Abstract
Objectives
Strontium (Sr2+) is a crucial trace element in humans, mainly present in the bones. We investigated the effects of Sr2+ on human periodontal ligament stem cell (hPDLSC) proliferation and osteogenesis and the relevant pathways.
Methods
hPDLSCs were harvested from extracted premolars and characterized by flow cytometry, then cultured and treated with various Sr2+ concentrations. Cell-counting kit-8 (CCK-8) assays were used to assess hPDLSC proliferation, with alkaline phosphatase (ALP) staining, Alizarin red S staining, and ALP activity assays used to analyze their osteogenic capacity. Quantitative reverse transcription polymerase chain reaction and western blots were used to examine the expression levels of relevant factors, such as collagen I (COL-1), ALP, and Runx family transcription factor 2 (RUNX2). Moreover, tankyrase inhibitor XAV939 treatment was used to investigate the role of Sr2+ in the canonical Wnt/β-catenin signaling pathway.
Results
The hPDLSCs were successfully isolated and cultured in vitro. A 0.01 mM Sr2+ concentration significantly enhanced hPDLSC proliferation and osteogenic differentiation. However, XAV939-mediated inhibition of the canonical Wnt/β-catenin pathway could reverse the Sr2+-induced osteogenic effects.
Conclusions
Sr2+ can enhance hPDLSC proliferation and osteogenesis by stimulating canonical Wnt/β-catenin signaling, suggesting it may play a critical role in periodontal regeneration and has clinical application potential.
Keywords
Introduction
Periodontal health is not only fundamental to oral hygiene, but also integral to general health and well-being. Recent trends have indicated an escalating prevalence of periodontitis, with research suggesting that over 50% of the population suffers from periodontitis and 11% of adults experience severe forms of the condition. Consequently, severe periodontitis is one of the most widespread diseases globally. 1 Periodontitis prevalence increases steadily with age and peaks at about 38 years of age, indicating a sharp increase between the ages of 30 and 40. 2 The clinical symptoms associated with periodontitis include the progressive development of periodontal pockets and loss of attachment, which eventually results in tooth mobility and loss. Periodontal disease can occur from localized causes, such as bacterial plaque biofilm formation, or from an impaired immune response. 3 Established therapeutic modalities include periodontal scaling and guided tissue regeneration, but their efficacy for fully ameliorating the periodontal inflammatory microenvironment to regenerate the periodontium remains limited. However, recent stem cell research advancements have shown significant promise for such regeneration.4,5 Human periodontal ligament stem cells (hPDLSCs), which are capable of regeneration and maintaining periodontal homeostasis, are found in the periodontal ligament. 6 HPDLSCs are undifferentiated mesenchymal cells with the potential for multidirectional differentiation and self-regeneration. They can differentiate into fibroblasts, osteoblasts, cementoblasts, and non-dental tissues. Tassi et al. indicated that hPDLSCs are the most appropriate mesenchymal stem cell population for regeneration of the periodontium after reviewing 22 pre-clinical studies on periodontal regeneration associated with mesenchymal stem cells. 7
Strontium ions (Sr2+) are a type of alkaline earth metal element that are abundantly present in nature and one of the vital trace constituents of the human body. 8 Mainly found in the bones and teeth, Sr2+ plays an indispensable role in fostering bone growth and averting bone resorption. 9 Multiple studies have revealed that Sr2+ can stimulate bone growth and development, as well as mitigate menopausal osteoporosis, coronary heart disease, hypertension, and other cardiovascular ailments. Additionally, Sr2+ can facilitate cell growth and development, deter dental caries, and serve as a supplement for diabetes treatment.10–14 Sr2+ deficiency can easily lead to metabolic disorder development, such as weakness in the extremities, sweating, and bone retardation, and may cause osteoporosis and other serious consequences. Furthermore, at least 34 medical conditions have been discovered to be associated with significantly reduced Sr2+ content in the body.15,16 Research on the drinking water in longevity villages in China has consistently demonstrated high Sr2+ content. Concurrently, trace element detection indicated higher Sr2+ content in the hair of long-lived elderly individuals compared with that of ordinary elderly individuals.17,18 Therefore, previous studies have called Sr2+ a “long-lived” element. The application of Sr2+ in the oral cavity is also relatively common. For example, a strontium-rich implant coating can effectively promote the adhesion, proliferation, and osteogenic differentiation of osteoblasts, as well as promote bone integration. 19
The role of Sr2+ in bone tissue has been extensively examined over the last few decades, prompted by research advancements on strontium ranelate. This anti-osteoporosis pharmaceutical drug has been used for decades to treat postmenopausal osteoporosis in women and has been approved as a medication in more than 70 countries. 20 Sr2+ can promote osteogenic development and inhibit bone marrow mesenchymal stem cell (BMSC) lipogenic differentiation by selectively blocking the ERK pathway. 21 Brennan et al. found that Sr2+ could promote the early adherence, growth, maturation, and matrix mineralization of human osteoblasts by activating calcium-sensing receptors (CaSRs). 22 Caudrillier et al. reported that Sr2+ can inhibit osteoclast differentiation by mediating CaSR by reducing the reactivity of tartrate resistant acid phosphatase in mouse osteoclasts. 23 In addition, Sr2+ can stimulate human adipose stem cells to differentiate into osteoblasts, human umbilical venous endothelial cells to proliferate and regulate macrophage M2 polarization to promote bone formation, and dental pulp stem cells to proliferate and mineralize the matrix.24–27 However, few studies have examined how Sr2+ affects the proliferation and osteogenic potential of hPDLSCs.
In this study, we aimed to determine the impact of Sr2+ on hPDLSC proliferation and osteogenic differentiation and explore the potential mechanisms underlying these effects. Previous research has shown that Sr2+ influences the Wnt signaling pathway, thereby regulating the osteogenic potential of BMSCs in mice.28,29 Consequently, it is possible that Sr2+ can modulate the ability of hPDLSCs to proliferate and differentiate osteogenically via canonical Wnt signaling. This hypothesis provides potential avenues for the medical application of Sr2+ and hPDLSCs for periodontal regeneration.
Methods
HPDLSC culture and identification
The ethics committee of the School of Shandong University authorized this study in compliance with the Declaration of Helsinki (Approval No. 20220401). Premolars were extracted from adolescent patients between 10 and 18 years of age who were treated in the Department of Orthodontics from September 2022 to September 2023. Written informed consent was obtained from both the patients and their guardians. For inclusion, the premolars were required to be free of periodontal tissue disease. The extracted teeth were quickly placed into a pre-prepared centrifugal tube containing 5% penicillin/streptomycin antibiotic (Solarbio, Beijing, China) and α-Minimal Essential Medium (α-MEM; Solarbio), then transferred to a laboratory bio-safety cabinet. The culture medium in the centrifugal tube was discarded and the teeth were rinsed with a phosphate buffer containing 5% penicillin/streptomycin. The risk of contamination was minimized by removing any saliva and blood from the tooth surface. The periodontal ligament tissue on the surface of the root was then gently removed after applying pressure to the crown. The obtained periodontal ligament was placed in culture medium containing 20% fetal bovine serum (FBS; Yeasen, Shanghai, China). The underside of the periodontal ligament was then placed facing upwards. After being incubated for 4 to 6 hours at 37°C with 5% CO2, the culture container was rotated to ensure the tissue mass was completely immersed. The medium was changed every 3 days. After about 1 to 2 weeks, cell emergence was observed. When the cells covered approximately 80% of the bottom of the culture container, the original culture medium was discarded, the cells were washed with PBS, and 2 mL trypsin-EDTA solution (Yeasen) was added to dissociate the cells. The culture container was placed in the incubator for 1 to 2 minutes, after which the cell suspension was transferred into a centrifuge tube and centrifuged at 1000 rpm for 5 minutes at room temperature. After the supernatant was removed and fresh medium was added, the cells were re-suspended and transferred at a ratio of 1:2 or 1:3. The medium was replaced every 3 days. The subsequent passage was initiated after the cell growth density reached 80%. Cells from the third to fifth generation were selected for subsequent experiments. Cell phenotypes were detected by flow cytometry using fluorescent isothiocyanate (FITC)-labeled antibodies against CD34 (clone 581), CD44 (clone HI44a), CD45 (clone HI30), CD90 (clone 5E10), and CD105 (clone SN6) (Beckman Coulter, Franklin Lakes, NJ, USA) at 1:200 dilutions.
HPDLSC differentiation ability assay
The multipotential differentiation of hPDLSCs was evaluated using osteogenic and adipogenic assays. For this purpose, 1 × 105 hPDLSCs were seeded in each well of six-well plates. The culture medium was an osteogenic induction medium, specifically α-MEM containing 10% FBS, 10 nM dexamethasone, 10 mM sodium β-glycerophosphate, and 50 µg/mL vitamin C (Sigma-Aldrich, Shanghai, China). The osteogenic induction medium was added approximately 24 hours after the cells were seeded. Following a 21-day induction period, the cells were fixed using 4% paraformaldehyde for 30 minutes at room temperature. After a PBS rinse, alizarin red staining solution (Cyagen Biosciences, Guangzhou, China) was applied for 30 minutes to stain the cells. After rinsing with triple distilled water, the staining results were observed.
For adipogenic induction, a lipogenic induction medium was used, specifically α-MEM containing 10% FBS, 500 μM 3-isobutyl-1-methylxanthine, 200 μM indomethacin, 10 μM dexamethasone, and 10 μg/mL insulin (Solarbio). Following a 21-day induction period, the cells were rinsed with PBS and stained with Oil Red O staining solution (Cyagen Biosciences) for 30 minutes. After rinsing with triple distilled water, the staining results were observed.
Cell counting kit-8 (CCK-8) cell proliferation assay
The hPDLSCs were seeded in 96-well plates at a density of 2 × 103 cells per well. Approximately 24 hours later, complete culture medium containing varying concentrations of Sr2+ (0, 0.001, 0.01, 0.1, or 1 mM; Sigma-Aldrich) was added. For each experimental condition, six replicate wells were prepared, each receiving 200 μL of medium. The original medium was replaced on days 1, 3, and 5, with 100 μL of CCK-8 detection reagent (Biosharp, Anhui, China) then added. After a 2-hour incubation period, the absorbance at 450 nm for each well was measured using a plate reader (BMG Labtech, Ortenberg, Germany) to assess cell viability.
Alkaline phosphatase (ALP) staining and activity assay
Approximately 1 × 105 hPDLSCs were seeded in each well of six-well plates. An osteogenic induction culture medium containing varying concentrations of Sr2+ (0, 0.001, 0.01, 0.1, or 1 mM) was applied for about 24 hours. The medium in each well was replaced every 3 days. After 7 days of incubation, the cells were lysed in RIPA buffer for 30 minutes on ice. The lysate was collected and centrifuged at 12,000 rpm for 15 minutes at 4°C, then the ALP activity was assessed following the suggested protocol of the alkaline phosphatase assay kit (NJJCBIO, Nanjing, China). ALP staining was performed by preparing ALP staining solution following the instructions of the 5-bromo-4-chloro-3-indolyl phosphate/nitro blue tetrazolium (BCIP/NBT) alkaline phosphatase colorimetric kit (NJJCBIO), then observing the color change.
Alizarin red staining
The hPDLSCs were fixed with 4% paraformaldehyde for 30 minutes. After a PBS rinse, Alizarin red staining solution was added. The coloration was observed after washing. Subsequently, 10% cetyl pyridinium chloride (Solarbio) was added, then the optical density value was measured using a spectrophotometer (SPECTRAstar, Nano, BMG Labtech). Three replicates were performed for each group.
Quantitative reverse transcription polymerase chain reaction (RT-qPCR)
The hPDLSCs were prepared as described in the ALP assay section. Following incubation for 7 days, total RNA was isolated using Trizol (Agbio, Hunan, China), then a reverse transcription kit (Yeasen) was used to produce cDNA. Next, RT-qPCR was conducted using the SYBR®Premix Pro Taq HS qPCR Kit (Agbio) and Roche Light Cycler® 480 Sequence Detection System (Roche Diagnostics GmbH, Mannheim, Germany). Each sample was assayed at least three times. Data analysis was conducted using the 2−ΔΔCt method to calculate the relative expression of target genes, with target gene expression levels normalized to GAPDH expression levels. The following primer sequences were used in this study: collagen I (COL-1) forward: 5′-TAAAGGGTCACCGTGGCTTC-3′, COL-1 reverse: 5′-GGGAGACCGTTGAGTCCATC-3′; ALP forward: 5′-GGCGGTGAACGAGAGAATGT-3′, ALP reverse: 5′-GGACGTAGTTCTGCTCGTGG-3′; Runx family transcription factor 2 (RUNX2) forward: 5′-GGAGTGGACGAGGCAAGAGT-3′, RUNX2 reverse: 5′-AGGCGGTCAGAGAACAAACT-3′; GAPDH forward: 5′-GCACCGTCAAGGCTGAGAAC-3′, GAPDH reverse: 5′-TGGTGAAGACGCCAGTGGA-3′.
Western blot analysis
Cell lysates were obtained after 7 days of incubation. The samples were lysed with RIPA buffer for 30 minutes on ice, then gently removed and centrifuged at 12,000 rpm for 15 minutes at 4°C. The BCA kit (Solarbio) was used to determine the protein concentrations of the supernatants. Protein samples were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis, then transferred to a polyvinylidene fluoride membrane. The membrane was blocked with TBS buffer containing skim milk powder and Tween-20 (TBST). The membrane was divided into strips according to the molecular weights of COL-1, ALP, RUNX2, and GAPDH. The strips were incubated with the following primary antibodies overnight at 4°C at 1:1000 dilutions: anti-COL-1 (EPR27415-30, Abcam, Cambridge, UK), anti-ALP (EPR27191-14, Abcam), anti-RUNX2 (EPR14334, Abcam), and anti-GAPDH (EPR16891, Abcam). Incubation with secondary antibodies (ab288151, Abcam, 1:1000) was performed for 1 hour at ambient temperature. An enhanced chemiluminescence substrate kit (Yeasen) was used to develop the strips, which were quantitatively analyzed using ImageJ software (National Institutes of Health, Bethesda, MD, USA) for grayscale values.
XAV939 treatment
Approximately 1 × 105 hPDLSCs were seeded per well in six-well plates and allocated into four treatment groups: (i) osteogenic induction solution, (ii) osteogenic induction solution containing 2.5 × 10−6 M XAV939, (iii) osteogenic induction solution containing 0.01 mM Sr2+, and (iv) osteogenic induction solution containing 2.5 × 10−6 M XAV939 and 0.01 mM Sr2+. The medium in each well was refreshed every 3 days. Western blot analysis and RT-qPCR were performed to identify the expression levels of osteogenesis markers, such as COL-1, ALP, and RUNX2, and markers associated with specific pathways, such as β-catenin, glycogen synthase kinase 3 (GSK3), and phosphorylated glycogen synthase kinase 3 P-GSK3 (Ser9) (Abcam).
Statistical analysis
The data are presented as the mean ± standard deviation. Each experiment was conducted in triplicate. The data were analyzed using GraphPad Prism 9.0 (GraphPad Software, Inc., La Jolla, CA, USA), with differences analyzed using the Student’s t-test or one-way analysis of variance (ANOVA). The Shapiro–Wilk test was used to evaluate the normality of the data and the F-test was used to evaluate the homogeneity of the variance. The Tukey’s test was used for post hoc analysis. P < 0.05 was considered statistically significant.
Results
hPDLSC isolation, identification, and culture
Approximately 10 days after sampling, hPDLSCs began to emerge from the tissue block, exhibiting long spindle or spindle-shaped cell morphology (Figure 1(a)). For subsequent experiments, cells from passages 3 to 5 were selected. Following the induction of osteogenesis and lipogenesis, red mineralized nodules and lipid droplets were observed, respectively, which demonstrated the multidirectional differentiation potential of these hPDLSCs (Figure 1(b), (c)). Additionally, flow cytometry analysis indicated that the hPDLSCs were positive for CD44, CD90, and CD105, while they were negative for CD34 and CD45, confirming their stem cell phenotype (Figure 1(d)–(h)).

Isolation, cultivation, and characterization of human periodontal ligament stem cells (hPDLSCs). (a) Morphology of the hPDLSCs; scale bar: 200 μm. (b) Osteogenic induction staining; scale bars: 100 μm. (c) Lipogenic induction staining; scale bars: 100 μm. (d–h) The expression levels of (d) CD44, (e) CD90, (f) CD105, (g) CD34, and (h) CD45 were examined by flow cytometry.
hPDLSC proliferation assay
The CCK-8 assay results suggested that during the first and third days of culture, the proliferation of hPDLSCs exposed to varying concentrations of Sr2+ (0, 0.001, 0.01, 0.1, 1 mM) did not exhibit significant differences. However, on the fifth day of culture, the 0.01 and 0.1 mM Sr2+ concentrations significantly enhanced hPDLSC proliferation (P < 0.0001). Within the 0.001 to 1 mM range, Sr2+ did not demonstrate a notable inhibitory effect on hPDLSC growth (Figure 2(a), (b)).

Effects of different Sr2+ concentrations on human periodontal ligament stem cell (hPDLSC) proliferation. (a) Proliferation of hPDLSCs at 1, 3, and 5 days and (b) at day 5, 0.01 mM Sr2+ significantly promoted hPDLSC proliferation compared with the control group; ****P < 0.0001.
ALP activity assay and staining test
The ALP activity was measured at 7 days to examine how Sr2+ affected the osteogenic potential of the hPDLSCs. The results revealed that 0.01 mM Sr2+ markedly increased the ALP activity. The ALP staining test revealed that 0.01 mM Sr2+ had a strong positive staining affinity for ALP and pronounced precipitation compared with the control group (P < 0.001, Figure 3(a), (b)).

Effects of different Sr2+ concentrations on human periodontal ligament stem cell (hPDLSC) alkaline phosphatase (ALP) activity. (a) ALP staining results at different Sr2+ concentrations and (b) treatment with 0.01 mM Sr2+ significantly promoted hPDLSC ALP activity compared with the control group; ***P < 0.001.
Alizarin red staining
The Alizarin red staining experiments and quantitative analysis at 21 days revealed that 0.01 mM Sr2+ significantly enhanced the formation of mineralized nodules (P < 0.0001, Figure 4(a), (b)).
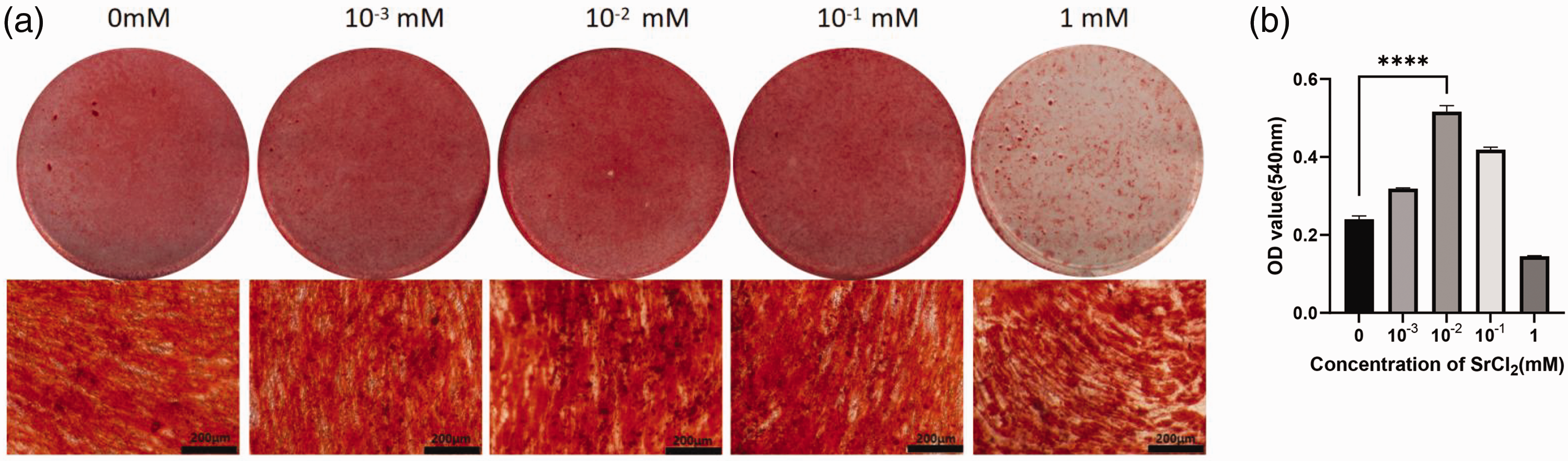
Alizarin red staining results. (a) Alizarin red staining results at different Sr2+ concentrations and (b) treatment with 0.01 mM Sr2+ significantly promoted mineralized nodule formation compared with the control group; ****P < 0.0001.
Sr2+ enhanced the expression of relevant osteogenic factors in hPDLSCs
The mRNA and protein expression levels of osteogenic-related factors, including COL-1, ALP, and RUNX2, were markedly increased in the 0.01 mM Sr2+ hPDLSC group after 7 days of incubation (P < 0.01, Figure 5(a)–(g)).

Sr2+ promoted the expression of osteogenic-related factors in human periodontal ligament stem cells (hPDLSCs). (a) Western blot strip. (b–d) Western blot analysis results for (b) alkaline phosphatase (ALP), (c) Runx family transcription factor 2 (RUNX2), and (d) collagen I (COL-1). (e–g) RT-qPCR results for (e) ALP, (f) RUNX2, and (g) COL-1; ****P < 0.0001, **P < 0.01.
Role of the Wnt signaling pathway in Sr2+-enhanced hPDLSC osteogenic differentiation
The results of the previous experiments revealed that a concentration of 0.01 mM Sr2+ significantly improved the hPDLSC osteogenic potential. Inhibition of the canonical Wnt signaling pathway with XAV939 treatment resulted in decreased expression levels of the osteogenic-related proteins COL-1, ALP, and RUNX2, as well as reduced expression patterns of proteins associated with the β-catenin and P-GSK3β (Ser9) pathways. No significant difference was observed for GSK3β protein expression, indicating that the canonical Wnt signaling pathway had been suppressed. In addition, simultaneous administration of XAV939 and 0.01 mM Sr2+ indicated that Sr2+ could partially reverse the XAV939-mediated inhibition of the canonical Wnt signaling pathway. These results suggest that the osteogenic-promoting effect of Sr2+ correlates with activation of the canonical Wnt signaling pathway (P < 0.05, Figure 6(a)–(j)).
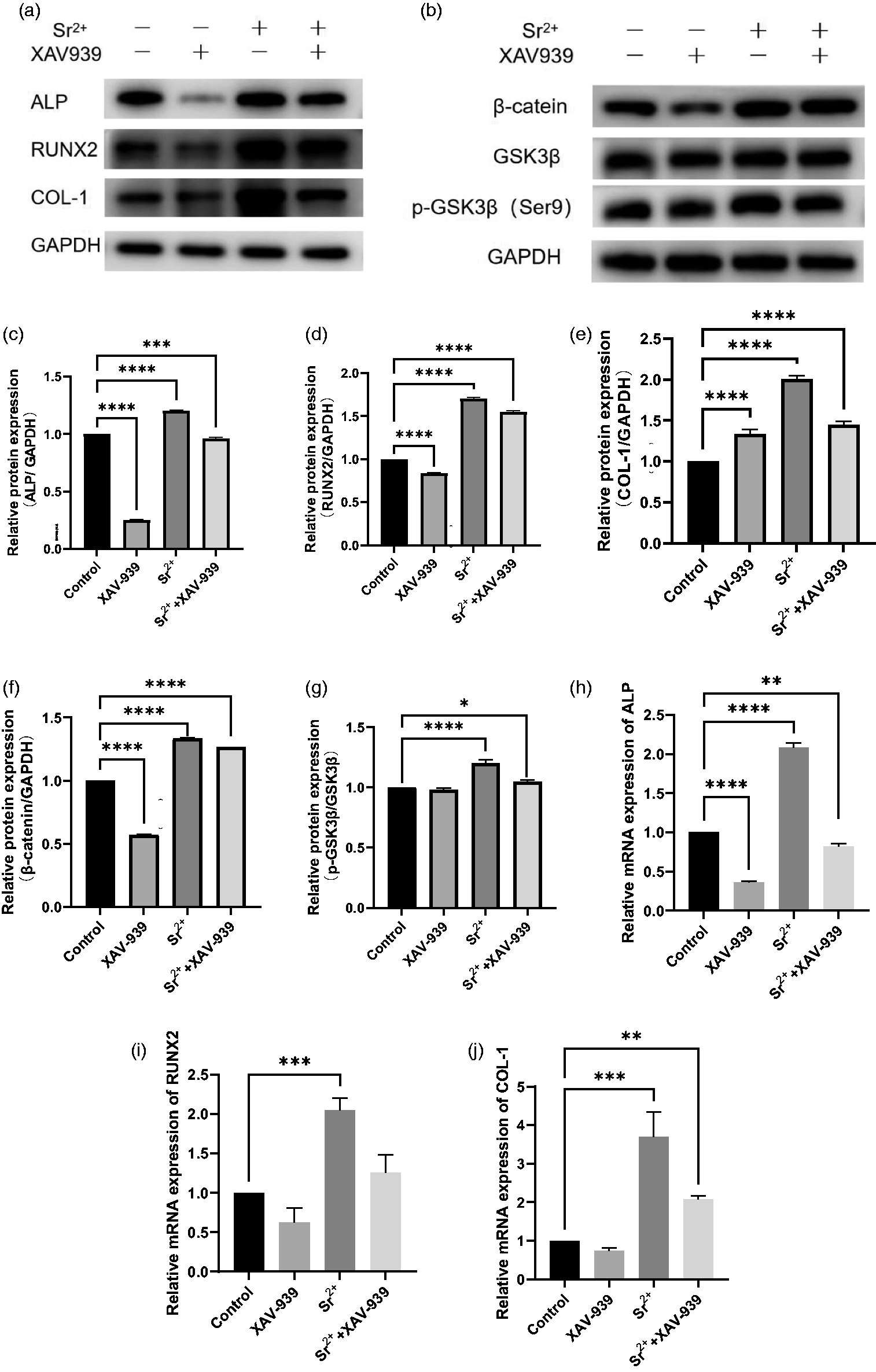
Effects of XAV939 treatment on Sr2+-promoted human periodontal ligament stem cell (hPDLSC) osteogenic differentiation. (a) XAV939 treatment inhibited the expression of osteogenic-related proteins. Continued.(b) XAV939 inhibited the expression of Wnt pathway-related proteins. (c–g) Western blot analysis results for (c) alkaline phosphatase (ALP), (d) Runx family transcription factor 2 (RUNX2), (e) collagen-1 (COL-1), (f) β-catenin, and (g) glycogen synthase kinase 3 (p-GSK3β/GSK3β). (h–j) RT-qPCR results for (h) ALP, (i) RUNX2, and (j) COL-1; *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Discussion
Periodontitis is a chronic inflammatory condition that leads to destruction of the supporting tissues of the periodontium, representing the principal cause of tooth loss in adults.30,31 Numerous studies have confirmed that hPDLSCs can maintain homeostasis of the periodontium microenvironment and support periodontal tissue damage repair. However, periodontitis results in a substantial accumulation of pro-inflammatory cytokines within the periodontium, which impair the capacity of hPDLSCs to synthesize new bone. Consequently, enhancing the osteogenic differentiation abilities of hPDLSCs to facilitate periodontal tissue regeneration has garnered considerable interest.32,33
Sr2+ is closely related to bone and can regulate osteoblasts and osteoclasts through multiple pathways. Strontium chloride and strontium ranelate have both been shown to improve bone tissue health. 34 Sr2+ has two main roles in bone tissue. First, it can promote osteoblast formation and upregulate the expression levels of RUNX2, ALP, bone sialoprotein (BSP), and bone Gla protein (BGP) to increase osteoblast proliferation and differentiation, as reported by Marx et al. 35 Second, it can suppress osteoclast formation and differentiation, as well as promote osteoclast apoptosis, thus reducing bone resorption. 36 These studies have suggested that Sr2+ has periodontium regeneration potential. Thus, it is crucial to investigate how Sr2+ affects hPDLSC proliferation and osteogenic differentiation.
In this study, we cultured hPDLSCs from premolars of young patients, then analyzed their phenotypic and multidirectional differentiation ability. Our data revealed that the hPDLSCs expressed BMSC surface markers, such as CD44, CD90, and CD105, but did not express hematopoietic stem cell surface markers, such as CD34 and CD45. We also verified the multi-directional differentiation function of these hPDLSCs. Next, we treated the hPDLSCs in culture with various Sr2+ concentrations, finding that 0.01 mM Sr2+ was the optimal concentration for promoting proliferation and osteogenesis. These results are consistent with those of Romagnoli et al. 8 Other studies have indicate that pre-mixed calcium, including the silicate cement-based sealers Bioroot Flow, AH Plus Bioceramic Sealer, Ceraseal, and Totalfill BC Sealer, can promote human periodontal stem cell osteogenic differentiation and downregulate inflammatory cytokine expression levels. These results demonstrate potential directions for the future application of Sr2+ in root canal therapy.37,38
ALP activity, mineralized nodules, and osteogenesis-related factor expression patterns are common indicators of the osteogenic differentiation ability of cells. Our experiments revealed that culturing hPDLSCs with 0.01 mM Sr2+ led to significantly increased levels of these indicators, further suggesting that this is the optimal Sr2+ concentration for promoting hPDLSC osteogenic differentiation.
Three classical osteogenesis-related genes, COL-1, ALP, and RUNX2, were selected for analysis in this study. ALP is involved in the extracellular matrix mineralization process, serving as an early marker of osteoblast maturation. 39 RUNX2, a crucial transcription factor in the conversion of mesenchymal stem cells to osteoblasts, is a member of the runt structural domain gene family of transcription factors that is involved in different stages of osteoblast differentiation and maturation. 40 COL-1 is a primary protein component of bone and most connective tissues and is closely related to osteogenesis. 41 Here, we used RT-qPCR and western blot experiments to examine the effects of various Sr2+ concentrations on the mRNA and protein expression levels of these osteogenesis-related factors in hPDLSCs. The results demonstrated that Sr2+ treatment could significantly increase the osteogenic-related factor expression levels in hPDLSCs, providing a basis for further examination of the specific mechanism of action.
Previous studies have revealed that Sr2+ can inhibit osteoclast activity by reducing activity of the nuclear factor-κB (NF-κB) receptor activator (RANK) signaling pathway. It can also promote osteoclast activity by triggering the Ras/MAPK signaling pathway and downstream transcription factor (RUNX2) to promote mesenchymal stem cell osteogenic differentiation.42,43 Wnt signaling includes both the canonical and non-canonical Wnt signaling pathways. Canonical Wnt signaling plays a critical role in regulating a wide range of biological processes. Research has shown that this pathway is intricately associated with the osteogenic differentiation of cells and maintenance of periodontal dynamic homeostasis. 44 Several studies have demonstrated that hPDLSC osteogenic differentiation involves the Wnt/β-catenin signaling pathway.45–47 β-catenin is an essential component of the Wnt signaling pathway and key player in signal transduction. GSK3β can mediate phosphorylation of β-catenin, with GSK3β activity negatively correlating with P-GSK3β (Ser9) activity. The small molecule XAV939 can potently inhibit the Wnt signaling pathway by promoting β-catenin degradation. Importantly, XAV939-mediated inhibition is specific to the Wnt/β-catenin pathway and does not affect other signaling pathways, such as NF-κB and TGF-β. 48 After treating hPDLSCs with XAV939, we observed decreased expression levels of proteins associated with the Wnt signaling pathway, demonstrating effective inhibition of this pathway. The expression levels of osteogenesis-related proteins were also downregulated in the XAV939 treatment group compared with the Sr2+ treatment group. This observation suggests that inhibiting the Wnt signaling pathway can affect the ability of Sr2+ to promote bone growth. Furthermore, treatment with Sr2+ and XAV939 in combination resulted in lower protein expression levels compared with Sr2+ treatment alone, indicating that the Sr2+-induced osteogenic stimulation was partially suppressed by XAV939-mediated inhibition of the Wnt signaling pathway. To the best of our knowledge, this is the first report using XAV939 to validate the critical role of the Wnt/β-catenin signaling pathway in enhancing the osteogenic differentiation capability of hPDLSCs by Sr2+.
Previous research has shown that three-dimensional-printed barium-doped calcium silicate/poly-ε-caprolactone composite scaffolds and sodium alginate mineralized collagen complex hydrogel can continuously release barium or calcium ions to alter the local osteogenic microenvironment and induce new bone tissue growth. These findings provide a new perspective for using Sr2+ for osteogenic applications.49,50
In conclusion, our study revealed that a 0.01 mM Sr2+ concentration could promote both the proliferation and osteogenic differentiation of hPDLSCs, with the underlying mechanism potentially involving activation of the Wnt/β-catenin signaling pathway. We believe that this study will provide a valuable reference for the clinical application of Sr2+ and hPDLSCs in periodontal tissue regeneration, suggesting promising avenues for future therapies aimed at restoring periodontal health. This study did have some limitations, including a lack of detailed descriptions of the properties of Sr2+ in vitro, in vivo or dentin contact studies, and clinical application experiments. These points will be addressed in future studies. We also plan to integrate Sr2+ into dental materials, such as implant surface coatings, dental defect filling materials, and drugs and materials for pulp treatment, to verify the ability of Sr2+ to promote periodontal regeneration in clinical applications.
Footnotes
Acknowledgements
We thank the reviewers and editors for their hard work, as their comments and suggestions have greatly helped to improve the quality of this article.
Author contributions
TS, DM, and JZ conceived and conceptualized the study, drafted the manuscript, and reviewed the manuscript. TS, YS, JH, ZY, and XW collected and analyzed the data, including the statistical analysis. DM obtained the funding. All authors read and approved the final version of the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Natural Science Foundation of Shandong Province (Grant/Award Number: ZR2021QH340) and Shandong Provincial Hospital Affiliated to Shandong First Medical University (Grant/Award Number: 2020FY026).
