Abstract
A study was conducted to determine the effect of feeding yeast cell wall (YCW) products on the physiological and acute phase responses of crossbred, newly-received feedlot heifers to an endotoxin challenge. Heifers (n = 24; 219 ± 2.4 kg) were separated into treatment groups receiving either a control diet (n = 8), YCW-A (2.5 g/heifer/d; n = 8) or YCW-C (2.5 g/heifer/d; n = 8) and were fed for 52 d. On d 37 heifers were challenged i.v. with LPS (0.5 µg/kg body mass) and blood samples were collected from −2 h to 8 h and again at 24 h relative to LPS challenge. There was an increase in vaginal temperature in all heifers post-LPS, with YCW-C maintaining a lower vaginal temperature post-LPS than control and YCW-A heifers. Sickness behavior scores increased post-LPS in all heifers, but were not affected by treatment. Cortisol concentrations were greatest in control heifers post-LPS compared with YCW-A or YCW-C heifers. Concentrations of IFN-γ and TNF-α increased post-LPS, but were not affected by treatment. Serum IL-6 concentrations increased post-LPS and were greater in control heifers than YCW-A and YCW-C heifers. These data indicate that YCW supplementation can decrease the physiological and acute phase responses of newly-received heifers following an endotoxin challenge.
Introduction
In animal production, there have been great strides in areas such as feeding and nutrition, reproductive management and estrus synchronization, and genetic selection. However, animal health needs additional attention as producers continue to have production losses (monetary due to loss of weight and medication costs, as well as mortality) that could be alleviated by implementing practices that enhance the immune system. In addition to changing management strategies to decrease stressors known to inhibit immunity, there is the potential to alter immune function through feed supplementation. Studies on the use of feed supplements to enhance animal health are in increasing demand as a result of the movement to decrease and potentially eliminate the use of sub-therapeutic concentrations of antibiotics in feedstuffs. Because of the assumption that feeding sub-therapeutic antibiotics in feed leads to the development of antibiotic-resistant bacteria, the European Union has banned the use of direct-fed antibiotics. 1 Therefore, it is essential that more research be conducted to evaluate viable alternatives to feed-grade antibiotics.
The receiving period at a feedlot is very stressful, as cattle are exposed to various stressors, including the handling associated with transportation and processing, and commingling with unfamiliar cattle. While these stressors alone can cause immunosuppression and increase the incidence of disease, the transfer of pathogens between unfamiliar cattle further heightens this risk. 2 Indeed, there is a high rate of morbidity in receiving cattle, mainly attributed to bovine respiratory disease. 2 Therefore, alternative methodologies that enhance the immune system of receiving cattle have the potential to decrease production costs associated with medication usage and the weight loss associated with illness, and, consequently, are in high demand.
Yeast and yeast cell wall (YCW) products have been demonstrated to improve productivity during several periods of cattle production, and have the potential to be a viable non-antibiotic alternative feed supplement. Yeast supplementation has been demonstrated to improve dry matter intake and average daily gain (ADG), while decreasing morbidity in cattle.3–5 In addition, a previous study using live yeast and/or YCW products reported lower rectal temperature and cytokine concentrations in response to endotoxin administration in steers, suggesting yeast products can improve cattle health. 6 Therefore, the present study was designed to determine the effect of supplementing two different YCW products on the physiological and acute phase responses of newly received heifers following LPS challenge.
Materials and methods
Experimental design
All experimental procedures were in compliance with the Guide for the Care and Use of Agricultural Animals in Research and Teaching, and approved by the Institutional Animal Care and Use Committee at Texas Tech University (approval number: 10079-11).
Diet composition.
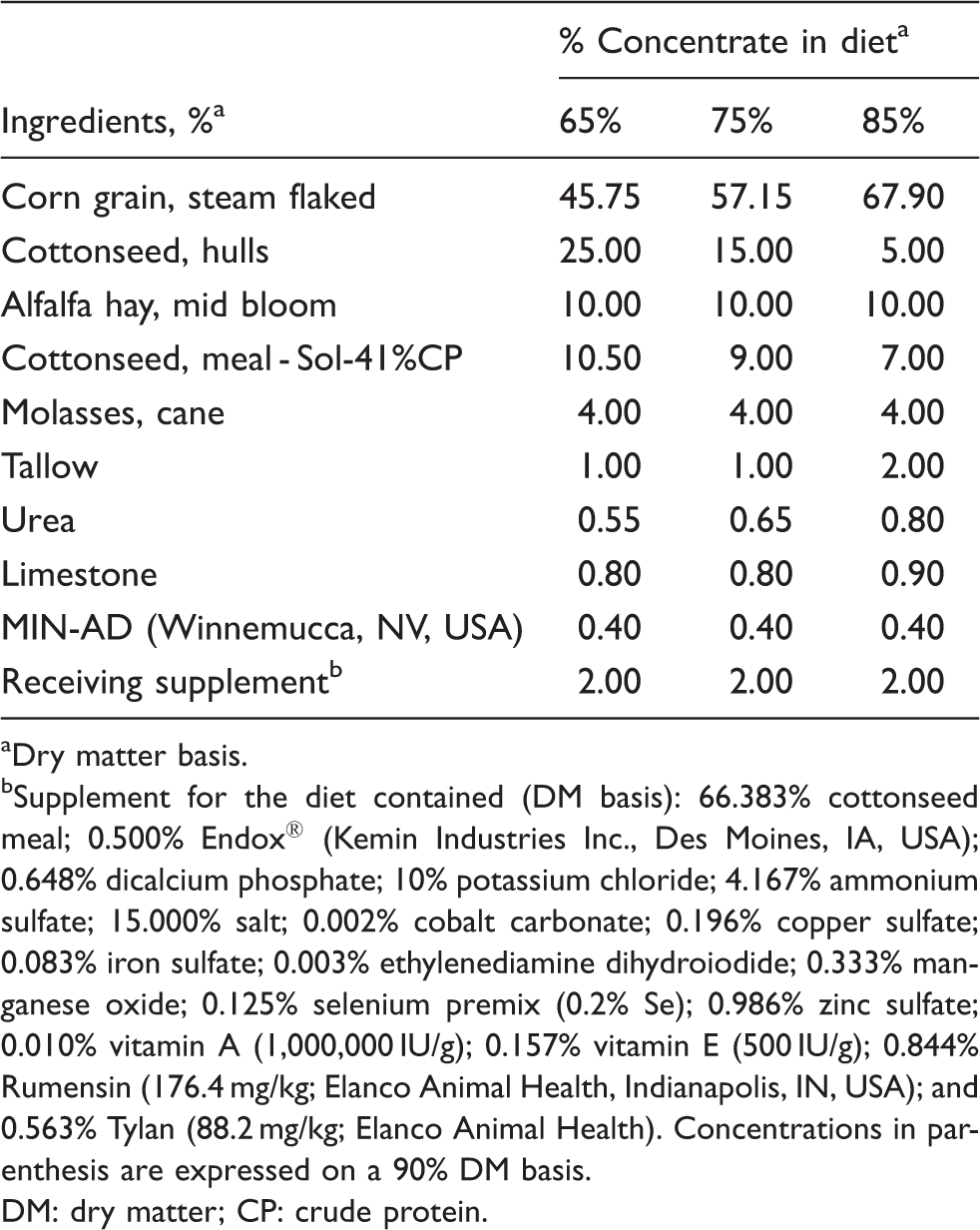
Dry matter basis.
Supplement for the diet contained (DM basis): 66.383% cottonseed meal; 0.500% Endox® (Kemin Industries Inc., Des Moines, IA, USA); 0.648% dicalcium phosphate; 10% potassium chloride; 4.167% ammonium sulfate; 15.000% salt; 0.002% cobalt carbonate; 0.196% copper sulfate; 0.083% iron sulfate; 0.003% ethylenediamine dihydroiodide; 0.333% manganese oxide; 0.125% selenium premix (0.2% Se); 0.986% zinc sulfate; 0.010% vitamin A (1,000,000 IU/g); 0.157% vitamin E (500 IU/g); 0.844% Rumensin (176.4 mg/kg; Elanco Animal Health, Indianapolis, IN, USA); and 0.563% Tylan (88.2 mg/kg; Elanco Animal Health). Concentrations in parenthesis are expressed on a 90% DM basis.
DM: dry matter; CP: crude protein.
All premixes were made at the Texas Tech University Burnett Center Feed Mill in a paddle type mixer (Marion Mixers Inc., Marion, IA, USA). The supplement premix included standard trace minerals, vitamins and monensin (Rumensin 90; Elanco Animal Health, Indianapolis, IN, USA). Ingredients for the YCW premix included ground corn, corn oil and yeast cell wall (excluded in the control premix). YCW was measured out into an individual clean bowl on a Mettler (Novatech UK Ltd, Portsmouth, UK) electronic balance (accuracy ± 4.5 g). Corn oil was measured in a similar fashion. Ground corn was measured on an Ohaus (Pine Brook, NJ, USA) electronic balance (accuracy ± 0.1 g). Ground corn was added first, followed by corn oil, and, finally, the appropriate quantity and type of YCW. All ingredients were mixed for 5 min. Once mixing was finished, premixes were divided evenly into three labeled barrels (per treatment). Samples were taken at the beginning, middle and end of allocation to barrels. The mixer was swept and blown out with pressurized air between each premix to help decrease contamination. YCW premixes were weighed out for each pen daily into plastic containers with corresponding numbered lids. The YCW premixes were top dressed at a rate of 91 g/heifer daily.
On d 36, heifers were fitted with jugular vein cannulas and indwelling vaginal temperature recording devices 7 that measured vaginal temperature continuously at 1-min intervals in the absence of a human operator. For the jugular cannulation procedure, a small (2–3 cm) incision was made in the skin to more easily access the jugular vein. Temporary indwelling jugular catheters, consisting of 30.48 cm of sterile Tygon® tubing (AAQ04133; US Plastics, Lima, OH, USA; 1.27 mm i.d. and 2.286 mm o.d.), were inserted into the jugular vein using a 14-gauge by 5.08-cm thin-walled stainless steel biomedical needle (o.d. = 3 mm). The catheter was held in place using tag cement and a 2.08-cm wide porous surgical tape around the incision site, and then the entire neck region of the heifers was wrapped with vet wrap (Vetrap™; 3 M Animal Care Products, St. Paul, MN, USA) to ensure stability of the catheterization site. The remaining tubing not inserted into the heifer served as the extension portion of the cannula for collection of blood samples. During these procedures, heifers were restrained in a working chute for approximately 10–15 min.
Following these procedures, heifers were moved to a facility that contained individual stalls (2.13 m long × 0.76 m wide) which housed the heifers through the duration of the LPS challenge. Heifers were placed so that treatments were alternated by stall. During the challenge the heifers had ad libitum access to feed and water. The extension tubing of the cannula was extended above the stall to allow researchers to collect blood throughout the study without disturbing the heifers, whether the heifers were standing or lying down.
On d 37, whole blood samples (10 ml) were collected into blood tubes containing no additive every 0.5 h, beginning 2 h before and continuing 8 h after administration of LPS (0.5 µg/kg body weight; Escherichia coli O111:B4; Sigma-Aldrich, St. Louis MO, USA) and again at 24 h. Whole blood was allowed to clot for 30 min and serum was collected after centrifugation at 1250 × g for 20 min at 4℃. Serum was stored at −80℃ until analyzed for cortisol and cytokine concentrations. Before administration of LPS, the jugular cannula became dislodged from 1 YCW-A heifer; therefore, data presented represent 23 heifers (Control, n = 8; YCW-A, n = 7; YCW-C, n = 8).
Sickness behavior
Sickness score definitions of visual signs of sickness.

Assays for cortisol and cytokines
All serum samples were analyzed in duplicate. Serum cortisol concentrations were determined using a commercially available enzyme immunoassay kit according to the manufacturer’s directions (Arbor Assays, Ann Arbor, MI, USA) by comparison of unknowns to standard curves generated with known concentrations of cortisol. The minimum detectable cortisol concentration was 45.4 pg/ml, and the intra- and inter-assay coefficients of variation were 7% and 20% respectively. Data are presented as ng/ml.
Serum cytokine concentrations (TNF-α, IFN-γ and IL-6) were determined by a custom bovine three-plex sandwich-based chemiluminescence ELISA kit (Searchlight-Aushon BioSystems Inc., Billerica, MA, USA). The minimum detectable concentrations were 0.5, 0.1 and 3.3 pg/ml for TNF-α, IFN-γ and IL-6, respectively. All intra-assay coefficients of variation were < 9% and all inter-assay coefficients of variation were < 21% for all assays. Data are presented as pg/ml.
Statistical analyses
Before analysis, vaginal temperature data were averaged into 1-h intervals. Data for vaginal temperature, sickness behavior scores, cortisol and cytokines were analyzed using the MIXED procedure of SAS (SAS, Inc., Cary, NC, USA) specific for repeated measures with treatment, time and time × treatment interaction included as fixed effects. Specific pre-planned treatment comparisons were made using Fisher’s protected least significant difference, with P < 0.05 considered significant. Data are presented as the least squares means ± the standard error of the mean.
Results
Vaginal temperature
There was no difference in vaginal temperature amongst treatments in the 12-h period before administration of LPS (treatment: P = 0.251; Figure 1), although vaginal temperature decreased over time (P < 0.001). In response to LPS administration at time 0 h, vaginal temperature increased (P = 0.010), reaching peak values within 2 h before decreasing. Average post-LPS vaginal temperatures were greater in Control (39.00 ± 0.03℃) and YCW-A heifers (38.99 ± 0.03℃) than YCW-C heifers (38.90 ± 0.03℃; P < 0.001).
Effect of yeast cell wall (YCW-A or YCW-C; 2.5 g/heifer/d) supplementation on the vaginal temperature response to an endotoxin (LPS; 0.5 µg/kg body weight i.v.) challenge (Control, n = 8; YCW-A, n = 7; YCW-C, n = 8). Data are presented as least squares means ± SEM.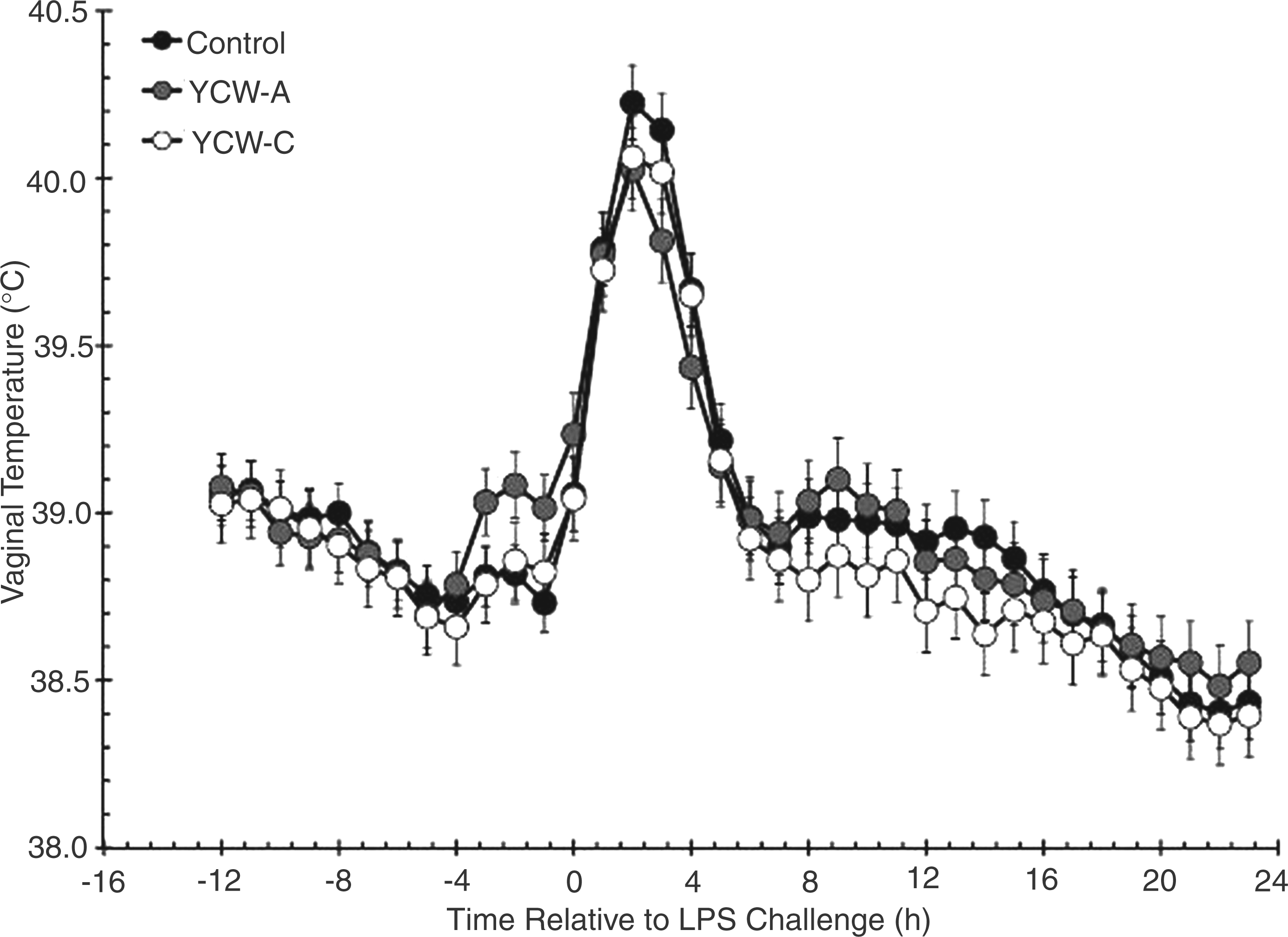
Sickness behavior scores
Before administration of LPS there was no difference in observed sickness behavior scores as a result of dietary treatment (Figure 2; P = 0.381). Although sickness behavior scores increased (P < 0.001) following administration of LPS at time 0 h, the heifers showed very limited sickness behaviors, as indicated by the low peak score between 1.3 and 1.5 (on a scale of 1–5). There was no effect of YCW supplementation on sickness behavior scores post-LPS (P = 0.539).
Effect of yeast cell wall (YCW-A or YCW-C; 2.5 g/heifer/d) supplementation on the sickness behavior response to an endotoxin (LPS; 0.5 µg/kg body weight i.v.) challenge (Control, n = 8; YCW-A, n = 7; YCW-C, n = 8). Heifers were scored on a scale of 1 (active or agitated), showing the least amount of sickness behavior, to 5 (laying on side with labored breathing), showing the greatest amount of sickness behavior. Data are presented as least squares means ± SEM.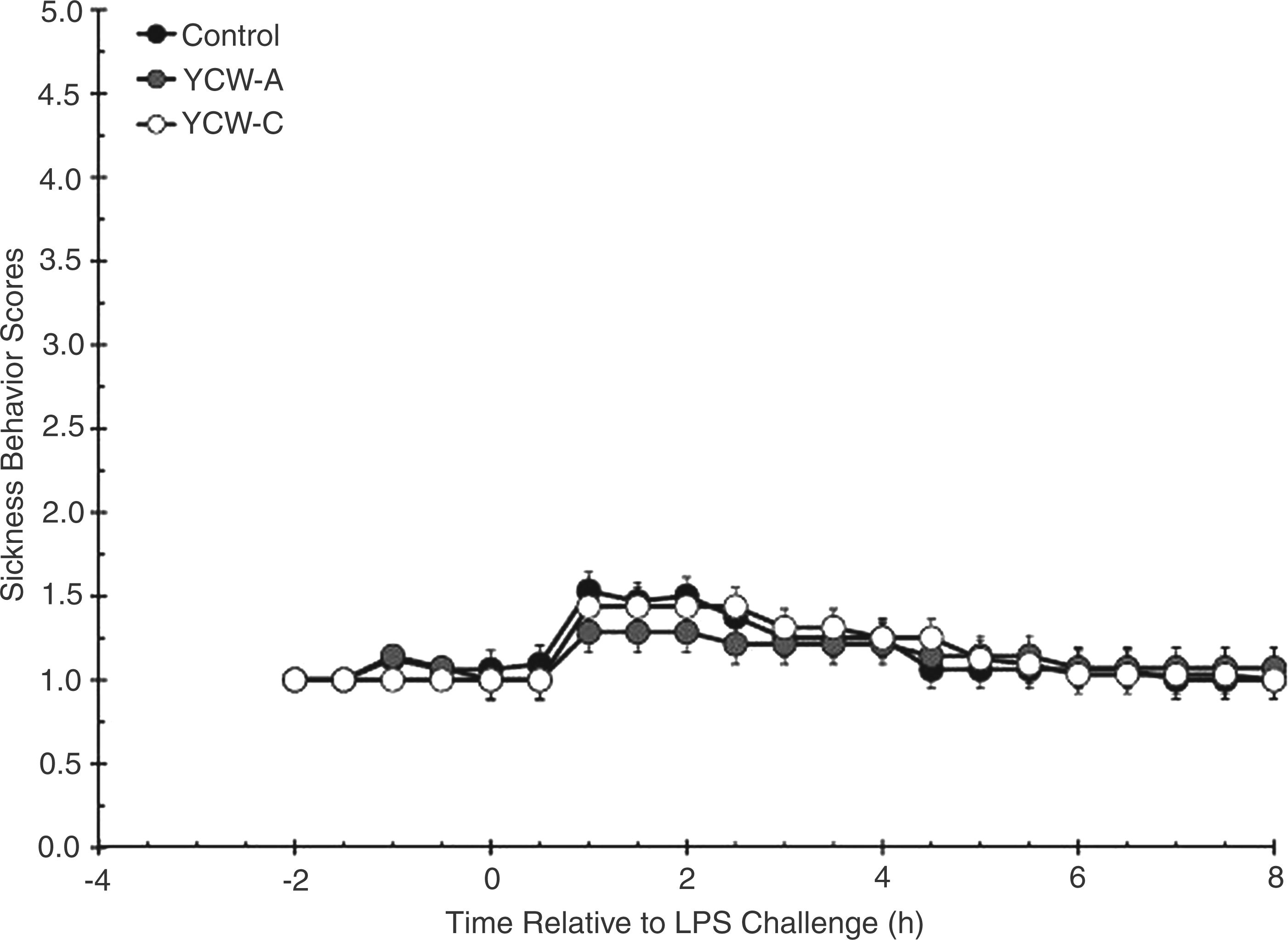
Serum cortisol concentration
There was no effect of YCW treatment on pre-LPS serum cortisol concentrations (Figure 3; P = 0.773). In response to administration of LPS at time 0 h, cortisol concentrations increased within 0.5 h (P < 0.001) in all treatment groups. Average post-LPS cortisol concentrations were greater in Control (39.7 ± 1.5 ng/ml) than in YCW-A (31.3 ± 1.7 ng/ml) and YCW-C heifers (32.0 ± 1.8 ng/ml; P < 0.001).
Effect of yeast cell wall (YCW-A or YCW-C; 2.5 g/heifer/d) supplementation on the serum cortisol response to an endotoxin (LPS; 0.5 µg/kg body weight i.v.) challenge (Control, n = 8; YCW-A, n = 7; YCW-C, n = 8). Data are presented as least squares means ± SEM.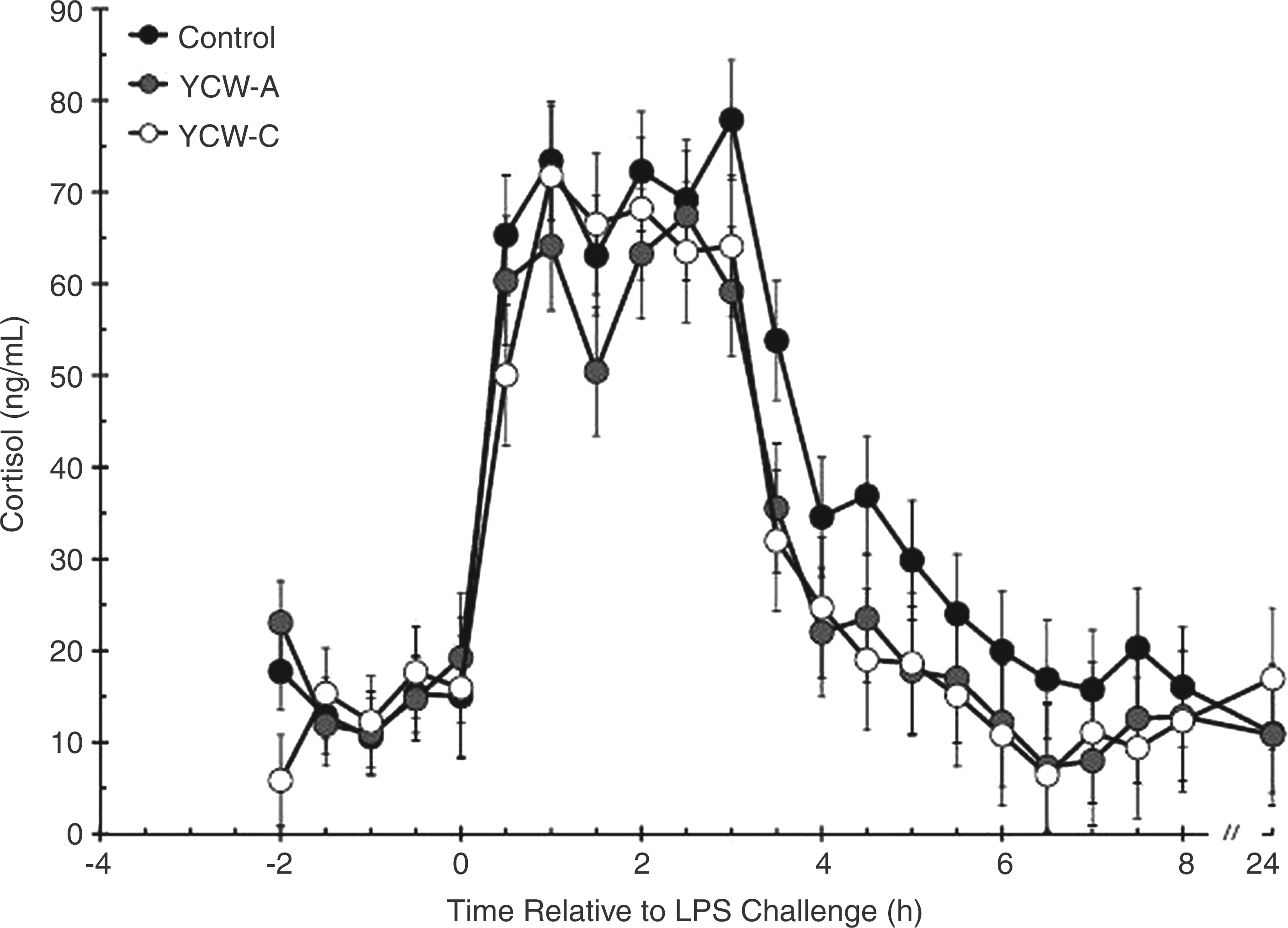
Serum cytokine concentrations
Serum concentration of IFN-γ was not affected by YCW treatment pre- (P = 0.612) or post-LPS administration (P = 0.497); however, concentrations changed over time both pre- (P = 0.028) and post-LPS (P < 0.001; data not shown). Similarly, serum concentration of TNF-α was not affected by YCW treatment pre- (P = 0.991) or post-LPS administration (P = 0.349; data not shown). Pre-LPS TNF-α concentrations were not affected by time (P = 0.775), yet post-LPS TNF-α concentrations increased 1 h post-LPS (P < 0.001) in all treatment groups.
There was no effect of YCW treatment (P = 0.683) or time (P = 0.672) on serum IL-6 concentration prior to LPS administration (Figure 4). Serum concentration of IL-6 increased within 1.5 h post-LPS (P < 0.001) in all treatment groups. However, average post-LPS IL-6 concentration was greater in Control heifers (351.5 ± 36.0 pg/ml) than in YCW-A (85.8 ± 42.9 pg/ml) and YCW-C heifers (136.2 ± 36.0 pg/ml; P < 0.001; Figure 4 inset).
Effect of yeast cell wall (YCW-A or YCW-C; 2.5 g/heifer/d) supplementation on the serum IL-6 response to an endotoxin (LPS; 0.5 µg/kg body weight i.v.) challenge (Control, n = 8; YCW-A, n = 7; YCW-C, n = 8). Data are presented as least squares means ± SEM. The figure inset represents treatment differences in average post-LPS IL-6 concentrations. a,bP < 0.05.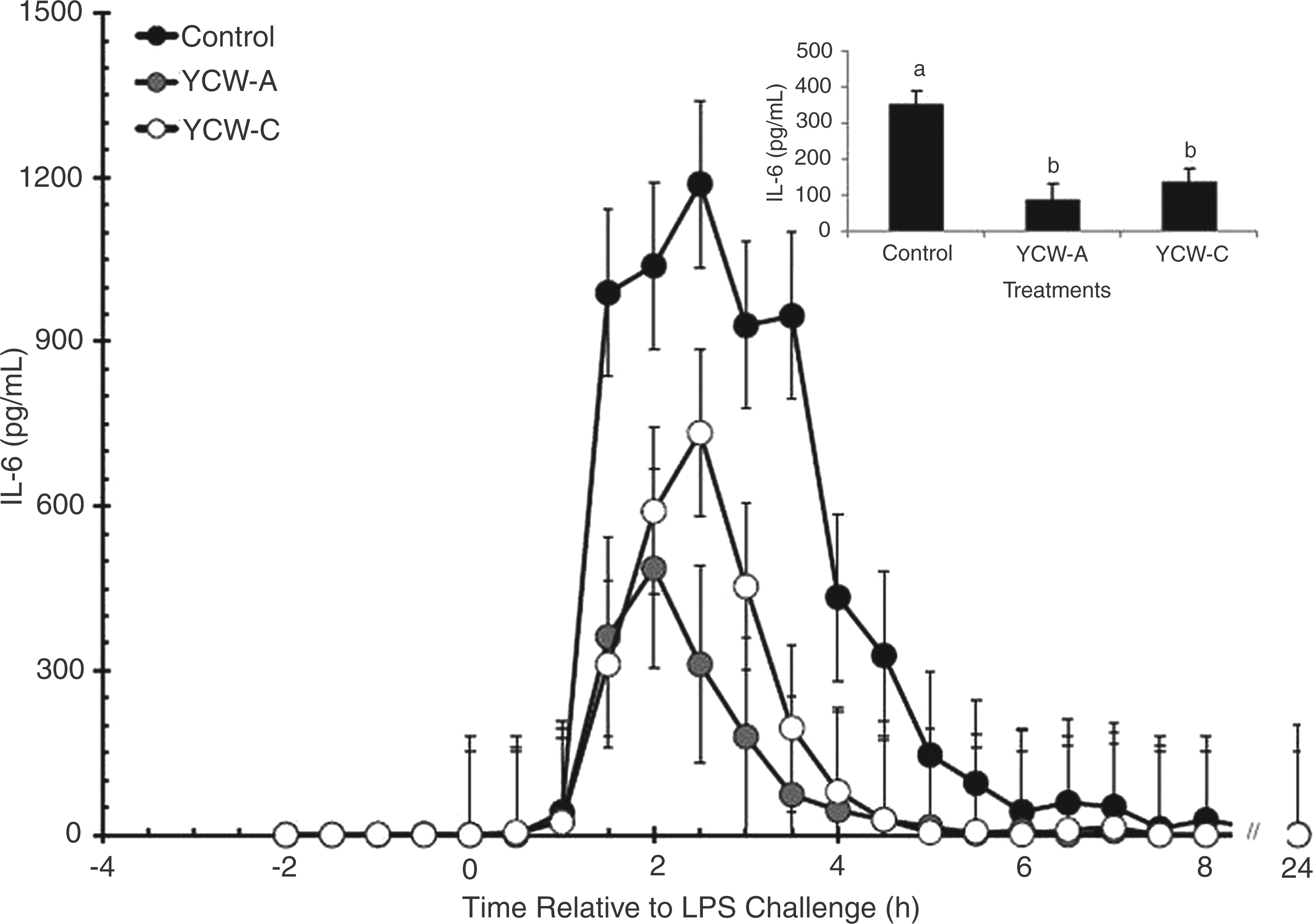
Discussion
The use of yeast supplements in cattle is a growing area of research; however, at the present time, there is limited information regarding the use of yeast product supplements on the health of beef cattle. A study in young dairy calves demonstrated that feeding a yeast culture during the first 70 d of age improved survival rate of calves. 5 A study in neonatal dairy calves also demonstrated that feeding of hydrolyzed yeast was able to enhance health and serum IgA concentrations following a vaccine challenge. 9 Additionally, Cajurigky et al. 10 found that supplementation of dairy cows in early lactation with live yeast increased serum IgA, and circulating numbers of T and B lymphocytes. These studies demonstrate that yeast supplementation can modulate the health of dairy cattle; however, more research is needed on the effects of yeast supplementation on immunity in beef cattle. An enhancement of the health of beef cattle during the receiving period is essential for the industry, as the stress endured during this period as a result of increased handling and commingling with unfamiliar cattle increases the susceptibility to immune challenges. 11 Results have indicated that nutritional supplementation can have a direct influence on the immune system.2,11,12 The present study aimed to examine the effect of YCW supplementation on the physiological and acute phase responses of newly-received heifers to an endotoxin challenge. The data from this study demonstrated that YCW supplementation can decrease both physiological and acute phase responses elicited following a LPS challenge, as indicated by changes in vaginal temperature, and cortisol and IL-6 concentrations.
The mechanism by which yeast supplementation alters the immune system is still under investigation. Some literature suggests that components of the YCW (i.e. β-glucans and mannans) prevent the binding of bacteria to the intestinal wall, thus preventing their translocation from the gastrointestinal tract to the body cavity. 13 As for systemic effects, Murphy et al. 14 demonstrated that β-glucans in oats were able to modulate neutrophil function in male mice. Additionally, a study in multiparous dairy cows found that feeding the live yeast-based supplement OmniGen-AF increased the expression of genes that enhanced neutrophil function, specifically in the IL-4 receptor and production of IL-1β. 15 In vitro studies utilizing human blood observed an increase in the activity of natural killer cells towards tumor cells, decreased the production of IFN-γ, and increased antioxidant activity when blood cell cultures were incubated with yeast and yeast extracts. 16 Therefore, it appears that components of the YCW are able to modulate immunity directly through actions on immune cells and may act as an antioxidant, as well as an anti-inflammatory agent.
Vaginal temperature is one of the most common and useful measurements to evaluate the health status of cattle. 7 As acute stress has been demonstrated to increase core body temperature, 17 it is not unusual to see a slight rise in vaginal temperature during the initial hours of sampling before the administration of LPS as humans will be entering the facility where the cattle were housed for the first time. Therefore, the slight increase in vaginal temperature likely represents a minor stress response to the increased activity in the animal facility.
The vaginal temperature response to LPS is similar to that observed in other studies in which LPS was administered to cattle.6,7,18 The lower vaginal temperatures observed following administration of LPS in YCW-supplemented cattle is similar to that observed in steers supplemented with live yeast and/or YCW. 6 An increase in core body temperature is a necessary response to a pathogen, as greater body temperatures contribute to pathogen clearance. 19 The lower vaginal temperature response observed in YCW-supplemented heifers may infer that these heifers were healthier than the typical cattle coming into a feedlot and did not need as great of a vaginal temperature response to dispatch the infectious agent. This is supported by the lower IL-6 concentrations observed in YCW-supplemented heifers following administration of LPS. The secretion of IL-6, as well as TNF-α and IL-1β, has been reported to increase core body temperature;20–22 however, there were no differences in the secretion of TNF-α in the current study.
There were very limited behavioral signs of sickness exhibited by the heifers in the present study in response to LPS administration. Perhaps the heifers were more fit prior to the LPS challenge, as stated earlier, which decreased the visible behavioral signs of sickness. The low cytokine concentrations observed in the current study further support this claim. The sickness scores observed in the current study are less than what has been previously observed in Brahman bulls; 8 however, Bos indicus-influenced cattle are more sensitive to LPS than Bos taurus-influenced cattle, as demonstrated in the necessity to administer a lower dose of LPS to Brahman-influenced cattle than Bos taurus cattle in order to prevent mortality. In addition, studies have demonstrated differences in sickness behavior between heifers and bulls, 23 which may further contribute to the limited sickness behaviors observed in the current study. Previous studies from our laboratory have found a greater magnitude of sickness behavior in calm and intermediate Brahman bulls, 8 and in crossbred steers (with or without chromium supplementation) when administered the same dose of LPS. 18 Therefore, there may be several factors that influenced the magnitude of the sickness behavior response observed in the current study.
Cortisol is well known for its negative role in regulation of the immune system; however, in response to a pathogen, cortisol is necessary to prevent a hyperinflammatory state caused by increased concentrations of pro-inflammatory cytokines. Serum cortisol concentrations were greater in control than in YCW-supplemented heifers following administration of LPS. This finding corresponds to lower concentrations of IL-6 observed in YCW-supplemented heifers and lower vaginal temperatures following administration of LPS. Therefore, a muted cortisol response may be a result of a lesser cytokine response. Collier et al. 24 reported lower cortisol concentrations in live yeast-supplemented pigs 1 h after administration of LPS. In cattle, Carroll et al. 6 reported a lower cortisol response in steers supplemented with a combination of live yeast and YCW, which is similar to results of the current study. It should be noted, however, that cortisol concentrations in the aforementioned study peaked at concentrations twice as high as observed in the current study. The differences observed between studies may be a result of the overall health/immune status of the cattle and prior pathogen exposure, as greater morbidity was observed in the aforementioned cattle study. Indeed, the cattle in the current study were not observed to have any illness while supplemented for the 37 d prior to the challenge.
As mentioned earlier, cytokine concentrations produced following LPS administration were relatively low in the present study compared with other published studies in which LPS was administered to cattle.25–29 Nonetheless, only one of the aforementioned studies 28 used the same dose of LPS and two of the aforementioned studies25,29 used a different serotype of LPS (O55:B5), which may have contributed to greater cytokine concentrations post-LPS administration. A previous study in yeast-supplemented cattle reported a tendency for greater IFN-γ concentrations prior to administration of LPS. 6 Because of the variability observed in pre-LPS IFN-γ concentrations in the current study, no differences were observed. In addition, no differences in IFN-γ concentrations were observed post-LPS administration in the current study. The current study also indicated no effect of dietary treatment on TNF-α concentration, which is in contrast to a study by Collier et al. 24 who reported that administration of live yeast to young pigs accelerated and increased the TNF-α response to LPS. The difference observed between the current study and the study by Collier et al., 24 in which pigs were used, may demonstrate differences between animal species or differences between a live yeast product and a YCW product.
In contrast to IFN-γ and TNF-α, differences in IL-6 concentrations were apparent following LPS administration in the current study. The supplementation of YCW products decreased serum IL-6 concentrations, which is consistent with the lower vaginal temperature and cortisol responses observed. The lower concentrations of IL-6 observed in YCW-supplemented heifers also agrees with the lower expression of IL-6 that has been observed in a porcine intestinal epithelial cell line stimulated with live yeast and enterotoxigenic E. coli. 30 The reason for observed differences in IL-6, but not IFN-γ and TNF-α, in the current study is not clear, but it might be related to the actions of IL-6 to stimulate release of acute phase proteins and stimulate the adaptive immune response, aspects which require further study.
Conclusions
Supplementation of newly-received heifers with two YCW products decreased aspects of the physiological and cytokine responses to a provocative endotoxin challenge. Specifically, vaginal temperatures were significantly lower in YCW-C-supplemented heifers, while cortisol and IL-6 responses were significantly lower in all YCW-supplemented heifers compared with Control heifers. These preliminary data suggest that YCW supplementation may enhance the overall health status of heifers, resulting in a reduced response to subsequent immune challenge. However, additional studies are warranted to determine if other aspects of the immune system that were not measured are altered as as result of YCW supplementation. While it can be debated whether or not a greater or lesser immune response is more beneficial, data from a companion article support the current conclusions due to the fact that YCW-supplemented heifers gained weight at a faster rate following the LPS challenge compared with control heifers, consequently enhancing recovery. Therefore, YCW supplementation may be a viable feed supplement for newly-received heifers in order to reduce the negative effects of illness on productivity while at a feedlot. Future studies are warranted to gain a better understanding of the effects of YCW supplementation on the innate immune response, and may require measuring factors such as complete blood counts and functional neutrophil assays.
Footnotes
Acknowledgments
Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture (USDA). The USDA prohibits discrimination in all its programs and activities on the basis of race, color, national origin, age, disability, and, where applicable, sex, marital status, familial status, parental status, religion, sexual orientation, genetic information, political beliefs, reprisal, or because all or part of an individual's income is derived from any public assistance program. (Not all prohibited bases apply to all programs.) Persons with disabilities who require alternative means for communication of program information (Braille, large print, audiotape, etc.) should contact USDA's TARGET Center at (202) 720-2600 (voice and TDD). To file a complaint of discrimination, write to USDA, Director, Office of Civil Rights, 1400 Independence Avenue, S.W., Washington, D.C. 20250-9410, or call (800) 795-3272 (voice) or (202) 720-6382 (TDD). USDA is an equal opportunity provider and employer. The authors would like to acknowledge the technical assistance of C. Wu, J.C. Carroll, J.W. Dailey, M.A. Jennings and J.T. Cribbs.
Funding
This study was supported, in part, by Lesaffre Feed Additives, Milwaukee, WI, USA, and the Gordon W. Davis Regents Chair Endowment at Texas Tech University.
