Abstract
X-DING-CD4 blocks HIV-1 long terminal repeat (LTR) and pathogen induced pro-inflammatory response. Increased activity of the X-DING-CD4 gene is associated with cellular resistance to virus; therefore, HIV-1 elite controllers (ECs) should have higher X-DING-CD4 and reduced pro-inflammatory mRNA activity than viremic or uninfected individuals. Also, depending on the cell stimulating factor, expression of X-DING-CD4 mRNA in ECs might be autonomous or contingent on IFN signaling. We compared expression of X-DING-CD4, IFN-α and IL-8 mRNAs in naive, phytohemagglutinin- or HIV-1 exposed PBMCs from ECs, HIV progressors and negative controls; tested correlation between X-DING-CD4 and IFN-α expression; sensitivity of the X-DING-CD4 gene to IFN-α regulation; and evaluated interactions between innate and pro-inflammatory genes. We found that expression of X-DING-CD4 and IFN-α was up-regulated in ECs and correlated in cells stimulated with mitogen, but not HIV-1. The X-DING-CD4 gene was more sensitive to HIV-1 than rIFN-α stimulation. ECs had significantly less IL-8 mRNA when PBMCs were exposed to exogenous HIV-1. Two-way ANOVA showed that control of HIV-1 and virus-induced pro-inflammatory response by ECs stemmed from interactions between expression of innate immunity and pro-inflammatory genes, the state of cell stimulation and the status of virus control. Consequently, interaction of multiple host innate immune responses rather than a single mechanism regulates restriction of HIV-1 in ECs.
Introduction
The effective long-term suppression of HIV-1 infection is a trait exclusive to a fraction of human population designated as HIV-1 elite controllers (ECs). A multitude of clinical and basic research studies contributed to the understanding of this biological phenomena.1–11 ECs maintain stable CD4+ T cell counts, have an exceptionally efficient CD8+ T cell responses to virus12–14 and restrict viremic dissemination, even in the absence of the antiretroviral therapy. 5 The higher frequency of B*57, B*5701 and B*27 HLA alleles,15–18 and unique expression patterns for leukocyte immunoglobulin-like receptors on myeloid dendritic cells are markers of exceptional Ag-presenting properties and successful restriction of HIV-1 infection in this group. 19 Interestingly, cells from ECs harbor a replication competent virus2,4 and, in an ex vivo setting, support HIV-1 entry and productive infection. 7
There is not a single definition for the overall mechanism regulating restriction of virus by ECs, but mounting evidence suggests that the innate immunity response is critical for a successful blockage of HIV-1 replication in this group.3,20–24 For example, Jin et al. 22 found that expression of APOBEC3G mRNA was elevated in long-term non-progressors compared with un-infected controls and AIDS patients. 22 Another study implicated a function of IFN-α in the innate immune regulation of HIV-1 latency by ECs, 23 while others have reported a positive correlation between the selective up-regulation of p21 cyclin-dependent kinase inhibitor and restriction of HIV-1 replication. 3 Finally, Ferre et al. 20 found that the mucosal IFN-γ, MIP-1β and TNF-α responses were particularly strong in ECs, 20 pointing to the greater complexity and poly-functional quality of their T cells.
Based on the unique proficiency of ECs to restrict replication of HIV-13 and precedence indicating an important role of the innate immunity factors in this restriction3,17,19,22–25 we postulated that the activity of the innate immunity genes might be significantly higher in this group when compared with HIV progressors or control uninfected population. We also posited that depending on the state of cell stimulation, the expression of these factors could be autonomous or orchestrated; for example, two or more factors could be expressed independently leading to a parallel blockage of various stages of HIV-1 replicative cycle, or transcription of one antiviral gene could depend on the activity of another antiviral mRNA. The increased activity of antiviral factors could also limit activation of the pro-inflammatory response and increase the health of the immune system.
To prove our theory we tested the expression of two antiviral genes: the IFN-α and HIV-1 restriction factor, X-DING-CD4, and one pro-inflammatory gene, IL-8. IFN-α is found in abundance in ECs 23 and might block HIV-1 infection indirectly by regulating expression of antiviral factors,26,27 and directly by targeting initiation of reverse transcription. 28 The X-DING-CD4 is a novel member of DING protein family 29 and acts as an inhibitor of HIV-1 transcription by blocking long terminal repeat (LTR)/NF-κB binding.29–32 The X-DING-CD4 protein also blocks the LPS-mediated activation of NF-κB-dependent IL-8 promoter in human macrophages and endothelial cells. 33 Interestingly, the serum content for another human DING variant, human phosphate binding protein (HPBP) was increased in HIV-1-infected people, 34 but the higher concentration of this protein was not correlated to the status of virus control.
First, we measured the mRNA levels for X-DING-CD4, IFN-α and IL-8 mRNA in PBMCs from ECs, HIV progressors and healthy HIV-negative individuals by quantitative RT-PCR (qRT-PCR) protocol. We established an interquartile range (IQR) for the expression of these genes in quiescent, mitogen-activated or HIV-1 challenged cells, and assessed variations in mRNA concentration between the three donor groups. Second, we determined if there was a linear dependence for the expression of X-DING-CD4 and IFN-α genes in cells from ECs under different cell stimulation status, and determined whether expression of X-DING-CD4 is regulated by IFN-α signaling. Finally, we analyzed the interactions between X-DING-CD4, IFN-α and IL-8 mRNA activity, the status of cell stimulation and the type of virus control, defined as elite HIV control, no HIV control or no HIV.
Materials and methods
Study groups
The HIV-1 related characteristics of three study groups.

ECs
ECs should have asymptomatic HIV-1 infection over 10 yrs after seroconversion, be antiviral therapy (ART)-naive, have a CD4+ T cell count > 500 and a HIV plasma load of < 50 copies/ml. The CD4+ T cell count in our EC study group ranged from 704 to 1634, and HIV plasma load was < 50 copies/ml (Table 1).
Progressors
Progressors should have symptomatic infection and have initiated ART within 10 yrs of seroconversion, have a CD4+ T cell count <200 and a HIV plasma load of >5000 copies/ml. Our group of HIV progressors consisted of AIDS patients treated with anti-retrovirals (ARVs) (n = 7) or who had not been treated (n = 3). The main criterion for patient selection in this study group was a low (<200) CD4+ T cell count, indicating ongoing HIV disease. The average CD4+ T cell count in this study group was 82 and HIV plasma load was 56,650 copies/ml (Table 1). Depending on the year of the sample collection, the cut-off for HIV-1 plasma load assay was 48 or 50 copies/ml. Our control group was selected from healthy, HIV-negative individuals.
Cell cultures, viruses and reagents
PBMCs were isolated by leukapheresis and cryopreserved immediately after isolation. For testing the expression of X-DING-CD4, IFN-α and IL-8 mRNA 5 × 106 PBMCs was collected upon arrival at the laboratory, pelleted by centrifugation and suspended in Trizol reagent (Life Technologies, Grand Island, NY, USA) for RNA isolation. This sample represented the un-induced cells. The remaining PBMCs were cultured for 24 h in RPMI medium supplemented with 10%/vol FBS, phytohemagglutinin (PHA; 5 mg/ml; Sigma, St. Louis, MO, USA), Il-2 (10 U/ml; Biotest Diagnostics, Denville, NJ, USA), antibiotics, and glutamine. Twenty-four h after PHA treatment cells were collected as described above. This sample is defined as mitogen-induced. For HIV-1 challenge cells we used the X4 NL4-3 HIV-1 clone; 36 cells were washed twice in sterile PBS and exposed for 1 h to virus at a multiplicity of infection (MOI) of 0.01. Following HIV-1 infection cells were washed twice in sterile PBS and cultured for 24 h in RPMI medium as described above but without PHA. This sample is defined as HIV-1 exposed. Separate cell cultures from each PBMC donor were maintained in RPMI medium supplemented with 10% FBS, IL-2, antibiotics and glutamine for 24 h. These cultures represented the mock medium control. The viability of cells in all samples was above 75% at any experimental time-point.
The HIV-1 resistant, X-DING-CD4 (+) T cells 31 were used for testing the X-DING-CD4 gene response to HIV-1 or rIFN-α cell stimulation (the experimental detail is described below). These cells were maintained in RPMI 1640 (Sigma, St. Louis, MO, USA) supplemented with 5%/vol FBS, antibiotics and glutamine, as described before. 29
RNA isolation and qRT-PCR analysis
Primers and probes used for qRT-PCR analysis.

The primers and probes were designed based on the X-DING-CD4 sequence (GenBank HQ586056), IFN-α (GenBank NM_024013.2) and IL-8 (GenBank NM_000584.3); all primers were synthesized by Invitrogen; the fluorogenic probes were synthesized by Life Technologies (Grand Island, NY, USA). The respective primer and probe sequences are listed in Table 2. The HIV-1 gag primers and probe were as published previously. 38 The real-time detection of mRNA expression was performed through the absolute (qRT-PCR) or relative (rRT-PCR) quantitation protocol using TaqMan Gene Expression Assay on a 7300 real-time PCR System (Life Technologies, Grand Island, NY, USA). Briefly, for the absolute quantitation of the X-DING-CD4, IFN-α and IL-8 genes triplicates of total reaction mixture of 25 µl were composed of 2 µl of cDNA, 12.5 µl of 2 × ABI TaqMan PCR Master Mix, 0.25 µl (90 µM) of both sense and antisense primers, and 0.5 µl (10 µM) of the probe (Table 2). After the initial denaturation at 95℃ (10 min), target genes were amplified through 40 cycles of universal cycling conditions (95℃/10 s, 60℃/1 min). All PCR reactions were normalized to β-actin mRNA amplified using pre-designed pair of primers and a probe in concentrations suggested by manufacturer (Life Technologies, Grand Island, NY, USA). Standard cell dilution curves were calibrated using serial dilutions of control pTA_X-DING-CD4, pTA_IFN-α or pTA_IL-8 plasmids representing 1 × 106, 1 × 105, 1 × 104, 1 × 103, 1 × 102 and 10 copies of respective cDNA per reaction; all values are expressed as means ± SD of the mean.
For detection of interferon regulatory factor 7 (IRF-7) mRNA activity we used the IRF-7 mRNA detection kit (Life Technologies, Grand Island, NY, USA) and applied the relative quantitation (rRT-PCR) protocol. 39 All samples were normalized to β-actin mRNA as described above.
The results of qRT-PCR analyses are presented as the IQR or error bars; the rRT-PCR results are presented as log10 relative quantitation. The P-values for the X-DING-CD4 and IFN-α mRNA IQRs were derived from the Mann–Whitney rank sum test calculated by Sigma Plot 11 (Systat Software, Inc. San Jose, CA, USA); the Pearson’s correlation coefficient for expression of X-DING-CD4 and IFN-α mRNAs, the Kruskal-Wallis test to calculate P-values for ART(+) vs ART(−) HIV progressors for expression X-DING-CD4, IFN-α and IL-8 mRNA, and two-way ANOVA were analyzed by GraphPad Prism5 (GraphPad Software, Inc., San Diego, CA, USA). The alpha level was set at 0.050.
Evaluation of X-DING-CD4 gene sensitivity to IFN-α and HIV-1 stimulation
The X-DING-CD4(+) cells were exposed for 24 h to X4 NL4-3 HIV-1 at a MOI of 0.01 or 0.1 ng/ml human rIFN-α (Universal Type I IFN human rIFN-alpha A/D; R&D Systems, Minneapolis, MN, USA) or cultured alone. Subsequently, cells were collected for total RNA isolation, and analysis of X-DING-CD4 mRNA was performed by qRT-PCR and IRF-7 by rRT-PCR as described above.
Results
The expression of X-DING-CD4 and IFN-α mRNA is elevated in PBMCs from ECs
We assessed the selected mRNA expressions in PBMCs that were either naive to exogenous stimuli or stimulated by PHA, or exposed to HIV-1 for 24 h. Both PHA and HIV-1 activate T cells,40,41 and the effect of these diverse stimulating factors on gene expression could be compared in a mixed cell population. Evaluations of gene expression in the individual cell subsets were beyond the scope of this study.
For HIV-1 challenge we used the laboratory strain NL4-3 HIV-1. 36 Based on our previous observations, the cellular exposure to R5 or X4 HIV-1 did not generate variations in X-DING-CD4 mRNA expression in human monocytes and macrophages. 38 Also, data published by Rabi et al. 7 suggested that diversity in the viral envelope did not affect fusion and productive infection in cells of ECs. For all experiments involving HIV-1 exposure, the 24-h time point was selected solely for the observation of virus-mediated innate immune response and not the virus replication itself. The IQR for HIV-1 gag mRNA at this time point was 0.0371–0.878 c/ng RNA with one outlier at 0.0012 c/ng RNA in samples from negative controls and 0.186–0.692 c/ng RNA in samples from ECs. We did not test the HIV-1 gag mRNA transcripts in samples from HIV progressors as these individuals have a high content of endogenous HIV-1.
Our qRT-PCR evaluations showed statistically significant up-regulation of X-DING-CD4 mRNA in cells from ECs compared with the other two study groups (Figure 1). For example, based on IQR the expression of X-DING-CD4 mRNA was 6.6- and 4.8-fold higher in the un-induced cells of ECs than in HIV progressors and HIV-negative population (Figure 1A); in cells treated with PHA expression of X-DING-CD4 was 16- and 31-fold higher (Figure 1B); and in cells exposed to HIV-1 it was elevated by 30- and 60-fold than viremic participants and negative controls (Figure 1C). The mock medium control for expression of this gene was 0.02–1.02 c/ng RNA for ECs, 0.1–0.9 c/ng RNA for HIV progressors and 0.06–0.15 c/ng RNA for HIV-1-negative controls. It is important to note that expression of X-DING-CD4 mRNA was similar in samples from HIV progressors who were receiving or not receiving ART (Supplementary Figure 1).
Evaluation of X-DING-CD4 mRNA expression in PBMCs from ECs, HIV progressors and HIV-negative participants. X-DING-CD4 mRNA levels were tested in the un-induced (A), PHA-treated (B) or HIV-1-infected (C) PBMCs. The IQR for each experimental system was calculated by SigmaPlot. Each data point for each individual donor is derived from three amplifications of the sample. Boxes indicate the range of the middle 50% of the data, with a horizontal line representing the median and vertical lines beyond the boxes indicating the upper and lower 25% of the data. Outliers are represented by closed circles. This figure summarizes results of three independent real-time PCR evaluations. The P-values were derived from the Mann–Whitney rank sum test; the alpha level was set at 0.050.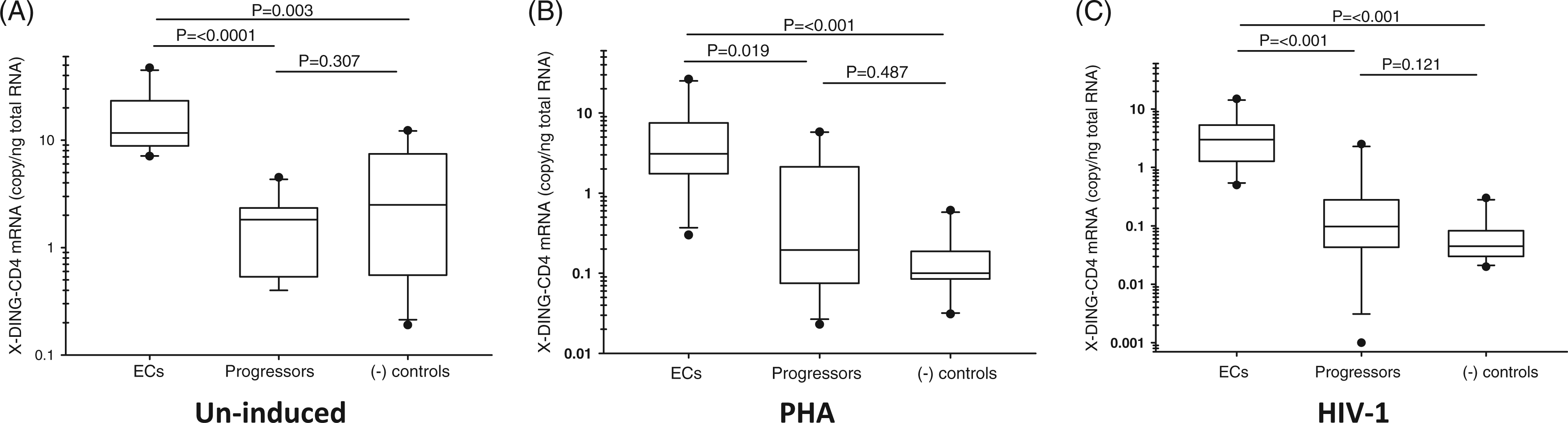
The activity of IFN-α mRNA was also enhanced in samples from ECs (Figure 2). In this regard, our data are in accord with the report by Machmach et al.,
23
which showed a higher concentration of this protein in plasmacytoid dendritic cells (pDCs) from ECs. In our study, the expression of IFN-α mRNA in quiescent cells was higher in ECs by 22- and 3-fold than in HIV progressors and negative controls respectively (Figure 2A). Mitogen stimulation increased the expression of IFN-α in ECs, but not in the other two study groups; IFN-α mRNA was higher by 47- and 40-fold in ECs than in viremic patients and negative controls (Figure 2B). In samples exposed to HIV-1 IFN-α mRNA was elevated by 30- and 14-fold compared with HIV progressors and negative controls (Figure 2C). The mock medium control for expression of this gene was 0.46–2.44 c/ng RNA for ECs, 0.02–0.35 c/ng RNA for HIV progressors and 0.021–0.47 c/ng RNA for HIV-1 negative controls. We noted some diversity in the expression of IFN-α in the PHA-treated cells between the HIV progressors receiving or not receiving ART. The ART(+) patients had higher level of IFN-α than the un-treated patients (Supplementary Figure 1); however, the HIV progressors as a group had lower activity of this gene than the ECs (Figure 2B).
Evaluation of IFN-α mRNA expression in PBMCs from ECs, HIV progressors and HIV-negative participants. The IFN-α mRNA levels were tested in the un-induced (A), PHA-treated (B) or HIV-1-infected (C) PBMCs. The statistical analysis is described in the legend to Figure 1. This figure summarizes results of two independent real-time PCR evaluations.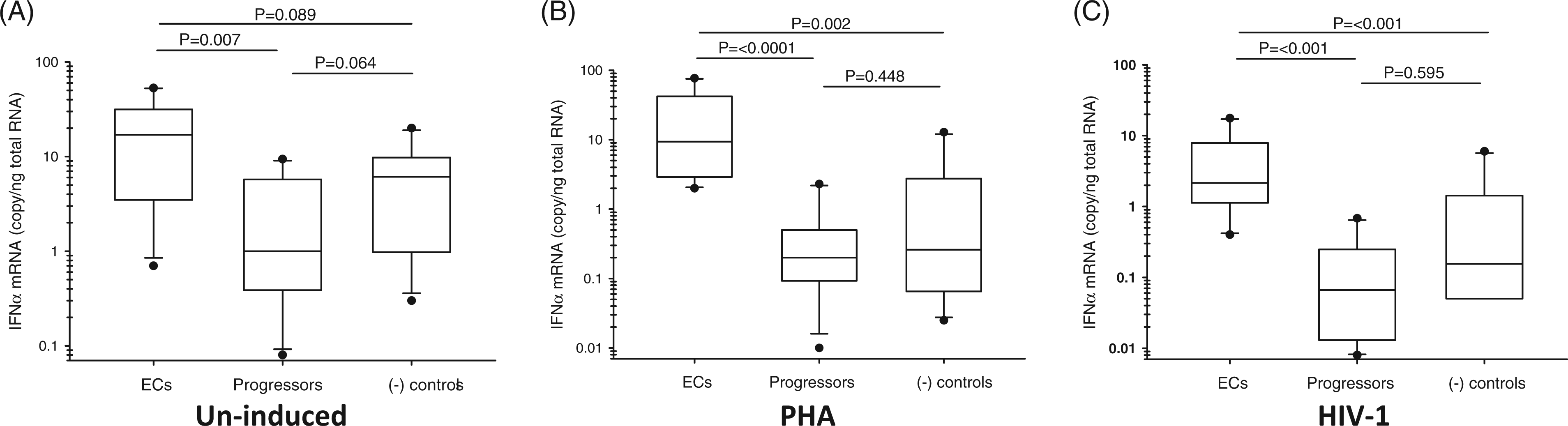
These observations confirmed our hypothesis that the ECs had much higher activity of innate immunity genes exemplified here by X-DING-CD4 and IFN-α than other two study groups; the expression pattern of these two genes was also similar to one observed previously for another HIV restriction factor—the APOBEC3G: ‘HIV-1 elite controllers > HIV(−) donors > HIV progressors. 22
The linear correlation for X-DING-CD4 and IFN-α mRNA expression in ECs depends on the type of cell stimulation
Based on our results both antiviral genes were significantly induced in PBMCs from ECs, but not from two other study groups indicating that (i) the enhanced expression of X-DING-CD4 and IFN-α in ECs could be controlled by independent signaling pathways initiated by mitogen or HIV-1 exposure, or (ii) one molecule controlled expression of another. To resolve this query we examined the linear dependence for the activity of both genes in this study group in relation to cell stimulation status. A strong correlation would indicate that expression of one gene could affect expression of another, while lack of correlation would indicate the independent regulatory mechanisms for both proteins.
The linear correlation for the expression of X-DING-CD4 and IFN-α mRNA in group of elite controllers. The correlation coefficient was calculated using GraphPad Prism version 5.00 for Windows.

The bold letters denote statistically relevant values.
We measured the expression of X-DING-CD4 and IFN-α mRNAs in a mixed cell population; therefore, it is reasonable to assume that the main source of the IFN-α in this pool was pDCs. Although expression of X-DING-CD4 and IFN-α mRNAs in cells exposed to virus did not correlate (Table 3), we considered the possibility that the increased concentration of IFN-α protein, and not HIV-1, influenced the activity of X-DING-CD4 mRNA. To solve this question we tested the sensitivity of the X-DING-CD4 gene to stimulation by extracellular IFN-α protein or HIV-1 in our T cell model of X-DING-CD4-mediated HIV-1 resistance.
38
These cells have many characteristics of HIV-1 infection seen in ECs: they are resistant to HIV-1 infection, restricting transcription of HIV-1 LTR promoter;
42
they constitutively express the X-DING-CD4 protein;29,30,38 and the expression of this gene might be enhanced rapidly by cellular exposure to HIV-1.
38
As shown in Figure 3 both IFN-α and HIV-1 induced the expression of X-DING-CD4 gene, but the sensitivity of this mRNA was greater in the system where cells were exposed to HIV-1. Control IFR-7 mRNA expression was up-regulated only in cells treated with the IFN-α (Figure 3, inset).
Testing the sensitivity of X-DING-CD4 mRNA to HIV-1 or IFN-α cell stimulation. X-DING-CD4 mRNA levels were tested in X-DING-CD4+ T cells exposed to HIV-1 or IFN-α. The untreated cells (Mock) were cultured in parallel without the stimulating factors. Inset: the efficacy of the IFN-α protein stimulation in X-DING-CD4+ cells was tested by amplification of IRF-7 mRNA.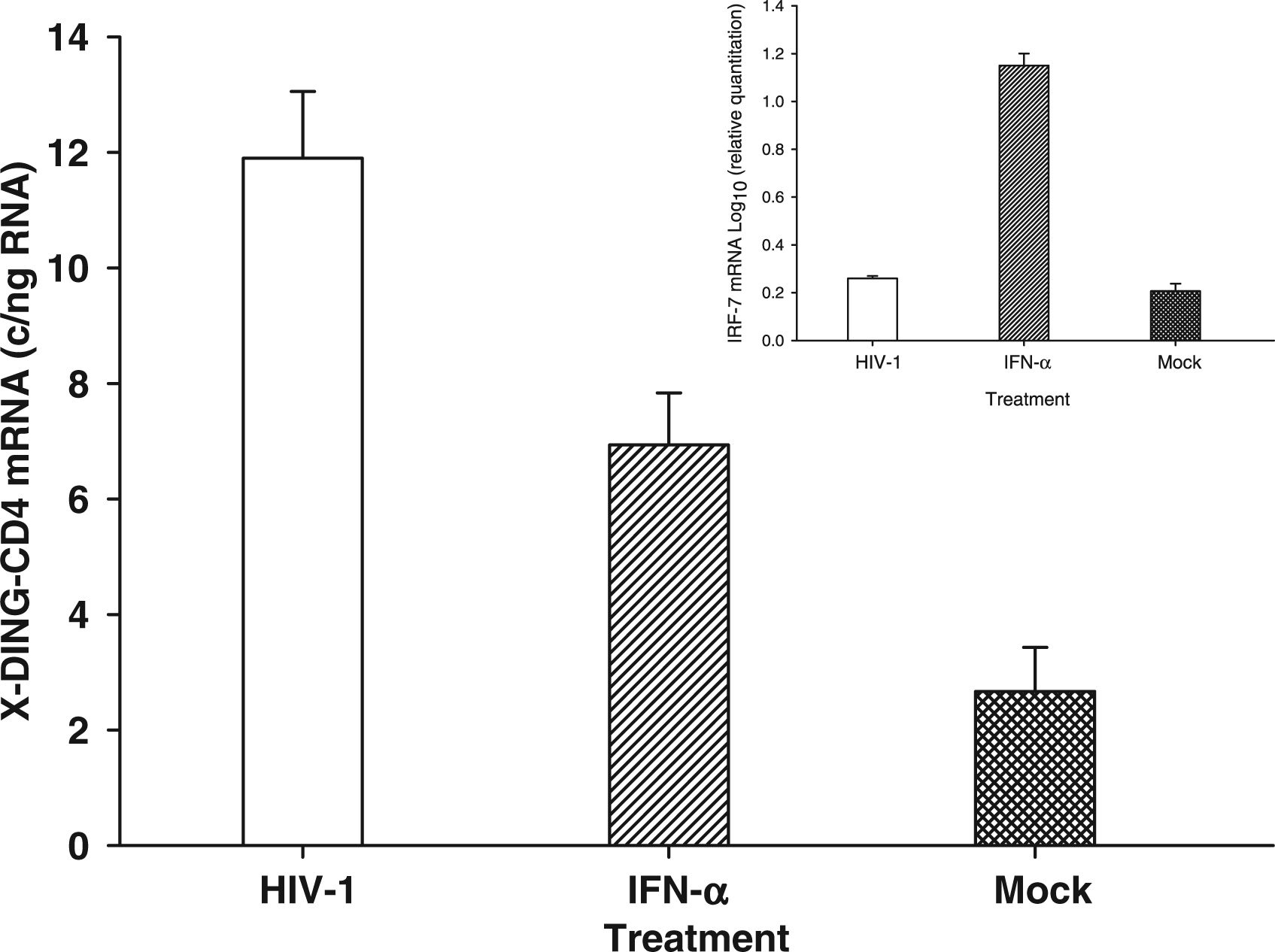
These data confirmed observations shown in Table 3, adding to our previous conclusion that, depending on the cell stimulating factor, the innate immune response might be regulated through diverse pathways distinguished by sensitivity to the IFN-α signaling.
Exposure of PBMCs to HIV-1 prompts interaction between X-DING-CD4, IFN-α and IL-8 mRNA leading to reduction of the pro-inflammatory response in ECs
We reported previously that the X-DING-CD4 protein blocked transcriptional activity of the IL-8 promoter in human macrophages and endothelial cells exposed to bacterial LPS, 33 and others have indicated that elevated levels of IFN-α in primary HIV-1 infection coincided with slow elevation of pro-inflammatory cytokines. 43
In agreement with these earlier reports we evaluated expression of IL-8 mRNA in our three study groups. As shown in Figure 4 (A, B) the expression of this gene in naive and PHA-treated cells was elevated in both HIV-1-infected samples compared with our uninfected control group. Opposite to this, when cells were exposed to HIV-1, the expression of IL-8 mRNA was significantly reduced in the group of ECs, and induced in HIV progressors and the HIV-1-negative control group (Figure 4C). We did not detect a significant difference in expression of IL-8 between ART(+) and (−) HIV progressors (Supplementary Figure 1).
Evaluation of the IL-8 mRNA expression in PBMCs from ECs, HIV progressors and HIV-negative participants. IL-8 mRNA levels were tested in the un-induced (A), PHA-treated (B) or HIV-1-infected (C) PBMCs. The statistical analysis is described in legend to Figure 1. This figure summarizes the results of two independent real-time PCR evaluations.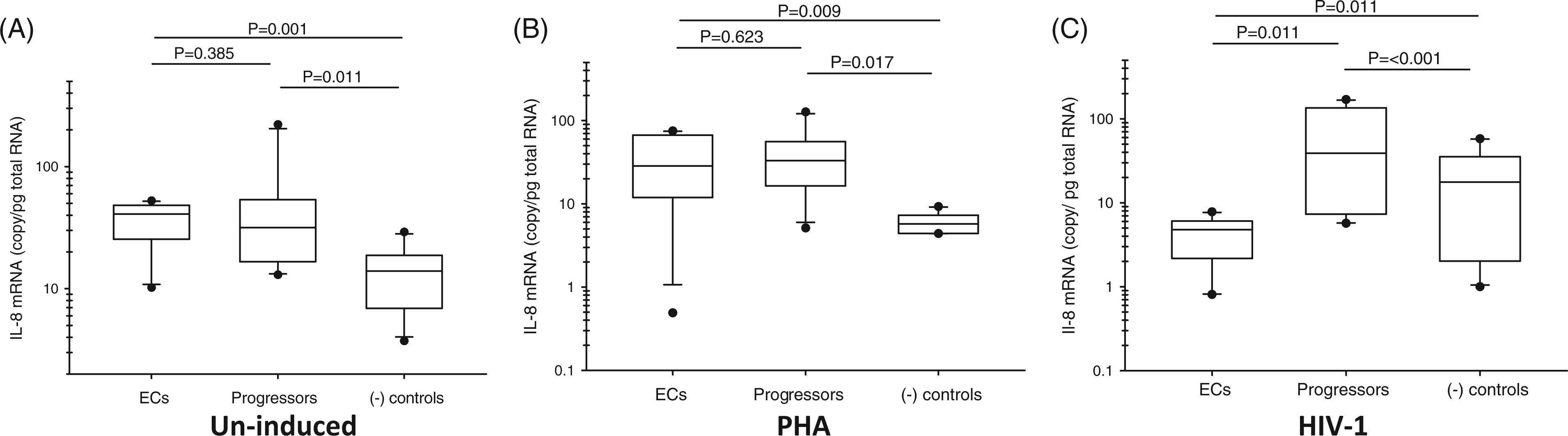
Based on this evidence we proposed that the reduction of the pro-inflammatory response in HIV-1 infected cells from ECs could be caused by interactions between activities of antiviral and pro-inflammatory genes, the type of cell stimulation and the patients’ ability to control the virus. To prove this hypothesis we performed a two-way ANOVA for three null hypotheses: (1) the expression of X-DING-CD4, IFN-α and IL-8 mRNA is equal in all samples; (2) all patients have the same proficiency to control HIV-1 infection; and (3) there is no interaction between expression of tested genes and the status of virus control defined as (i) elite HIV control, (ii) no HIV control or (iii) no HIV. The statistical significance for hypotheses 1 and 2 would mean that the expression of the tested genes is not equal and not all patients control the virus with the same efficacy, while significance for the interaction (hypothesis 3) would mean that the expression activity for all tested genes interacts with the type of virus control.
Results of the two-way ANOVA with respect to the relative expression of X-DING-CD4, IFN-α and IL-8 versus virus control, such as elite control, no control or no HIV infection. Two-way ANOVA for the un-induced, PHA-induced or HIV-1 exposed PBMCs was performed using GraphPad Prism version 5.00 for Windows.
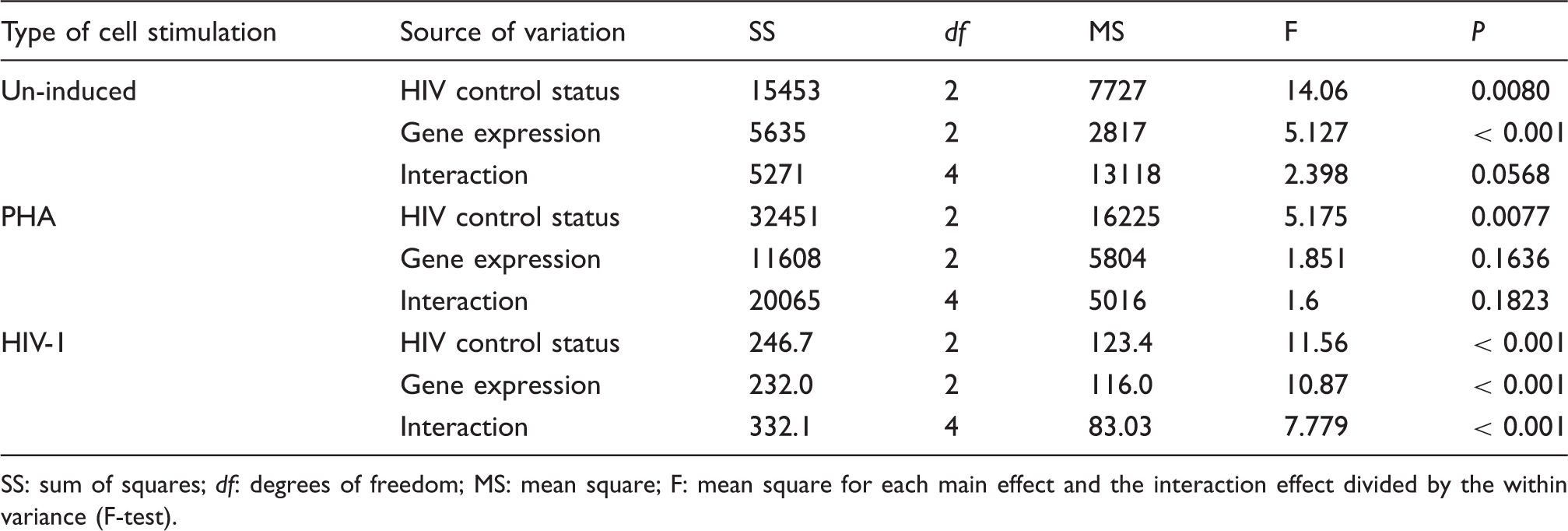
SS: sum of squares; df: degrees of freedom; MS: mean square; F: mean square for each main effect and the interaction effect divided by the within variance (F-test).
The outcome of our analysis was quite different in cells exposed to the exogenous HIV-1 (Table 4). There was a significant effect for all tested variables, and strong interaction between expression of these genes and the status of virus control (all P < 0.0001).
These data suggested that induction of X-DING-CD4 and IFN-α mRNAs, and a reduction of IL-8 in ECs or a reduction of X-DING-CD4 and IFN-α mRNAs and induction of IL-8 in HIV progressors in the presence of the exogenous virus, is an effect of a complex interplay between all tested genes and patients’ ability to control HIV infection.
Our experimental setting included PBMCs that had already been induced by the endogenous HIV-1 (such as ECs and HIV progressors) and cells from HIV-1-negative participants that were naive to such stimulation. This triggered the question of whether we could observe similar interactions between tested genes in cells that had not been pre-stimulated with endogenous HIV-1. To correlate the data presented in Figures 1, 2 and 4 to a sample that was naive to the previous HIV-1 stimulation we examined expression of X-DING-CD4, IFN-α and IL-8 mRNAs in PBMCs from our HIV-1-negative study group, which had been exposed to exogenous HIV-1 or to PHA. Our earlier investigation showed a significant negative correlation between expression of X-DING-CD4 and HIV-1 gag mRNAs in monocytes and macrophages from healthy HIV-1-negative donors, 38 and the induction of the X-DING-CD4 gene coincided with the blockage of HIV-1 LTR transcription. Based on these previous observations we looked first at the diversity in the expression of HIV-1 gag mRNA in PBMCs from healthy donors exposed to NL4-3 HIV-1.
One donor in this group had significantly lower activity of the gag gene than the remaining study participants (Figure 5A, HIV-1 gag). Based on this observation we cautiously designated this sample as HIV-1 ‘resistant’ (R), emphasizing that this donor was capable of suppressing replication of the virus within the 24 h window, and the remaining nine samples as HIV-1 ‘susceptible’ (S). Then we compared the expression of X-DING-CD4, IFN-α and IL-8 mRNAs in cells exposed to HIV-1 or PHA stimulation.
Comparison of X-DING-CD4, INF-α, IL-8 and HIV-1 gag mRNA levels in PBMCs from HIV-1 negative donors. (R) and (S) denote samples that had reduced (R) or not (S) levels of HIV-1 gag mRNA. Cells were exposed to HIV-1 (A) or PHA (B). The statistical analysis is described in legend to Figure 1. This figure summarizes the results of two independent real-time PCR evaluations.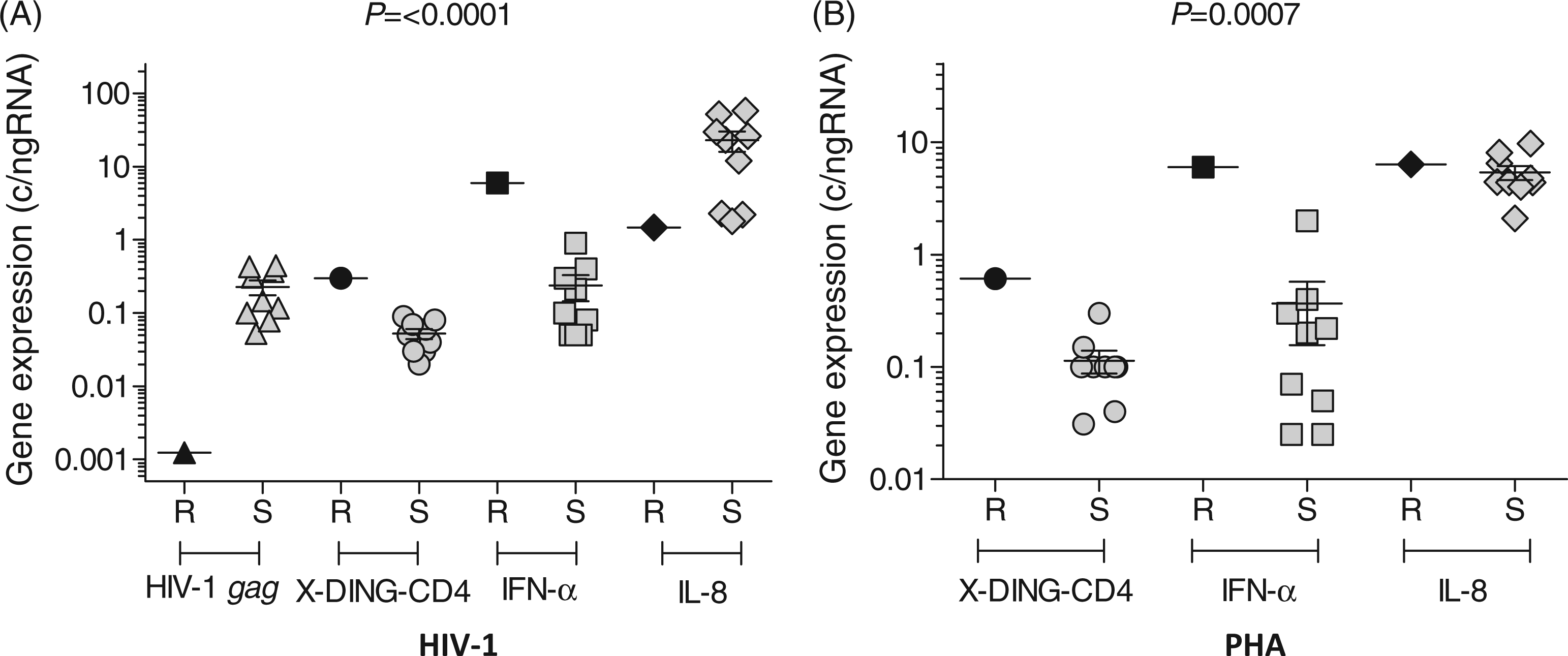
Upon exposure of cells to HIV-1 the expression of X-DING-CD4 and IFN-α was elevated, and the activity of IL-8 mRNA was reduced in the donor that restricted transcription of HIV-1 (Figure 5A). The lower levels of IL-8 mRNA could also be observed in three donors from the ‘susceptible’ group, but they did not restrict HIV-1 replication, and their expression of X-DING-CD4 and IFN-α mRNA was comparable with other ‘susceptible’ donor samples.
The expression of X-DING-CD4 and IFN-α was also enhanced in the ‘resistant’ donor when cells were stimulated by PHA (Figure 5B), but the activity of IL-8 mRNA was similar in both HIV-1 ‘resistant’ and ‘susceptible’ samples.
These data suggested that activation of X-DING-CD4, IFN-α and IL-8 mRNA follows a similar pattern in PBMCs from patients pre-stimulated with endogenous virus and those isolated from HIV-negative people. Therefore, the general profile of ‘resistant’ versus ‘susceptible’ response to HIV-1 infection can be distinguished early after virus exposure. The exceptional control of HIV-1 by ECs, however, is achieved by rapid, persisting and above average activity of the innate immunity genes (Figures 1, 2 and 4).
Discussion
The mechanism by which ECs restrict virus replication is still not fully understood. A number of studies delineated various characteristics and possible pathways through which the ECs could control viremia,3,6,10,11,20–24,44 and strong evidence points to a function of innate immunity molecules in this group of people.3,20,22,23
In this work we posed a question: What is the difference in the response of selected innate immunity genes between the ECs and HIV progressors allowing the first group a successful restriction of HIV-1 infection? Our control HIV-1-negative study group provided a background of these gene responses in an undifferentiated, uninfected population that could include both types of virus control. We looked at the expression of two genes (X-DING-CD4 and IFN-α) whose protein products have well defined HIV-1 blocking activity23,26–29,32,38 and pro-inflammatory IL-8 mRNA, which is induced in AIDS patients. 45
An earlier study indicated that the X-DING-CD4 mRNA was up-regulated in monocytes and macrophages from a fraction of the HIV-1-negative human population, and the higher activity of this gene coincided with restriction of HIV-1 when cells were challenged with the virus. 38 Based on this evidence we anticipated a considerably higher activity of the X-DING-CD4 gene in cells from ECs, who represent a positive selection for HIV-1 resistance, and our evaluations confirmed this hypothesis. The obvious interpretation of these data could be that the ECs have a healthier structure of their immune cells and a higher quantity of X-DING-CD4-expressing CD4 + T cells than AIDS patients. But to counter this view our HIV-negative controls also had healthy immune systems and equally high quantities of CD4 + T cells as the ECs; their cells, however, produce significantly less X-DING-CD4 or IFN-α mRNA, and, as we showed before, are highly susceptible to HIV-1 infection. 38 Consequently, not the quantity of cells expressing the X-DING-CD4 or IFN-α, but the efficacy of X-DING-CD4-mediated restriction of HIV-1 justifies the unique control of virus by ECs. The effect of X-DING-CD4-enhanced mRNA activity could be also seen in sample from one HIV-1-negative donor whose cells restricted replication of HIV-1 within the 24 h window.
Knowing that ECs express, in general, higher levels of several intrinsic molecules3,22,23 we explored additional scenarios for their host–pathogen response: (i) two anti-viral genes could interact under specific cell activation status and (ii) this interaction could restrict virus replication and cellular activation.
The response of two innate immune genes in PBMCs from ECs varied on mitogen stimulation or HIV-1 challenge. The expression of IFN-α and X-DING-CD4 genes was correlated only when cells were exposed to PHA and autonomous when cells were exposed to HIV-1. The functional analysis indicated that X-DING-CD4 mRNA could be up-regulated by either HIV-1 or IFN-α protein, but the sensitivity of this gene was at least twofold higher when cells were exposed to HIV-1. This could suggest a two-way activation of the innate immunity response: (i) the coordinated and, possibly generic, X-DING-CD4 and IFN-α response in ECs is sensitive to mitogen stimulation and (ii) specific un-coordinated anti-viral response can be induced only by pathogen invasion. The un-coordinated transcriptional activity of various host molecules in case of cell invasion provides a multifunctional inhibitory response that blocks diverse stages of the HIV-1 replicative cycle.
Diversified response to the extracellular stimuli might also mark the fundamental difference for the unmatched virus control by ECs. We propose that the ECs have high reactivity of the innate immunity genes, exemplified here by X-DING-CD4 and IFN-α, and reported by other groups.3,19,20,22–24 It is reasonable to believe that at the onset of pathogen invasion, these individuals have readily available host proteins that can block its replication. Subsequently, the extremely slow replication of the endogenous HIV-1 causes chronic X-DING-CD4 and IFN-α and possible other innate immune factor’s mRNA up-regulation, which, through their protein products, restrict back the replication of HIV-1. The enhanced expression of X-DING-CD4 and IFN-α in the un-stimulated PBMCs from ECs seems to support this proposition. This particular scenario could also explain the higher ability of ECs to control the HCV viremia. 46
The reduction of virus-induced pro-inflammatory molecules is much more favorable for cellular survival in the course of HIV-1 infection. We posited that the decrease of the pro-inflammatory response in ECs could stem from the interaction between the antiviral and pro-inflammatory genes, and the status of virus control only in the presence of the exogenous HIV-1. In this way the elevated expression of the antiviral genes and reduced pro-inflammatory response could be seen only in ECs, and the reduced expression of antiviral genes and elevated expression of pro-inflammatory mRNA could be observed in HIV progressors. Our data gave ground to the conclusion that exposure to HIV-1 caused an innate immunity response in ECs resulting in the inhibition of virus replication, but also prevention of virus-mediated activation of the pro-inflammatory response.
Antiviral activity was also detected in other DING variants derived from human and plant cells; these proteins blocked transcription of HIV-1 LTR with efficiency comparable to X-DING-CD4.34,47,48 While much of their biology is still unknown, this might suggest that DING proteins participate in the innate immunity responses to pathogens in diverse species. Interestingly, the serum levels of the HPBP DING homologue were elevated in AIDS patients, 34 which is contrary to our previous findings 38 and the research presented herein. One explanation of this discrepancy could be the association of HPBP with human paraoxonase-1, 49 which renders the protein inactive against HIV-1. 47
Footnotes
Funding
This work was supported by NS062649 grant from the National Institutes of Health. This publication resulted (in part) from research supported by the International HIV Controllers Study, the Bill and Melinda Gates Foundation and the Harvard University Center for AIDS Research (CFAR), an NIH funded program (P30 AI060354), which is supported by the following NIH co-funding and Participating Institutes and Centers: NIAID, NCI, NICHD, NHLBI, NIDA, NIMH, NIA, FIC and OAR.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
