Abstract
Objective
The growth factor progranulin (PGRN) is widely expressed and plays important roles in anti-inflammatory signaling and bone regeneration. However, the anti-inflammatory and pro-osteogenic roles of PGRN in periodontitis are seldom studied. We used an in vitro model to investigate whether PGRN can promote osteogenic differentiation of periodontal ligament stem cells (PDLSCs).
Methods
PDLSCs were treated with PGRN (0 to 100 ng/mL) and the optimal concentrations required to induce proliferation and osteogenesis were identified. PDLSCs were cultured with 10 ng/mL tumor necrosis factor (TNF)-α, 25 ng/mL PGRN, or 10 ng/mL TNF-α + 25 ng/ml PGRN; untreated PDLSCs were used as controls. The effects of PGRN on PDLSC proliferation and osteogenic differentiation were assessed.
Results
PGRN (5, 25, and 50 ng/mL) promoted PDLSC proliferation and osteogenic differentiation, with the 25-ng/mL dose showing the largest effect. Furthermore, 25 ng/mL PGRN reversed inhibition of osteogenic differentiation by TNF-α.
Conclusion
PGRN promotes PDLSC proliferation, osteogenic differentiation, and mineralization in both inflammatory and non-inflammatory conditions. The 25-ng/mL PRGN dose was the most suitable for inducing proliferation and osteogenesis. Further studies using animal models will be required to obtain pre-clinical evidence to support using PGRN as a treatment for periodontitis.
Keywords
Introduction
Periodontitis is a chronic inflammatory disease resulting from multiple underlying factors. The main pathological changes of periodontitis comprise the destruction of periodontal soft and hard tissue, ultimately leading to tooth loss. 1 The long-term objective of periodontal therapy is reconstruction of destroyed periodontal tissue. Stem cells and tissue engineering technologies are effective methods for treatment of periodontitis and biological regeneration of periodontal tissue. Periodontal ligament stem cells (PDLSCs) are members of a larger group of oral mesenchymal stem cells that includes dental pulp stem cells, stem cells of human exfoliated deciduous teeth, dental follicle stem cells, and stem cells of the dental apical papilla. PDLSCs are adult stem cells located in periodontal tissue and can differentiate into alveolar bone, periodontal ligament, and cementum. Thus, they are considered good candidates for use in periodontal tissue regeneration. 2
Microorganisms are the initial cause of periodontitis. The ecological imbalance between host cells and bacteria results in progressive destruction of periodontal tissue. 3 , 4 Dysregulation of the immune inflammatory response can cause hypersecretion of proinflammatory cytokines including tumor necrosis factor-α (TNF-α), a factor involved in tissue destruction in periodontitis. 5 Nagasawa et al. 6 found elevated concentrations of TNF-α in gingival crevicular fluid from patients with periodontitis; the concentration of TNF-α was correlated with periodontitis activity. TNF-α is also involved in osteoclastogenesis and interferes with alveolar bone regeneration. 7 There is considerable evidence that TNF-α inhibits osteogenesis. 8 , 9 Lee et al. 10 showed that exposure to TNF-α led to restrained osteoblast differentiation under various inflammatory conditions. Chen et al. 11 demonstrated that osteogenic capability of PDLSCs from patients with periodontitis was markedly diminished compared with individuals with good periodontal health. Therefore, strategies to reduce inflammation and accelerate osteogenesis by targeting TNF-α or TNF receptors (TNFRs) are critical for bone regeneration efforts in the treatment of periodontitis.
Progranulin (PGRN) is a growth factor present in numerous tissues (e.g., epithelial, nervous, immune, and tumor tissues) that participates in diverse physiological and pathological processes. 12 , 13 TNF-α activity is mediated through its receptors, TNFR1 and TNFR2. 14 PGRN is a physiological antagonist of TNFRs 15 and suppresses the inflammatory action of TNF-α. 16 In addition, PGRN strongly promoted regeneration of bone defects in a mouse model. 17 PGRN antagonizes the negative effects of TNF-α while promoting osteogenic activity, suggesting that it may represent a therapeutic candidate for periodontitis control and periodontal regeneration. In a previous study, we showed that PGRN protected against progression of experimental periodontitis 18 and aided in the regeneration of periodontal bone defects. 19 The present study investigated the in vitro effects of PGRN on osteogenic differentiation of PDLSCs under both inflammatory and non-inflammatory conditions. Optimal concentrations of PGRN required for proliferation and osteogenesis of PDLSCs were identified.
Materials and methods
Human PDLSC culture
Collection of human PDLSCs was approved by the ethics committee of Shandong University Stomatological School (approval number 2010015). Each participant provided written informed consent. Healthy premolars were extracted during orthodontic treatment from individuals aged 12 to 18 years. For each premolar tooth, the periodontal membrane was scraped and cultured using an enzymatic tissue block method as previously described. 20 The medium was changed at 3-day intervals until cells dissociated from the tissue block. Subsequently, the cells were grown to 80% confluence, then transferred into fresh culture flasks. Isolation and purification of PDLSCs by limiting dilution was performed as described previously. 1 Cells were used for experiments at the third passage. The immunophenotypes and pluripotency of PDLSCs were evaluated using an existing method. 1 , 21
Assessment of the effects of PGRN on PDLSC proliferation
The effects of PGRN on PDLSC proliferation were assessed using a CCK-8 assay (Dojindo, Rockville, MD, USA). PDLSCs were treated with 0, 5, 25, 50 or 100 ng/mL PGRN in minimum essential medium with α-modification (α-MEM) containing 0.1% fetal bovine serum for 24, 48, or 72 hours. Cells were then treated with CCK-8 solution for 2.5 hours at 37°C. Absorbance values at 450 nm were recorded.
Culture of human PDLSCs with PGRN
First, the optimal concentration of PGRN for osteogenic differentiation was identified. PDLSCs were cultured in six-well plates in medium containing various concentrations of PGRN (R&D Systems, Minneapolis, MN, USA). After culture in osteogenic-induction medium for 7 or 14 days, PDLSCs were subjected to analyses of alkaline phosphatase (ALP) activity. Additionally, after 3 and 7 days of culture, PDLSCs were subjected to real-time polymerase chain reaction to assess mRNA levels of osteogenic markers. Subsequently, adherent PDLSCs were seeded in six-well plates (1 × 105 cells/well). The cells were treated as follows: no PGRN or TNF-α (untreated control group); 10 ng/mL TNF-α (inflammatory microenvironment); 10 ng/mL TNF-α + 25 ng/mL PGRN (PGRN stimulation in an inflammatory microenvironment); and 25 ng/mL PGRN (PRGN stimulation in a non-inflammatory microenvironment). Osteogenic induction medium (α-MEM containing 10% fetal calf serum, 50 U/mL penicillin, 50 mg/mL streptomycin, 0.1 mM dexamethasone, 10 mM β-glycerophosphate, and 50 mg/mL ascorbate-2-phosphate; Sigma-Aldrich, St. Louis, MO, USA) was used to induce osteoblastic differentiation.
ALP activity assay
After PDLSCs had been cultured for 7 or 14 days, cells were lysed with 400 µL of 0.1% Triton X-100 (Solarbio, Beijing, China). The supernatant was collected following centrifugation at 12,000 ×g for 5 minutes. Total protein was extracted from PDLSCs and ALP activity was analyzed following the instructions of the ALP activity kit (Nanjing Jiancheng, Nanjing, China). Absorbance at 520 nm was measured using a microplate reader (Bio-Rad, Hercules, CA, USA) and ALP activity was calculated. The results were normalized to total protein concentrations determined using a bicinchoninic acid (BCA) assay.
Real-time reverse transcription polymerase chain reaction
Total RNA was extracted from PDLSCs and reverse transcribed into cDNA. Real-time polymerase chain reaction assays were carried out using the SYBR PremixTaq™ II kit (Tli RNaseH Plus, Takara, Dalian, China). The reaction conditions were as follows: pre-heating at 93°C for 5 minutes, followed by 40 cycles of denaturation at 93°C for 1 minute, annealing at 55°C for 30 s, and extension at 72°C for 3 minutes. Primers were designed using gene sequences available in GenBank. The sequences of the primers were as follows: glyceraldehyde-3-phosphate dehydrogenase (GAPDH) forward: 5′-
Western blotting
Total protein was extracted from PDLSCs and homogenized with 100 μL of radioimmunoprecipitation assay buffer. Protein concentrations were determined using a BCA assay kit (Nanjing Jiancheng). The loading buffer and protein solution were mixed at a ratio of 1:4 and heated at 95°C for 5 minutes. Each sample (20 µg protein) was then subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride membranes. After blocking with 5% milk for 2 hours, the membranes were incubated overnight with primary antibodies against ALP, Runx2, and OPN (Abcam, Cambridge, UK). The next day, the membranes were incubated with secondary antibodies at room temperature for 1 hour. As a reference, blotting was also conducted by incubation with an anti-β-actin antibody as a reference (Proteintech, Wuhan, China) for 1 hour. Color development was then performed. The integrated optical density values of each band were measured.
Alizarin red staining
After PDLSCs were incubated in osteogenic induction medium for 28 days, they were stained with 1% Alizarin red at room temperature. Formation of mineralized nodules was observed by microscopy. A total volume of 300 µL cetylpyridinium chloride (Sigma-Aldrich) in 10 mM sodium phosphate buffer (pH 7.0) was added to each well and incubated at 37°C for 30 minutes. The destaining solution was collected and absorbance was measured.
Statistical analysis
All data were expressed as means ± standard deviations. Each experiment included five replicates. Statistical significance was determined using one-way analysis of variance. Values of P < 0.05 were considered statistically significant.
Results
PGRN influences PDLSC proliferation
After PDLSCs were cultured for 24 hours, proliferation of cells treated with 25 and 50 ng/mL PGRN was significantly greater than proliferation of control cells (P < 0.05). After PDLSCs were cultured for 48 and 72 hours, proliferation of cells treated with 5 and 25 ng/mL PGRN was significantly greater than proliferation of control cells (P < 0.05). The greatest extent of proliferation was observed in PDLSCs treated with 25 ng/mL PGRN (P < 0.05). However, treatment with 100 ng/mL PGRN led to the inhibition of PDLSC proliferation at three time points (P < 0.05) (Figure 1).

Determination of the optimal concentration of progranulin (PGRN) for promoting periodontal ligament stem cell (PDLSC) proliferation. The greatest extent of proliferation was observed in PDLSCs treated with 25 ng/mL PGRN. Different symbols indicate statistically significant differences among groups (P < 0.05), while the same symbols indicate no statistically significant differences among groups (P > 0.05).
PGRN influences osteogenic differentiation of PDLSCs
PDLSCs were stimulated with various concentrations of PGRN and osteogenic differentiation markers were examined. At day 7, treatment with 5, 2.5, and 50 ng/mL PGRN significantly promoted ALP activity compared with untreated control cells (P < 0.05). At day 14, treatment with 5 and 25 ng/ml PGRN significantly promoted ALP activity compared with untreated control cells (P < 0.05). Treatment with 50 ng/mL PGRN did not result in a significant change in ALP activity. At both days 7 and 14, treatment with 100 ng/mL PGRN significantly reduced ALP activity compared with the control group (P < 0.05) (Figure 2).

Determination of the optimal concentration of progranulin (PGRN) for promoting alkaline phosphatase (ALP) activity in periodontal ligament stem cell (PDLSCs). Treatment with 5 and 25 ng/mL PGRN significantly enhanced ALP activity and treatment with 100 ng/mL PGRN significantly reduced ALP activity compared with untreated control PDLSCs. Different symbols indicate statistically significant differences among groups (P < 0.05), while the same symbols indicate no statistically significant differences among groups (P > 0.05).
Real-time reverse transcription polymerase chain reaction analysis showed that treatment with 5, 25, and 50 ng/mL PGRN significantly enhanced the mRNA expression of ALP and Runx2 (P < 0.05). At days 3 and 7, the greatest effects were observed following treatment with 25 ng/mL PGRN. However, treatment with 100 ng/mL PGRN significantly reduced mRNA expression of ALP and Runx2 at both time points (P < 0.05) (Figure 3).

Determination of the optimal concentration of progranulin (PGRN) for promoting osteogenic gene expression in periodontal ligament stem cell (PDLSCs). (A) Treatment of PDLSCs with 5, 25, and 50 ng/mL PGRN significantly enhanced ALP mRNA expression. (B) Treatment of PDLSCs with 5, 25, and 50 ng/mL PGRN significantly enhanced Runx2 mRNA expression. The greatest effects on ALP and Runx2 mRNA expression were observed following treatment with 25 ng/mL PGRN. Different symbols indicate statistically significant differences among groups (P < 0.05), while the same symbols indicate no statistically significant differences among groups (P > 0.05).
PGRN antagonized TNF-α-mediated inhibition of PDLSC osteogenic differentiation
The above results indicated that 25 ng/mL PGRN was the optimal concentration for enhancing PDLSC proliferation and osteogenic differentiation. Thus, this concentration was used to evaluate the antagonistic effects of PGRN on TNF-α-mediated suppression of PDSLC osteogenic differentiation.
PGRN reversed TNF-α-mediated inhibition of ALP activity and extracellular matrix calcification
ALP activity assays revealed that on days 7 and 14, treatment of PDLSCs with 10 ng/mL TNF-α significantly reduced ALP activity (P < 0.05). By contrast, PDLSCs treated with 10 ng/mL TNF-α + 25 ng/mL PGRN showed no significant difference in ALP activity compared with untreated control cells (P > 0.05). Notably, cells treated with 25 ng/mL PGRN exhibited the highest ALP activity (P > 0.05) (Figure 4).
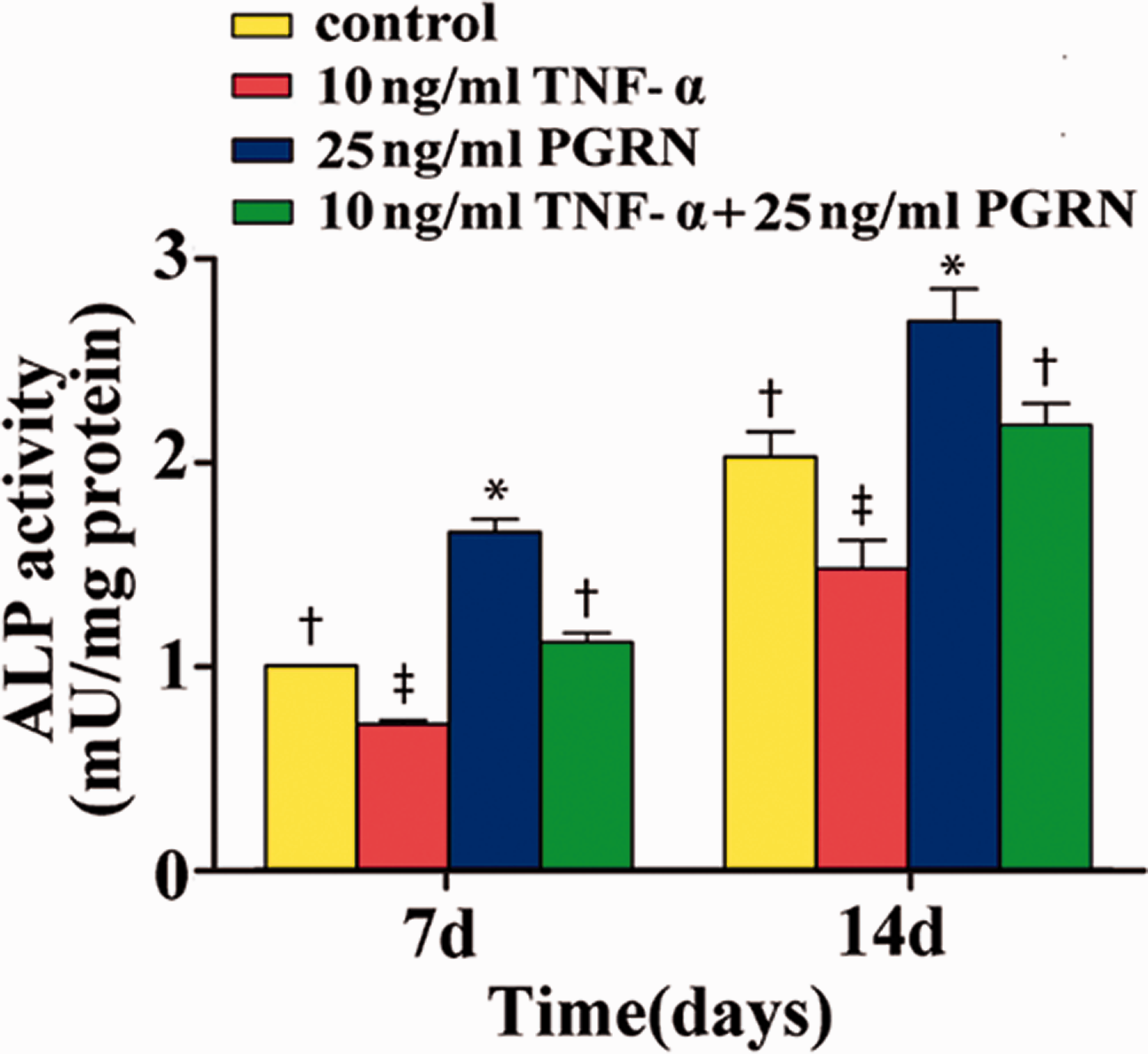
Effect of progranulin (PGRN) on alkaline phosphatase (ALP) activity in periodontal ligament stem cell (PDLSCs). PDLSCs treated with 25 ng/mL PGRN exhibited the highest ALP activity, while PDLSCs treated with 10 ng/mL tumor necrosis factor (TNF)-α exhibited significantly lower ALP activity. Different symbols indicate statistically significant differences among groups (P < 0.05), while the same symbols indicate no statistically significant differences among groups (P > 0.05).
Alizarin red staining was performed to assess extracellular matrix calcification and compare calcium content in different treatment groups. Compared with control cells, PDLSCs treated with 10 ng/mL TNF-α exhibited significantly lower mineralization (P < 0.05). By contrast, PDLSCs treated with 10 ng/mL TNF-α + 25 ng/mL PGRN exhibited similar mineralization and PDLSCs treated with 25 ng/mL PGRN exhibited significantly higher mineralization than control cells (P < 0.05) (Figure 5).

Effect of progranulin (PGRN) on mineralization of periodontal ligament stem cell (PDLSCs) as shown by Alizarin red staining (A–D, 100× magnification) and determination of calcium content (E). PDLSCs treated with 10 ng/mL TNF-α + 25 ng/mL PGRN showed similar mineralization, while PDLSCs treated with 25 ng/mL PGRN exhibited significantly greater mineralization. Different symbols indicate statistically significant differences among groups (P < 0.05), while the same symbols indicate no statistically significant differences among groups (P > 0.05)
PGRN reversed TNF-α-mediated inhibition of osteogenic gene expressions in PDLSCs
Real-time reverse transcription polymerase chain reaction assays showed that at days 7, 14, and 21, mRNA expression of ALP, Runx2 and OPN were significantly lower in PDLSCs treated with 10 ng/mL TNF-α compared with control cells. However, ALP, OPN (days 7, 14, and 21) and Runx2 (day 21) mRNA expression levels were similar in PDLSCs treated with 10 ng/mL TNF-α + 25 ng/mL PGRN compared with control cells. The mRNA expression levels of Runx2 were significantly higher in PDLSCs treated with 10 ng/mL TNF-α + 25 ng/mL PGRN compared with control cells at days 7 and 14 (P < 0.05). At all three time points, PDLSCs treated with 25 ng/mL PGRN exhibited the highest mRNA expression levels of osteogenic genes (Figure 6).

Effect of progranulin (PGRN) on osteoblastic gene expressions in periodontal ligament stem cell (PDLSCs). At all three time points, PDLSCs treated with 25 ng/mL PGRN exhibited the highest mRNA expression levels of osteogenic genes. (A) ALP mRNA expression in PDLSCs treated with 10 ng/mL tumor necrosis factor (TNF)-α + 25 ng/mL PGRN were not significantly different compared with untreated control cells at days 7, 14, and 21. (B) Runx2 mRNA expression was significantly higher in PDLSCs treated with 10 ng/mL TNF-α + 25 ng/ml PGRN compared with untreated control cells at days 7 and 14 but was similar at day 21. (C) OPN mRNA expression in PDLSCs treated with 10 ng/mL TNF-α + 25 ng/mL PGRN was not significantly different compared with untreated control cells at days 7, 14, and 21. Different symbols indicate statistically significant differences among groups (P < 0.05), while the same symbols indicate no statistically significant differences among groups (P > 0.05).
PGRN reversed TNF-α-mediated inhibition of osteogenic protein expression in PDLSCs
Western blotting indicated that at days 7, 14, and 21, levels of ALP, Runx2, and OPN proteins were significantly lower in PDLSCs treated with 10 ng/mL TNF-α compared with control cells (P < 0.05). However, ALP and OPN protein levels (days 7, 14, and 21) and Runx2 protein levels (days 14 and 21) were not significantly different in cells treated with 10 ng/mL TNF-α + 25 ng/mL PGRN group compared with control cells (P > 0.05). Notably, Runx2 protein levels were significantly higher in PDLSCs treated with 10 ng/mL TNF-α + 25 ng/mL PGRN group compared with control cells at day 7 ( < 0.05). At all three time points, the 25 ng/mL PGRN group showed the highest levels of osteogenic proteins (Figure 7).

Effect of progranulin (PGRN) on osteoblastic protein expression in periodontal ligament stem cell (PDLSCs). At all three time points, PDLSCs treated with 25 ng/mL PGRN group showed the highest levels of osteogenic proteins. (A) ALP protein expression in PDLSCs treated with 10 ng/mL tumor necrosis factor (TNF)-α + 25 ng/mL PGRN was not significantly different compared with untreated control cells at days 7, 14, and 21. (B) Runx2 protein expression was significantly higher in PDLSCs treated with 10 ng/mL TNF-α + 25 ng/mL PGRN compared with untreated control cells at day 7, but no significant differences were observed at days 14 and 21. (C) OPN protein expression in PDLSCs treated with 10 ng/mL TNF-α + 25 ng/mL PGRN group were not significantly different compared with untreated control cells at days 7, 14, and 21. Different symbols indicate statistically significant differences among groups (P < 0.05), while the same symbols indicate no statistically significant differences among groups (P > 0.05).
Discussion
PDLSCs possess the capacity for proliferation, self-renewal, and pluripotent differentiation (e.g., into new alveolar bone, periodontal membrane, and cementum). Thus, PDLSCs are regarded as critical factors in periodontal regeneration. 3 In the context of periodontal tissue engineering, PDLSC function is affected by periodontal microenvironment. Invasion by periodontal pathogens can directly damage periodontal tissues and activate host defense cells, which subsequently produce and secrete pro-inflammatory cytokines, (e.g., TNF-α) that contribute to the destruction of periodontal tissues. 18 TNF-α is an important mediator in the development of periodontitis, stimulating the emergence of chemokines and other inflammatory mediators, accelerating the destruction of connective tissue, enhancing the activity of osteoclasts, and promoting bone resorption. Our findings are consistent with those of prior studies showing that TNF-α adversely influenced both osteodifferentiation and osteogenesis in various stem cells. 7 , 9 , 22 , 23
PGRN is a growth factor with multiple effects that has a wide-ranging cellular expression profile. PRGN plays important roles in diverse disease processes such as tumor formation, inflammatory responses, proliferation and differentiation of cartilage cells, and endochondral ossification during cartilage development. Here, we speculated that PGRN may participate in osteogenesis, particularly in the presence of inflammation. Previous studies demonstrated that 500 ng/mL PGRN accelerated the formation of multinucleated osteoclasts among mouse bone marrow cells and stimulated subsequent bone resorption. 24 Moreover, PGRN inhibits TNF-α-induced osteoclastogenesis and promotes osteoblastogenesis. 25 PGRN reportedly stimulates the proliferation of islet cells 26 and promotes the proliferation of retina precursor cells. 27 In our previous study, PGRN promoted osteogenesis within inflamed periodontal intrabony defects in rats. 19 In the present study, we showed that physiological concentrations of PGRN (5, 25, and 50 ng/ml) upregulated osteogenic markers in PDLSCs, thus promoting proliferation, osteogenesis, and mineralization. A dose of 100 ng/mL PRGN attenuated the expression of osteogenic markers. These results clearly indicate that an appropriate concentration of PGRN is required to promote proliferation and osteogenesis in PDLSCs.
PGRN is a natural TNF-α antagonist that can inhibit the biological effects of TNF-α and TNFRs. PGRN suppresses the inflammatory action of TNF-α and inhibits the activation of β-catenin signaling in cartilage and chondrocytes. 16 Our previous study showed that treatment with recombinant human PGRN alleviated alveolar bone loss and inhibited production of TNF-α in the periodontium of rats with experimental periodontitis. 18 The combination of PGRN treatment with TNFR modification can effectively inhibit NF-κB signaling in the presence of inflammation. 15 , 28 In the present study, PDLSCs treated with both PGRN and TNF-α showed improvements in various osteogenic indexes compared with cells treated with TNF-α alone. This finding indicated that PGRN could alleviate the effects of TNF-α on PDLSCs. Alizarin red staining showed that PDLSCs treated with both PGRN and TNF-α formed significantly larger mineralized nodules than PDLSCs treated with TNF-α alone, suggesting that PGRN reversed the effects of TNF-α. These findings imply that PGRN can antagonize the inhibitory effects of TNF-α on osteogenic differentiation of PDLSCs and may improve periodontal regeneration.
TNF-α has two distinct receptors. TNFR1 is the main signaling receptor of TNF-α in all human tissues, while TNFR2 is mainly expressed in immune cells and mediates a limited array of biological responses. PGRN has been shown to directly bind to TNFR, thereby inhibiting TNF-α-mediated inflammation.29–32 Tang et al. 15 demonstrated that PGRN has a higher affinity for TNFR2, thus blocking their interactions with TNF-α. Treatment with recombinant PGRN greatly accelerated repair of bone defects in rats; this induction of bone formation was mediated by effects on TNFR2 rather than TNFR1. Previous studies have indicated that PGRN promotes osteogenesis and chondrogenesis by interactions with TNFR2. Further research is needed to confirm whether PGRN directly promotes osteogenic differentiation of PDLSCs by binding to TNFR2 and activating MAPK signaling.
Conclusion
This in vitro study showed that PGRN promotes PDLSC proliferation, osteogenic differentiation, and mineralization in both inflammatory and non-inflammatory conditions. We found that a dose of 25 ng/mL PGRN was the most suitable for inducing proliferation and osteogenesis of PDLSCs. However, further research using animal models is required to provide preclinical evidence in support of PGRN as a promising treatment for periodontitis.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Programs for Medical & Health Science and Technology Development of Shandong Province of China (No. 2017WS732) and the Scientific Research Project of Weifang Health Committee (No. wfwsjk_2019_034).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
