Abstract
Recent studies have demonstrated the interplay of human periodontal ligament cells (hPDLs) with immune cells, such as macrophages, during tissue repair. High-mobility group box protein-1 (HMGB1) is released into the extracellular milieu by damaged cells and functions as an alarmin to mediate the inflammatory host response. The present study addressed the role of HMGB1 released by hPDLs in the regulation of macrophage differentiation, migration and activity. The aim was to examine the inflammatory potential of HMGB1 itself and in combination with other mediators. The induction of sterile necrosis by thermal insult of hPDLs resulted in HMGB1 translocation from the nucleus to the cytoplasm and on to the extracellular space, as determined by immunocytochemistry/ELISA. Exposure of human macrophages to the conditioned PDL cell medium increased the expression of macrophage differentiation/activation markers CD14, CD23, CD64 and CD163. Chemotactic migration and osteoclastic differentiation of macrophages were also enhanced. Supplementation of the conditioned medium with a saturating concentration of HMGB1-Ab reduced these effects. Challenge with recombinant HMGB1 protein induced less migration and osteoclast differentiation than thermal insult. These data point to the immune modulatory capacity of hPDLs by the release of mediators, including HMGB1, which modify macrophage differentiation, migration and activity during periodontal repair, and indicate an enhanced HMGB1 activity when acting in concert with other mediators.
Introduction
Orthodontic tooth movement, trauma or inflammatory periodontal disease result in the degradation of soft and bony tissue and, subsequently, in a remodeling process to resolve necrotic tissues and induce a healing process of the tooth-supporting apparatus. 1 Several studies indicate that this repair process is dependent on remaining periodontal ligament (PDL) cells in the defect area, which then interact with the innate immune system. 2 PDL cells represent a mixed population that, amongst other cell types, hosts progenitor cells that bear the potential to differentiate into the multiple periodontal tissues and initiate the formation of new soft tissue and bone.3,4 Furthermore, PDL cells function as a link between periodontal tissues and the innate immune system by the expression of pro-inflammatory cytokines, which, in turn, mediate macrophage attraction from the blood to the injured site. 5 Likewise, the maturation and fusion of macrophages to multinucleated osteoclasts underlie a regulatory influence of PDL cells.
High-mobility group box protein-1 (HMGB1) is a 215-Aa chromatin-binding protein with a highly conserved sequence among species. HMGB1 has been demonstrated to have two distinct functions in cellular systems comprising an intracellular role as a regulator of gene transcription in addition to its extracellular immune modulatory capacity during sterile and bacterial inflammation. 6 HMGB1 has been suggested to function as an ‘alarmin’ outside the cell to modulate innate immune activity and mediate a wide range of physiological and pathological responses. In the course of tissue necrosis and/or inflammation, HMGB1 translocates from the nucleus to the extracellular milieu or is actively secreted by immune competent cells. In the extracellular space, it binds to the receptor for advanced glycation end-products (RAGE) and TLR2 and TLR4, 7 thus initiating tissue repair and remodeling processes. 8
Similar to the RANK/RANKL/OPG system in bony tissues, HMGB1 regulates immune processes by addressing chemotaxis, proliferation, differentiation and the expression of pro-inflammatory cytokines in target cells. In this context, HMGB1 has been reported to be chemotactic to osteoclasts and osteoblasts during enchondral ossification, as it is to monocytes and other immune and non-immune cells. 9
Little is known about the role of HMGB1 in the metabolism of periodontal tissues. In a recent study, a basal expression of HMGB1 was demonstrated in human (hPDL) cells and a HMGB1-induced stimulation of the pro-inflammatory and osteoclastogenic cytokines IL-1β, IL-6, IL-17 and RANKL. 10 In gingival crevicular fluid from patients with periodontitis, high amounts of HMGB1 were measured, whereas it was undetectable in healthy individuals. These findings point to a certain importance of HMGB1 as a marker for the presence or even progression of periodontal diseases. 11 Studies on bone marrow neutrophils suggested that the pro-inflammatory capacity of HMGB1 strongly depends on the synergistic action with other inflammatory mediators. 12 Regarding the necessity of HMGB1 for the RANKL-induced differentiation of osteoclast precursors, 13 a regulatory cross-talk of PDL cells and immune cells under inflammatory conditions with HMGB1 and RANKL representing key players in the control of periodontal bone resorption might be proposed. However, the ability of HMGB1 itself to function as a pro-inflammatory mediator in periodontal tissues and its potential to modulate the immune response to initiate periodontal remodeling remains unclear.
The present study aimed to throw further light on the influence of HMGB1 as a pro-inflammatory mediator released from hPDL cells undergoing necrosis on the activation, migration and osteoclastic differentiation of monocyte-derived macrophages to initiate periodontal tissue repair. We hypothesized that HMGB1 would exert enhanced modulatory effects on macrophage physiology thereby acting in concert with additional mediators.
Materials and methods
All experimental protocols were reviewed and approved by the ethics committee of the University of Bonn.
General experimental approach
Three different in vitro experimental approaches were carried out to examine the role of HMGB1 and other mediators released by hPDL cells on macrophage physiology. These comprised (a) the use of conditioned media from hPDL cells to culture macrophages; (b) inhibition of HMGB1 in the conditioned medium using a HMGB1-Ab in excess; and (c) supplementation of the culture medium with recombinant HMGB1 protein at a concentration found under necrotic conditions.
PDL cell explantation and cell characterization
Human PDL cells were explanted from the middle third of premolar roots without pathological periodontal findings of three different donors aged between 12 and 14 yrs. All teeth had been extracted for orthodontic reasons and with written parental consent. Following cell characterization as described previously, fifth passage PDL cells were cultured to confluence in DMEM according to standard protocols. 14
Induction of sterile cell necrosis
At confluence, cells were cultured under serum-free conditions for 8 h. To induce sterile necrosis, PDL cells were exposed to three repetitive freeze/thaw cycles at −80℃ followed by thawing at 37℃ for 30 min. 15 Thereafter, supernatants were collected and cleared of cell debris by centrifugation at 15,000 g for 5 min. The supernatant of non-thermally insulted (untreated) cells served as control.
Immunocytochemistry
To demonstrate the basal expression of HMGB1 and its translocation from the nucleus to the cytoplasm upon induction of necrosis, immunohistochemical staining of hPDL cells on cover slips was performed using a 1:200 working dilution of the primary Ab (anti-HMGB1 2600-1; Epitomics, Burlingame, CA, USA) in combination with secondary fluorescence Ab (Invitrogen, Germany) and analyzed microscopically. Specificity of the HMGB1-Ab was verified by Western blots of both hPDL cell supernatants and pure HMGB1 protein (GenWay-Biotech, San Diego, CA, USA) using a 1:10,000 working solution of the Ab (data not shown). 9 According to the manufacturer, HMGB1 protein was produced in Escherichia coli as a single, non-glycosylated, polypeptide chain containing 223 Aas and purified by proprietary chromatographic techniques.
HMGB1 ELISA
To quantify the amount of HMGB1 release by hPDL cells into the cell culture medium under non-thermally insulted (untreated) conditions compared with thermal insult-induced necrotic conditions, a commercially-available HMGB1 ELISA (Chondrex Inc., Redmond, WA, USA) was performed according to the manufacturer’s instructions.
Macrophage culture
Monocyte-derived macrophages were induced by isolation and purification of human monocytes from the blood of two healthy donors obtained from buffy coats from the blood bank followed by stimulation with RPMI1640 medium supplemented with 10% FCS and 100 ng/ml GM-CSF (Berlex Laboratories, Seattle, WA, USA) for 7 d. Macrophages were not combined, but employed in separate preparations to account for inter-individual variation.
Macrophage activation assay
Macrophage activation was determined by flow cytometry (Navious, Beckmann-Coulter, Krefeld, Germany) expression analysis of the specific maturation markers CD14, CD23, CD64, CD163 (Biolegend, Fell, Germany) 16 after 48 h of culture in the presence of either the conditioned medium (a) from hPDL cells or with 8 ng/ml recombinant HMGB1 protein (c), respectively.
Chemotactic migration assay
To address the influence of HMGB1 and other hPDL cell-derived mediators on chemotactic cell migration of macrophages, transwell assays were performed. Briefly, the lower chamber of 24-transwell-plates (Corning, Lowell, MA, USA) was filled with either macrophage culture medium (as described above) for control purposes or, alternatively, with conditioned medium (a) or with recombinant HMGB1 c) protein, respectively. Carboxyfluorescein diacetate succinimidyl ester-labeled macrophages were cultured in the upper chamber. After 24 h of incubation, the number of cells migrated to the lower chamber was determined using fluorescence imaging. 17
Osteoclast differentiation assay
To investigate the effect of HMGB1 and other hPDL cell-derived mediators on osteoclastic differentiation of macrophages, macrophages were cultured with standard macrophage differentiation medium containing 100 ng/ml M-CSF or an additional supplementation with either the conditioned medium (a) from hPDL cells or cell culture medium containing recombinant HMGB1 protein (c) all for 8 d. Subsequently, staining for tartrate-resistant acid phosphatase (TRAP) was performed. 18
HMGB1 inhibition
For inhibition of HMGB1 in the conditioned supernatants (c), they were pre-incubated with 500 ng/ml HMGB1-Ab for 16 h (concentration proven to result in a reduction of HMGB1 concentration to control level in preliminary experiments) and administered to the experiments as described above.
Statistical analysis
For any given experiment, each data point represents the mean ± SEM of six independent cultures per group (n = 6). Variance and statistical significance were analyzed using Bonferroni's modification of Student's t-test. P-Values < 0.05 were accepted to be significant.
Results
Immunofluorescence staining of hPDL cells verified the basal expression of HMGB1 protein and its translocation from the nucleus to the cytoplasm, and further on to the extracellular space upon induction of a thermal insult (Figure 1a).
(a) Immunocytochemical staining (anti-HMGB1 2600-1; Epitomics) of PDL cell cultures. Thermal insult-induced translocation of HMGB1 from the nucleus (arrows) to the cytoplasm (open arrow heads). (b) Quantification of HMGB1 released into the supernatant of cultured human PDL cells by ELISA showed a basal expression of the protein under control conditions which was ∼twofold increased following thermal insult. The bar graph shows the mean ± SEM of six independent cultures that were assayed in duplicate. *P < 0.05, experimental group versus cell-free culture medium (no cells); #P < 0.05, experimental group versus untreated cell cultures (conditioned medium).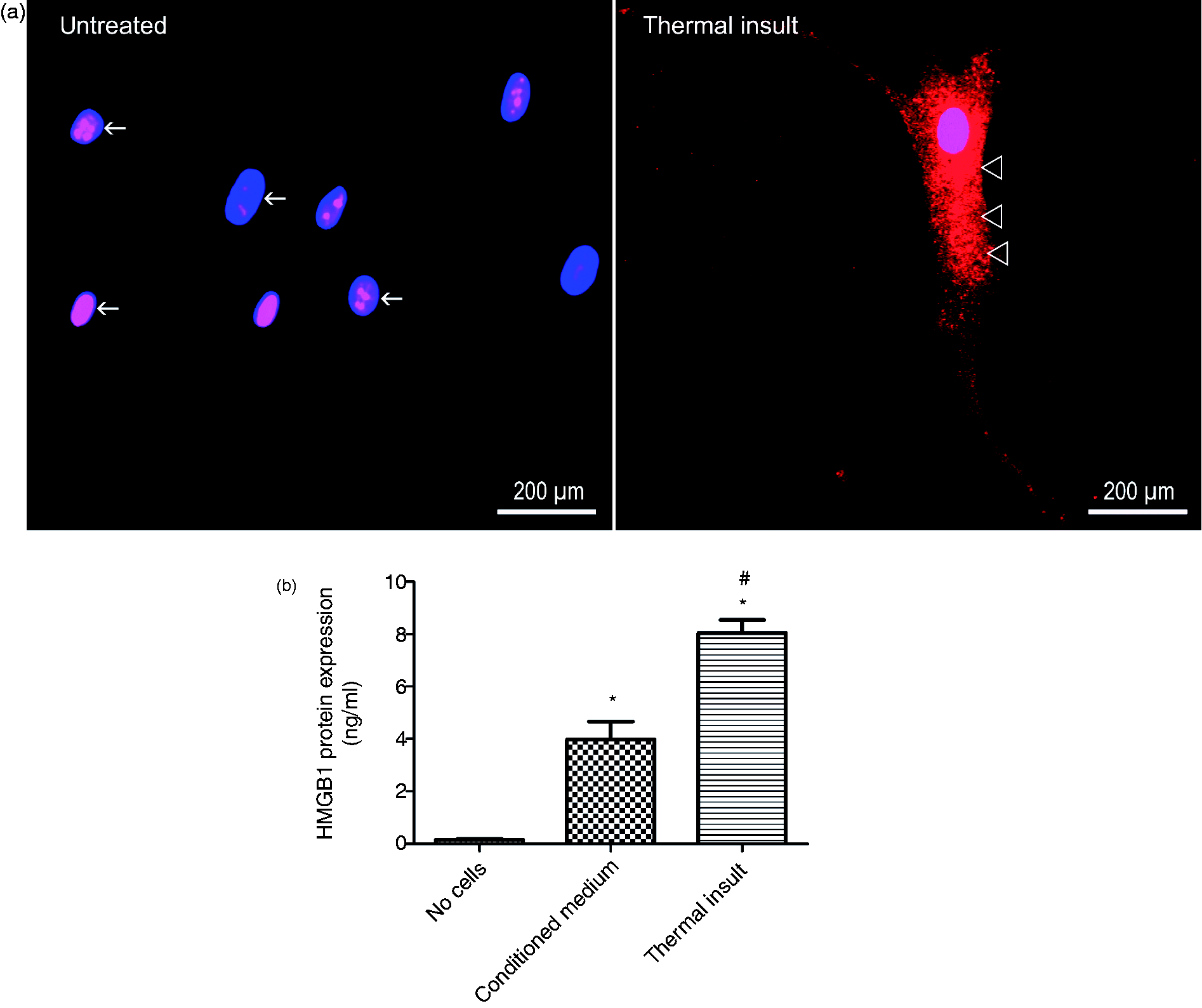
The HMGB1 content of the cell-free medium was 0.16 ± 0.03 ng/ml compared with 4.91 ± 1.07 ng/ml and 7.37 ± 0.76 ng/ml that were released by hPDL cells under untreated or thermal insult conditions, respectively (Figure 1b).
Culturing of macrophages in the presence of the conditioned medium from thermally-insulted hPDL cells resulted in a significant up-regulation of the macrophage activation markers CD14, CD23, CD64 and CD163. Blocking of HMGB1 in the conditioned media with a saturating concentration of HMGB1-Ab led to a reduction of the number of immunopositive cells to control levels. Application of recombinant HMGB1 protein had no effect on macrophage activation. Supplementation of control media with HMGB1 antibody did not have a significant effect (Figure 2).
Flow cytometry analysis of typical markers of macrophage activation (CD14, CD23, CD64, CD163) after 48 h of incubation with 8 ng/ml HMGB1 alone or with the various conditioned media obtained from the PDL cells. Macrophages were either cultured in the presence of cell culture medium from untreated (untreated) or thermally-insulted hPDL cells (thermal insult). In addition, the conditioned medium from thermal insult experiments was supplemented with a saturating concentration of 500 ng/ml HMGB1-Ab (thermal insult + HMGB1-Ab). The role of recombinant HMGB-1 was determined by the addition of 8 ng/ml HMGB1 protein to the cell culture medium (HMGB1 protein). The bar graph shows the mean ± SEM of six independent cultures that were assayed in duplicate. *P < 0.05, experimental group versus untreated control; #P < 0.05, experimental group versus conditioned medium from thermally-insulted specimens.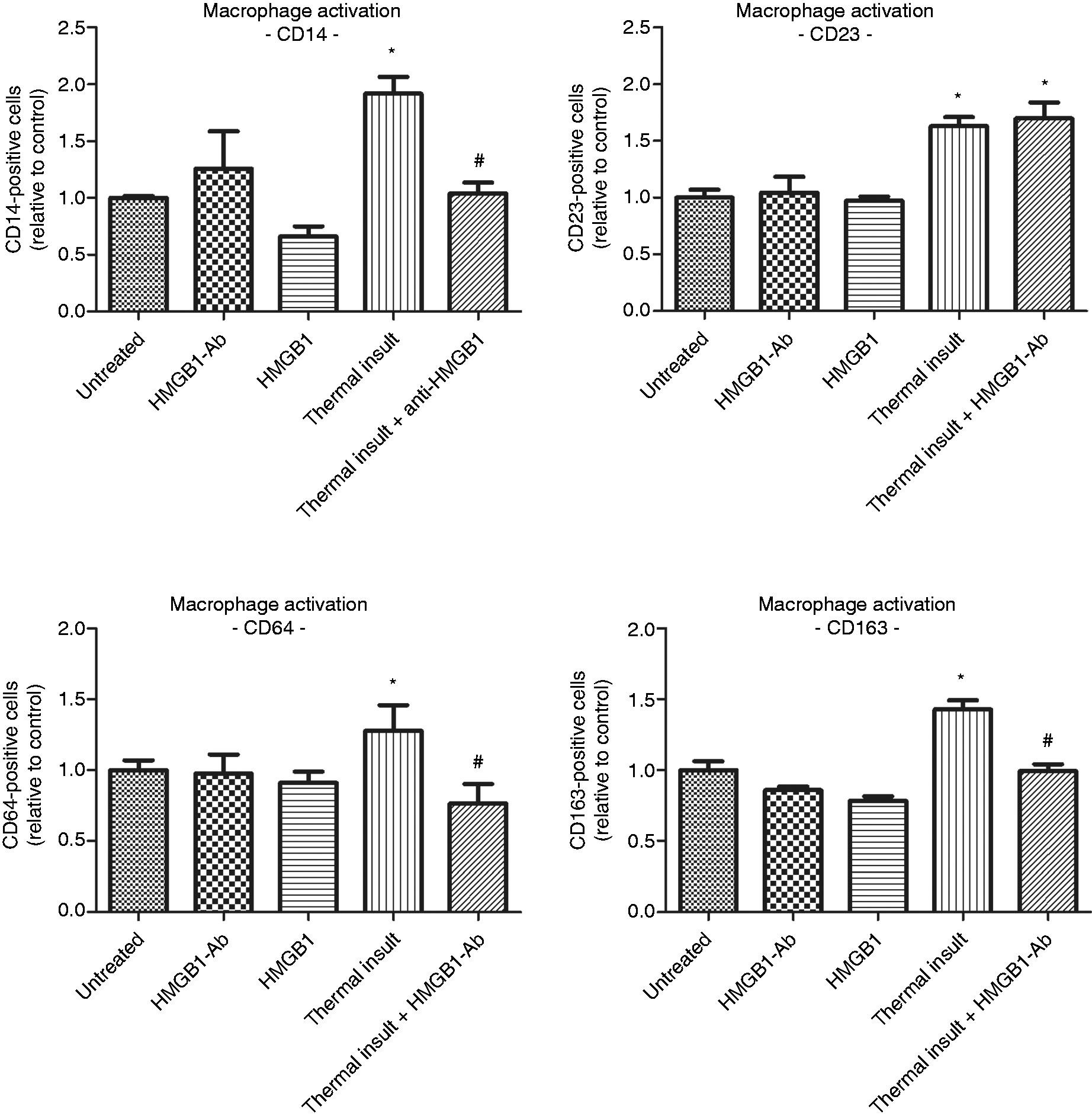
Regarding chemotaxis, in the transwell assays, there was an ∼ eightfold increase in the percentage of macrophages that migrated towards the lower chamber in the presence of the conditioned medium compared with the control. Inhibition of HMGB1 in the conditioned medium significantly reduced macrophage migration, but the number of migrating cells still remained significantly above control levels. Application of recombinant HMGB1 protein also increased migration of macrophages, although to a lower extent than the conditioned medium from the thermally-insulted PDL cells. Likewise in the macrophage activation experiments, Ab application to control media had no effect (Figures 3a, b).
Stimulatory effect of HMGB1 released by the PDL cells on macrophage migration. Transwell assays were performed to investigate chemotactic cell migration of macrophages. The lower chamber was filled with either chemotactic supernatants from untreated (untreated) or thermally-insulted hPDL cells (thermal insult). Alternatively, the conditioned medium from thermal insult experiments was supplemented with a saturating concentration of 500 ng/ml HMGB1-Ab (thermal insult + HMGB1-Ab). The addition of 8 ng/ml HMGB1 protein to the culture medium addressed the role of HMGB1 under basic conditions (HMGB1 protein). Carboxyflourescein diacetate succinimidyl ester-labeled macrophages were cultured in the upper chamber. After 24 h of incubation, migrated cells were visualized microscopically (a) and their number quantified histomorphometrically (b). The bar graph shows the mean ± SEM of six independent cultures that were assayed in duplicate. *P < 0.05, experimental group versus untreated control; #P < 0.05, experimental group versus conditioned medium from thermally-insulted specimens.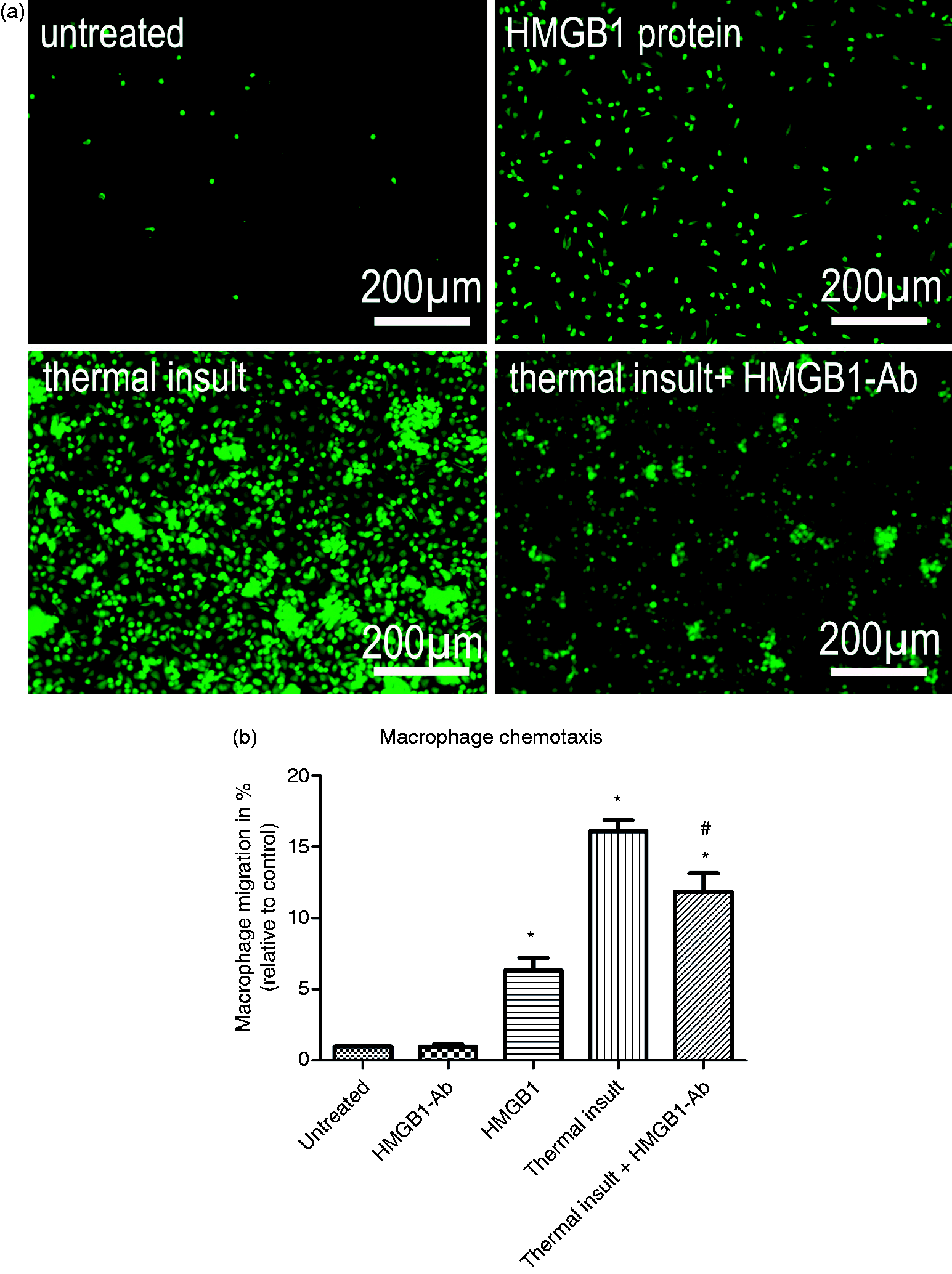
The osteoclastic differentiation properties of macrophages were also enhanced by the various media with the most pronounced effect observed for the conditioned media from thermally-insulted hPDL cells. The addition of HMGB1-Ab to the conditioned medium inhibited, but did not abolish, the increase in the number of TRAP-positive cells (Figures 4a, b). HMGB1 demonstrated to induce significantly fewer osteoclasts than the conditioned medium. Ab treatment in control experiments did not alter this target parameter.
(a) Increased osteoclastic differentiation of macrophages visualized by TRAP staining. (b) Following culture of macrophages in the presence of cell culture medium from untreated (untreated) or thermally-insulted PDL cells (thermal insult), two photographs of each of six independent cultures were taken at 20 × magnification and TRAP-positive cells were quantified using Image J software. Supplementation of the conditioned medium with excess HMGB1-Ab (500 ng/ml) reduced this effect markedly (thermal insult + HMGB1-Ab). Addition of 8 ng/ml HMGB1 protein to the PDL culture medium did not exert a significant effect on macrophage differentiation. The bar graph shows the mean ± SEM of six independent cultures that were assayed in duplicate. *P < 0.05, experimental group versus untreated control; #P < 0.05, experimental group versus conditioned medium from thermally-insulted specimens.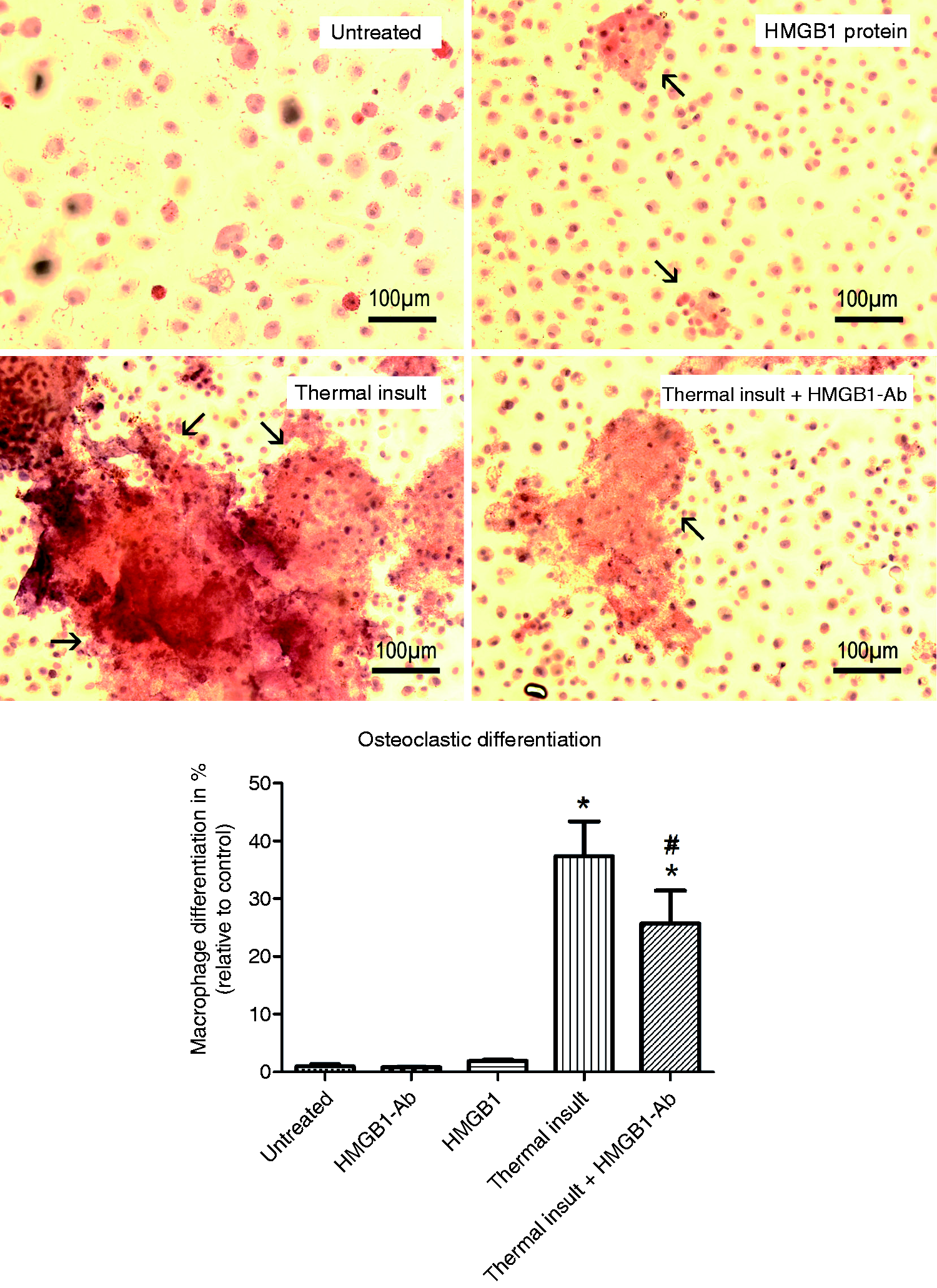
Discussion
The present study demonstrated that culturing macrophages in the presence of conditioned medium from thermally-insulted hPDL cells resulted in an enhanced expression of the macrophage differentiation/activation markers CD14, CD23, CD64 and CD163, and also stimulated chemotactic migration and osteoclastic differentiation of macrophages. Supplementation of the conditioned medium with a saturating concentration of HMGB1 Ab reduced these effects. Challenge with recombinant HMGB1 protein induced less migration and osteoclast differentiation than thermal insult.
Physiological binding of HMGB1 to chromatin depends on the viability of the cell and the release of the cytokine by necrotic cells is an important signal for neighboring cells to activate an appropriate response. 8 Cell death is usually the result of trauma or infection, each of which requires prompt reaction, damage damping and/or repair. In this study, thermal insult was established as a valid model for the induction of sterile PDL cell necrosis by the visualization of HMGB1 translocation from the nucleus to the cytoplasm, and on to the extracellular space. These findings corroborate observations from other tissues and cells establishing the role of HMGB1 as an alarmin during cell necrosis, 15 which contrasts the retention of HMGB1 in the nucleus during programmed cell death by apoptosis. 8 The quantitative data on HMGB1 release into the culture medium under our thermally-induced experimental conditions correlate well with values obtained from experiments with gingival fibroblasts where similar levels of the protein were detected following thermal heating or LPS exposure of the cells. 19
To further test directly whether the release of HMGB1 by necrotic PDL cells might be an immediate trigger for an inflammatory response, human monocyte-derived macrophages were stimulated with a low dose of HMGB1, corresponding to the concentration produced by the hPDL cells under necrotic conditions in the present experimental model. Such a treatment regimen had only a small effect on the measured parameters, indicating that the enhanced HMGB1 concentrations released incident to necrosis were of relevance to the microenvironment only in combination with other released cytokines. This conclusion is strengthened by experimental data on fibroblasts showing that the inflammatory response to IL-1 was markedly reduced when IL-1 was administered alone compared with HMGB1 plus IL-1. 20 In contrast to the present data, stimulation of macrophages with HMGB1 protein, but at a concentration 1000-fold higher than the one used in the present experiments, demonstrated the ability of recombinant HMGB1 to induce significantly enhanced osteoclastic differentiation. 21 Sha et al. 17 also demonstrated that HMGB1 induced only minimal cytokine production by macrophages and neutrophils unless bound to other pro-inflammatory mediators, upon which it acquired markedly enhanced cell-activating properties. 12 In those experiments, stimulation with HMGB1 protein purified from cytokine-treated cells induced enhanced inflammatory effects, as evidenced by increased TNF-α and MIP-2 expression. In contrast, when HMGB1 was purified without cytokine pretreatment, it was not able to modulate TNF-α expression of bone marrow neutrophils. However, it was also shown that the HMGB1 effect as an alarmin is controlled by the oxidation status of the microenvironment.22,23 Harris et al. 22 as well as Yang et al. 13 demonstrated that post-translational redox modifications of HMGB1, including certain cysteine residues, controlled the activity and receptor interaction of HMGB1.13,22 As a possible explanation for the failure of HMGB1 cytokine activity when administered alone, Harris et al. 22 discussed that commercially-available HMGB1 may possibly contain certain reducing agents. A possible explanation for the HMGB1-induced increase in macrophage migration, but not activation, might be derived from findings in the literature. According to those reports, a redox state-dependent effect of HMGB1 protein has been demonstrated with disulfide-HMGB1, which induces the affect activation of macrophages, whereas all-thiol-HMGB1 stimulates chemotaxis and migration. 24
Following stimulation with conditioned medium from necrotized PDL cells, the expression of different macrophage activation markers increased significantly, but could be antagonized by an access of HMGB1-Ab, except in the case of CD23; nevertheless, this points to an importance of HMGB1 in the activation process of host immune response. The lack of an effect of the HMGB1 antibody in case of CD23 suggests that other pro-inflammatory cytokines in the conditioned medium of hPDL cells contribute, in particular, to the stimulation of CD23 expression by macrophages. This conclusion is strengthened by our data on macrophage migration and osteoclastic differentiation where the blocking of HMGB1 by antibody administration likewise resulted only in a partial, but not complete, inhibition of the increased number of migrated or TRAP-positive cells that were induced by the conditioned medium.
HMGB1 was shown to be a prerequisite for the RANKL-induced differentiation of osteoclast precursors in vitro and in vivo, 13 and Kim et al. 10 linked HMGB1-mediated periodontal bone resorption to an altered RANKL expression in hPDL cells. Our group demonstrated that RANKL released by hPDL cells induced osteoclastic differentiation, 25 and, in a previous study, using an animal model of orthodontic tooth movement, we provided evidence for the involvement of the HMGB1/RANKL axis in the regulation of host response in the early and late phase of periodontal repair. 26 An intense HMGB1 protein expression in the compressed periodontal ligament following the application of orthodontic force was reported to follow the typical occurrence of necrotic tissue, thus indicating the HMGB1-assisted initiation of host response to the local tissue stress, as well as the onset of the early phase of repair.26,27
Together with the above findings, the data obtained in the present study support the conclusion that multiple mediators are involved in the regulation of the immune response to periodontal injury, with HMGB1 not being a single factor, but an important co-factor. In studies using rigorous purification techniques, highly purified HMGB1 did not appear to be capable of inducing cellular activation unless binding to DNA-containing immune complexes or to bacterial products.28,29 More proof of the fact that HMGB1 itself does not induce pro-inflammatory response is obtained from reports on patients recovering from severe pneumonia. In those patients, circulating levels of HMGB1 remained elevated, even though there was an improvement in organ system dysfunction and overall clinical status. 30 Again, this indicates that HMGB1 does not produce continuing inflammatory injury and strengthens the present data that HMGB1 induces fewer inflammatory effects when acting without other mediators. However, given the concern that HMGB1 could lose activity as a result of its purification process, the present data provide additional information for the understanding of the mechanism by which HMGB1 released by hPDL cells acts as a pro-inflammatory mediator.
As well as the present data, which attribute a role to HMGB1 as a pro-inflammatory cytokine enhancing macrophage activity, recent reports have also found HMGB1 to exert chemotactic effects on skin fibroblasts and keratinocytes in vitro and to enhance wound-healing in diabetic mice. 31 Likewise, in another investigation using 3T3 fibroblasts, HMGB1 stimulated wound-healing by enhancing cell proliferation and migration. 32 Thus, HMGB1 make, at the same time, a contribution as a chemoattractant and proliferative signal for PDL cells to promote the wound-healing process.
In summary, our findings add further support to the idea that PDL cells do not represent just structural components of the PDL, but actively orchestrate host immune responses by interacting with cells of the innate immune system. Furthermore, these results confirm previously published data obtained from other cell systems that HMGB1, amongst other mediators, is a co-factor in the regulation of the cell/macrophage interplay and transfer this information to the periodontal remodeling process. In the light of recent developments in arthritis therapy, which suggest HMGB1-Ab administration as a treatment option, 33 or its already-proven anti-inflammatory effects in lung disease as presented in the mouse model of experimental asthma, 33 the present data warrant further research in the fields of periodontology and orthodontics to provide a basis for the development of strategies to prevent tissue damage caused by an excessive immunological response to the typically associated local necrosis of the PDL tissues.
Footnotes
Acknowledgements
We thank Peter Wurst (Flow Cytometry Core Facility, University of Bonn) and Tina Schaffrath for expert technical assistance.
Funding
We thank the German Research Foundation (DFG) and the Medical Faculty of the University of Bonn for providing a research grant (KFO 208, TP8, LO-1181/2-2).
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
