Abstract
Clinical reports indicate that some infected individuals control HIV-1 replication through undefined mechanisms. Our group reported that a human protein named X-DING-CD4 holds a potent antiviral activity, blocking transcription of HIV-1 LTR through the inhibition of NF-κB/DNA binding. Based on observations that transformed HIV-1 resistant CD4+ T cells produce higher levels of soluble X-DING-CD4 protein upon their exposure to virus, we hypothesized that resistance to HIV-1 in these cells may be regulated through function of the X-DING-CD4 gene. Real-time PCR evaluations of X-DING-CD4 mRNA expression confirmed our hypothesis; HIV-1 exposure caused rapid up-regulation of X-DING-CD4 mRNA in resistant, but not susceptible, cells; and the burst of X-DING-CD4 mRNA expression correlated with restriction of HIV-1 transcription. Subsequently, we examined the activity of the X-DING-CD4 gene in monocytes and macrophages from (n = 13) HIV-negative donors. The assessment of HIV-1 gag mRNA showed that the majority of cells were permissive to virus replication; however, macrophages from four donors were refractory to HIV-1 infection. In response to virus, these cells up-regulated X-DING-CD4 gene expression by 2- to 1000-fold. These data provide evidence that the X-DING-CD4 gene contributes to early cellular protection from HIV infection in some individuals and this protection depends solely on the unique genetic regulation of the host.
Introduction
The phenomenon of natural resistance to HIV has challenged researchers for many years now. In time, it has become clear that the long time non-progressors (LTNPs) and particularly the subset of this group—the elite controllers (ECs)—effectively suppress virus replication through an undefined mechanism(s). Despite being infected with the replication competent virus, 1 – 3 the ECs maintain stable CD4+ T cell counts and are frequently naïve to antiviral treatments.4,5 The HIV target cells from ECs are susceptible to virus entry and productive infection, 6 but gene expression signatures, although indistinguishable in antiretroviral therapy (ART)-treated patients, are significantly different from those in HIV-negative individuals. 7 This indicates that resistance to HIV in ECs is not related to cell physiology or defects in the infecting virus, but to genetic regulation resulting in intrinsic control of the pathogen.
We recently reported the isolation and identificationof a novel human protein, the X-DING-CD4 (e
To evaluate this proposition we employed a quantitative real-time PCR (qRT-PCR) for analysis of X-DING-CD4 gene activity in transformed CD4+T cells and primary human monocytes and macrophages exposed, or not, to HIV-1. The goal of this study was to observe the correlation between X-DING-CD4 gene activity and restriction of HIV-1 transcription. First, we employed our HIV-1- resistant T cell-model to measure kinetics of X-DING-CD4 mRNA expression and to establish experimental parameters for subsequent analysis of this gene expression in primary human cells. Then, we evaluated the expression of the X-DING-CD4 gene in naïve monocytes and macrophages derived from 13 HIV-negative individuals; we established an interquartile range (IQR) for X-DING-CD4 mRNA expression in our donor cells in the absence and presence of virus exposure. Finally, we correlated X-DING-CD4 activity in the virus-exposed human macrophages to HIV-1 gag mRNA transcription rate at the early stage of infection.
Materials and methods
Cell cultures, viruses and reagents
HIV-1-resistant cells [here called X-DING-CD4(+)] have been previously described by the name HRF(+); 9 – 15 these cells express and secrete the X-DING-CD4 protein upon their exposure to HIV-1. 8 The control HIV-1-susceptible cells used for this research were CEM, 16 obtained from the American Type Culture Collection (Rockville, MD, USA). CEM cells tested negative for the production of the soluble X-DING-CD4 protein (not shown) and, based on this evidence, we annotated this control cell system as X-DING-CD4(-). The continuous cultures of X-DING-CD4(+) or control X-DING-CD4(-) CEM cells were maintained in RPMI 1640 (Sigma, St Louis, MO, USA) supplemented with 5%/vol FBS, antibiotics and glutamine at 37°C in a 5% CO2 95% air-humidified incubator.
Human monocytes from (n = 13) healthy, HIV-1-negative volunteers were obtained by elutriation from whole blood; the detailed culture conditions are described in the next section. The studies reported here, using human peripheral blood monocytes, weregranted exempt status by the Institutional ReviewBoard under qualifications listed in section 45.101(b) (4).
Testing the expression of X-DING-CD4 and HIV-1 mRNAs in transformed and primary cells
For X-DING-CD4, HIV-1 vif and gag mRNA, or infectivity studies, the X-DING-CD4(+) and control X-DING-CD4(-) cells were exposed to NL4-3 HIV-1 isolate, 17 while the primary human macrophages were infected with R5 HIV-1-ADA 18 or NL4-3 HIV-1, all at multiplicity of infection (MOI) of 0.01. After 1 h incubation at 37°C, virus was removed by washing in ice cold PBS. Infected cells were cultured in the respective media as described above and below and, at the designated time-points, cells were harvested for RNA isolation and evaluation of viral proteins by immunofluorescence (IF) and ELISA. The IF staining of HIV-1-infected transformed cells was performed with pooled serum from AIDS patients (gifted from Dr Volsky) followed by goat anti-human FITC antibody (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA). The concentration of HIV-1 p24 core antigen in supernatants of HIV-1-infected cells was established by ELISA (Perkin Elmer, Waltham, MA, USA).
For testing the expression of X-DING-CD4 mRNA in primary cells, 5 × 106 human monocytes were selected upon arrival to the laboratory, pelleted by centrifugation and suspended in Trizol reagent (Invitrogen, Carlsbad, CA, USA) for RNA isolation. This sample represented the mitogen- and HIV-1-naïve monocytes. The remaining monocytes were allowed to differentiate into macrophages as described previously. 13 Briefly, cells were seeded into six-well plates (Corning Scientific, Santa Clara, CA, USA) at a concentration of 5 × 106/well in DMEM (Sigma) supplemented with 10%/vol endotoxin-free, heat-inactivated human serum (Cambrex, Rutherford, NJ, USA), 10%/vol giant cell tumor conditioned medium (BioVertis, Vienna, Austria), 2 mM glutamine and antibiotics. Cells were cultured for 5–7 d until fully adherent. Thereafter they were cultured in DMEM with 10%/vol endotoxin-free FBS, glutamine and antibiotics. Based on previous examinations of gene expression profiles in human macrophages cultured under the same conditions, 19 we are confident that cells were fully differentiated. After a change of medium, cells were cultured for another 24 h and then exposed, or not, to HIV-1. Samples for RNA analysis were collected in 24 h intervals; the uninfected macrophages cultured along the experimental time-line were collected at the respective time-points to observe background X-DING-CD4 mRNA expression.
Primers and probes used for qRT-PCR analysis and cDNA synthesis.

Quantitative real-time PCR (qRT-PCR) analysis
The Q-PCR primers and probes were designed based on the X-DING-CD4 sequence [GenBank (HQ586056)] and HIV-1 NL4-3 gag and single spliced vif mRNA sequence; all primers were synthesized by Invitrogen; probes were synthesized by Applied Biosystems (Foster City, CA, USA), and all sequences are listed in Table 1. The real-time detection of each tested mRNA expression was performed through the absolutequantification protocol using TaqMan Gene Expression Assay on 7300 real-time PCR System (Applied Biosystems). Briefly, triplicates of total reaction mixture of 25 μl were composed of 2 μl of cDNA, 12.5 μl of 2 × ABI TaqMan PCR Master Mix, 0.25 μl (90 μM) of both sense and antisense primers, and 0.5 μl (10 μM) of the probe. After the initial denaturation at 95°C (10 min), target genes were amplified through 40 cycles of universal cycling conditions (95°C/10 s, 60°C/1 min). All PCR reactions were normalized to GAPDH or β-actin mRNA in transformed and primary cells respectively. The GAPDH and β-actin target genes were amplified using a predesigned pair of primers and a probe in concentrations suggested by the manufacturer (Applied Biosystems). Standard cell dilution curves were calibrated using serial dilutions of control pTA_X-DING-CD4, pTA_gag or pTA_vif plasmids representing 1 × 106, 1 × 105, 1 × 104, 1 × 103, 1 × 102 and 10 copies of X-DING-CD4 cDNA per reaction. All values are expressed as means ± standard deviation of the mean.
Depending on the experimental setting, the results of qRT-PCR analyses are presented as the X-DING-CD4 mRNA fold change, the X-DING-CD4 mRNA copy/ng total RNA or the IQR. The fold change was calculated as a fold of the X-DING-CD4 mRNA increase or decrease between the experimental sample and the uninfected control; the IQR for X-DING-CD4 mRNA expression was calculated by Sigma Plot 11 (Systat Software Inc., Chicago, IL, USA) for all 13 samples measured at the specific time-points and represent copy of X-DING-CD4 mRNA/ng total RNA; the correlation between the expression of X-DING-CD4 and HIV-1 gag mRNAs was analyzed by GraphPad Prism5 (GraphPad Software Inc., La Jolla, CA, USA).
Results
X-DING-CD4 mRNA is rapidly up-regulated in HIV-1-resistant X-DING-CD4(+) cells and the enhanced expression of the X-DING-CD4 gene coincides with inhibition of HIV-1 transcription
We previously reported that X-DING-CD4-mediated restrictive activity in HIV-1-resistant X-DING-CD4(+) cells [previously called HRF(+)] 10 did not affect virus cell entry or reverse transcription but blocked HIV-1 at the stage of LTR transcription.10,11,13 This data suggested that X-DING-CD4-mediated antiviral protection was induced shortly after cell exposure to HIV-1 and could be previewed by changes in the activity of the X-DING-CD4 gene. To test this hypothesis, we analyzed levels of X-DING-CD4 transcripts in HIV-1-resistant and control HIV-1-susceptible cells exposed to a low virus dose.
Cells were infected with HIV-1 at MOI of 0.01 and total RNA was harvested at short time intervals. The activity of X-DING-CD4 mRNA was evaluated by the qRT-PCR and data from respective experimental systems were compared with those obtained from respective uninfected controls to gauge the variations in gene expression. As shown in Figure 1, transcription of X-DING-CD4 mRNA in HIV-resistant cells increased by 1.8–4-fold within the first 30 min of HIV-1 exposure; expression of this gene in the control HIV-1-susceptible cells increased by 1.3-fold at the 30-min time-point and was then suppressed by 0.6-fold at the 6-h time-point. Twenty-four hours after cell exposure to virus, the expression of the X-DING-CD4 gene further increased by 11–23-fold in HIV-1 resistant cells and was suppressed 0.7-fold in HIV susceptible cells. At 48 and 72 h after infection, the expression of this gene was still induced 2.5–5 and 4–7-fold in HIV-resistant cells and was unchanged in HIV susceptible cells.
Expression of X-DING-CD4 mRNA is induced in HIV-1 resistant cells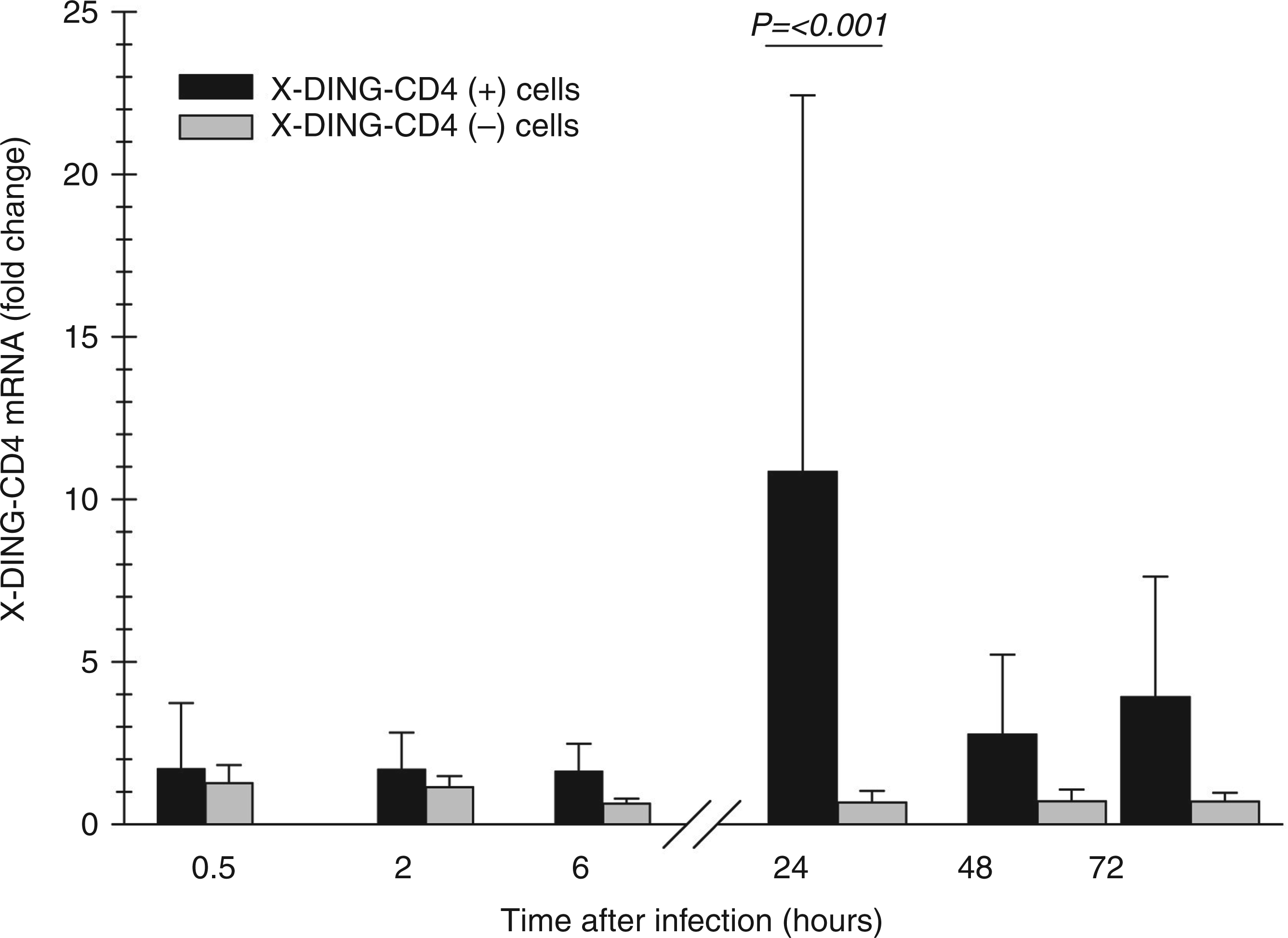
Subsequently, we used the same experimental setting and evaluated the expression of X-DING-CD4 and single-spliced HIV-1 vif transcripts during 10 d of infection. As previously observed, 24 h after exposure to virus, expression of X-DING-CD4 mRNA was induced in HIV-1-resistant cells (Figure 2A, bars, left axis) and suppressed in HIV-1-susceptible cells (Figure 2B, bars, left axis). At the subsequent time-points, expression of the X-DING-CD4 gene normalized to range between 60–80 copies/ng total RNA in HIV-1-resistant cells and 50–80 copies/ng total RNA in HIV-susceptible cells (Figure 2B, bars, left axis).
The elevated expression of X-DING-CD4 mRNA associates with inhibition of HIV-1 vif gene transcription. X-DING-CD4 and HIV-1 vif mRNA expression was tested by qRT-PCR in (A) HIV-1-resistant [X-DING-CD4(+)] and (B) HIV-1-susceptible cell lines. Levels of X-DING-CD4 mRNA (bars) are calculated on the left y axis; levels of HIV-1 vif mRNA (lines) are calculated on the right y axis. Expression of HIV-1 proteins was tested by immunofluorescence (IF). This figure summarizes the results of three independent experiments.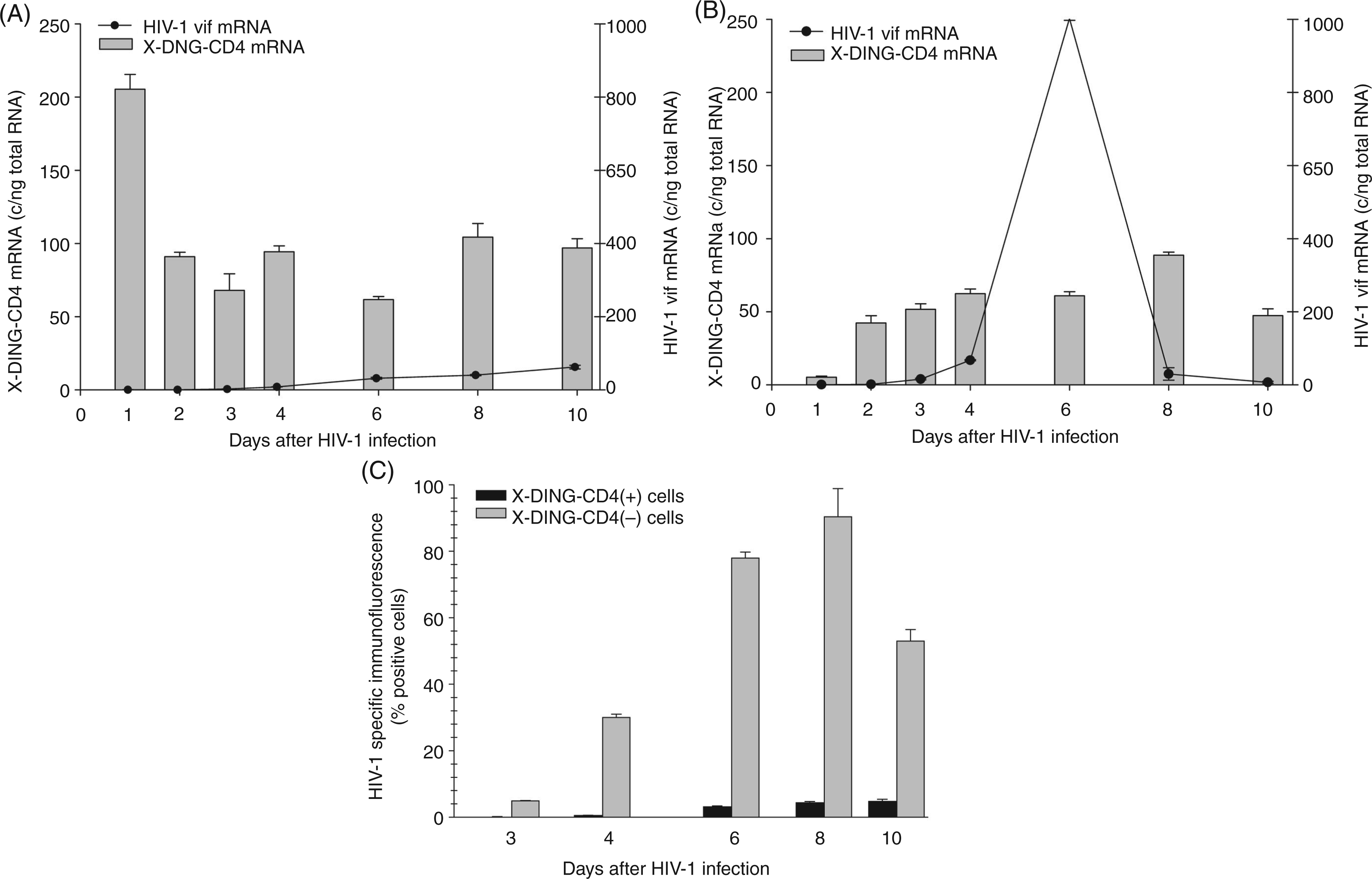
vif mRNA was undetectable in HIV-1-resistant cells for up to 3 d after infection and, at the 6 d time-point, was still blocked by 97% (Figure 2A, line, right axis). Opposite to this, the control HIV-1-susceptible cells supported virus replication; the vif transcripts could be detected as early as 1 d after HIV-1 exposure; transcription of this gene peaked at day six (Figure 2B, line, right axis). The qRT-PCR data were confirmed by evaluation of HIV-1 proteins by IF (Figure 2C) and ELISA (not shown). Viral proteins could be detected as early as 3 d after infection in the control HIV-1-susceptible cellsand maximum viral infectivity was observed 8 d after infection. Concurrent with the qRT-PCR data, the X-DING-CD4(+) HIV-1-resistant cells did not support virus replication. Viral proteins were not detected until 6 d after infection when the IF value reached 3%. At the following 8- and 10-d time-points, IF was measured at 5%.
Over-expression of the X-DING-CD4 gene in HIV-infected macrophages correlates with the inhibition of HIV-1 transcription
Based on the evidence presented in Figure 2, we hypothesized that the X-DING-CD4 gene-mediated antiviral activity may be effective in the human population and lend an explanation as to why some individuals are capable of controlling HIV replication despite being infected with the replication competent virus. To evaluate this proposition, we tested the activity of the X-DING-CD4 gene and transcription of HIV-1 gag mRNA in macrophages from 13 HIV-1-negative individuals.
First, we collected the mitogen- and HIV-1-naïve monocytes to obtain the X-DING-CD4 mRNA expression background. Then, we tested the expression of the X-DING-CD4 gene in respective macrophages exposed, or not, to HIV-1. We measured the expression of HIV-1 gag and X-DING-CD4 mRNA in 24-h intervals for the first 3 d after infection.
To exclude natural resistance to HIV infection through a genetic defect in the CCR5 receptor, we used the X4 NL4-3 HIV-1 strain. Prior evaluations on a smaller, randomly-selected sample showed that infections with R5 ADA and X4 NL4-3 HIV-1 induced similar levels of X-DING-CD4 mRNA in human macrophages and produced a similar quantity of HIV-1 gag mRNA transcripts during the first 3 d after infection (not shown).
To observe variations in X-DING-CD4 mRNA, the qRT-PCR data were subjected to IQR analysis (Figure 3A). Half of the sample readings for mitogen-naïve monocytes were between 1.3 and 9.76 X-DING-CD4 mRNA copies/ng total RNA; the outlier sample reached the expression of this gene at 27 copies/ng total RNA and the median value for all monocytes was 4.5 X-DING-CD4 mRNA copy/ng total RNA (Figure 3A—monocytes).
Evaluation of the X-DING-CD4 and HIV-1 gag mRNA expression in human monocytes and monocyte-derived macrophages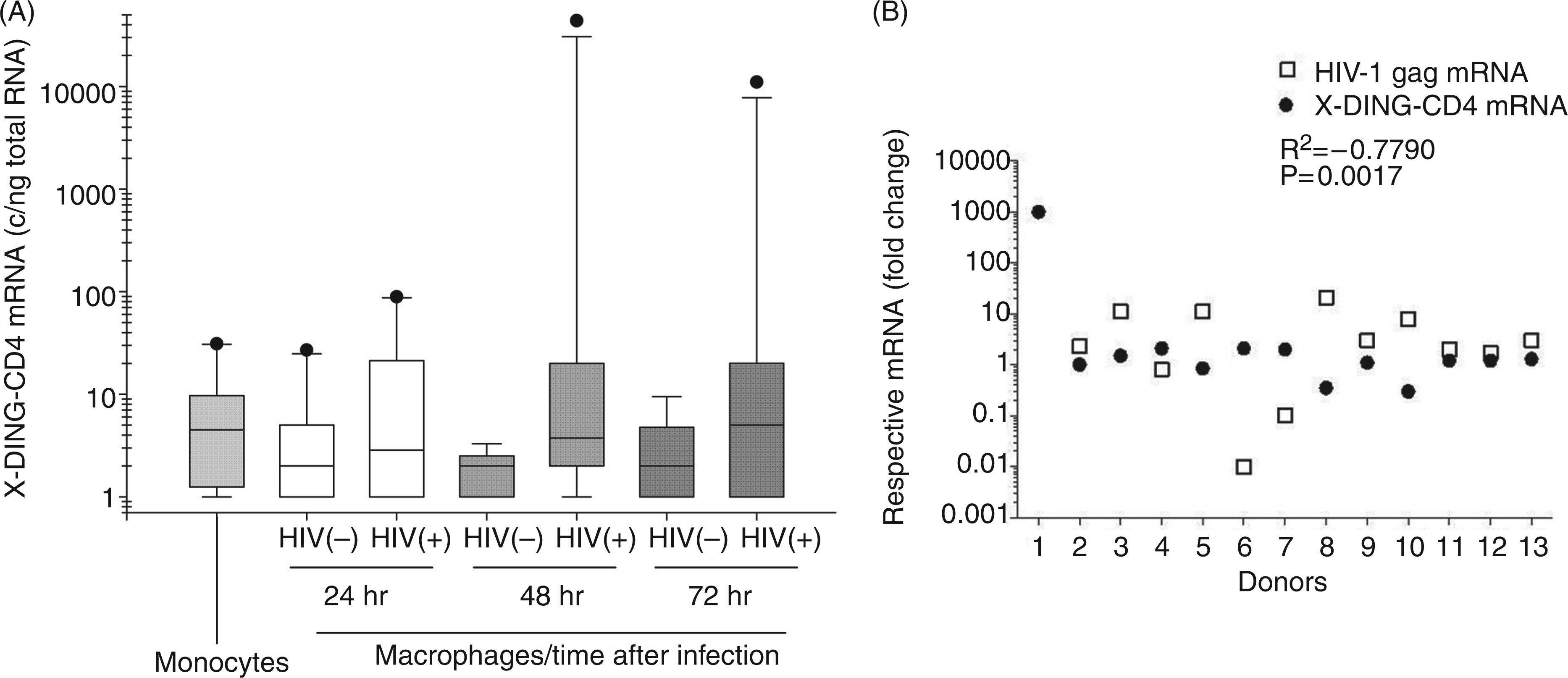
The exposure of monocytes to mitogens and their differentiation to macrophages resulted in a modest 1.9-fold decrease of X-DING-CD4 expression; half of the sample readings ranged from 1–4.9 copies/ng total RNA; however, similarly to the monocyte group, the outlier samples reached 27 copies/ng total RNA [Figure 3A—HIV(−) macrophages 24 h].
HIV-1 exposure induced the expression of X-DING-CD4 mRNA at all time-points tested (Figure 3A HIV(+)). At the 24-h time-point, half of the sample readings placed in the range of 1–21 copies/ng total RNA and the outlier samples reached 87 copies/ng total RNA. At the 48 h time-point, the expression ofthis gene ranged from 2–19.4 copies/ng total RNA in the infected macrophages and 1–3.3 copies/ng total RNA in the control cells tested alongside them. Interestingly, the expression of X-DING-CD4 in the outlier samples reached 45,143 copies/ng total RNA in cells from donor 1. At the 72-h time-point, the range of X-DING-CD4 expression in the uninfected sample was comparable to that tested at the 24-h time-point, expanding from 1–4.7 copies/ng total RNA; the expression of the gene in 50% of the infected samples ranged between 1–20 copies/ng total RNA and expression in the outlier sample from donor 1 was reduced to 11,545 copies/ng total RNA.
Next, we evaluated the correlation between the expression of X-DING-CD4 and HIV-1 gag mRNAs in the infected macrophages. Based on X-DING-CD4 mRNA expression kinetics in transformed and primary cells (Figures 2 and 3A), and the assumption that activation of the X-DING-CD4 gene should precede the blockage of HIV-1 LTR, we selected the qRT-PCR data from the 24- and 48-h time-points for the X-DING-CD4 gene and the 48- and 72-h time-points for HIV-1 gag mRNA. We established the numerical value representing a fold change in X-DING-CD4 and HIV-1 gag mRNA transcripts for the selected time-points and calculated the correlation between the expressions of both genes tested.
This analysis showed a significant negative correlation between the expression of X-DING-CD4 and HIV-1 gag mRNAs (R2 = −0.7790); the induction of X-DING-CD4 gene coincided with the blockage of HIV-1 LTR transcription (Figure 3B). For example, macrophages that did not alter expression of the X-DING-CD4 gene by more than twofold supported replication of HIV-1. Donors 1, 4, 6 and 7, whose cells had increased levels of X-DING-CD4 mRNA, restricted the expression of HIV-1 gag mRNA. In particular, cells from donor 1, who had very high X-DING-CD4 mRNA activity, restricted replication of the virus from 9 copies at the 48-h time-point to an undetectable level 3 d after infection (Figure 3B donor 1). Macrophages from donors 4, 6 and 7 had a modest 2–2.5-fold increase of X-DING-CD4 mRNA activity and also restricted the replication of HIV-1, but with a dissimilar efficiency. For example, cells from donor 4 blocked the replication of the HIV-1 gag gene by 1.3-fold, while cells from donors 6 and 7 blocked transcription of this viral gene by 122- and 12-fold, respectively (Figure 3B).
Discussion
The identity of eukaryotic DING genes is controversial 21 and, although fragments of cDNA were found in diverse organisms, lack of their chromosomal assignment has puzzled scientists for years. It has been suggested that DING proteins arise from bacterial species and do not exist in eukaryotes. 22 We, and others, have provided evidence that the eukaryotic DING proteins are coded by plant or animal genomes and exert specific biological functions in the host cells.20,23,24
The antiviral activity of the X-DING-CD4 protein has been reported before8,9,11 and, in this study, we tested the hypothesis that the activity of the X-DING-CD4 gene may be linked to the cellular restriction of HIV-1 transcription in human cells. In these studies, we showed that exposure of cells to HIV-1 altered the expression of the X-DING-CD4 gene. It was expected in our X-DING-CD4(+) cells,which are refractory to HIV-1 infection. We previously observed the enhanced activity of the X-DING-CD4 protein upon their exposure to HIV-1. 13 Interestingly, we found a similar restriction of HIV-1 promoter activity in primary macrophages of four donors whose cells also reacted to virus exposure with an enhanced transcription of the X-DING-CD4 gene.
The induction of the X-DING-CD4 gene always correlated to the inhibition of HIV transcripts in transformed cells, but it was not so obvious in primary macrophages. Although four donors had elevated levels of the X-DING-CD4 gene, only one restricted transcription of HIV-1 to an undetectable level. The other three donors had a modest 2–2.5-fold increase in expression of the X-DING-CD4 mRNA and, although all had diminished levels of gag mRNA, there was a notable unevenness in the LTR restriction level; study in a larger sample and future evaluations will clarify the current observations. However, based on the data in hand, we believe that the protective status of X-DING-CD4 can be achieved in cells that produced a very high concentration of protein preceded by high activation of its gene. The sensitivity of the X-DING-CD4 gene to cellular exposure to HIV-1 may be a part of the unique host mechanism regulating cellular resistance to virus—we showed that some individuals who have the inducible X-DING-CD4 gene responses also have a higher capacity to restrict HIV-1.
Our data showed that all tested cells had a transcriptionally active X-DING-CD4 gene. In this sense, all cells should have the capacity to restrict virus replication; however, this is not what we observed. We consider that X-DING-CD4 has a dual function in human cells: physiological—as a phosphatase and antiviral—and as HIV-1 LTR transcription inhibitor. Based on our previous reports, we hypothesize that the antiviral function of X-DING-CD4 involves a rare methylation of glutamic acid 69 present in about 10% of its molecules; 8 we postulate that target cells from some individuals, in the event of virus infection, induce the expression of X-DING-CD4 mRNA followed by methylation of X-DING-CD4 protein. This modification facilitates a ‘switch’ into X-DING-CD4 protective mode and further stimulates the induction of the X-DING-CD4 gene. Considering that methylation of glutamic acid 69 is a leading key for a switch of X-DING-CD4 activity from physiological to protective, we hypothesize that a larger concentration of a substrate for this modification warrants a more successful outcome.
We observed two opposite trends in the expression of X-DING-CD4 mRNA when transformed CD4+ T cells were exposed to HIV-1: (i) rapid induction; or (ii)rapid suppression. The induction of X-DING-CD4 activity occurred in cells that resisted HIV-1 infection, while reduced activity of this gene was seen in cells susceptible to virus. The response of the X-DING-CD4 gene was more diverse in human macrophages. This observation brings forth a question if one of the HIV-1 evasive techniques also includes a mechanism that down-regulates the expression of X-DING-CD4. This concept is true for other intrinsic proteins, such APOBEC3g—deactivated by HIV-1 Vif protein 25 —and Daxx, neutralized by CMV pp71 protein. 26
The mobilization of the X-DING-CD4 gene upon virus exposure was rapid in primary and transformed cells, which is a characteristic trait of the intrinsic response to pathogen invasion; however, X-DING-CD4 activity peaked at 24 h in infected, CD4+-transformed cells; while in primary macrophages, activity peaked 48 h after infection. This difference in the responsiveness of X-DING-CD4 mRNA to virus invasion in these two cell systems could be related to the clonal selection of transformed X-DING-CD4 (+) cells.
The phenomenon of restricting HIV-1 infection by host target cells is an exciting avenue for novel therapeutic interventions. The introduction of native X-DING-CD4 protein into HIV-1-susceptible, infected CD4+ T cell-culture, blocks infection of HIV-1 as effectively as the endogenous protein expressed in the X-DING-CD4(+) cell system.8,11 Moreover, another DING homologue, p27SJ isolated from Hyperforicum perforatum, was found to have an anti-HIV activity 24 and phosphatase activity. 23 The phosphatase activity of the p27SJ plant DING protein was associated with its antiviral activity. Future research on the X-DING-CD4 mechanism will show if its phosphatase activity is also indispensable for blocking HIV-1 LTR transcription. This highly conserved antiviral activity in two distinct members of the DING protein family makes a human X-DING-CD4 an attractive target for the development of a novel biotherapeutic.
It is reasonable to believe that the overall inhibition of HIV-1 in cells of the LTNPs and, particularly in ECs, may be regulated by a complex cellular immunity involving multiple proteins. Our results show that the X-DING-CD4 mechanism is a part of the complex cellular activities against the pathogen invasion. Some of the intrinsic molecules with an anti-viral activity have been already identified,25,27 and it will be interesting to gauge their expression levels in cells of LTNPs, AIDS patients and the healthy HIV-1-negative population.
Footnotes
Funding
This work was supported by NS062649 grant from the National Institutes of Health.
Acknowledgements
We thank Dr Potash for providing human monocytes for these studies.
