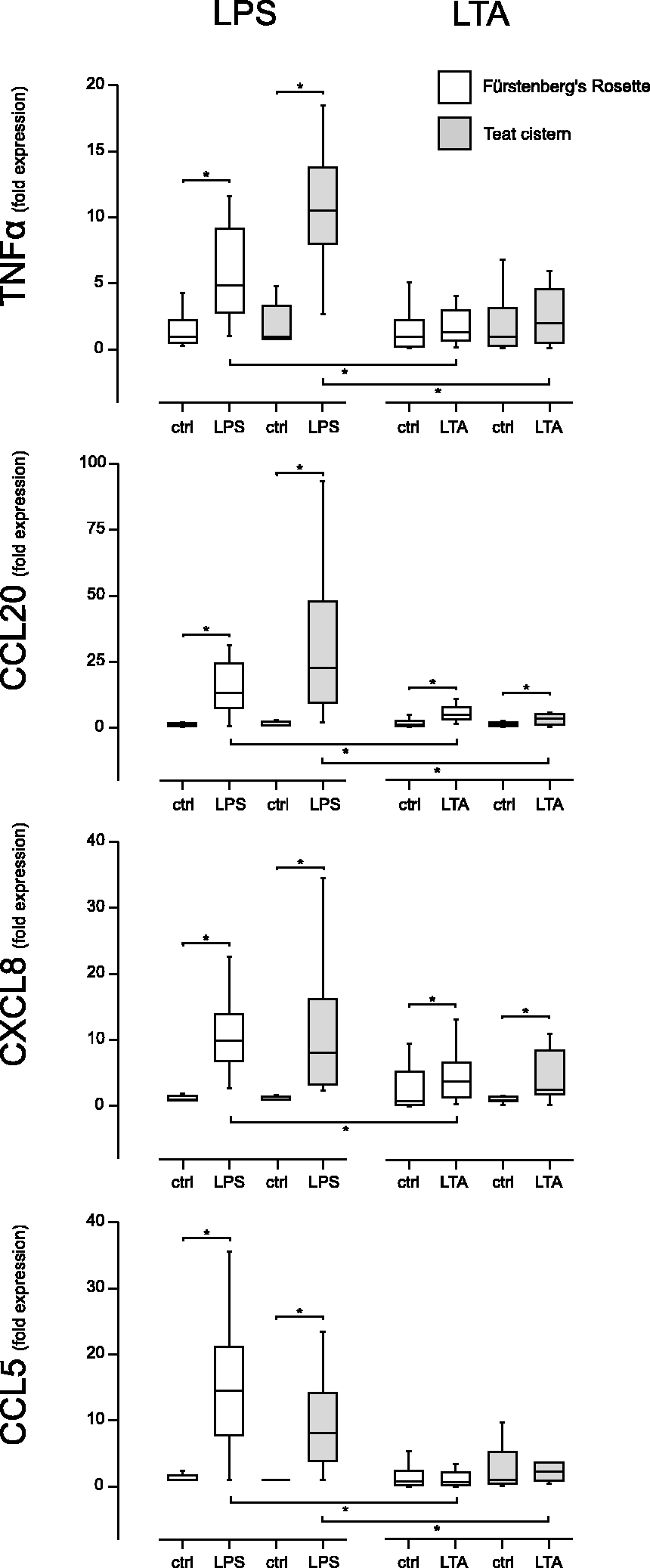
CCL20 was strongly induced after LPS stimulation (FR: 13.5-fold; P < 0.01; TC: 22.6-fold; P < 0.01). Furthermore, CCL20 was significantly upregulated in LTA-stimulated FR (5.1-fold, P < 0.01) and TC explants (3.8-fold, P = 0.01) (Figure 2).
LPS stimulation induced strong induction of CXCL8 (FR: 10.1-fold, P < 0.01; TC: 7.9-fold; P < 0.01). LTA stimulation resulted in a significant but weaker upregulation of CXCL8 mRNA expression (FR: 3.9-fold; P = 0.03; TC: 2.7-fold; P < 0.01).
LTA stimulation failed to upregulate CCL5 mRNA expression, whereas LPS stimulation resulted in a significant up-regulation (FR: 14.7-fold; P < 0.01; TC: 8.4-fold; P < 0.01).
With the exception of CXCL8 in TC, LPS stimulation resulted in significantly higher mRNA expression levels (P < 0.05) of the analysed chemokines and TNF-α compared with LTA stimulation (Figure 2).
After LTA stimulation, TC explants showed a statistically higher TNF-α (P = 0.0022) and CCL5 (P < 0.01) mRNA abundance compared with FR explants.
CCL20 and CXCL8 mRNA expression did not differ significantly between TC and FR explants, neither in LPS- nor in LTA-stimulated set-ups.
Calgranulin mRNA is expressed constitutively higher in FR explants, but only inducible in TC explants
Fold expression (RT-qPCR) of S100 calcium-binding proteins in FR compared with the TC in untreated explants (n = 42).

In TC explants, S100A8, S100A9 and S100A12 mRNA abundance was significantly upregulated after LPS stimulation (fold expression; S100A8: 3.83, P < 0.01; S100A9: 5.82, P < 0.01, S100A12: 2.44, P < 0.01). In contrast, LTA stimulation only induced the upregulation of S100A8 mRNA expression (fold expression: 3.03, P < 0.01) (Figure 3).
Fold expression of mRNA for S100A8, S100A9 and S100A12 measured by RT-qPCR in explants of the teat cistern after stimulation for 3 h with 1 µg/ml LPS or 10 µg/ml LTA, and in control explants without stimulation [control (ctrl), median = 1]; n = 22 udder quarters from six animals. Asterisks mark statistically significant differences (P < 0.05).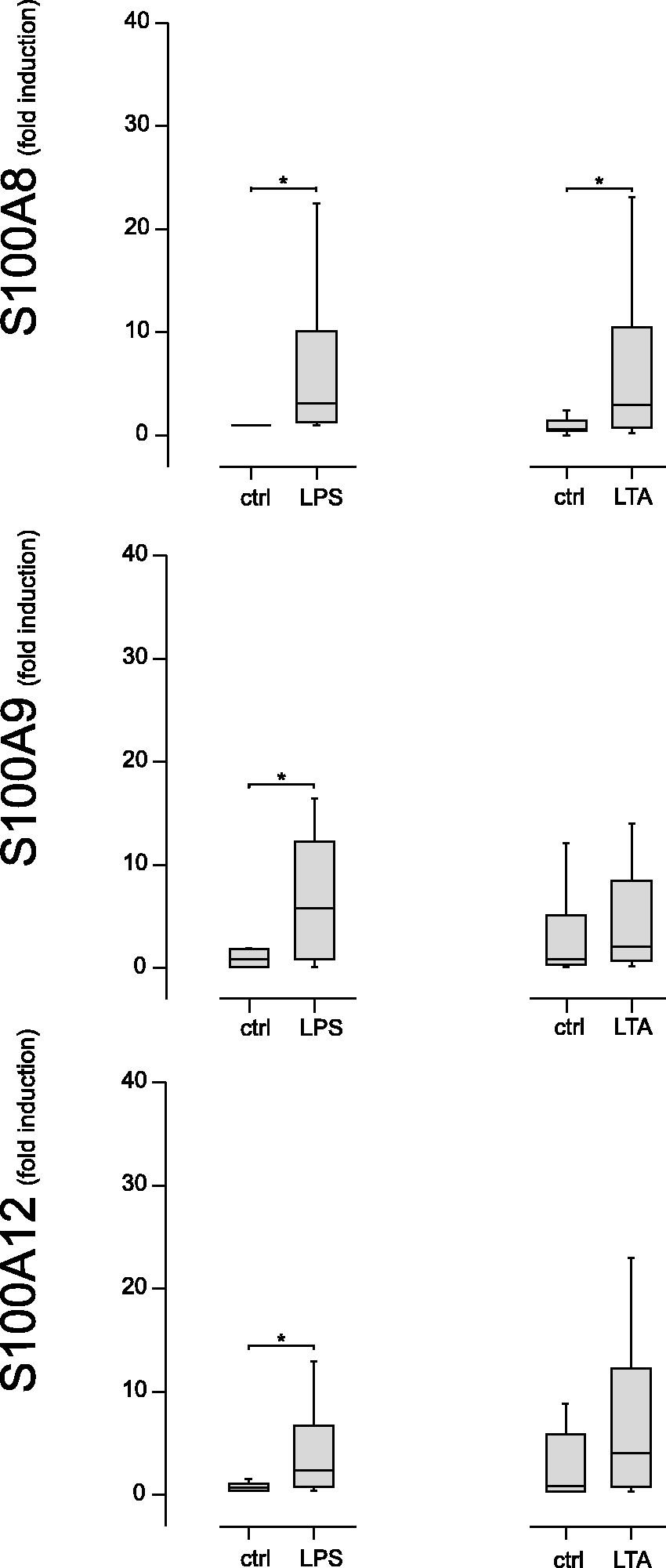
LPS- and LTA-stimulated teat explants secrete chemokines but not TNF-α
CXCL8 and CXCL3 concentration (pg/ml) in explant culture supernatants (ELISA).
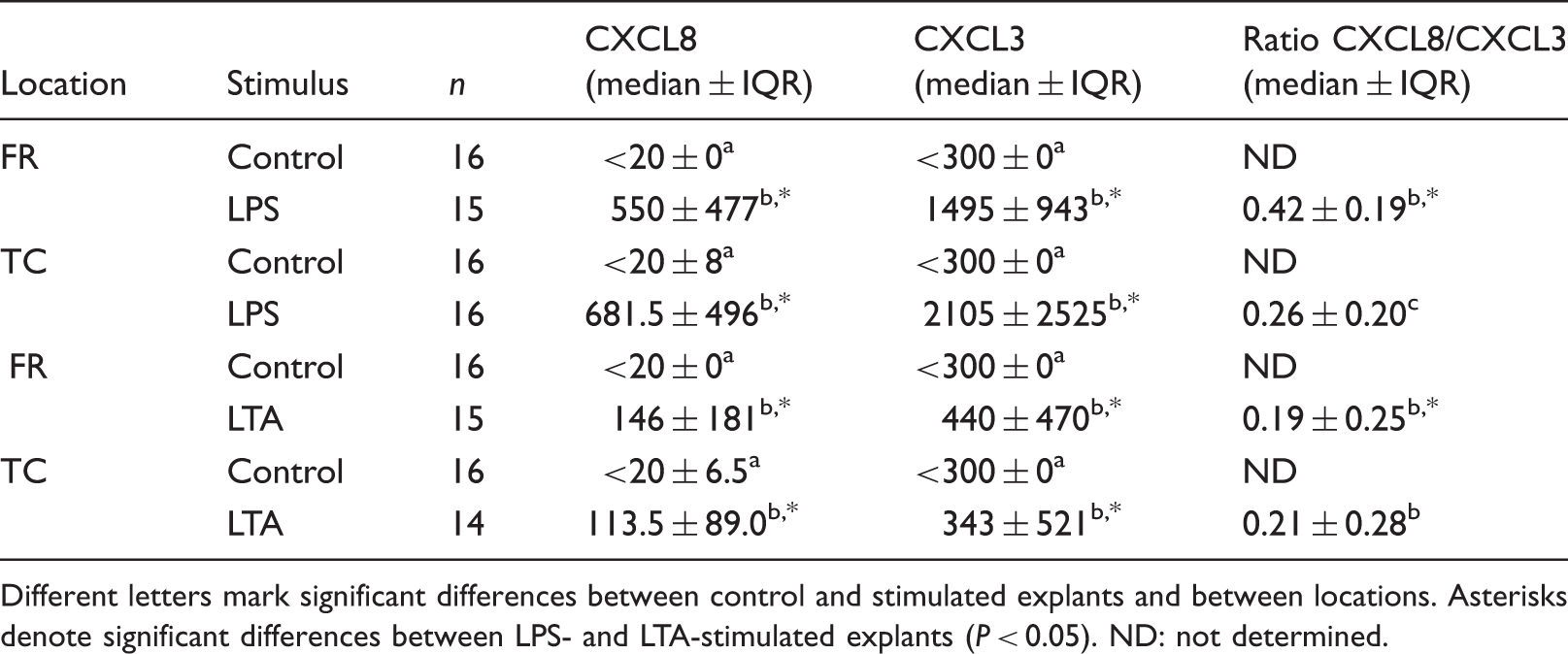
Different letters mark significant differences between control and stimulated explants and between locations. Asterisks denote significant differences between LPS- and LTA-stimulated explants (P < 0.05). ND: not determined.
Supernatants of LPS- and LTA-stimulated FR and TC explants attracted significantly higher numbers of PMN in chemotaxis assays compared with unstimulated explant supernatants (Figure 4). Supernatants of LPS-stimulated explants had a significantly (P < 0.01) stronger chemotactic effect than supernatants of LTA-stimulated explants (Figure 4). Stimulated FR and TC explant supernatants did not differ significantly in their potency to attract PMN.
PMN migration induced by culture supernatants of explants of FR (white) and the TC (grey) after stimulation for 18 h with 1 µg/ml LPS (LPS) or 10 µg/ml LTA, or without stimulation [control (ctrl)]. The rate of PMN actively migrating through the pore membrane towards 100 ng/ml rhIL-8 was set equal to 100%. The rates of PMN migrating towards explant culture supernatants were set in relation to this rate (relative rate of migration in %); n = 20 udder quarters from five animals. Asterisks mark significant differences (P < 0.01).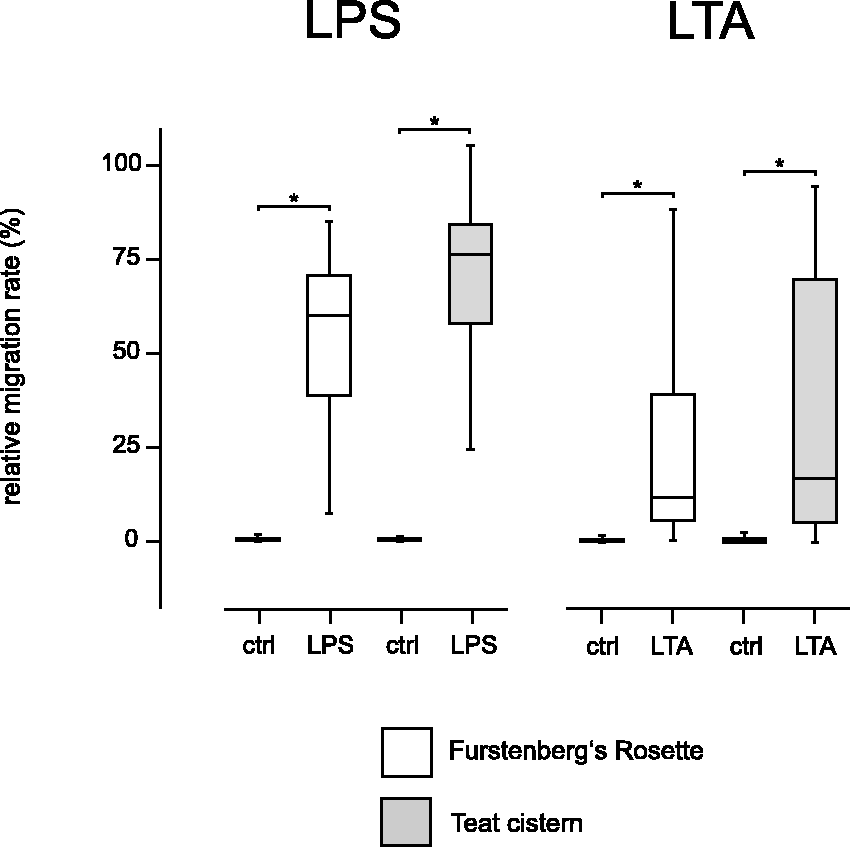
Discussion
S. aureus and E. coli are both highly relevant pathogens affecting the dairy industry; however, the clinical picture of mastitis caused by these two pathogens differs greatly. Mastitis-causing pathogens primarily invade the udder through the streak canal. Although the teat, with its different locations, acts anatomically as an important barrier against the cow’s environment, knowledge of early host–pathogen interactions during bovine mastitis is mainly limited to cells from lobulo-alveolar tissue and milk. After invading the mammary gland through the teat, both pathogens are recognised by the host’s innate immune system. However, E. coli elicits a much stronger inflammatory response than S. aureus.12,31 It is not known whether this distinction in the immune response is already decided at the site of the pathogen entry in the teat.
Animal models and in vitro culture of MECs have frequently been used for analysing host–pathogen interactions in the bovine udder. However, there have been few studies carried out with bovine mammary explants and these have been limited to lobulo-alveolar parenchyma.21,32 The objective of this study was to design a novel explant culture system for the bovine teat and to examine whether the different teat locations FR and TC respond quantitatively and qualitatively differently towards PAMPs of S. aureus and E. coli.
Quantitative stereological analysis could demonstrate comparable tissue compositions of explants within one location and differences between FR and TC with regard to muscular tissue, connective tissue, epithelium and CD11a/18-positive leukocytes. The higher percentage of CD11a/18-positive leukocytes in FR explants is in line with previous observations showing increased leukocyte infiltrates in the location of the FR compared with TC. 10 The higher amount of epithelium, including keratinocytes in FR explants, is most likely due to a denser and more complex folding of the mucosal surface compared with TC. Thus, the locations of the FR and the TC have to be examined separately with respect to their function in responding to pathogen threats.
We chose LPS and LTA isolated from mastitis-causing strains of E. coli and S. aureus. Both strains have been repeatedly applied to in vivo and in vitro models, showing that E. coli strain 1303 causes a clear-cut inflammatory response and acute clinical mastitis, whereas S. aureus 1027 causes a mild cellular response and subclinical mastitis.8,12,33 Similar observations are reported for their predominant PAMPs: intramammary infusion of LPS of E. coli strain 1303 induces a stronger inflammatory response than LTA of S. aureus 1027. 34 In the current study, we could also confirm these observations in the bovine teat. Stimulation with LPS significantly increased mRNA expression of TNF-α and the analysed chemokines CCL20, CCL5 and CXCL8, whereas LTA stimulation only led to an upregulation of CCL20 and CXCL8 (Figure 2). These results indicate that FR and TC are both capable of responding differently to Gram-positive and Gram-negative threats after pathogen entry.
The weaker and differential host response towards LTA, which has been reported for lobulo-alveaolar tissue and MEC,32,34,35 was also present in FR and TC. Absent or delayed activation of TNF-α and CCL5 after challenge with S. aureus or LTA has been previously reported for mammary tissue and MEC, whereas CXCL8 and CCL20 are invariantly upregulated.8,34 Whether this PAMP-specific response contributes to the predominant mild clinical picture of mastitis and the development of persistent S. aureus infection remains speculative. It would appear that the selective host inflammatory response to S. aureus could be induced after bacterial entry of the teat.
The response of the explants was not limited to mRNA expression. Secretion of functionally active proteins was validated by ELISA and chemotaxis assay. Both LPS and LTA stimulation induced the secretion of CXCL8 and CXCL3 into culture supernatants (Table 3). The stronger induction of CXCL8 mRNA expression (Figure 2) after LPS stimulation was reflected in significantly higher concentrations of the protein compared with LTA stimulation (Table 3), as well as the chemotactic capacity of culture supernatants (Figure 4).
Besides the clear-cut difference of mRNA induction in LPS- and LTA- stimulated teat explants, TNF-α was not detectable in culture supernatants 18 h after stimulation with LPS and LTA (data not shown). Whether this is due to limitations in sensitivity of the ELISA or whether produced TNF-α has been bound to receptors on explant cells has not been further investigated. Expression of TNF-α by mammary epithelial cells at the mRNA level but not at the protein level in response to LTA has previously been reported, 36 and the present study extends this observation to mammary epithelial and subepithelial tissue. These observations suggest that, in isolation, PAMPs such as LPS and LTA are not sufficient to overcome the regulation of the post-transcriptional regulation of TNF-α secretion in the mammary epithelium.
The mRNA expression of TNF-α, CCL20, CCL5 and CXCL8 (Figure 2), secreted CXCL3 and CXCL8 (Table 3) and the chemotactic activity of supernatants (Figure 4) showed no location-specific difference, which shows that the observed differences in cell composition between TC and FR had no influence on the expression of these inflammation-related genes. Only the CXCL8/CXCL3 ratio was significantly higher in LPS-stimulated TC explant supernatants compared with FR (Table 3), demonstrating subtle location-specific responses.
The observed higher volume densities of epithelium including keratinocytes and CD11a/18-positive leukocytes in the FR most likely accounts for the significantly higher constitutive mRNA abundance of S100A8, S100A9 and S100A12 compared with explants of the TC (Table 2), as these cell types are a major source of S100 proteins. 37 However, the specific cell types responsible for the high constitutive expression of analysed S100 proteins still remain unidentified. We assume, that the high constitutive expression of S100 proteins in the location of the FR can be regarded as a natural defence mechanism against invading microorganisms into the teat. This is supported by a previous report on the expression of S100A7 (psoriasin) on the bovine teat skin, known to be an active antimicrobial peptide against E. coli. 38 The mRNA expression of S100A8, S100A9 and S100A12 was only inducible in TC explants (Figure 3). This location-specific difference concerning inducibility has been previously reported for S100A12. 9 However, the vast differences of the constitutive mRNA expression have been unknown up to now but may also be explained by the observed location dependent tissue composition of FR and TC.
LPS stimulation of TC explants induced increased mRNA abundance for all three examined calgranulins, whereas LTA stimulation led only to a significant up-regulation of S100A8 (Figure 3). Transcriptomic profiling of mammary tissues during E. coli IMI repeatedly showed a strong regulation of distinct S100 proteins in the early stage of inflammation.9,33,38 Little is yet known about S. aureus and its associated LTA, but a recent study shows that S100A8 and S100A12 are marginally up-regulated in neighbouring quarters during experimental S. aureus challenge. 15 Overall, this suggests that upregulation of S100A8, S100A9 and S100A12 in the teat depends on the extent of the inflammatory response.
The presented teat explant model has the advantage that cells remain in their original location-specific composition and resemble more closely the in vivo situation. We could show that pathogen- and location-specific responses are already reflected in tissues located at the site of entry into the udder. The current study underpins previous observations concerning host response to PAMPs of mastitis-causing E. coli and S. aureus.
Footnotes
Funding
This work was supported by the German Research Foundation (DFG FOR 585).
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Acknowledgements
Our sincere gratitude is expressed to Mr Harald Kyri, Fa. Moksel AG, Buchloe, for his great support at the abattoir. We thank Dr Frank Weber, Dr Eva Kalivoda, Dr Katharina Zimprich, Dr Katharina Mühlbauer, Dr Anna Düvel, Dr Tanja Poth, Ms Lisa Pichl and Ms Sandra Kirsch for valuable technical assistance.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
