Abstract
Onsets of bacterial infections devastate the compromised immune system in AIDS patients. Damaged gut mucosa permits dissemination of bacterial toxins into deeper layers and hyper-activation of the immune system. We previously reported that the unfractionated supernatants of HIV-resistant CD4+ T cells impeded the NF-κB/DNA binding in macrophages induced by either HIV-1 or LPS. The active component of this soluble material was identified as X-DING-CD4 (e
Introduction
Chronic diarrhea associated with progressive HIV disease may be caused by common bacterial, parasitic or opportunistic infections. The bacterial species Salmonella, Shigella and Campylobacter are the most frequent causative agents of diarrhea in the AIDS patients. 1 – 4 In the absence of immune protection, these pathogens may disseminate from the gut and cause severe systemic infections. Some AIDS patients present to emergency rooms with the onset of diarrheal disease of undetermined etiology where the pathogen cannot be identified. 5 Severe deregulation of intestinal cytokines may contribute to this condition. 6
HIV disease is marked by high levels of inflammatory molecules secreted by blood monocytes 7 and increased NF-κB activation in T cells is a driving factor for HIV transcription. During bacterial infection of the gut, the intestinal lining is rapidly activated through enhanced secretion of adhesion molecules by endothelial cells. 8 – 10 Subsequently, the IL-8, IL-6 and MCP-1 chemokines secreted by the activated endothelium cause influx of leukocytes to the gut wall. 10 In response to these homing signals, recruited immune cells release pro-inflammatory molecules, such as TNF-α and IL-1β which, in turn, further evoke the up-regulation of the endothelium and surrounding tissues. In this view, productive HIV infection is enhanced by the inflammatory environment. The interruption of these inflammatory events could have a favorable effect in AIDS patients; (i) blocking the propagation of HIVand (ii) dissemination of bacterial antigens from the gut.
Recently, we reported the identification of a novel human protein, X-DING-CD4 [e
In our previous study, we showed that levels of LPS activated TNF-α protein were reduced in human macrophages treated with the X-DING-CD4 protein. 14 This particular insensitivity of cells to LPS stimulation in the presence of native X-DING-CD4 suggested that the function of this protein might extend to pathogen-activated NF-κB-dependent promoters, such as IL-8, and block hyperactivation of the immune system in the case of HIV-related diarrheic disease.
To provide proof for our hypothesis that native X-DING-CD4 protein could have a therapeutic effect in blocking the inflammatory pathway associated with bacterial endotoxin, we selected the IL-8 protein as a model pro-inflammatory chemokine secreted by both immune and non-immune cells in response to the LPS exposure. We reasoned that the X-DING-CD4 protein could be developed into a novel biotherapeutic, utilizing its potential to interfere with the LPS-mediated induction of cytokines in the course of AIDS. We show that Salmonella Typhimurium LPS activation of the IL-8 promoter was selectively blocked by the X-DING-CD4 protein at NF-κB, but not at the Ap-1 binding site. The X-DING-CD4-mediated blockage of the IL-8 promoter also reduced levels of the extracellular IL-8 protein in endothelial cells and human macrophages infected with enteric bacteria that frequently cause diarrheal disease in AIDS patients. The assessment of X-DING-CD4 mRNA activity in human macrophages and HIV-resistant, CD4+-transformed T cells exposed to various bacterial LPS or HIV-1, showed that X-DING-CD4-mediated blockage of LPS-induced IL-8 protein was regulated through the exogenous X-DING-CD4 protein and not through the endogenous activity of the X-DING-CD4 gene.
Materials and methods
Cells, reagents and bacterial cultures
The studies reported here using human peripheral blood monocytes were granted exempt status by the St Luke’s-Roosevelt Institutional Review Board under qualifications listed in section 45.101 (b) (4). Human monocytes from healthy, HIV-1 negative volunteers were obtained by elutriation from whole blood. For all experiments, monocytes were allowed to differentiate in 24- or 6-well plates (Corning Scientific, Santa Clara, CA, USA) under culture conditions we described previously. 13 Briefly, cells were seeded at a concentration of 2 × 105 (for ELISA evaluations) or 5 × 106/well (for ChIP assay) and cultured in DMEM (Sigma, St Louis, MO, USA) supplemented with 10%/vol endotoxin-free, heat-inactivated human serum, giant cells tumor conditioned medium (Sigma), glutamine and antibiotics. Following 5 d of the initial culture, the medium was changed to DMEM (Sigma) supplemented with 10%/vol FBS, glutamine (Sigma) and antibiotics.
HUVEC were obtained from the American Type Tissue Collection (ATCC). For routine propagation, HUVEC cells were cultured until 75–80% confluence in Kaighn’s F12 medium with 2 mM glutamine (Sigma), 1.5 g/l sodium bicarbonate, 0.1 mg/ml heparin (Sigma) and 0.05 mg/ml endothelial cell growth supplement (ECGS; BD Biosciences, San Jose, CA, USA). Three days before each experiment, cells were seeded in 24-well plates at a concentration of 2 × 105/well (for ELISA evaluations) or 6-well plates at a concentration of 5 × 106/well (for ChIP assay) and cultured under the same conditions as above.
HIV-1-resistant X-DING-CD4 (+) T cells 15 used for the real time X-DING-CD4 mRNA evaluations were maintained in RPMI 1640 (Sigma) supplemented with 5%/vol FBS, antibiotics and glutamine at 37°C in a 5% CO2 95% air-humidified incubator.
Salmonella enterica subsp. enterica serovar Typhimurium, Shigella flexneri and Camplylobacter jejuni subsp. jejuni were obtained from the ATCC. For infections with Salmonella and Shigella, bacteria were inoculated in a nutrient broth with 0.5%/vol NaCl (BD Biosciences, Sparks, MD, USA) and cultured overnight at 37°C with moderate shaking. Subsequently, bacterial cultures were diluted 100-fold with nutrient broth and cultured under the same conditions until the optical density measured on Ultrospec 10 (Amersham Biosciences, Piscataway, NJ, USA) at a wavelength of 588 nm reached 0.45; this OD value corresponded to ∼5 × 108 bacteria/ml. The above protocol was determined experimentally to produce the sufficient amount of bacteria inducing a detectable quantity of IL-8 protein in supernatants of endothelial cells within the first 24 h of cell exposure to pathogen. Subsequently, 1 ml of each culture was pelleted, washed twice in PBS and re-suspended in 1 ml of PBS. Campylobacter jejuni was cultured on Campy CSM agar plates (BD Biosciences, Sparks, MD, USA) under microaerophilic conditions until full confluence. Subsequently, bacteria were suspended in PBS until the OD value reached 0.45.
Virus isolates used for this study were X4 HIV-1-NL4-3 16 and R5 HIV-1-ADA. 17
Purification of native X-DING-CD4 protein
Briefly, X-DIN-CD4 (+) cells were cultured in protein-free hybridoma medium (Sigma), as previously described; 11 , 18 2 × 106/ml cells were cultured overnight in protein-free Hybridoma medium (Sigma) supplemented with glutamine (Sigma). Subsequently, cells were removed by centrifugation and supernatants were filtered through 0.45 mM membranes (Millipore, Bedford, MA, USA) and concentrated by lyophilization from 30-ml aliquots using a Labconco Lyophilizer. Protein powder was re-suspended in 10 ml distilled water and subjected to dialysis against 10 mM Tris-Cl, pH 7.4, using benzoylated cellulose tubing with MM cut-off of 1.2 KDa (Sigma). Dialyzed material was concentrated, again by lyophilization, and separated during 40 min 0–100% linear gradient of B [Tris-Cl (pH 8.1)+1 M NaCl] at a flow of 3.0 ml/min through a 5-ml ion-exchange High Trap Q XL column by Acta Prime chromatography system (all from Pharmacia, Peapack, NJ, USA). The X-DING-CD4 fraction was dialyzed and its antiviral activity was confirmed by rapid suppression assay (RSA). RSA measures the expression of HIV-1 LTR-promoted luciferase gene and has been our standard assay to detect the transcription inhibitory activity of X-DING-CD4.11,18
Evaluation of Salmonella, Shigella or Campylobacter—induced extracellular IL-8 protein in human macrophages and HUVEC cells treated with native X-DING-CD4
Each experimental system was set up in triplicate. Human monocyte-derived macrophages and HUVEC cells were cultured in respective culture medium supplemented with 100 ng/ml native X-DING-CD4 protein; 24 or 48 h later, cells were infected with 100 μl of respective bacteria/PBS solution and incubated for 30 min at 37°C. Then, bacteria were removed by thorough washing in PBS and macrophages and HUVEC cells were cultured for another 24 h in respective culture media. The extracellular IL-8 was measured in cell culture supernatants by ELISA (Cell Sciences, Canton, MA, USA) according to the manufacturer’s protocol.
Evaluation of NF-κB DNA binding on IL-8 promoter in human macrophages and HUVEC cells upon exposure to native X-DING-CD4
ChIP (chromatin immunoprecipitation) assay
We focused our evaluations on the availability of IL-8 promoter for p65 NF-κB or control Ap-1 proteins in the presence of X-DING-CD4. Briefly, HUVEC cells or human monocyte-derived macrophages were cultured for 3 h in hybridoma medium (HM) (Invitrogen, Carlsbad, CA, USA) supplemented with 1%/vol FBS, glutamine and 100 ng native X-DING-CD4 protein. Subsequently, cells were washed in PBS and medium was replaced by HM supplemented with 1%/vol FBS, glutamine and 30 ng/ml LPS from Salmonella Typhimurium (Sigma). At designated time-points, all proteins were cross-linked with 1%/vol HCHO for 10 min at 25°C followed by 5 min incubation in 0.125 M glycine. Cells were washed in PBS and re-suspended in ice-cold lysis buffer [50 mM Tris, pH 8.0, 0.2 mM EDTA, 0.1%/vol NP40, 10%/vol glycerol and standard cocktail of protease inhibitors: Aprotinin 1 µg/ml, Leupeptin 1 µg/ml and AEBSF 5 µg/ml (all from Sigma)]. After 10 min, nuclei were collected by centrifugation at 500 × g and incubated for 10 min in 100 μl of ice-cold 50 mM Tris, pH 8.0, supplemented with 0.1%/vol SDS, 5 mM EDTA + protease inhibitors (see above). Chromatin was sheared by sonication to the point that 800–1000 bp fragments were produced (titration data not shown). All samples were subjected to centrifugation for 10 min at 6700 × g and supernatants containing DNA-protein fragments were transferred into new tubes and mixed with 400 μl of dilution buffer (50 mM Tris, pH 8.0, 0.5%/vol NP40, 0.2 M NaCl, 0.5 mM EDTA + protease inhibitors). To reduce the nonspecific background, all samples were precleared for 30 min with 80 μl of salmon sperm DNA-protein-A agarose slurry. Protein A-agarose beads (Sigma) were collected by brief centrifugation and 80 μl of each sample (input control) was set aside. For evaluation of IL-8 promoter, the rest of the material was divided into two portions and incubated overnight with 4 µg each of α-p65NF-κB or α-AP1 Abs (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA). Immune complexes were collected by salmon sperm DNA-protein-A sepharose beads after 1 h of incubation followed by three washes in high salt buffer (20 mM Tris, pH 8.0, 0.1%/vol SDS, 1%/vol NP40, 2 mM EDTA, 0.5 M NaCl), three washes with 1 × TE buffer and elution with 1 × TE supplemented with 2%/vol SDS. Eluted samples were heated at 65°C for 10 min. Formaldehyde cross-links were reversed in all samples (including input control) by adding NaCl to the final concentration of 0.3 M and incubated at 65°C for 5 h.
Chromatin-associated protein was digested for 1 h with 100 µg/ml Proteinase K in the presence of 10 mM EDTA and 20 mM Tris-HCl, pH 6.5, at 45°C. DNA samples were extracted with phenol/chloroform and precipitated with 95%/vol ETOH.
Real time PCR (qRT-PCR) analysis of immunoprecipitated DNA
DNA concentration was calculated based on the OD reading measured by the spectrophotometer at a wavelength of 260 nm; all samples were subjected to 40 cycles of amplification using respective pairs of primers and probes: IL-8: sense 5’-TGCCCCCTAAGAGCAGTAAC-3’, antisense 5’-TGAAGATAAGCCA GCCAATCATTT-3’, probe 5’-AGAAACTCTCTAAAATGCT-3’.
The qRT-PCR detection of NF-κB-binding DNA templates was performed using TaqMan Gene Expression Assay on a 7300 real-time PCR System (Applied Biosystems, Foster City, CA, USA). Briefly, triplicates of a total reaction mixture of 25 μl were composed of DNA standardized by input control, 12.5 μl of 2 × ABI TaqMan PCR Master Mix and 0.25 μl (90 μM) of both sense and antisense primers (synthesized by Invitrogen, Carlsbad, CA, USA) and 0.5 μl (10 μM) of the probe (synthesized by Applied Biosystems). After the initial denaturation at 95°C (10 min), target genes were amplified through 40 cycles of universal cycling conditions (95°C/10 s, 60°C/1 min). Standard dilution curves were calibrated using serial dilutions of control plasmid pTA_IL-8 representing 1 × 106, 1 × 105, 1 × 104, 1 × 103, 1 × 102 and 10 copies of DNA per reaction and all valuesare expressed as means ± standard deviation of the mean. The qRT-PCR data for each experimental system were divided by data for their respective input control providing the numerical value (arbitrary units), representing the extent of IL-8 promoters available for p65 NF-κB or AP-1 binding.
Evaluation of X-DING-CD4 mRNA expression by qRT-PCR analysis
For testing the induction of the X-DING-CD4 gene, each 5 × 106 human macrophage or HIV-1-resistant CD4+T cells were cultured for 1 h with 30 ng/ml S. Minnesota, S. Typhimurium or V. cholerae LPS (all from Sigma) or 0.01 pg/cell NL-4-3 HIV-1 16 for X-DING-CD4(+) CD4 T cells, 0.01 pg/cell ADA 17 for human macrophages or medium alone. Following LPS or HIV activation, cells were washed in cold PBS and cultured in appropriate media, as described above. Total RNA was isolated 24 h later by Trizol reagent (Invitrogen). Total of 2 µg total RNA pretreated withDNAse I was reverse transcribed using random hexamers and gene-specific primer (RD1-5’-TC GTTCAGCTTGGTCATCACGCCTTGGCTGGTCACGGC-3’), and Transcriptor First Strand cDNA Synthesis kit (Roche, Mannheim, Germany) according to manufacturer’s protocol with the exception that annealing temperature was set at 65°C.
The qRT-PCR primers and probe were designed based on X-DING-CD4 GenBank sequence [HQ586056] and synthesized by Applied Biosystems, and are described in detail in Sachdeva et al. 19 The real-time detection of X-DING-CD4 and human β-actin gene expression was performed using a TaqMan Gene Expression Assay on 7300 real-time PCR System (Applied Biosystems). Briefly, triplicates of a total reaction mixture of 25 μl were composed of 2 μl of cDNA, 12.5 μl of 2 × ABI TaqMan PCR Master Mix, and 0.25 μl (90 μM) of both sense and antisense primers, and 0.5 μl (10 μM) of the probe. After initial denaturation at 95°C (10 min), target genes were amplified through 40 cycles of universal cycling conditions (95°C/10 s, 60°C/1 min). The β-actin target gene was amplified using a predesigned pair of primers and a probe (Applied Biosystems). Standard cell dilution curves were calibrated using serial dilutions of control plasmid pTA_X-DING-CD4 representing 1 × 106, 1 × 105, 1 × 104, 1 × 103, 1 × 102 and 10 copies of X-DING-CD4 cDNA per reaction and all values are expressed as means ± standard deviation of the mean. All PCR reactions were normalized to the β-actin gene.
Statistical analyses
Standard deviation was calculated by Sigma Plot 11 (Systat Software Inc., Chicago, IL, USA). The P values were derived from the Mann-Whitney rank sum test; the alpha level was set at 0.050.
Results
The X-DING-CD4 protein prevents early LPS activation of the IL-8 promoter in human endothelial cells and macrophages by blocking the recruitment of NF-κB to its cognate binding site
The activation of NF-κB is a common response to cellular exposure to many pathogens. For example, people infected with HIV have greatly induced levels of the IL-8 protein. 7 Monocyte-derived macrophages and vascular endothelial cells undergo a rapid activation to LPS stimulation resulting in the expression of several chemokines, including the IL-8 protein. 20
We previously reported that the unfractionated X-DING-CD4 protein did not impede the nuclear translocation of the NF-κB p50/p65 dimer but interfered with its effective binding to the HIV LTR promoter. 11 Building on these observations, we examined the activation of the IL-8 protein by assessing the efficiency of NF-κB binding to the regulatory sequences on the IL-8 promoter. The nuclear translocation of NF-κB was initiated by cellular exposure to LPS from S. Typhimurium and activation of the IL-8 gene and function of native X-DING-CD4 protein in this process was assayed by chromatin immunoprecipitation (ChIP). We chose a 3 h window to observe the early recruitment of NF-κB and control AP-1 proteins to the IL-8 promoter based on the previous report of biphasic pattern of IL-8 activation, including an early and rapid response to bacterial endotoxin. 21
As shown in Figure 1A and B, the binding of the NF-κB dimer to the IL-8 promoter was rapidly induced by LPS in both cell types, with the maximum promoter occupancy at the 60-min time-point. Interestingly, the activity of the IL-8 promoter in response to S. Typhimurium LPS was, on average, twofold higher at the 30- and 60-min time-points in the endothelial cells. The X-DING-CD4 treatment reduced the recruitment of NF-κB to the IL-8 promoter at all time-points tested; at 30 and 60 min after LPS stimulation, NF-κB/DNA binding was reduced by 95% and 64% in endothelial cells respectively (Figure 1A). In human macrophages exposed to LPS from S. Typhimurium, the X-DING-CD4 treatment reduced the induction of the IL-8 promoter by 95% at both these time-points (Figure 1B). The examination of the control AP-1 binding site under the same experimental conditions showed no statistical significance between the LPS-induced and un-induced systems. Also, the X-DING-CD4 protein treatment did not affect the AP-1/DNA binding (Figure 1C and D). Similar effect of LPS on the induction of AP-1 proteins was observed in murine aortic endothelial cells.
22
Exposure to native X-DING-CD4 protein blocks recruitment of NF-κB to the IL-8 promoter. Five × 106/well of HUVEC (A and C) and monocyte-derived macrophages (MQ) (B and D) were cultured in the presence of 100 ng/ml native X-DING-CD4 for 3 h; following X-DING-CD4 treatment, cells were exposed to 30 ng/ml LPS from S. Typhimurium. The experimental samples were collected at 30, 60 and 180 min after LPS exposure. NF-κB and Ap-1 binding to the IL-8 promoter was evaluated by ChIP and qRT-PCR as described in methods. The P values were derived from the Mann-Whitney rank sum test; a P value of <0.050 represents statistical significance. (C and D) The P values for the control and respective experimental samples are >0.050. The results are representative of at least two separate experiments.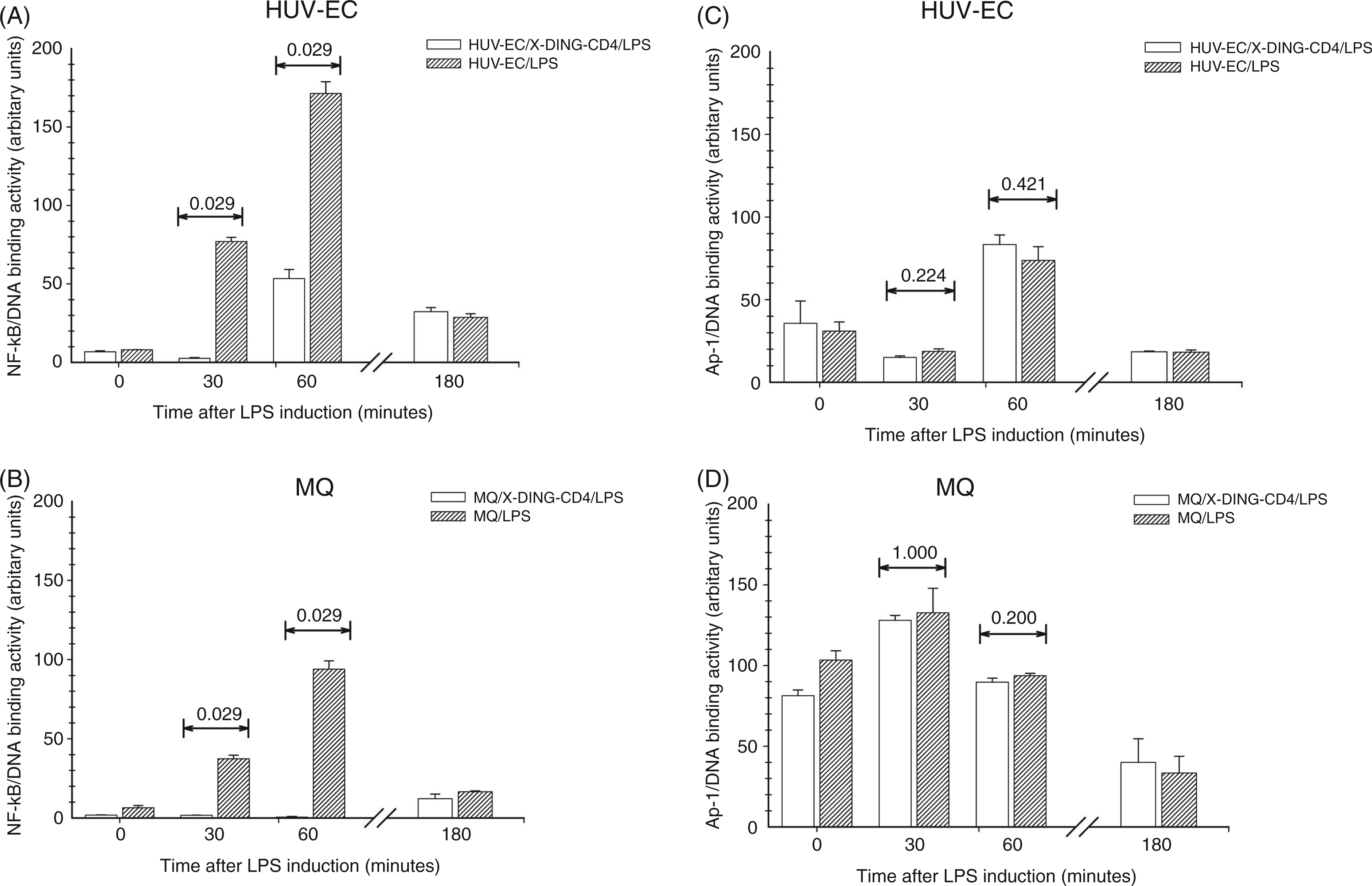
Levels of soluble IL-8 protein are reduced in enteric bacteria-infected endothelial cells and human macrophages upon their exposure to native X-DING-CD4
The expression of the IL-8 gene depends on the recruitment of the NF-κB transactivator. In a previous study, we showed that X-DING-CD4 blocked the NF-κB/DNA binding in cells exposed to LPS from V. cholerae, S. Minnesota and Escherichia coli. 14 Data shown in the previous section provided evidence that the X-DING-CD4 treatment blocked also the recruitment of the NF-κB dimer to S. Typhimurium LPS-activated IL-8 promoter in immune and non-immune cells. Knowing that bacterial pathogens employ diverse invasive mechanisms that action results in a similar cellular response,1,23– 25 we investigated X-DING-CD4 anti-inflammatory potential in the experimental setting where cells were infected with S. Typhimurium or other two enteric bacteria also causing chronic diarrhea in AIDS patients.
Both human endothelial cells and macrophages were exposed to S. Typhimurium, S. flexneri or C. jejuni in the presence, or absence, of the X-DING-CD4 protein. The effect of bacterial infection on the IL-8 promoter was evaluated by levels of the soluble IL-8 protein by ELISA in 24-h intervals. In general, bacterial infection induced high levels of extracellular IL-8 protein in the systems where cells were cultured alone [Figure 2A–F, (+) control]. In stark contrast, prior exposure to native X-DING-CD4 reduced the concentration of this pro-inflammatory mediator in both cell types infected with S. flexneri and macrophages infected with C. jejuni (Figure 2B, E and F, X-DING-CD4). Variations of IL-8 expression in cells infected with S. Typhimurium did not reach statistical significance, suggesting that X-DING-CD4-mediated blockage of S. Typhimurium LPS-induced IL-8 promoter observed by ChIP assay (Figure 1) was less obvious in the decrease of the soluble IL-8 protein (Figure 2 A and D). We believe that diminished activity of the X-DING-CD4 treatment in cells infected by S. Typhimurium could be related to redundant invasive mechanisms utilized by this bacterium, also resulting in the induction of the IL-8 protein.
23
–
25
The X-DING-CD4 treatment of endothelial cells infected with C. jejuni did not show a statistically significant reducing effect on extracellular IL-8 protein (Figure 2C). We consider the possibility that endothelial cell polarization
26
could interfere with X-DING-CD4/cell penetration.
Exposure of endothelial cells and human macrophages to native X-DING-CD4 protein inhibits bacteria-induced secretion of pro-inflammatory cytokine IL-8. Two × 105 of HUVEC (upper panel, A, B and C) or monocyte-derived macrophages (MQ) (lower panel, D, E and F) were cultured in the presence of 100 ng/ml of native X-DING-CD4 protein for 24 or 48 h; following X-DING-CD4 treatment, cells were exposed to 5 × 108/ml S. Typhimurium (A and D), S. flexneri (B and E) or C. jejuni (C and F) for 30 min. The concentration of IL-8 protein was measured in cell culture supernatants by ELISA as described in the ‘Materials and methods’. The P values were derived from the Mann-Whitney rank sum test; a P value of<0.050 represents statistical significance. The results are representative of at least three separate experiments.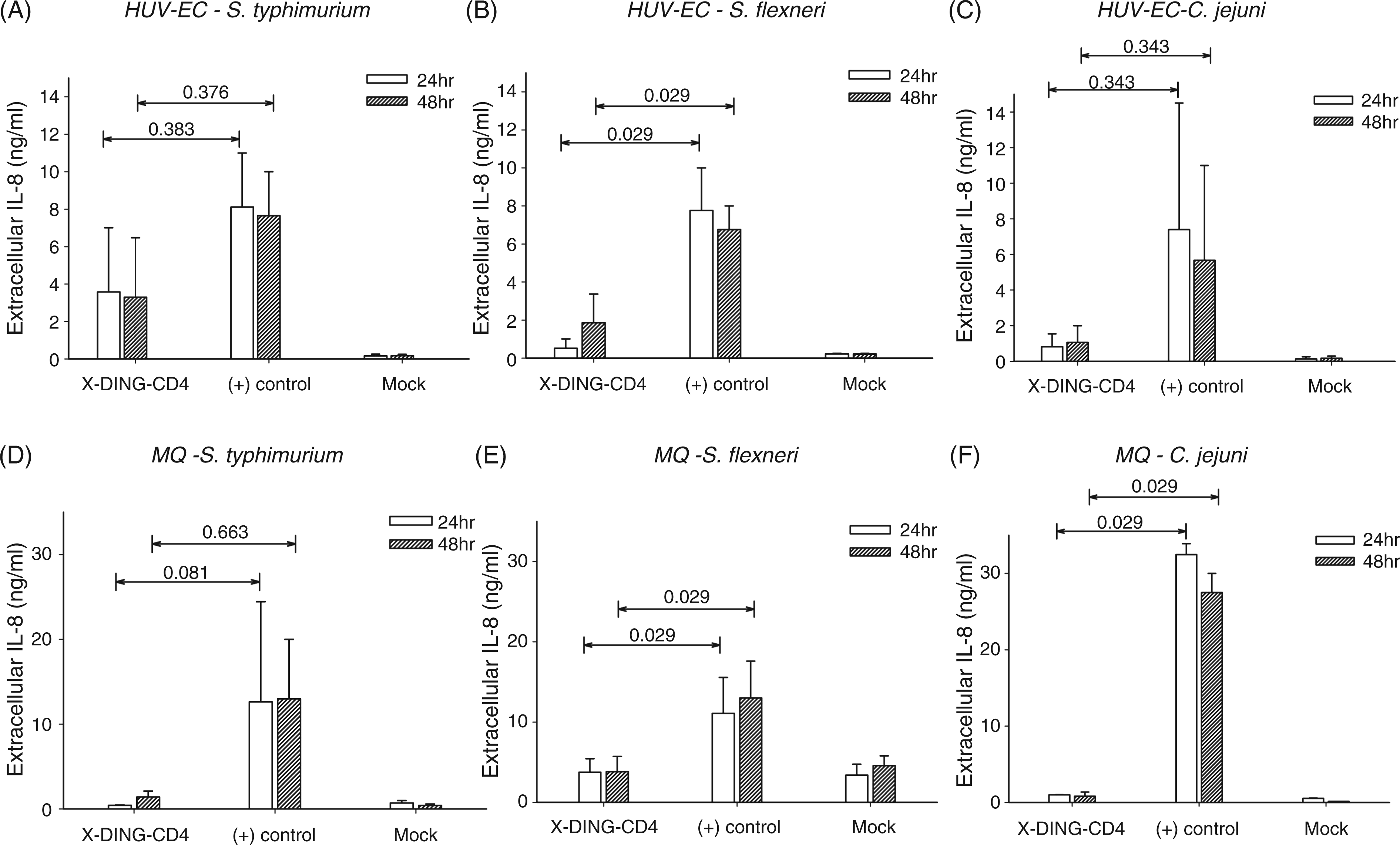
X-DING-CD4-mediated blockage of IL-8 promoter activation does not require de novo X-DING-CD4 protein synthesis
In parallel studies, we found that primary and transformed T cells refractive to HIV-1 rapidly activate the expression of X-DING-CD4 mRNA in response to virus exposure. 12 This inducible responsiveness of the X-DING-CD4 gene to HIV-1 is a distinguishing marker of the natural resistance to virus in these cells.Building on this observation, we investigated if X-DING-CD4-mediated inhibition of LPS-induced NF-κB/IL-8 promoter-binding could be regulated through the function of the X-DING-CD4 gene. We tested this hypothesis in human macrophages susceptible to HIV-1 and X-DING-CD4(+) T cells refractive to HIV-1 infection.
Cells were treated with various bacterial LPS or HIV-1; variations in the X-DING-CD4 mRNA level were assessed by qRT-PCR. Considering the cell entry receptor requirements, human macrophages were infected with R5 ADA HIV-1 and X-DING-CD4(+) cells with X4 NL-4-3 HIV-1. As shown in Figure 3, the exposure of HIV-1-resistant X-DING-CD4(+) cells to HIV-1 resulted in a rapid 20–25-fold induction of X-DING-CD4 mRNA while treatment of these cells with LPS from S. Minnesota, S. Typhimurium or V.cholerae had only a mild effect on X-DING-CD4 mRNA activity, inducing it by 1.8–2.7-fold.
Evaluation of X-DING-CD4 mRNA expression in human macrophages and HIV-resistant, X-DING-CD4(+) cells. The monocyte-derived macrophages from HIV-negative donor (MQ) and HIV-1 resistant X-DING-CD4(+) cells were exposed to 30 ng/ml of LPS from S. Minnesota, S. Typhimurium or V. cholera, respectively, or 0.01 pg/cell respective HIV-1; MQ were infected with the macrophage tropic R5 HIV-1 ADA and X-DING-CD4(+) T cells with X4 NL4-3 HIV-1. The expression of X-DING-CD4 mRNA was tested by qRT-PCR as described in the ‘Materials and methods’. The results are representative of at least three separate experiments.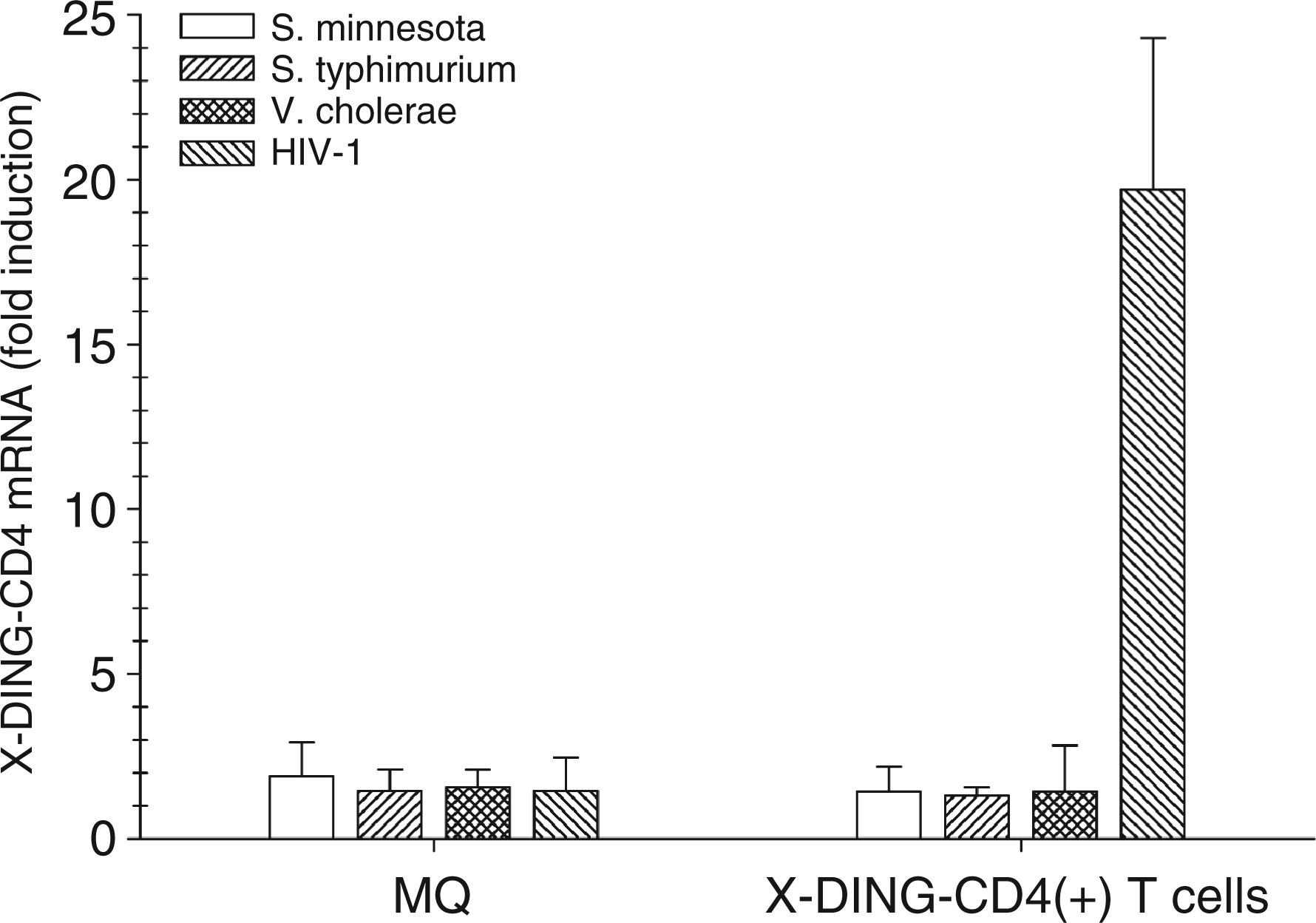
A similar effect of LPS or HIV-1 ADA clone on the X-DING-CD4 gene was observed in human macrophages (Figure 3), indicating that X-DING-CD4 mediated blockage of IL-8 transcription is mediated by the function of exogenous X-DING-CD4.
Discussion
The immune functions of T cells are continuously regulated through the array of soluble mediators secreted by the effector cells.27,28 In the course of AIDS, these immune functions are severely impaired and one reason for this disarray may be a massive depletion of CD4+ T cells from mucosal surfaces in the acute phase of HIV infection. 29 This hypothesis was corroborated by data from animal studies showing the translocation of intestinal bacterial antigens through a compromised mucosa followed by systemic immune activation. 30 In this sense, the anti-inflammatory property of native X-DING-CD4 could be beneficial in AIDS-related diarrheal disease and the main rationale for our study was to obtain proof of the principle of our hypothesis that the X-DING-CD4 protein has therapeutic potential to reduce the LPS-mediated immune activation in the immune and non-immune cells.
This hypothesis originated from our previous reports which showed that X-DING-CD4 treatment interfered with the production of TNF-α protein in LPS-induced macrophages. 14 We also showed that the X-DING-CD4 protein blocked HIV-1 transcription through interference with NF-κB/LTR-binding, but not Sp-1- or YB-1/LTR-binding,11,13 thus providing grounds forthe proposition that the anti-inflammatory effect of X-DING-CD4 may extend to non-immune cells, for example endothelial cells undergoing persistent endotoxin stimulation in the course of advanced HIV disease. To evaluate our theory that the effect of X-DING-CD4 protein extends to LPS–activated, NF-κB-dependent promoters, we selected the exemplary IL-8 chemokine secreted by activated endothelial cells and macrophages. We showed that native X-DING-CD4 protein blocked the IL-8 protein through interference with the recruitment of the NF-κB dimer but not with Ap-1 to its promoter.
The similar blockage of NF-κB, but not SP-1 binding, was observed during evaluations of X-DING-CD4 activity on the activation of the HIV-1 LTR promoter. 13 It is interesting that the end-point of X-DING-CD4 treatment in cells infected with pathogenic bacteria (an inhibition of NF-κB/DNA binding) is similar to that observed in cells infected with HIV-1,11,13,15 suggesting that native X-DING-CD4 protein is a broad spectrum NF-κB inhibitor in the pathogen-activated cells and provides grounds for possible utility of this protein or its synthetic peptides as a biotherapeutic.
The analysis of the X-DING-CD4 inhibition of S. Typhimurium LPS-mediated induction of IL-8 showed statistical relevance in the evaluations of NF-κB/IL-8 promoter binding, but no significance was observed for X-DING-CD4-mediated blockage of the soluble IL-8 protein in cells infected by this pathogen. This discrepancy can be related to a complex mechanism employed by this bacterium in the process of cellular invasion. In addition to its endotoxin, other virulence factors, such as SipA and flagellin, also induce IL-8 expression. 23 – 25 The effect of the X-DING-CD4 protein on the activity of these factors was not studied in this work.
The lack of statistical significance for X-DING-CD4 treatment of endothelial cells infected with C. jejuni wasunexpected. The production of soluble IL-8 was reduced by X-DING-CD4 treatment in human macrophages, but not in endothelial cells infected with this bacterium. The complex interactions of C. jejuni with simple monolayer cultures could offer an explanation for this diminished activity of X-DING-CD4 on infected cells. It was suggested that interactions of this bacterium with a simple monolayer could be interrupted by the polarized surface of the epithelial cells. 31 We hypothesize that cell polarization could also interfere with X-DING-CD4 cellular translocation, however, these parameters were not studied in this work.
Based on a recent study, we postulated that the X-DING-CD4-mediated inhibition of HIV-1 LTR promoter in HIV-resistant, but not HIV-susceptible cells, is regulated through a function of the X-DING-CD4 gene, 12 suggesting that the intrinsic response to virus in these cells requires de novo synthesis and possibly methylation of the X-DING-CD4 protein. About 10% of the native soluble X-DING-CD4 protein secreted by HIV-resistant cells had a methyl group on the glutamic acid in position 69. 32 We postulated that the responsiveness of X-DING-CD4 mRNA to HIV-1 infection is a marker of cellular resistance to virus 12 and, for the purpose of this study, we tested the sensitivity of the X-DING-CD4 gene to LPS stimulation.
Bacterial LPS did not induce expression of X-DING-CD4 mRNA in human macrophages or HIV-resistant, X-DING-CD4(+) T cells. Lack of X-DING-CD4 gene up-regulation in cells exposed to bacterial endotoxin suggests that the X-DING-CD4-mediated blockage of LPS-induced IL-8 promoter is regulated through a mechanism that relies on the exogenous X-DING-CD4 protein transported to cells through a receptor-independent pathway (unpublished). These data show a distinction between the ‘active’ role of the X-DING-CD4 gene in cellular resistance to HIV-1 and a ‘passive’ protection of the X-DING-CD4 protein from the pathogen-activated NF-κB pathway.
Footnotes
*
These authors contributed equally to this work.
Funding
This work was supported by AI 061286-01 and NS062649 from the National Institutes of Health.
Acknowledgements
We thank Dr Potash for providing human monocytes for these studies.
