Abstract
The immune response of cows against the core oligosaccharide of Escherichia coli rough mutants (core types R1-R4, K-12 and J-5) was investigated after immunization with a synthetic glycoconjugate composed of deacylated LPS conjugated to hemocyanine (22 animals). Ab formation was measured by ELISA using LPS or deacylated LPS conjugated to BSA as an Ag. The glycoconjugate immunogens were used to vaccinate cows (36 animals), which were then challenged intramammarily with E. coli O 157 (K1 negative, R1 core type). Compared with control groups no protection was observed, although high titers against the R1 core type were detected in vaccinated animals. Western blots using the immune sera showed that the Ab response was directed against the core region and not against the O-antigen, which may explain the failure of the vaccine.
Introduction
Bovine mastitis is a frequently occurring infection of lactating cows, which is an important economical factor in milk industry.1–9 Staphylococcus aureus and Escherichia coli are the major pathogens causing mastitis in cows and several attempts have been made to develop vaccines against this disease whereby a vaccine using killed E. coli J-5 bacteria is currently used in North America with differing success.10–19 The J-5 strain is a rough mutant, which has a defect in LPS biosynthesis and does not contain in its LPS the O-side chain and the outer core. 20 This strain has been claimed, since the late 1960s, to induce Abs in mice, rabbits and humans that have a broad serological cross-reactivity and a broad cross-neutralizing activity against LPS of Gram-negative bacteria in general. 21 Even various mAbs have been obtained after immunization with J-5 LPS with unequivocal results in protection experiments (reviewed in Müller-Loennies, et al. 22 ).
In the 1990s, a mAb (WN1 222-5) was developed that was able to neutralize the endotoxin of all E. coli strains of all O-serotypes, and was considered useful for the treatment of human Gram-negative septicemia. 23 However, this Ab was not introduced into clinical use as a major percentage of human Gram-negative sepsis is caused by Klebsiella pneumoniae and Pseudomonas aeruginosa, in addition to E. coli.
We therefore hypothesized that the induction of Abs similar to WN1 222-5 could be the basis for the development of a new vaccine against bovine mastitis.
Materials and methods
Bacterial LPS and neoglycoconjugates
LPS from rough mutants of E. coli Rc (strain J-5), R1 (strain F470), R2 (strain F576), R3 (strain F653), R4 (strain F2513) and K-12 (strain 3110) were extracted with phenol/chloroform/light petroleum ether as described previously. 24 LPS was deacylated (LPSdeac) and conjugated to BSA as reported elsewhere; it will be abbreviated as LPSdeac-BSA. 25 For the preparation of neoglycoconjugates used for immunization, oligosaccharides were conjugated to hemocyanine (HC) following the same protocol; these conjugates will be abbreviated as LPSdeac-HC.
Immunization with LPSdeac-HC glycoconjugates
In the first experiment, 3 cows (conventional Dutch dairy cows, 10–20 months of age before first immunization) were each immunized with LPSdeac-HC containing the LPSdeac of E. coli R1, R2, R3, R4, K-12 or J-5 and alu-oil as adjuvant (alu-oil is a water-in-oil emulsion containing aluminium hydroxid). The animals received 2 ml of vaccine intramuscularly containing 250 µg of conjugate per dose and alu-oil as adjuvant on d 0, 42 and 126. Blood samples were taken at d 0, 28, 42, 56, 84, 126 and 140 by puncture of the tail vein using vacutainers; sera were stored at −20℃.
In a second experiment, 4 cows (conventional Dutch dairy cows, 10–20 months of age before first immunization) were immunized with a mixture of LPSdeac-HC containing the LPSdeac of E. coli R1, R2, R3, R4 and K-12. The animals were vaccinated intramuscularly with 3 ml of vaccine containing 150 µg of each per dose and alu-oil as adjuvant on d 0 and 42. Blood samples were taken at d 0, 28, 42 and 56 by puncture of the tail vein using vacutainers and sera were stored at −20℃.
Vaccination and challenge
In the final experiment, we immunized cows (conventional Dutch dairy cows; first parity, in lactation for 2–3 months) in groups of 12 animals with LPSdeac-HC glycoconjugate of E. coli R1 (150 µg per dose in 2 ml) or a mixture of R1, R2 and R3 (150 µg of each per dose in 2 ml) on d 0, 42 and 84 intramuscularly; both vaccines contained alu-oil as adjuvant. A control group of 12 animals was injected with the adjuvant alone. Two wk later the animals were challenged intramammarily with an E. coli wild type strain (O-157, K1 negative, R1 core type) isolated from a mastitis case. Sera were collected over a period of 119 days and tested by LPS-ELISA or by conjugate-ELISA using LPS or LPSdeac of E. coli R1, R2 or R3 as antigen.
Clinical measurements
For clinical evaluation, rectal temperatures were measured twice daily from 1 d before challenge, up to 3 d after challenge or until rectal temperatures were <39.0℃. To determine if vaccination prevented or reduced intramammary infection after challenge with E. coli, somatic cell counts of quarter foremilk and the number of CFU of E. coli in fore milk samples were determined regularly from the day before and up to 21 d after challenge.
Core typing of clinical bovine mastitis isolates
In 50 clinical isolates of E. coli mastitis strains the core type of LPS was determined. Bacterial strains were grown on solid media overnight, a loopful of bacteria was suspended in lysing buffer as used for SDS-PAGE and digested with proteinase K. Aliquots (1–5 µl) were dotted onto nitrocellulose. After drying (1 h, 37℃) and fixation (5 min, room temperature, 10% isopropyl alcohol, 10% acetic acid), the nitrocellulose was washed twice shortly (immediate removal of the washing fluid) and twice (5 min each) with distilled water. All the following steps were performed at room temperature. After blocking in blotting buffer (50 mM Tris-HCl, 0.2 M NaCl, pH 7.4) supplemented with 10% non-fat dry milk for 1 h, core-specific mAbs were added. After incubation overnight and washing (6 times, 5 min each) in blotting buffer, alkaline phosphatase-conjugated goat-anti mouse IgG (heavy and light chain specific; Dianova) was added (diluted 1:1,000 in blotting buffer supplemented with non-fat dry milk) and incubation was continued for another 2 h. After washing as before, 5-bromo-4-chloro-3-indoylphosphate and p-toluidine p-nitroblue tetrazolium chloride (Bio-Rad, Munich, Germany) were added as substrates according to the manufacturer’s instructions. After 15 min the reaction was stopped by the addition of distilled water.
The following mAb were used for the development: mAb X1SW1, mAb S37-20 and mAb S31-14, being specific for the R1, R3 and K-12 core type respectively. MAb H7 41-76 recognized the R2 and K-12 core type; therefore, strains being positive with this mAb, but negative with the K-12 specific Ab S31-14, were classified as R2 core type. Those which did not react with any of the Abs were classified as R4 core type.
Serology
ELISA using LPSdeac-BSA neoglycoconjugate antigens
Neoglycoconjugates in carbonate buffer (50 mM, pH 9.2) were coated onto Maxisorb microtiter plates (96-well, U-bottom; Nunc, Wiesbaden, Germany) at 4℃ overnight. Antigen solutions were adjusted to equimolar concentrations of 50 pmol per well based on the amount of ligand present in the respective glycoconjugate. If not stated otherwise, 50 µl volumes were used. Plates were washed twice in PBS supplemented with Tween 20 (0.05%; Bio-Rad, Munich, Germany) and thimerosal (0.01%, PBS-T) and were then blocked with PBS-T supplemented with casein (2.5%, PBS-TC) for 1 h at 37℃ on a rocker platform followed by two washings. Appropriate serum dilutions in PBS-TC supplemented with 5% BSA (PBS–TCB) were added and incubated for 1 h at 37℃. After two washings, peroxidase-conjugated goat anti-bovine IgG [heavy- and light-chain specific (Dianova) diluted 1:1000 in PBS–TCB] was added and incubation was continued for 1 h at 37℃. The plates were washed three times with PBS-T. Commercial 3,3′,5,5′-tetramethylbenzidine (TMB; Uptima Interchim, Montluçon, France) substrate solution (100 µl) was added. After 30 min at 37℃, the reaction was stopped by the addition of aqueous oxalic acid (2%) and the plates were read by a microplate reader (Tecan Sunrise, Crailsheim, Germany) at 405 nm. Tests were run twice in quadruplicates with confidence values not exceeding 10%.
ELISA using LPS antigens
When LPS was used as an antigen in ELISA, microtiter polyvinyl plates (96-well, Falcon 3911; Becton Dickinson, Heidelberg, Germany) were coated with LPS (250 ng/well) diluted in PBS (pH 7.2) and were incubated overnight at 4℃. PBS and PBS-containing solutions were supplemented with thimerosal (0.01%). Further incubation steps were performed at 37℃ under gentle agitation. The coated plates were washed three times with PBS and blocked for 1 h at 37℃ with PBS supplemented with casein [2.5% (Sigma, Munich, Germany) PBS-C, 200 µl per well]. Serial serum dilutions in PBS-C were added subsequently and the mixture was incubated for 1 h at 37℃. After three washings in PBS, secondary Abs diluted in PBS-C (same source and dilution as above) were added and incubation was allowed for 1 h at 37℃. After three washings in PBS, substrate solution [freshly prepared and composed of azino-di-3-ethylbenzthiazolinsulfonic acid (1 mg) dissolved in substrate buffer (0.1 M sodium citrate, pH 4.5; 1 ml)] was added followed by the addition of hydrogen peroxide (25 µl of a 0.1% solution). After 30 min at 37℃, the reaction was stopped by the addition of aqueous oxalic acid (2%) and the plates were read by a microplate reader (Tecan Sunrise, Crailsheim, Germany) at 405 nm. Tests were run twice in quadruplicates with confidence values not exceeding 10%.
SDS-PAGE and Western blots
Proteinase K-digested whole cell lysates of E. coli O157 were applied to one large slot (12.5 cm) and separated by SDS-PAGE on a 5% stacking and 15% separating gel at a constant voltage of 150 V. The gels were transferred overnight onto polyvinylidene difluoride membranes (pore size 0.45 µm; Millipore, Schwalbach, Germany) by tank blotting (Bio-Rad, Munich, Germany). Prior to use, the membranes were wetted in methanol for 10 s, after which they were washed in distilled water for at least 5 min. Following transfer, the blots were cut into stripes (0.5 cm width) and placed in Mini-incubation trays (Bio-Rad, Munich, Germany). The following steps were performed at room temperature: after blocking in blotting buffer (50 m
Results and discussion
Immunization of cows with LPSdeac-HC
Observed seroconversion in cows after immunization with LPSdeac-HC glycoconjugate of E. coli rough mutants.

Homologous reactivities printed in italics.
Data not valid owing to background activity with BSA alone.
This shows that the LPSdeac-HC conjugate is a potent immunogen resulting in the formation of Abs which bind equally to LPS and deacylated LPS and which are thus directed against carbohydrate epitopes of LPS.
Homologous Ab titers in cow sera after immunization with LPSdeac-HC glycoconjugate of E. coli rough mutants.
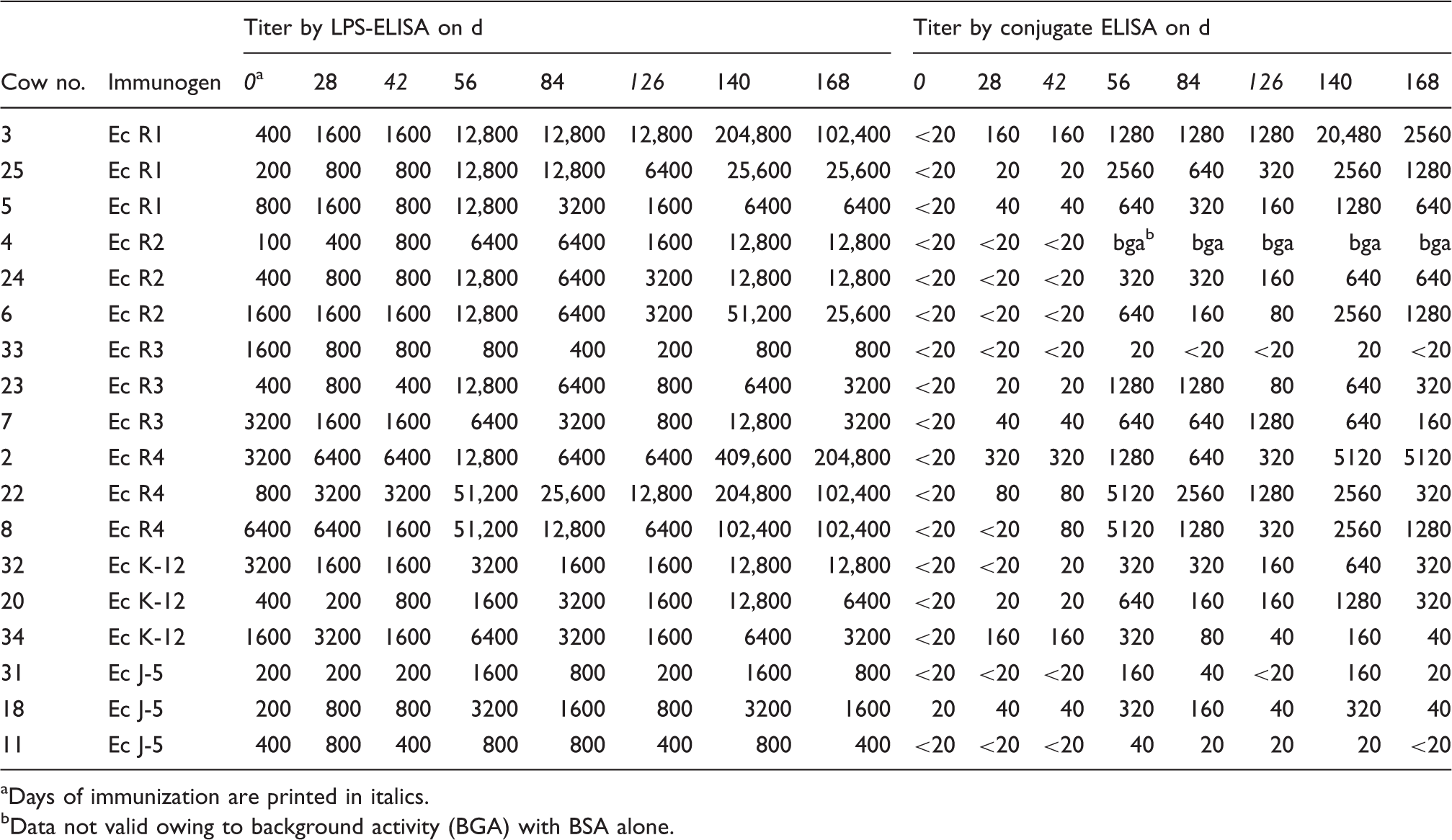
Days of immunization are printed in italics.
Data not valid owing to background activity (BGA) with BSA alone.
At first glance absolute titers in LPS-ELISA seem to be higher than in conjugate-ELISA. However, in the former assay significant titers of up to 6400 were observed already in pre-immune sera whereas no reactivity except animal number 18 was observed in conjugate-ELISA.
Heterologous Ab titers of cow sera after immunization with E. coli core oligosaccharides conjugated to hemocyanine (HC) as tested by conjugate ELISA (left) and LPS ELISA (right).
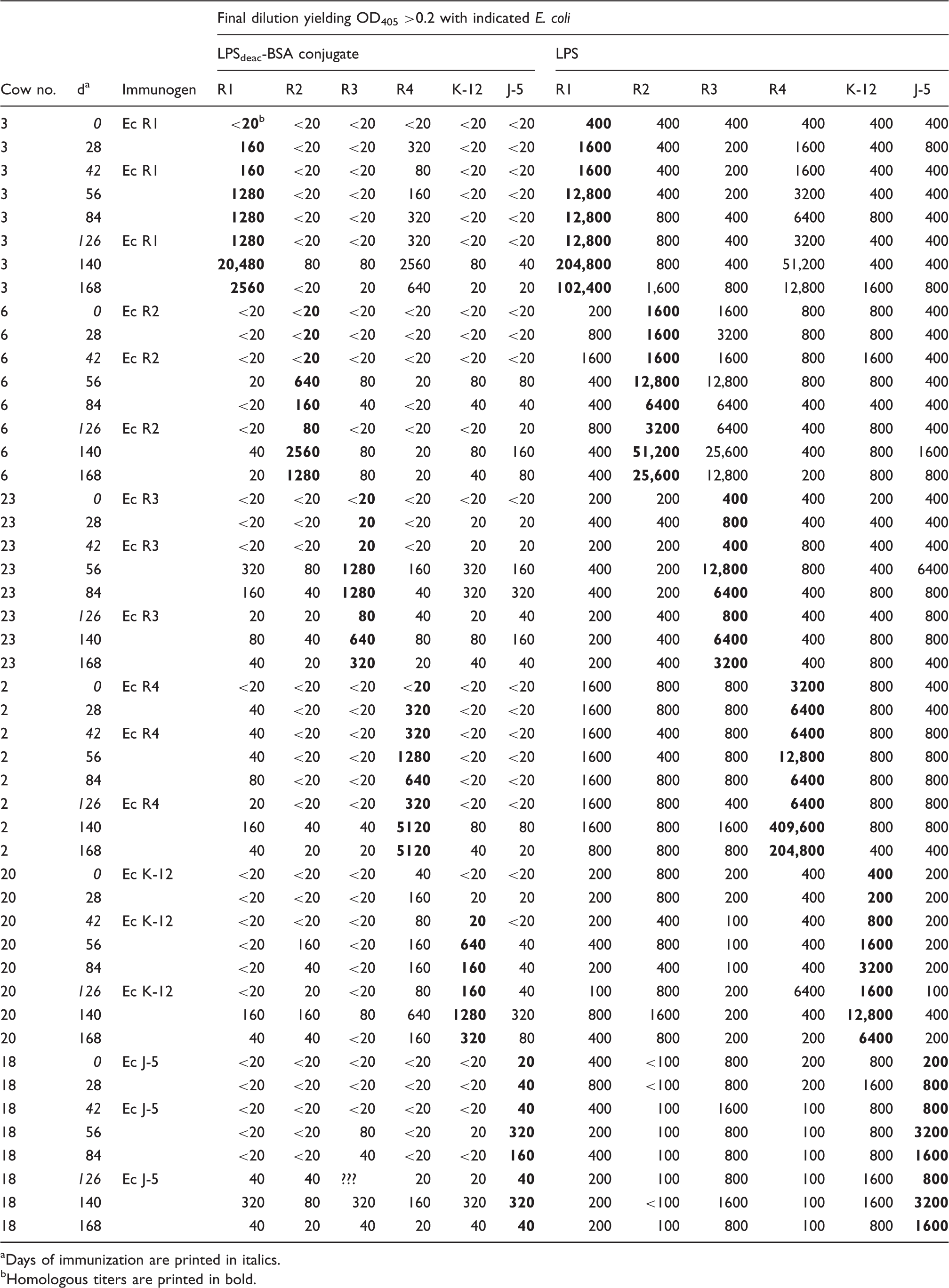
Days of immunization are printed in italics.
Homologous titers are printed in bold.
Core-typing of clinical E. coli mastitis strains
As it was planned for the next step to design a vaccination protocol in which cows should be immunized with an LPSdeac-HC glycoconjugate vaccine and be challenged with an E. coli wild type strain, we first determined which core type occurs most frequently in clinical isolates from bovine mastitis. Fifty isolates were typed using monoclonal Abs against the different E. coli core types, as described in the Materials and Methods. Twenty strains (40%) had the R1 core, 17 strains (34%) the R2 core, 11 strains (22%) the R3 core and two strains (4%) the R4 core. The K-12 core was not detected in any of the clinical isolates. These results are in accordance with those published. 26
Immune response to a mixture of glycoconjugate vaccines
Ab titers of cow sera after immunization with LPSdeac-HC glycoconjugate containing a mixture all E. coli core types.
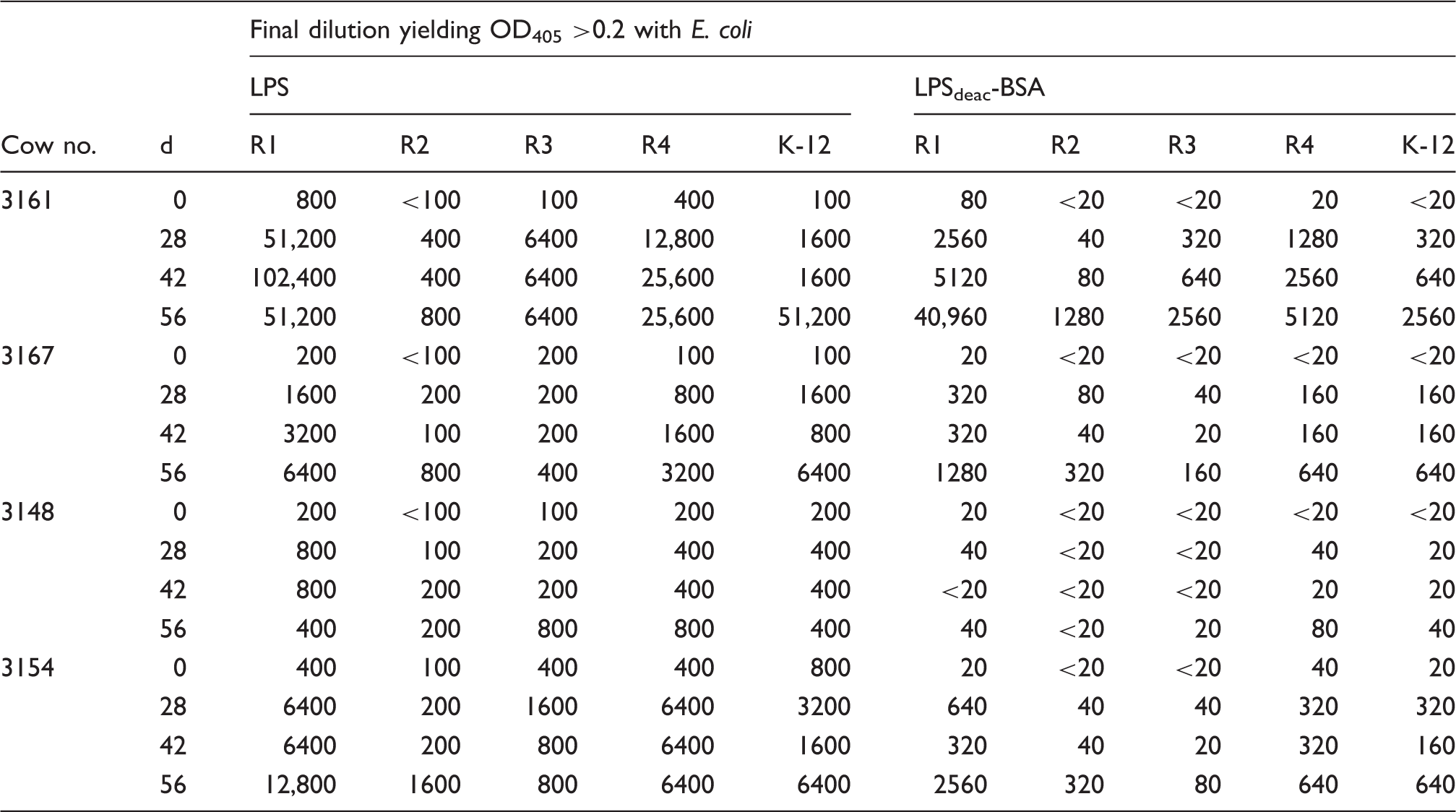
Clinical vaccination challenge study
Seroconversion of cows immunized with LPSdeac-HC glycoconjugate of E. coli R1 or a mixture of R1, R2 and R3, and challenged with an E. coli wild type strain with the R1 core type.
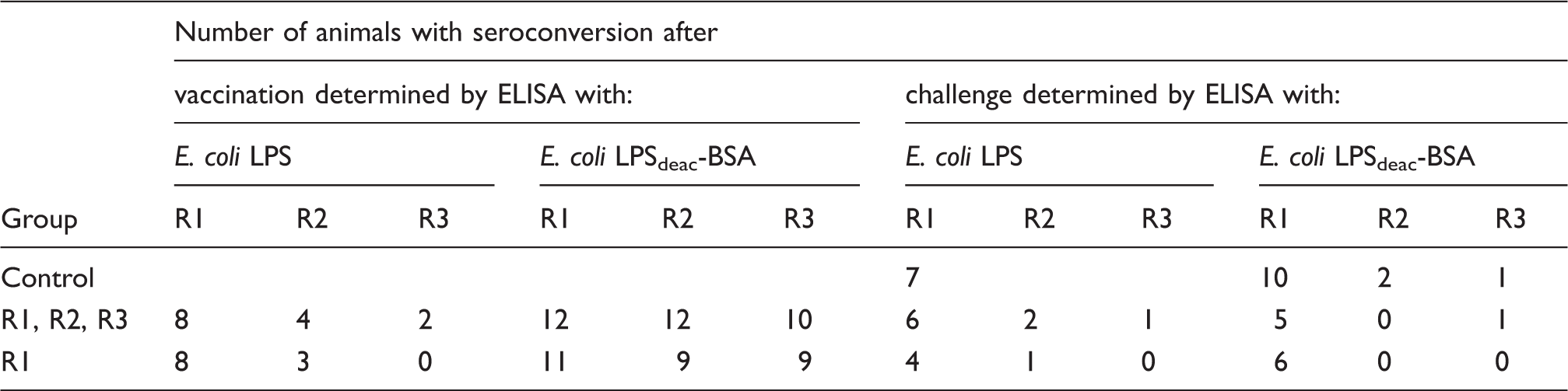
In the group immunized with the mixture of R1, R2 and R3 conjugates, 12, 12 and 10 animals showed seroconversion in conjugate-ELISA against R1, R2 and R3, respectively, and 8, 4 and 2 animals, respectively, when Abs were measured by LPS-ELISA. After challenge, a second seroconversion was observed, most often against the R1 core type (for details see Table 5). The results obtained in the R1 group were similar to those obtained for the mixture group.
Clinical evaluation showed that all cows developed clinical mastitis within 12 h of challenge, as evidenced by increased rectal temperature, increased somatic cell counts and the presence of E. coli in milk. The morning after challenge rectal temperatures peaked in all 3 groups; the average increase was 2.2 ± 0.5, 2.5 ± 0.6 and 2.6 ± 0.5℃ for the groups vaccinated with the mixture of (R1, R2 and R3), with R1 alone or with the adjuvant only respectively. Thereafter, the rectal temperatures of all groups decreased.
On the first day after challenge E. coli was isolated from the milk of all challenged quarters from the three groups. Thereafter, the number of quarters positive for E. coli decreased gradually; on day 7 E. coli was isolated from 4 out of 12, 6 out of 11 and 4 out of 12 quarters of groups immunized with the mixture (R1, R2 and R3), with R1 alone or with the adjuvant respectively. On d 14 this was 2 out of 12, 3 out of 11 and 2 out of 12 quarters for the 2 vaccinated groups and the placebo group respectively. The average colony counts of E. coli per group is shown in Figure 1A. These data show that there is no beneficial effect of vaccination on the average colony counts of E. coli in milk.
Average bacterial counts (A) (cfu/ml) and average somatic cell counts (SCC) (B) of 12 cows each vaccinated with E. coli R1 conjugate (triangles), E. coli R1, R2, R3 mixture conjugate (squares) or receiving a placebo (diamonds) and challenged 2 wk after the last immunization with E. coli O157 (R1 core type). Samples were taken during the morning (am) and in the afternoon (pm).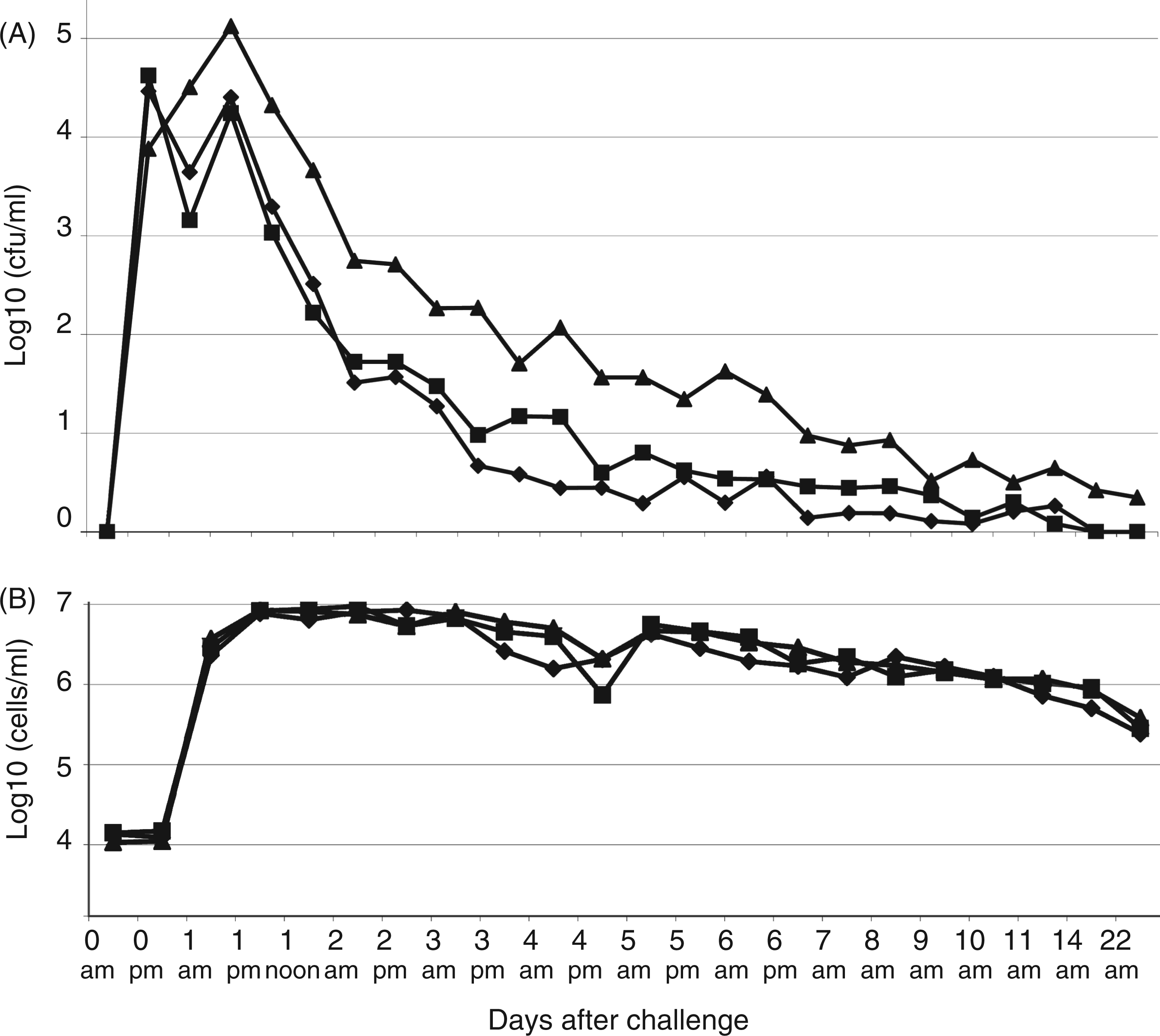
The somatic cell counts (SCC) of all challenged quarters increased after challenge. At 24 h after challenge the average SCC was 106.9 cells/ml for all 3 groups. Average somatic cell counts remained high up to d 21 (day of last observations, see Figure 1B). No beneficial effect of vaccination on the level of SCC was observed.
To understand the failure of the vaccination we analyzed sera from nine cows vaccinated with the R1 vaccine by Western blots to see whether Abs against the rough and smooth fraction of the E. coli O157 LPS were present. As seen in Figure 2, Abs against the rough fraction were detected in all cows at dilutions of up to 1:10,000 whereas no Abs against the smooth fraction were found even at the lowest dilution of 1:100. This shows that the vaccination did not lead to the formation of Abs similar to the broadly cross-reactive Ab WN1 222-5.
23
Western blots of proteinase K-digested whole cell lysates of E. coli O157 separated by SDS-PAGE on a 15% separating gel. Blots were developed with sera collected 14 wk after the first immunization at different dilutions (a, 1:100; b, 1:1,000; c, 1:10,000).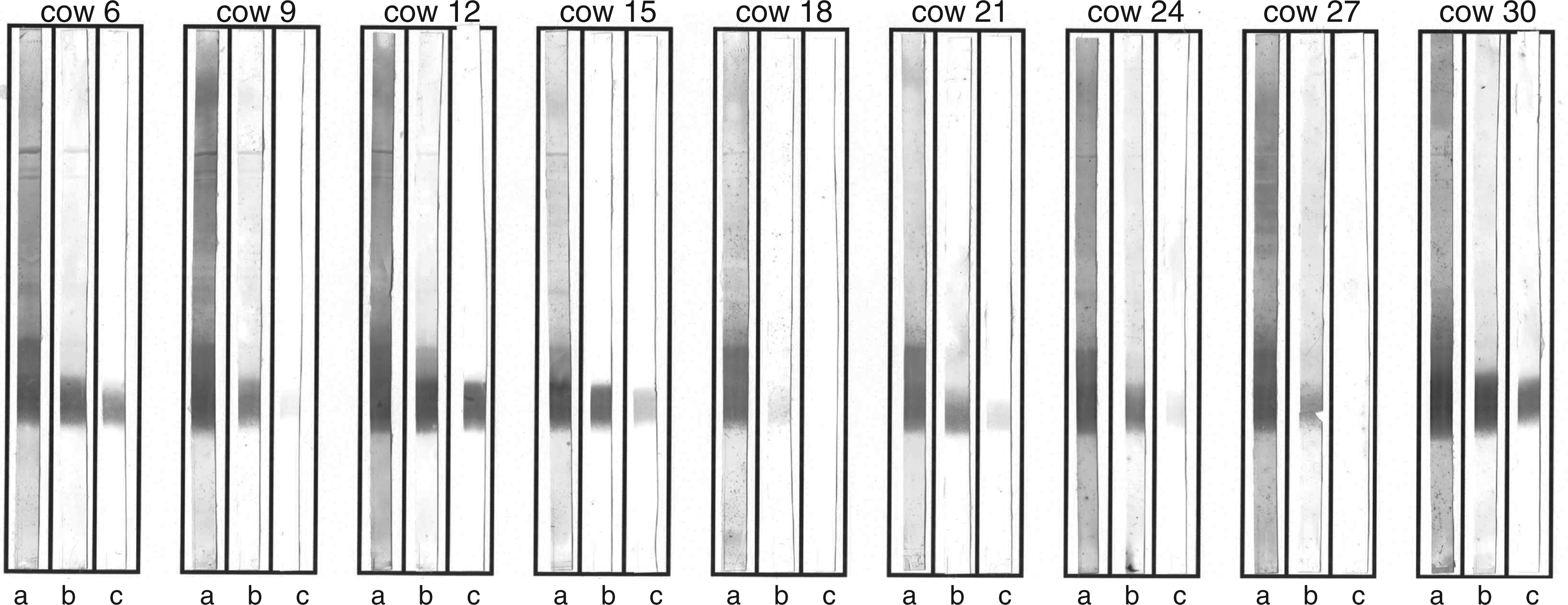
The analysis of the clinical data led to the conclusion that vaccination did not prevent, or reduce, the intramammary infection after challenge with E. coli.
Conclusions
The present study gives insight into the bovine immune response to carbohydrate epitopes in LPS of E. coli rough mutants. Cows, as many other mammalians, produce anti-LPS Abs after immunization with whole bacteria, but it has been shown earlier that the immunization with a deep rough (Re-type) bacterium of E. coli K-12 failed to protect cows from intramammary challenge with an E. coli wild type strain. 15
This study focused on the immune response against complete core oligosaccharides from the five different core types of E. coli as it has been shown that the mAb WN1 222-5 could be induced in mice which bound equally to all core types of E. coli and, at the same time, bound also to all E. coli wild type strains. 23 After immunization with glycoconjugates representing all core types the immune response was directed mainly against the homologous immunizing antigen with a low degree of cross-reactivity. Therefore, Abs with similar binding properties as WN1 222-5 could not be induced.
Nevertheless, we investigated whether the homologous Abs would protect cows against the intramammary challenge with an E. coli wild type strain with the same core type. The vaccination experiment (carried out on a total of 36 cows) showed that neither the vaccination with R1 core type alone nor with a mixture of all clinically relevant core types (R1, R2 and R3 core) failed to protect from intramammary challenge with E. coli wild type strain O157 with the R1 core, although the animals developed high titers against the R1 core.
We conclude that deacylated LPS conjugated to hemocyanine cannot be used as a vaccine to prevent bovine mastitis.
Footnotes
Acknowledgment
We thank U. Agge, S. Cohrs, I. von Cube, C. Schneider and V. Susott for technical assistance.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
